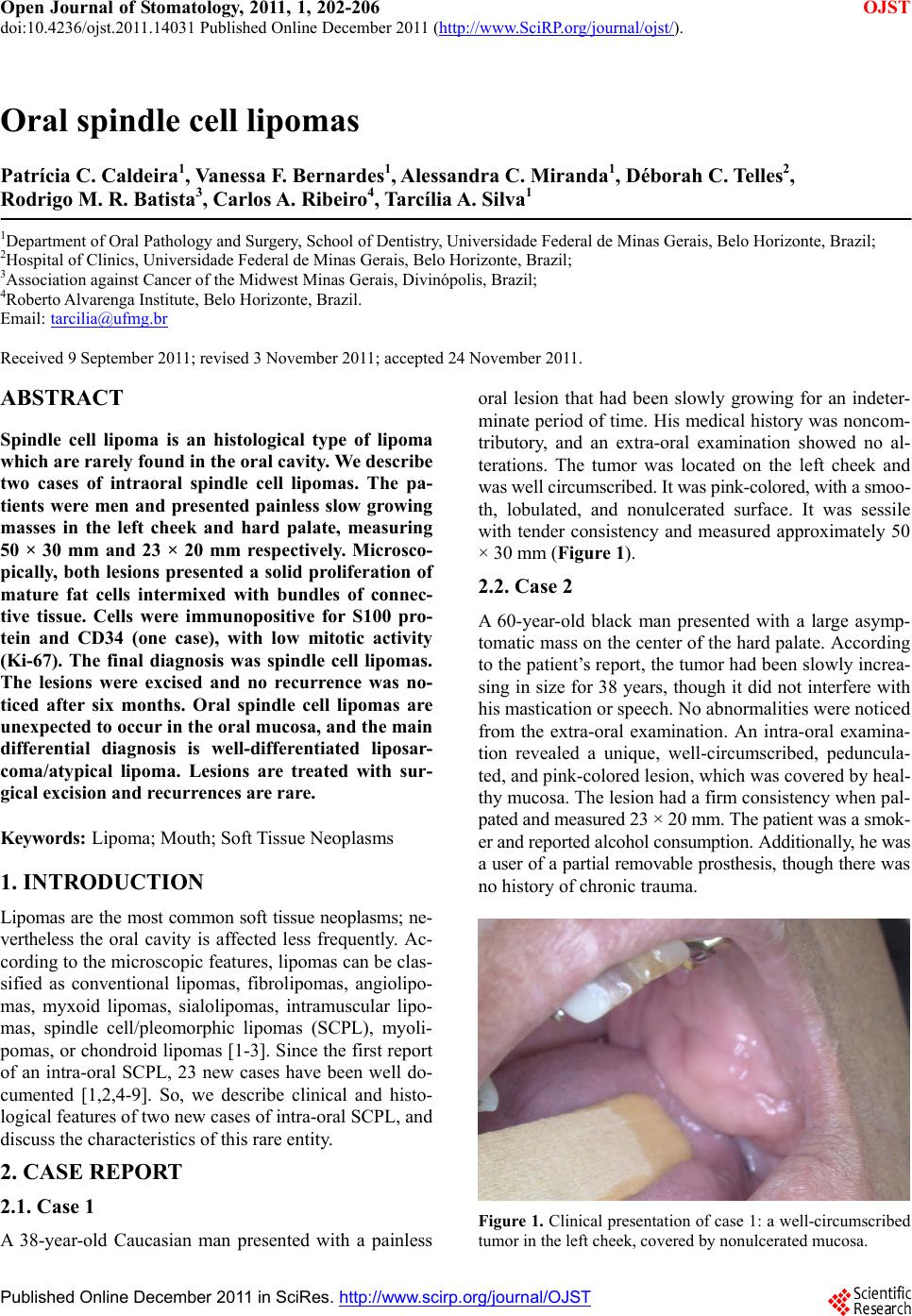
 Open Journal of Stomatology, 2011, 1, 202-206 doi:10.4236/ojst.2011.14031 Published Online December 2011 (http://www.SciRP.org/journal/ojst/ OJST ). Published Online December 2011 in SciRes. http://www.scirp.org/journal/OJST Oral spindle cell lipomas Patrícia C. Caldeira1, Vanessa F. Bernardes1, Alessandra C. Miranda1, Déborah C. Telles2, Rodrigo M. R. Batista3, Carlos A . Ribeiro4, Tarc ília A . Silva1 1Department of Oral Pathology and Surgery, School of Dentistry, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil; 2Hospital of Clinics, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil; 3Association against Cancer of the Midwest Minas Gerais, Divinópolis, Brazil; 4Roberto Alvarenga Institute, Belo Horizonte, Brazil. Email: tarcilia@ufmg.br Received 9 September 2011; revised 3 November 2011; accepted 24 November 2011. ABSTRACT Spindle cell lipoma is an histological type of lipoma which are rarely found in the oral cavity. We describe two cases of intraoral spindle cell lipomas. The pa- tients were men and presented painless slow growing masses in the left cheek and hard palate, measuring 50 × 30 mm and 23 × 20 mm respectively. Microsco- pically, both lesions presented a solid proliferation of mature fat cells intermixed with bundles of connec- tive tissue. Cells were immunopositive for S100 pro- tein and CD34 (one case), with low mitotic activity (Ki-67). The final diagnosis was spindle cell lipomas. The lesions were excised and no recurrence was no- ticed after six months. Oral spindle cell lipomas are unexpected to occur in the oral mucosa, and the main differential diagnosis is well-differentiated liposar- coma/atypical lipoma. Lesions are treated with sur- gical excision and recurrences are rare. Keywords: Lipoma; Mouth; Soft Tissue Neoplasms 1. INTRODUCTION Lipomas are the most co mmon soft tissue neoplasms; ne- vertheless the oral cavity is affected less frequently. Ac- cording to the microscopic features, lipomas can be clas- sified as conventional lipomas, fibrolipomas, angiolipo- mas, myxoid lipomas, sialolipomas, intramuscular lipo- mas, spindle cell/pleomorphic lipomas (SCPL), myoli- pomas, or chondroid lipomas [1-3]. Since the first report of an intra-oral SCPL, 23 new cases have been well do- cumented [1,2,4-9]. So, we describe clinical and histo- logical features of two new cases of intra-oral SCPL, and discuss the characteristics of this rare entity. 2. CASE REPORT 2.1. Case 1 A 38-year-old Caucasian man presented with a painless oral lesion that had been slowly growing for an indeter- minate period of time. His medical history was noncom- tributory, and an extra-oral examination showed no al- terations. The tumor was located on the left cheek and was well circumscribed. It was pink-colored, with a smoo- th, lobulated, and nonulcerated surface. It was sessile with tender consistency and measured approximately 50 × 30 mm (Figure 1). 2.2. Case 2 A 60-year-old black man presented with a large asymp- tomatic mass on the center of the hard palate. According to the patient’s report, the tumor had been slowly increa- sing in size for 38 years, though it did not interfere with his mastication or speech. No abnormalities were noticed from the extra-oral examination. An intra-oral examina- tion revealed a unique, well-circumscribed, peduncula- ted, and pink-colored lesion, which was covered by heal- thy mucosa. The lesion had a firm consistency wh en pal- pated and measured 23 × 20 mm. The patient was a smok- er and reported alcohol consumption. Additionally, he was a user of a partial removable prosthesis, though there was no history of chronic trauma. Figure 1. Clinical presentation of case 1: a well-circumscribed tumor in the left cheek, covered by nonulcerated m u co s a.  P. C. Caldeira et al. / Open Journal of Stomatology 1 (2011) 202-206 203 2.3. Microscopic Findings In case 1, an incisional biopsy was performed, while in case 2 the lesion was completely excised. Both proce- dures were performed under local anesthesia. Specimens were routinely processed and stained with hematoxylin and eosin (HE). Microscopically, both lesions presented a solid prolif- eration of mature fat cells intermixed with bundles of connective tissue (Figure 2(a)). Fat cells showed a large univacuolated cytoplasm with a small, flattened nucleus in the periphery. Spind le cells with single, vesicular, oval or fusiform nuclei, as well as a poorly defined cytoplasm, were noticed to intermix with the fat cells. The underly- ing connective tissue presented short bundles of collagen (Figure 2(b)). Moreover, floret-like multinucleated cells could be observed (Figure 2(c)). Mitotic figures, nuclear pleomorphism, and hyperchromatism were absent. Vas- cularization and mast cells were evident, though lym- phocytes were scarce. Spindle cells in case 1 were im- munopositive for CD34 (Figure 2(d)), whereas the equi- valent cells from case 2 were negativ e. Fat cells showed immunoreactivity for the S100 protein in both cases (Figure 2(e)). Additionally, Ki-67 immunostaining re- vealed low mitotic activity (not shown). The final diagnosis for both cases was spindle cell/ pleomorphic lipoma. A complete excision was perfor- med in case 1. For both cases, no recurrence was noticed after six months of follow-up. 3. DISCUSSION SCPL usually affects the posterior neck, shoulder, and back regions of middle-aged and elderly men [5]. Occur- rence in the oral mucosa is uncommon. According to previous reported cases of oral SCPL and the present re- port, the most affected intra-oral sites are the tongue and buccal mucosa, although the floor of the mouth, lip, pal- ate, and other regions may also be involved [1,2,4-9]. The male to female ratio is 1.7:1, and the mean age is 56.2 years. Oral SCPL usually presents as a solitary, well- circumscribed, rubbery, painless, and slow-growing tu- mor, and it is covered by a healthy mucosa with sizes ranging from 3 to 50 mm (Ta b le 1 ) [1,2,4-9]. The most affected intra-oral sites are the buccal mucosa and ton- gue, although the lip, floor of the mouth, palate, and other regions may also be involved. The male to female ratio is 1.36:1, and the mean age is 56 years [1,2,4-9]. Oral SCPL usually presents as a solitary, well-circum- scribed, rubbery, painless, and slow-growing tumor, and it is covered by a healthy mucosa with sizes ranging from 3 to 50 mm [1,2 ,4-9]. Figure 2. Microscopic features: (a) solid proliferation of mature fat cells intermixed with bundles of connective tissue (HE, ×50); (b) Fat cells show a large univacuolated cytoplasm with a small flattened nucleus in the periphery. Spindle cells have single, vesicular, oval or fusiform nuclei, as well as a poorly defined cytoplasm. The underlying connective tissue presents short bundles of collagen (HE, ×100); (c) Floret-like multinucleated cell (arrow) (HE, ×400); (d) spindle cells are immunopositive for CD34 (streptavidin-biotin, ×400); (e) Fat cells show immunoreactivity for S100 protein (streptavidin-biotin, ×400). C opyright © 2011 SciRes. OJST  P. C. Caldeira et al. / Open Journal of Stomatology 1 (2011) 202-206 204 Table 1. Features of the 27 cases of spindle cell lipoma reported in the oral cavity. Case number Author/Year of publication Site Gender Age (years) Size (mm) 1 McDaniel et al., 1984 Floor of mouth Female 33 10 2 McDaniel et al., 1984 Tongue Male 52 Not available 3 Christopoulos et al., 1989 Hard palate Male 58 20 4 Lombardi and Odell, 1994 Tongue Female 63 15 5 Levy and Goding, 1989 Floor of mouth Female 74 45 6 Tosios et al., 1995 Cheek Male 55 40 7 Piattelli et al., 1999 Cheek Male 75 20 8 Piattelli et al., 2000 Cheek Male 63 25 9 Said-Al-Naief et al., 2001 Tongue Male 66 30 10 Said-Al-Naief et al., 2001 Tongue Female 53 7 11 Dutt et al., 1999 Tongue Female 42 30 12 Khoo and Lian, 1995 Cheek Male 23 50 13 Atik et al., 2002 Tongue Male 45 20 14 Darling et al., 2002 Alveolar mucosa Male 69 5 15 Piattelli et al., 2005 Floor of mouth Male 50 10 16 Billings et al., 2006 Lower lip Female 55 6 17 Billings et al., 2006 Floor of mouth Female 84 10 18 Billings et al., 2006 Cheek Male 88 10 19 Billings et al., 2006 Tongue Male 45 9 20 Billings et al., 2006 Tongue Male 67 10 21 Billings et al., 2006 Tongue Female 31 3 22 Billings et al., 2006 Tongue Female 75 5 23 Coimbra et al., 2 006 Floor of mouth Female 29 15 24 Imai et al., 2008 Tongue Male 72 15 25 Vecchio et al., 2009 Cheek Male 52 25 26 27 Caldeira et al., 2011 (present cases) Cheek Hard palate Male Male 38 60 50 23 Generally, clinical differential diagnoses include clas- sic lipomas, fibromas, fibroepithelial polyps, benign ner- ve sheath tumors, salivary gland neoplasms, and herni- ated buccal fat pads [1]. Diagnostic hypotheses of case 1 comprised two categories: mesenchymal tumors of soft tissues (lipoma, neurilemmoma, neurofibroma) and non- neoplastic proliferative lesions (fibrous hyperplasia/tra- umatic fibroma). The most common location for oral lipomas is the buccal mucosa, and it was also considered because of the tender consistency of the lesion [4-6]. Considering the clinical aspects the herniated buccal fat pad, also called “traumatic pseudolipoma”, must be in- cluded in differential diagnosis [1]. It presents as a ses- sile or pedunculated mass originated by trauma and is common in children but may also be present in adults. When it become epithelialized after trauma is not possi- ble to distinguish from a true lipoma [1]. Because of clinical features, other lesions, such as oral dermoid, epidermoid cysts, and oral lymphoepitelial cysts should be considered in the differential diagnostic of oral lipo- mas [5]. In addition, in both cases the neurilemoma and neurofibroma were included in the differential diagnosis because of their similar clinical findings. In the case 1, an incisional biopsy was performed due the extent of the lesion. Referring to case 2, the differential diagnosis of the palatal mass includes the traumatic or irritation fibroma, benign and malignant salivary gland neoplasms, benign neural tumors and chronic abscess. These lesions have many characteristics in common and may appear clini- cally indistinguishable. Emphasis is placed on the im- portance of obtaining a thorough, comprehensive health and dental history and collecting relevant information. The period of duration of the lesion suggested a benign lesion. The hypothesis of chronic abscess was excluded after the dental clinical evaluation and the considered clinical diagnostic hypotheses were benign mesenchy- mal neoplasms, including neurilemmoma and neurofi- broma, as considered in the former case, benign salivary gland tumor (pleomorphic adenoma), and fibrous hyper- plasia/traumatic fibroma. Considering the clinical as- pect and the long period of evolution of the lesion an ex- cisional biopsy was performed in this case. Imaging examination using ultrasonography, compu- ted tomography, and magnetic resonance may aid the diagnosis of oral lipomas. Unfortunately, there is quite C opyright © 2011 SciRes. OJST  P. C. Caldeira et al. / Open Journal of Stomatology 1 (2011) 202-206 205 little information about specific imaging features of oral SCPL and we could not access images of the present cases. However, as mention above, SCPL is only a his- topathological type of a lipoma, which may share imag- ing features with lipomas overall. On ultrasonography, oral lipomas are usually hypoechoic. On the computed tomography images, lesions have a density ranging from –123 to –83 Hounsfield units, with ill defined margins and with a characteristic homogeneous appearance with the same density as subcutaneous fat. Lesions tend to present high signal intensity on T1-weighted images with proportional reduction in signal intensity in T2. It has been stated that imaging by magnetic resonance can be useful in the delimitation of oral lipomas [10-12]. Microscopically, oral SCPL presents as a well-cir- cumscribed but rarely encapsulated lesion composed of a mixture of mature adipocytes and spindle-shaped cells in a fibrocollagenous and/or myxoid stromal background. The proportion of lipomatous, spindle cell, and stromal components may vary from one case to another. Fat cells are univacuolated with a small peripheral nucleus. Atro- phic changes in the adipocytes can be noticed, giving these cells a pseudo-lipoblastic appearance, though true lipoblasts are absent in SCPL. Spindle cells are usually uniform in size, shape and staining intensity, and they show pale-staining, vesicular, oval or compressed nuclei along with a sparse cytoplasm. The collagenous back- ground is typically composed of thick, ropey bundles. Mitosis, cellular pleomorphism, and hyperchromatism are extremely rare or absent. M ast cells are usually found, and lymphocytes may be present as well. Moreover, multi- nucleated floret-like cells can be observed, and vascular- ity is often inconspicuous [1,2,5]. In the present cases, only few differences regarding the histopathological fea- tures could be observed. In case 1, the spindle cell com- ponent was more exuberant than the adipose component, while in case 2, both were equally present. Moreover, multinucleated floret-like cells, myxoid stromal areas, and some atrophic changes in the fat cells were observed only in case 2. The spindle cells usually show positivity for CD34 and vimentin, while the adipocytes express the S100 protein [4,8]. Nevertheless, no reactivity to CD34 could be observed in case 2. Mast cells can be detected by the immuno-expression of tryptase [8] and the Ki-67 index shows a low mitotic activity. Our immunohistochemical findings are in accordance with previously reported findings. Additionally, SCPL exhibits cytogenetic aber- rations of chromosomes 13 and/or 16 [2,9]. Moreover, some authors have observed that SCPL expresses an- drogen receptors, pointing to a possible role of sex hor- mones on its pathogenesis [4]. Nevertheless, this could not be observed wi t h oral SC PL [2]. The main histopathological differential diagnosis to be considered is well-differentiated liposarcoma/atypical lipoma, a rare intra-oral lesion, which may present as a slow growing and painless mass that is found mainly in the tongue and cheeks [2]. This distinction, though dif- ficult, must be made in order to avoid overtreatment and a long-standing follow-up of the patient [2]. Some fea- tures, such as the well-circumscribed form; superficial location; slow growth; absence of lipoblasts; uniform spindle cells associated with mature, thick, and regular collagen bundles; sparse vascularity; and the absence of atypical cells are more likely to be presen t in SCPL than in well-differentiated liposarcomas/atypical lipomas [1,8, 9]. On the other hand, the most important characteristics needed to establish the diagnosis of a liposarcoma are the presence of variations in adipocyte size, atypical and enlarged adipocyte nuclei, and hyperchromatic stromal cells [13]. Moreover, spindle cells, if present, may show nuclear atypia and hypercromasia [13]. An immunohis- tochemical study is not contributory in this distinction [13]. Depending on the predominant microscopic compo- nent of the lesion, SCPL may resemble many other le- sions. If the spindle cell component is pro minent, fibrol- ipomas, neurilemmomas, neurofibromas, solitary fibrous tumors, nodular fasciitis, and malignant histiocytomas may be considered. Fibrolipomas are less cellular than SCPL and show large bundles of collagen. In cases where spindle cells present palisading nuclei, S100 stai- ning may be useful in distinguishing SCPL from a neu- rilemmoma. The possibility of a lipomatous neurofibro- ma or even a neurofibroma can be ruled out by the ab- sence of wavy nuclei in the spindle cells and their im- munonegativity for the S100 protein in SCPL [5,6] Soli- tary fibrous tumors may also show spindle cells that are positive for CD34; however, fat cells are absent in this lesion [8,9]. In nodular fasciitis, despite the plump be- nign spindle cells, there usually is little collagen. Fi- nally, malignant fibrous histiocytomas can present spin- dle cells and multinucleated giant cells, though pleo- morphisms, typical and atypical mitoses, and storiform arrangements are observed as well [3]. On the other hand, SCPL, with localized spindle cells, may be similar to conventional lipomas and lipomatous hemangiopericytomas. In lipomatous hemangiopericy- toma, the collagenized matrix and adipocytes are associ- ated with prominent hemangiopericytoma-like vessels [3]. Finally, a highly myxoid matrix may pose some con- fusion with myxolipomas and myxoid liposarcomas. Both lesions lack the wiry collagen; additionally, my- xolipoma lacks positivity for CD34, whereas myxoid liposarcoma shows ev ident lipoblastic differentiation [5]. Oral SCPL is treated with surgical excision and recur- C opyright © 2011 SciRes. OJST  P. C. Caldeira et al. / Open Journal of Stomatology 1 (2011) 202-206 Copyright © 2011 SciRes. 206 [7] Darling, M., Thompson, I. and Schneider, J. (2002) Spin- dle cell lipoma of the alveolar mucosa: A case report. Oral Surgery, Oral Medicine, Oral Pathology, Oral Ra- diology & Endodontics, 93, 171-173. doi:10.1067/moe.2002.120520 rences are not expected [7]. 4. ACKNOWLEDGEMENTS This study was supported in part by grants from PRPq-UFMG; FAPEMIG, CAPES, and CNPq, Brazil. Dr. Silva and Dr. Bernardes are research fellows of CNPq. [8] Imai, T., Michizawa, M., Shimizu, H., Imai, T., Yama- moto, N. and Yura, Y. (2008) Bilateral multiple spindle cell lipomas of the tongue. Oral Medicine, Oral Pathol- ogy, Oral Radiology & Endodontics, 106, 264-269. REFERENCES [9] Piattelli, A., Perrotti, V., Fioroni, M. and Rubini, C. (2005) Spindle cell lipoma of the floor of the mouth: Report of a case. Auris Nasus Larynx, 32, 205-207. doi:10.1016/j.anl.2005.01.013 [1] Said-Al-Naief, N., Zahurullah, F.R. and Sciubba, J.J. (2001) Oral spindle cell lipoma. Annals of Diagnostic Pathology, 5, 207-215. doi:10.1053/adpa.2001.26973 [10] Chikui, T., Yonetsu, K., Yoshiura, K., Miwa, K., Kanda, S., Ozeki, S. and Shinohara, M. (1997) Imaging findings of lipomas in the orofacial region with CT, US, and MRI. Oral Medicine, Oral Pathology, Oral Radiology & En- dodontics, 84, 88-95. [2] Vecchio, G., Amico, P., Caltabiano, R., Colella, G., Lan- zafame, S. and Magro, G. (2009) Spindle cell/pleomor- phic lipoma of the oral cavity. Journal of Craniofacial Surgery, 20, 1992-1994. doi:10.1097/SCS.0b013e3181bd2dad [11] Bandéca, M.C., De Pádua, J.M., Nadalin, M.R., Ozório, J.E., Silva-Sousa, Y.T. and Da Cruz, P.D.E. (2007) Oral soft tissue lipomas: A case series. Journal of the Cana- dian Dental Association, 73, 431-434. [3] Barnes, L., Eveson, J., Reichart, P., Sidransky, D., Eds. (2002) Pathology and genetics of tumours of soft tissue and bone. World Health Organization Classification of Tumours, IARC Press, Lyon, 427. [12] Cappabianca, S., Colella, G., Pezzullo, M.G., Russo, A., Iaselli, F., Brunese, L. and Rotondo, A. (2008) Lipoma- tous lesions of the head and neck region: Imaging find- ings in comparison with histological type. Radiology Me- dicine, 113, 758-770. doi:10.1007/s11547-008-0258-5 [4] Atik, E., Usta, U. and Aydin, N.E. (2002) Pleomorphic lipoma of the tongue. Otolaryngol Head Neck Surg, 126, 430-431. doi:10.1067/mhn.2002.123919 [5] Billings, S.D., Henley, J.D., Summerlin, D.J., Vakili, S. and Tomich, C.E. (2006) Spindle cell lipoma of the oral cavity. American Journal of Dermatopathology, 28, 28- 31. doi:10.1097/01.dad.0000189615.13641.4b [13] Nascimento, A.F., McMenamin, M.E. and Fletcher, C.D. (2002) Liposarcomas/atypical lipomatous tumors of the oral cavity: A clinicopathologic study of 23 cases. Annals of Diagnostic Pathology, 6, 83-93. doi:10.1053/adpa.2002.32375 [6] Coimbra, F., Lopes, J.M., Figueiral, H. and Scully, C. (2006) Spindle cell lipoma of the floor of the mouth. A case report. Medicina Oral Patologia Oral y Cirugia Bucal, 11 , E401-E403. OJST
|