Materials Sciences and Applications
Vol.3 No.12(2012), Article ID:25533,7 pages DOI:10.4236/msa.2012.312128
Removal of 17β-Estradiol by Electro-Fenton Process
![]()
1Department of Chemistry and Biology, National Institute of Applied Sciences and Technology, Tunis, Tunisia; 2Laboratory of Research, Electrochemical, Catalysis, Nanomaterials and Their Application and Didactique, National Institute of Applied Sciences and Technology, Tunis, Tunisia.
Email: *imenaimi@gmail.com
Received August 31st, 2012; revised September 29th, 2012; accepted October 27th, 2012
Keywords: 17β-Estradiol; Endocrine Disrupting Chemicals; Electro-Fenton; Hydroxyl Radicals; Degradation
ABSTRACT
The electrochemical advanced oxidation method “Electro-Fenton” has been applied to remove 17β-estradiol (17β) estra-1,3,5(10)-triéne-3,17-diol) in aqueous-acetonitrile mixture. This endocrine disrupting is a steroid hormone, releases from humans, animals and residual pharmaceuticals into the environmental water and usually causes suspected undesirable effects in aquatic organisms. The degradation of this organic compound by Electro-Fenton process was showed using a carbon felt cathode and platinum anode. The evolution of the concentration during treatment was followed up by high performance liquid chromatography (HPLC). The influence of operating conditions on the degradation of 17β-estradiol by Electro-Fenton step, such as initial concentration and catalyst concentration, has been investigated and discussed. We showed that the degradation reaction obeyed apparent first-order reaction kinetics, with absolute rate constant determined as 5.12 × 109 M−1·s−1 by competitive kinetics method taking Benzoic Acid as reference compound. The results confirm the efficiency of the Electro-Fenton process to degrade organic pollutant in aqueous-acetonitrile mixture.
1. Introduction
Endocrine disrupting chemicals (EDCs) are a recognized group of environmental emerging contaminants which have been proven to induce estrogenic effects in aquatic organisms exposed to them. Low removal efficiency in wastewater treatment plants results in the presence of this type of contaminants in surface waters and also even in finished drinking water. Among the promising technology for the treatment of wastewater containing pharmaceuticals and endocrine disrupting chemicals, during the last decade, electrochemical advanced oxidation processes (EAOPs) have been developed. Based on the outstanding oxidation ability of •OH, these techniques have been applied successfully to the destruction of toxic and persistent organic pollutants.
Several studies, based on (AOPs) such as: photocatalytic degradation [1-5], ozonation [6,7], H2O2 photolysis (UV/H2O2) [8], photo-Fenton (H2O2/Fe2+/UV) [9-12], have investigated and showed the degradation of endocrine disrupting chemicals (EDCs).
Among the EDCs, 17β-estradiol (E2) (Figure 1) is the natural and most potent intracellular estrogen, reported to be harmful due to high potent estrogenic activity even at very low concentration. E2 may also enter the aquatic environment from humans and animals either excreted in urine as glucuronide or sulfated conjugates or simply eliminated in feces in unconjugated forms [13].
In order to decrease risks of E2 toxicity in water and wastewater, the degradation of this molecule until its removal were investigated by several authors [1-15]. Except that according to literature, no study has shown the degradation of the 17β-estradiol using Electro-Fenton process.
This new electrochemical advanced oxidation process was applied successfully to the total destruction of different organic pollutants, recently, in the framework of various methods developed for destruction of organic pollutants in water [16-25].

Figure 1. Molecular structure of 17β-estradiol.
During Electro-Fenton process, hydroxyl radicals are formed from the Fenton’s reagent (Fe2+ and H2O2) which are electrochemically generated in situ. Oxygen and ferric ions are simultaneously reduced at the cathode to generate hydrogen peroxide and ferrous ions according to the following reactions:
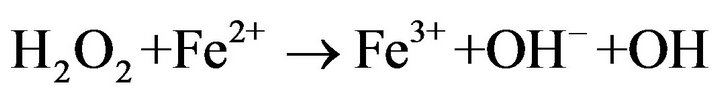 (1)
(1)
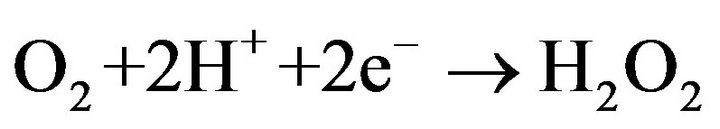 (2)
(2)
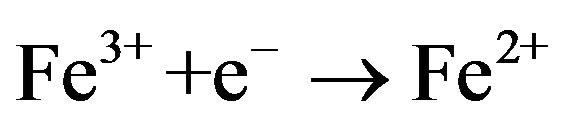 (3)
(3)
Hydrogen peroxide is produced continuously in aqueous medium by two-electron reduction of Oxygen gas at the cathode [23]. Fe2+ is usually added to the treated zing power of electro-generated H2O2. The produced Hydroxyl radicals are non-selective and highly reactive species. They react efficiently with organic compounds leading to their degradation until total mineralization.
In the present work, we have applied the electro-Fenton process to degrade, in aqueous-acetonitrile mixture, the 17β-estradiol by the action of hydroxyl radicals. The effect of some operational parameters such as effect of organic co-solvent, hormone and catalyst concentration on the efficiency of degradation E2, was investigated. The rate constant of the hydroxylation of E2 was determined by using competition kinetics.
2. Materials and Methods
2.1. Chemicals Reagent
17β-estradiol,(C18H24O2,(17β)-estra-1,3,5(10)-triéne-3,17-diol (E2) purity ≥ 98%) was purchased from Sigma-Aldrich and used without further purification. Fe2(SO4)3·7H2O (purity 99%) were purchased from Prolabo, Na2SO4 (purity 99%) used as inert supporting electrolyte were obtained from Acros organics. Acetonitrile (HPLC grade) was also obtained from Sigma-Aldrich. The solution pH was adjusted with sulfuric acid which was analytical grade, from Merck. Organic solvents and the other chemicals used were either HPLC or analytical grade from Prolabo and Aldrich.
2.2. Experimental Set Up and Analytical Methods
The carbon felt piece (80 cm2) (Carbone Lorraine RVG 4000 Mersen, Paris La Défense, France) is used as a cathode. It is placed on the inner wall of the cell covering the totality of the internal perimeter. The anode is a platinum electrode of 16 mm2 (Radiometer), placed on the centre of the cell. The initial pH of solutions is adjusted to 3 by Sulphuric Acid (H2SO4). Prior to the electrolysis, a moderate compressed air is bubbled during 10 min through the cell to saturate the solution, which is agitated continuously by a magnetic stirrer.
Fe2(SO4)3·7H2O is introduced to the cell just before the beginning of the electrolysis. Electrolyses are carried out under current controlled conditions. The current remained constant during electrolysis and samples are withdrawn at regular electrolysis times. The ionic strength is maintained constant by additions of 50 mM Na2SO4.
2.3. High Performance Liquid Chromatography (HPLC)
The evolution of 17 β-estradiol concentrations was detected using a TSP (Thermo Separation Products) High Performance Liquid Chromatography (HPLC) using Spectra system (P1000), equipped with an isocratic pump and UV/Visible detector model Spectra series UV100, fitted with reversed phase C-18 analytical column (Agilent, 5 μm; 4.6 mm × 250 mm).
The detection of E2 was carried out at 280 nm. The mobile phase was a mixture of water/acetonitrile (50:50, v/v). It was eluted with a rate of 1 mL·min−1. The injection volume was 20 µL.
3. Results and Discussion
3.1. Effect of the Experimental Parameters on Degradation of E2
The effect of some important system parameters on the degradation of E2 in the Electro-Fenton process was investigated such as effect of organic solvent, initial concentration of E2 and metal catalyst.
3.1.1. Effect of Organic Solvent
The solubility of E2 in water is very low (1.51 mg/L) [26] so we resort to the use of an organic co-solvent. However, in the presence of an organic solvent, the process will be accompanied by oxidation reactions combined. Indeed, it is known that hydrogen peroxide decomposition occurs as a radical chain process [25], in the presence of an organic solvent, chain process of H2O2 decomposition will be accompanied with conjugated solvent oxidation reactions because solvent molecules usually react with hydroxyl radicals leading to the formation of secondary radicals which are less reactive in the propagation stage [25]. This could reduce the effective •OH concentration and causes a decrease in oxidative capacity.
To evaluate the nature of co-solvent effect on ElectroFenton efficiency, we have conducted degradation tests of E2 in the presence of two different organic co-solvents: methanol and acetonitrile. The comparative results are shown in Figure 2.
Degradation of E2 is very slow in the presence of methanol as a co-solvent. This behavior may be explained as being due to the inhibitory effect of the solvent which react with •OH scavengers [27] by the following reaction:
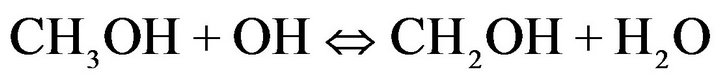 (4)
(4)
The consumption of hydroxyl radicals delays degradation of the product and leads to the formation of seconddary radicals less reactive. In the presence of acetonitrile as co-solvent, it was shown that degradation of E2 was more rapid than with methanol.
As can be seen in Figure 3, degradation decreased with decreasing co-solvent fractions. Although the detailed mechanism of acetonitrile oxidation by •OH remains unexplained at present [28], the observed behavior could be attributed to his well-known stability in anodic oxidation, and the occurrence of •OH regeneration in the presence of O2 according to the following reaction [29]:
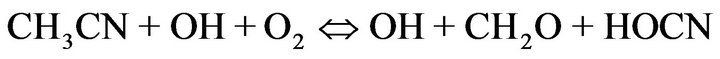 (5)
(5)
Consequently, all further experiments have been carried in acetonitrile-water mixture (30/70).
3.1.2. Effect of Initial Concentration on Degradation
The concentration of estrogenic hormones in wastewater effluents and in natural waters varies with the seasons
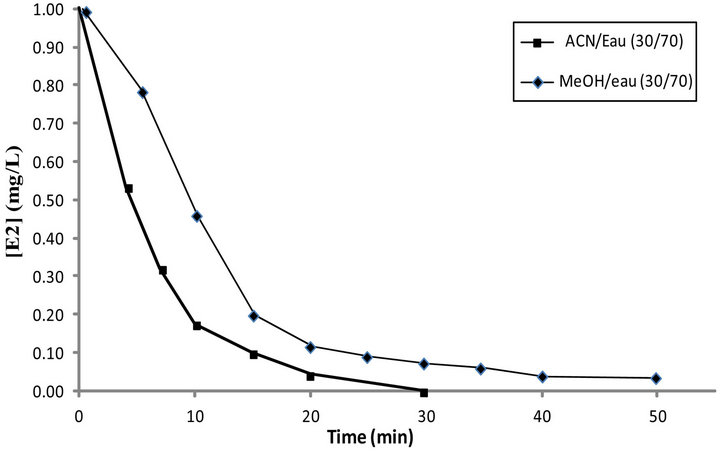
Figure 2. Effect of organic solvent type on Degradation of E2 during Electro-Fenton ([Fe2+] = 0.2 mM, [Na2SO4] = 0.05 M, pH = 3, V0 = 250 mL, [E2]0 = 1 mg·L–1, I = 200 mA).

Figure 3. Effect of fraction organic solvent on Degradation of E2 during Electro-Fenton treatment ([Fe2+] = 0.2 mM, [Na2SO4] = 0.05 M, pH = 3, V0= 250 mL, [E2]0=1 mg·L–1, I = 200 mA).
and the number of habitant. Thus, it is necessary to investigate E2 degradation during the Electro-Fenton treatment under different initial concentrations, in order to gain a better characterization of the oxidative power of the process.
Degradation of E2 was studied by changing the initial concentration levels (1, 5 and 10 mg·L−1) as depicted in Figure 4. The disappearance of E2 was fast. The hormone in acetonirile-water solutions with 1, 5 and 10 mg·L−1 concentration were completely degraded after 25, 30 and 40 min respectively. A higher concentration of E2 required a longer electrolysis time. This might be explained by the competitive consumption of hydroxyl radicals by the generated intermediates at high initial concentration [13]. In fact, the increase of initial concentration of E2 increases the number of hormone molecules present in the solution for the same number of hydroxyl radicals produced. So the amount of hydroxyl radicals will be enough to degrade high pollutant concentration leading to a decrease of the treatment efficiency [13].
In the other side, tracking the residual concentrations during the electrolysis showed that the initial concentration does not appear to affect the hormone degradation rate. In all cases, the degradation of E2 is totally completed after 40 min of electrolysis. The concentration decay fit well with the pseudo-first order kinetics. In conclusion, the results show that the Electro-Fenton process is applicable for a wide range of concentrations.
3.1.3. Effect of Metal Catalyst
The Fenton reaction could be catalyzed by several metal ions such as Fe2+, Co2+, Ag+, Cu2+... In order to optimize reaction conditions of the Electro-Fenton, hormone degradation was investigated in the presence of three different metal ions: Fe2+, Co2+ and Cu2+. The initial metal ion concentration is 0.2 mM.

Figure 4. Degradation kinetics of E2 with different initial concentrations during Electro-Fenton treatment in acetonitrile-water (30/70, v/v). Curves show pseudo-first order kinetic model analysis. ([Fe2+] = 0.2 mM, [Na2SO4] = 0.05 M, pH = 3, V0 = 250 mL, I = 200 mA).
As shown in Figure 5, degradation of E2 in the function of treatment time is exponential; the disappearance of the hormone in the presence of Acetonitrile-water mixture was fast. It was completely disappeared after 30 min in the presence of Fe2+ and after 50 min with Cu2+ and Co2+.
As it can be seen, in the presence of Fe2+, the removal of E2 was faster than the Electro-Fenton reaction with Co2+.
This behavior can be explained by scavenging reactions between Ferrous, cobalt (II) ions and hydroxyl radicals [30].
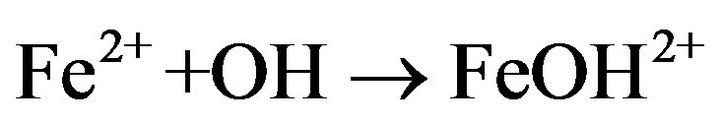 K = 4.3 × 108 M−1·s−1 (6)
K = 4.3 × 108 M−1·s−1 (6)
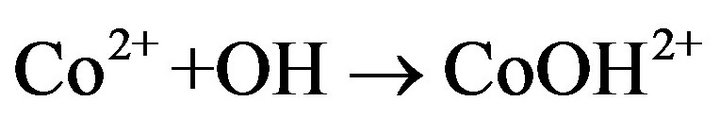 K = 8 × 105 M−1·s−1 (7)
K = 8 × 105 M−1·s−1 (7)
Using Co2+ as catalyst leads to faster formation of complexes, and changes its concentration in the media. Then, scavenging reactions harmed the treatment efficiency. Using Cu2+ as a catalyst or co-catalyst in this case does not lead to an improvement in the rate of degradation of E2. Curves clearly show that the ferrous ions lead to best degradation rates compared to Cu2+ as a catalyst with the same concentration. This can be explained by the higher oxidative potential of Fe3+/Fe2 + (E0 = 0.77 V /SHE) compared to Cu2+/Cu+ (E0 = 0.16 V/SHE) and consequently, low production of hydroxyl radicals from Fenton reaction between the generated Cu2+ and H2O2 [30].
According to several studies, amount of Fe2+ is one of important parameters which influence the efficiency of the Electro-Fenton process [16-21,25]. In this study, to determine the influence of Fe3+ concentration on the destruction rate of 17β-estradiol, a series of electrolysis was carried out with solutions of pH = 3, containing 5 mg·L−1 of E2 at 200 mA and varying Fe2+ concentrations in the range of 0.1 - 1 mM. The kinetics of the reaction was
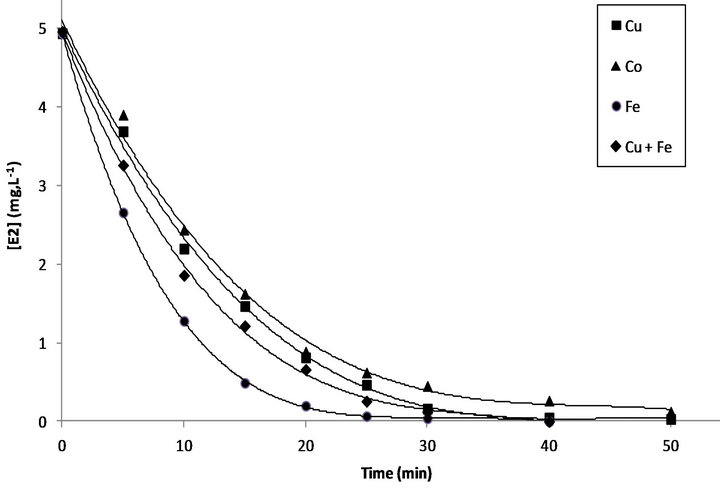
Figure 5. Degradation of E2 in acetonitrile-water solution with different metal catalyst by Electro-Fenton process. [E2]0 = 5 mg.L−1, [metal catalyst] = 0.2 mM, [Na2SO4] = 50 mM, I = 200 mA, V = 250 mL, pH = 3.
followed by HPLC chromatography.
As shown in Figure 6, the degradation rate was found to increase with increasing Fe2+ concentration up to 0.1 mM. There was no considerable change in the degradation rate for the concentration values between 0.1 and 0.2 mM. In contrast, the degradation rate was drastically decreased when the concentration value reached 0.5 mM. The degradation rate was significantly slow for the case of 1 mM. The negative influence of the higher catalyst concentration on degradation kinetics can be explained by the increase of the rate of parasitic reaction occurred during the Electro-Fenton process as follows [31]:
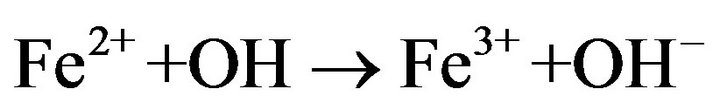 (8)
(8)
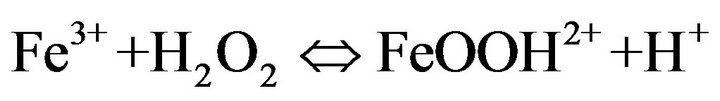 (9)
(9)
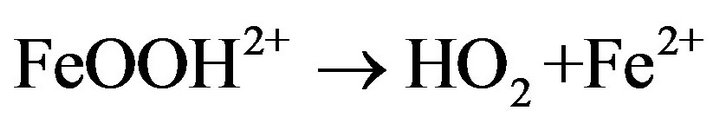 (10)
(10)
The excess ferrous ions can react with •OH, lead to the consumption of the hydroxyl radicals with a high oxidative potential [31].
This causes a decrease of the ferric reduction efficiency and affects hormone degradation rate during Electro-Fenton process. However, it is not a good idea to use over high concentration of Fe2+, because the ferrous ions are not only generate hydroxyl radicals but also are scavengers of •OH [32]. According to the obtained results, 0.2 mM was chosen as the optimal concentration value under these conditions.
3.2. Degradation Kinetics of 17β-Estradiol (E2)
Hydroxyl radicals produced in-situ by Electro-Fenton process, react on 17β-estradiol and lead to its degradation. To investigate the kinetics of this degradation, electrolyses were performed in acetonitrile-water solution with pH = 3, saturated in oxygen, without any reagent addition except ferric iron as catalyst and Na2SO4 as
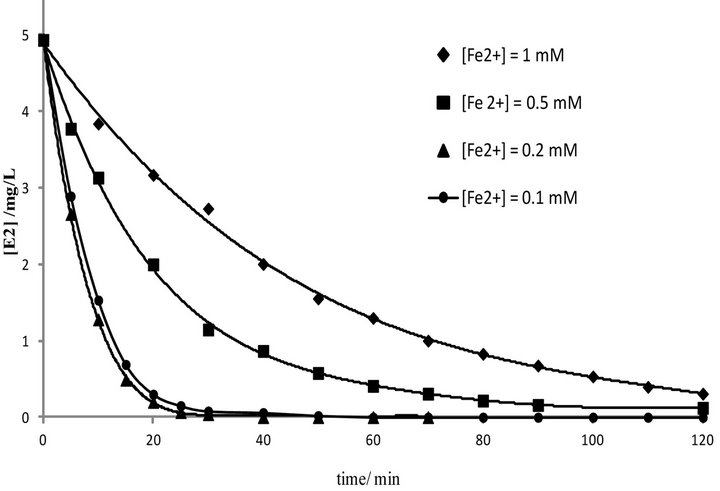
Figure 6. Degradation of E2 in acetonitrile-water solution at several Fe2+ concentrations by Electro-Fenton process. [E2]0 = 5 mg·L−1, [Na2SO4] = 50 mM, I = 200 mA, V = 250 mL, pH = 3.
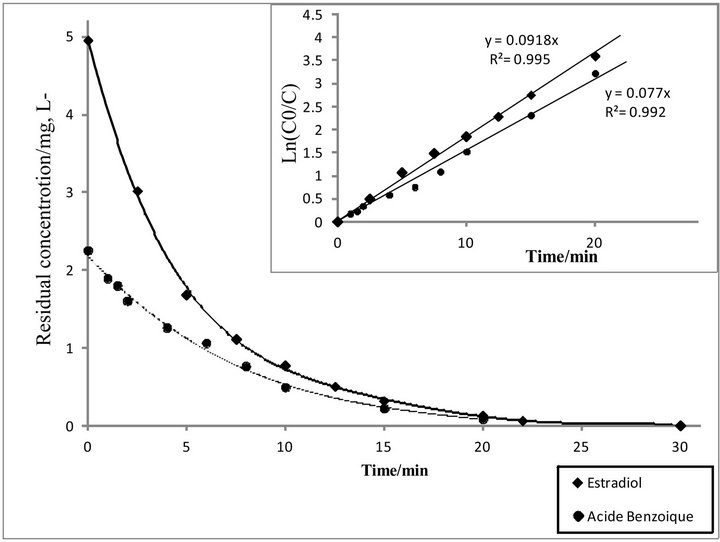
Figure 7. Competitive degradation kinetics of E2 and Benzoic Acid by Electro-Fenton treatment acetonitrile-water (30/70, v/v), [Na2SO4] = 0.05M, [Fe2+] = 0.2 mM, I = 200 mA, pH = 3, V0 = 250 mL, [E2]0 = 5 mg·L−1, [BA]0 = 2.25 mg·L−1.
supporting electrolyte. The analysis by HPLC during the entire electrolysis allowed following the evolution of hormone concentration.
As can be seen in Figure 7, the kinetics curves of the hormone were characterized by a regular and relatively fast exponential decrease of E2 concentration with electrolysis time. A pseudo-first-order kinetics model was applied to investigate the kinetics of degradation of E2. The kinetic curves, obtained for I = 200 mA, found to be best fitted with a pseudo-first order model as proved by the high correlation coefficient (0.99). Apparent rate constant value was determined analytically with Equation (1) from the slope of Ln(C0/C) vs. time (inset of Figure 7) and it was found to be 0.0918 min−1 under given experimental conditions.
In the Electro-Fenton process, the •OH formation rate is controlled by the applied current during electrolysis and the dissolved oxygen concentration. These parameters being maintained constant during electrolysis, the •OH production rate was also kept constant. As •OH is a very reactive species, it does not accumulate in the solution, and its concentration takes a steady-state value during treatment [15,24-26]. Therefore, the following equation can be written [17,25,33,34]
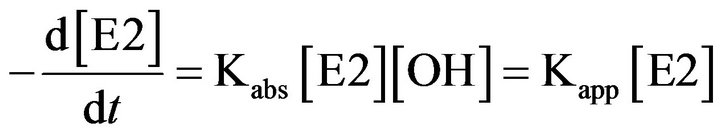 (11)
(11)
where [E2] is the hormone concentration.
In order to evaluate the apparent rate constant (Kapp) for the reaction between •OH and E2, the competitive kinetics method was applied. This method is based on simultaneous degradation of target compound and a well-characterized standard competition substrate such as benzoic acid (BA). The literature value of absolute rate constant of BA with hydroxyl radical is 4.3 × 109 M−1·s−1 [35]. Solutions containing equimolar concentrations of E2 and benzoic acid were electrolyzed at 200 mA. Apparent rate constant (Kapp) values of E2 and BA were determined as 0.0918 min−1 and 0.077 min−1 respectively.
The absolute rate constant was calculated by using the following equation
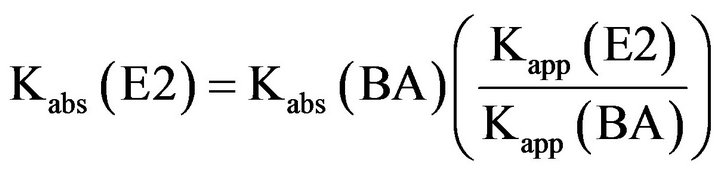 (12)
(12)
The absolute rate constant (Kabs) of the reaction between 17 β-estradiol and hydroxyl radicals was determined as 5.12 × 109 M−1·s−1 according to Equation (2). This value is in agreement with Kabs values for hydroxyllation reactions of aromatic compounds with hydroxyl radicals, and it is of the same order of magnitude (between 109 and 1010 M−1·s−1) as was reported by many authors [26,36].
4. Conclusion
In this work, we have investigated the degradation of 17β-estradiol by electrochemically generated hydroxyl radicals in acetonitrile-water mixture. The efficiency of this process is confirmed by the hormone concentration decrease during the treatment until disappearing. The results show that the Electro-Fenton process is applicable for a wide range of concentrations and able to remove E2, not only in aqueous media, but also in mixture of water and organic solvent. The degradation kinetic by hydroxyl radicals follows a pseudo-first order reaction kinetics. By means of the method of competitive kinetics using benzoic acid as standard competition substrate, the absolute second-order rate constant was determined as 5.12 × 109 M−1·s−1. This value is in agreement with Kabs values for hydroxylation reactions of aromatic compounds with hydroxyl radicals, and it is of the same order of magnitude (between 109 and 1010 M−1·s−1) of those reported by many authors. We plan in further studies to identify the degradation by-products formed during the Electro-Fenton process and to assess its toxicity. In conclusion, the treatment with Electro-Fenton process of surface waters is a significant removal pathway that needs to be taken into account, mainly for endocrine disrupting chemicals.
REFERENCES
- J. Mai, W. Sun, L. Xiong, Y. Liu and J. Ni, “Titanium Dioxide Mediated Photocatalytic Degradation of 17β-Estradiol in Aqueous Solution,” Chemosphere, Vol. 73, No. 4, 2008, pp. 600-606. doi:10.1016/j.chemosphere.2008.05.073
- Y. Zhao and J. Hu, “Photo-Fenton Degradation of 17β- Estradiol in Presence of α-FeOOHR and H2O2,” Applied Catalysis B: Environmental, Vol. 78, No. 3-4, 2008, pp. 250-258. doi:10.1016/j.apcatb.2007.09.026
- Y. Zhang, J. L. Zhon and B. Ning, “Photodegradation of Estrone and 17β-Estradiol in Water,” Water Research, Vol. 41, No. 1, 2007, pp. 19-26. doi:10.1016/j.watres.2006.09.020
- H. M. Coleman, V. Vimonses, G. Leslie and R. Amal, “Removal of Contaminants of Concern in Water Using Advanced Oxidation Techniques,” Water Science and Technology, Vol. 55, No. 12, 2007, pp. 301-306. doi:10.2166/wst.2007.421
- T. Malygina, S. Preis and J. Kallas, “The Role of pH in Aqueous Photocatalytic Oxidation of β-Estradiol,” International Journal of Photoenergy, Vol. 7, No. 4, 2005, pp. 187-191. doi:10.1155/S1110662X05000280
- R. O. Pareira, M. De Alda, J. Joglar, L. A. Daniel and D. Barcelo, “Identification of New Ozonation Disinfection Byproducts of 17 b-Estradiol and Estrone in Water,” Chemosphere, Vol. 84, No. 11, 2011, pp. 1534-1541.
- S. Irmak, O. Erbatur and A. Akgerman, “Degradation of 17β-Estradiol and Bisphenol A in Aqueous Medium by Using Ozone and Ozone/UV Techniques,” Journal of Hazardous Materials, Vol. 126, No. 1-3, 2005, pp. 54-62. doi:10.1016/j.jhazmat.2005.05.045
- E. J. Rosenfeldt, P. J. Chen, S. Kullman and K. G. Linden, “Destruction of Estrogenic Activity in Water Using UV Advanced Oxidation,” Science of the Total Environment, Vol. 377, No. 1, 2007, pp. 105-113. doi:10.1016/j.scitotenv.2007.01.096
- Y. Zhao, J. Hu and H. Chen, “Elimination of Estrogen and Its Estrogenicity by Heterogeneous Photo-Fenton Catalyst β-FeOOH/Resin,” Journal of Photochemistry Photobiology A: Chemistry, Vol. 212, No. 2-3, 2010, pp. 94- 100. doi:10.1016/j.jphotochem.2010.04.001
- Z.-H. Liu, K. Yoshinori and M. Satoshi, “Removal Mechanisms for Endocrine Disrupting Compounds (EDCs) in Wastewater Treatment—Physical Means, Biodegradation, and Chemical Advanced Oxidation: A Review,” Science of the Total Environment, Vol. 407, No. 2, 2009, pp. 731-748. doi:10.1016/j.scitotenv.2008.08.039
- M. Klavarioti, D. Mantzavinos and D. Kassinos, “Removal of Residual Pharmaceuticals from Aqueous Systems by Advanced Oxidation Processes,” Environment International, Vol. 35, No. 2, 2009, pp. 402-417. doi:10.1016/j.envint.2008.07.009
- X. H. Feng, J. F. Tu, S. M. Ding, F. Wu and N. S. Deng, “Photodegradation of 17β-Estradiol in Water by UV-Vis/ Fe(III)/H2O2 System,” Journal of Hazardous Materials, Vol. 127, No. 1-3, 2005, pp. 129-133. doi:10.1016/j.jhazmat.2005.06.039
- H. Adlercreutz, T. Fostis, C. Bannwart, E. Hamalainen, S. Bloigu and A. Ollus, “Assay of Estrogens in Human Feces,” Journal of Steroid Biochemistry, Vol. 17, No. 6, 1992, pp. 639-645. doi:10.1016/0022-4731(82)90565-9
- M. Murugananthan, S. Yoshihara, T. Rakuma, N. Uehara and T. Shirakashi, “Electrochemical Degradation of 17β- Estradiol (E2) at Boron-Doped Diamond (Si/BDD) Thin Film Electrode,” Electrochimica Acta, Vol. 52, No. 9, 2007, pp. 3242-3249. doi:10.1016/j.electacta.2006.09.073
- S. Esplugas, D. M. Bila, L. Gustavo, T. Krause and M. Dezotti, “Ozonation and Advanced Oxidation Technologies to Remove Endocrine Disrupting Chemicals (EDCs) and Pharmaceuticals and Personal Care Products (PPCPs) in Water Effluents,” Journal of Hazardous Materials, Vol. 149, No. 3, 2007, pp. 631-642. doi:10.1016/j.jhazmat.2007.07.073
- D. Mansour, F. Fourcade, N. Bellakhal, M. Dachraoui, D. Hauchard and A. Amrane, “Biodegradability Improvement of Sulfamethazine Solutions by Means of an ElectroFenton Process,” Water, Air and Soil Pollution, Vol. 223, No. 5, 2012, pp. 2023-2034. doi:10.1007/s11270-011-1002-7
- M. Murati, N. Oturan, J. J. Aaron, A. Dirany, B. Tassin, Z. Zdravkovski and M. A. Oturan, “Degradation and Mineralization of Sulcotrine and Mesotrine in Aqueous Medium by the Electro-Fenton Process: A Kinetic Study,” Environmental Science and Pollution Research, Vol. 19, No. 5, 2012, pp. 1563-1573. doi:10.1007/s11356-011-0667-1
- A. K. Abdessalem, N. Bellakhal, N. Oturan, M. Dachraoui and M. A. Oturan, “Treatment of a Mixture of Three Pesticides by Photoand Electro-Fenton Processes,” Desalination, Vol. 250, No. 1, 2010, pp. 450-455. doi:10.1016/j.desal.2009.09.072
- M. A. Oturan, M. C. Edelahi, N. Oturan, K. El Kacemi and J. Aaron, “Kinetics of Oxidative Degradation/Mineralization Pathways of the Phenylurea Herbicides Diuron, Monuron and Fenuron in Water during Application of the Electro-Fenton Process,” Applied Catalysis B: Environmental, Vol. 97, No. 1-2, 2010, pp. 82-89. doi:10.1016/j.apcatb.2010.03.026
- S. Hammami, A. Ouejhani, N. Bellekhal and M. Dachraoui, “Application of Doehlert Matrix to Determine the Optimal Conditions of Electrochemical Treatment of Tannery Effluents,” Journal of Hazardous Materials, Vol. 163, No. 1, 2009, pp. 251-258. doi:10.1016/j.jhazmat.2008.06.124
- S. Hammami, N. Oturan, N. Bellakhal, M. Dachraoui and M. A. Oturan, “Oxidative Degradation of Direct Orange 61 by Electro-Fenton Process Using a Carbon Felt Electrode: Application of the Experimental Design Methodology,” Journal of Electroanalytical Chemistry, Vol. 610, No. 1, 2007, pp. 75-84. doi:10.1016/j.jelechem.2007.07.004
- I. Sirés, J. A. Garrido, R. A. Rodríguez, E. Brillas, N. Outran and M. A. Oturan, “Catalytic Behavior of the Fe3+/ Fe2+ System in the Electro-Fenton Degradation of the Antimicrobial Chlorophene,” Applied Catalysis B: Environmental, Vol. 72, No. 3-4, 2007, pp. 382-394. doi:10.1016/j.apcatb.2006.11.016
- S. Hammami, M. A. Oturan, N. Oturan, N. Bellakhal and M. Dachraoui, “Comparative Mineralization of Textile Dye Indigo by Photo-Fenton Process and Anodic Oxidation Using Boron-Doped Diamond Anode,” Desalination and Water Treatment, Vol. 45, No. 1-3, 2012, pp. 297- 304. doi:10.1080/19443994.2012.692059
- M. A. Oturan, “An Ecologically Effective Water Treatment Technique Using Electrochemically Generated Hydroxyl Radicals for in Situ Destruction of Organic Pollutants: Application to Herbicide 2,4-D,” Journal of Applied Electrochemistry, Vol. 30, No. 4, 2000, pp. 475-482. doi:10.1023/A:1003994428571
- A. Dhaouadi, L. Monser and N. Adhoum, “Anodic Oxidation and Electro-Fenton Treatment of Rotenone,” Electrochimica Acta, Vol. 54, No. 19, 2009, pp. 4473-4480. doi:10.1016/j.electacta.2009.03.023
- A. Shareef, M. J. Angove, J. D. Wells and B. B. Johnson, “Aqueous Solubilities of Estrone, 17β-Estradiol, 17α-Ethynylestradiol, and Bisphenol A,” Journal Chemical Engineering Data, Vol. 51, No. 3, 2006, pp. 879-881. doi:10.1021/je050318c
- M. G. Gonzalez, E. Oliveros, M. Wörner and A. M. Braun, “Vacuum-Ultraviolet Photolysis of Aqueous Reaction Systems,” Journal Photochemistry Photobiology A: Chemistry, Vol. 5, No. 3, 2004, pp. 225-246. doi:10.1016/j.jphotochemrev.2004.10.002
- G. S. Tyndall, J. J. Orlando, J. T. Wallington and M. D. Hurley, “Products of the Chlorine-Atomand HydroxylRadical-Initiated Oxidation of CH3CN,” Journal of Physical Chemistry A, Vol. 105, No. 21, 2001, pp. 5380-5384. doi:10.1021/jp004318p
- A. J. Hynes and P. H. Wine, “Kinetics and Mechanism of the Reaction of Hydroxyl Radicals with Acetonitrile under Atmospheric Conditions,” Journal of Physical Chemistry, Vol. 95, No. 3, 1991, pp. 1232-1240. doi:10.1021/j100156a037
- M. Pimental, N. Oturan, M. Dezotti and M. A. Oturan, “Phenol Degradation by Advanced Electrochemical Oxidation Process Electro-Fenton Using a Carbon Felt Cathode,” Applied Catalysis B: Environmental, Vol. 83, No. 1-2, 2008, pp. 140-149. doi:10.1016/j.apcatb.2008.02.011
- A. Ozcan, M. A. Oturan, N. Oturan and Y. Sahin, “Removal of Acid Orange 7 from Water by Electrochemically Generated Fenton’s Reagent,” Journal of Hazardous Materials, Vol. 163, No. 2-3, 2009, pp. 1213-1220. doi:10.1016/j.jhazmat.2008.07.088
- W. P. Ting, M. C. Lub and Y. H. Huanga, “Kinetics of 2,6-Dimethylaniline Degradation by Electro-Fenton Process,” Journal of Hazardous Materials, Vol. 161, No. 2-3, 2009, pp. 1484-1490. doi:10.1016/j.jhazmat.2008.04.119
- S. G. Huling, R. G. Arnold, P. K. Jones and R. A. Sierka, “Predicting the Rate of Fenton-Driven 2-Chlorophenol Transformation Using a Contaminant Analog,” Journal of Environmental Engineering, Vol. 126, No. 4, 2000, pp. 348-353. doi:10.1061/(ASCE)0733-9372(2000)126:4(348)
- L. Kong and A. T. Lemley, “Kinetic Modeling of 2,4-D Degradation in Soil Slurry by Anodic Fenton Treatment,” Journal Agricultural and Food Chemistry, Vol. 54, No. 11, 2006, pp. 3941-3950. doi:10.1021/jf060046x
- G. V. Buxton, C. L. Greenstock, W. P. Helman and A. B. Ross, “Critical Review of Rate Constants for Reactions of Hydrated Electrons, Hydrogen Atoms and Hydroxyl Radicals (•OH/O–) in Aqueous Solutions,” Journal Physical Chemistry, Vol. 17, No. 2, 1988, pp. 513-886.
- I. Sires, E. Guivarch, N. Oturan and M. A. Oturan, “Efficient Removal of Triphenylmethane Dyes from Aqueous Medium by in Situ Electrogenerated Fenton’s Reagent at Carbon-Felt Cathode,” Chemosphere, Vol. 72, No. 4, 2008, pp. 592-600. doi:10.1016/j.chemosphere.2008.03.010
NOTES
*Corresponding author.

