Journal of Diabetes Mellitus
Vol.4 No.1(2014), Article ID:42856,4 pages DOI:10.4236/jdm.2014.41009
Inverse relationship between glomerular hyperfiltration and C-peptide level in Type 1 diabetes
![]()
1Diabetology Clinic, University Children’s Hospital Queen Fabiola, Free University of Brussels, Brussels, Belgium; *Corresponding Author: anissa.messaaoui@huderf.be
2Statistics Department, Erasme Academic Hospital, Free University of Brussels, Brussels, Belgium
Copyright © 2014 Anissa Messaaoui et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accordance of the Creative Commons Attribution License all Copyrights © 2014 are reserved for SCIRP and the owner of the intellectual property Anissa Messaaoui et al. All Copyright © 2014 are guarded by law and by SCIRP as a guardian.
Received 20 December 2013; revised 18 January 2014; accepted 26 January 2014
KEYWORDS
Type 1 Diabetes, Hyperfiltration, Nephropathy, C-Peptide, Glycated Hemoglobin
ABSTRACT
Background: Increased glomerular filtration rate (GFR) commonly develops in early diabetes and is closely correlated with the development of diabetic nephropathy. Objective: The aim was to study the relationship between GFR, C-peptide level and other parameters at diagnosis of Type 1 diabetes. Methods: We determined GFR, Cpeptide level, glycated hemoglobin (HbA1c), body mass index (BMI) SDS and loss of weight at diagnosis of Type 1 diabetes in 495 children (231 females). Linear and multiple regression analysis was used to test for the associations between GFR and other parameters. Results: In the 495 patients, GFR median (interquartile range) was increased vs normal values (p = 0.0001). GFR was significantly negatively correlated with age (p < 0.001) and C-peptide level (p = 0.001), and positively correlated with weight loss (p = 0.02). The multiple regression analysis showed that age (p = 0.001) and C-peptide level (p = 0.05) were independently and negatively related to GFR. Conclusions: This study shows that, at onset of Type 1 diabetes, higher the GFR, younger the age and lower the C-peptide level are. The role of this hyperfiltration in the development of later nephropathy and the putative preventive effect of C-peptide administration need to be evaluated.
1. INTRODUCTION
Glomerular hyperfiltration in diabetes mellitus was first describe by a Belgian physician, P. Cambier, in 1934 [1,2]. During the first year of Type 1 diabetes, increased glomerular filtration rate (GFR) is a predictor of nephropathy in adults [3] and in adolescents [4]. Hyperfiltration has been explained as the result of a reduced vascular resistance in the afferent arteriole, due to increased proximal electrolyte and glucose reabsorption as seen in hyperglycemia [5].
C-peptide, cleavage product of insulin synthesis, is secreted from the islets of Langerhans together with insulin. In the eighties, it was suggested that C-peptide may influence biological functions [6]. It has been shown that C-peptide, at physiological concentrations, reduces glomerular hyperfiltration mainly via constriction of the afferent arteriole, dilatation of the efferent arteriole, and inhibition of tubular Na+ reabsorption [7].
The relationship between GFR and C-peptide level at diagnosis of Type 1 diabetes has been understudied in children and adolescents. By studying GFR, C-peptide level and other parameters in a large population of young Type 1 diabetic patients, we tested the hypothesis that this relationship is true also in children.
2. METHODS
2.1. Subjects
Between 1995 and 2013, five hundred thirty-nine patients were diagnosed for diabetes at the Diabetology Clinic of the University Children’s Hospital Queen Fabiola in Brussels. Inclusion criteria were patients with Type 1 diabetes (509) and without other pathology (495). All data were collected during the first week after diagnosis of Type 1 diabetes.
2.2. Measurements
GFR was determined by single bolus intravenous injection of 51Cr-EDTA. Blood samples were taken at 90, 150 and 210 minutes and the non-compartemental Russell method was used [8]. Glycated hemoglobin (HbA1c) was measured by ion exchange, high-performance liquid chromatography, DCCT aligned (normal value < 6.0%). C-peptide level was measured on a chemi-luminescence analyzer Advia Centaur. Body weight and height were measured. Body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared and converted to standard deviation scores (SDS) based on the 1990 English references [9]. The loss of weight at diagnosis was expressed in percentage of body weight.
2.3. Statistical Analysis
Data were reported as mean (95% IC) for normally distributed values or as median (interquartile range) for nonnormally distributed values. The Mann-Whitney U test was used to test for the difference between median GFR in the 495 diabetic patients and the normal institution reference values and the difference between genders. Linear regression analysis was used to test for the associations between GFR and HbA1c, C-peptide level, age, BMI SDS and the loss of weight at diagnosis. Multiple linear regression analysis with GFR as the dependent variable was used to assess independent relationships. P ≤ 0.05 was considered to be significant.
3. RESULTS
The clinical characteristics of the study population are listed in Table 1. In the 495 patients, median age was 8.2 (4.7 - 11.6) years. There were 231 females and 264 males. GFR (Figure 1) was increased: in children aged less than 12 years [157.0 (134.0 - 179.7) ml/min/1.73m2 vs normal values 127.0 (109.1 - 147.3) ml/min/1.73m2 (p < 0.001)]; in girls aged more than 12 years [151.7 (121.8 - 171.5) ml/min/1.73m2 vs normal values 117.0 (102.2 - 133.0) ml/min/1.73m2 (p < 0.001)] and in boys aged more than 12 years [138.6 (123.5 - 159.7) ml/min/1.73m2 vs normal values 130.7 (109.2 - 149.1) ml/min/1.73m2 (p = 0.002)] (Table 2). In 36% of the patients, GFR was superior to upper normal limit (mean + 2 SD). There were no significant difference between males and females. GFR was significantly negatively correlated with age (p < 0.001, Figure 2) and C-peptide level (p = 0.001, Figure 3), and positively correlated with weight loss at diagnosis (p = 0.02). There was no significant correlation with gender, HbA1c and BMI SDS.
Variables entered into the multiple regression analysis (Table 2) were age, C-peptide level and weight loss at diagnosis. Age (p = 0.001) and C-peptide level (p = 0.05) were independently and negatively related to GFR.
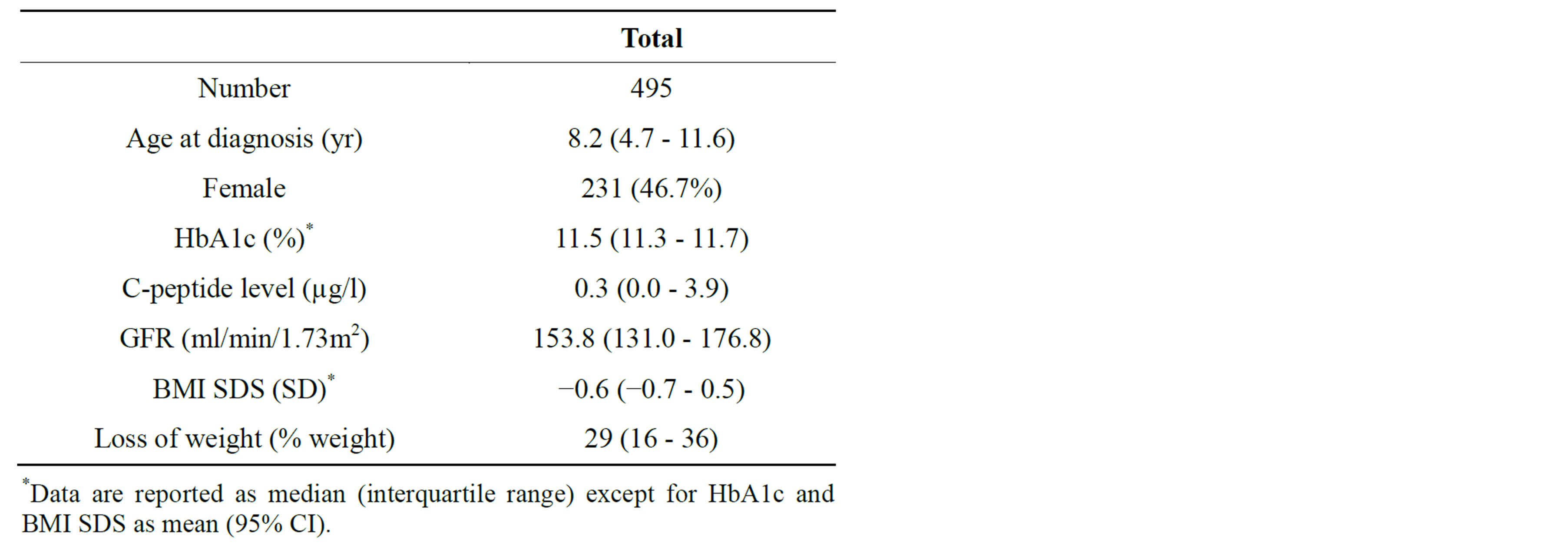
Table 1. Characteristics of the study population.
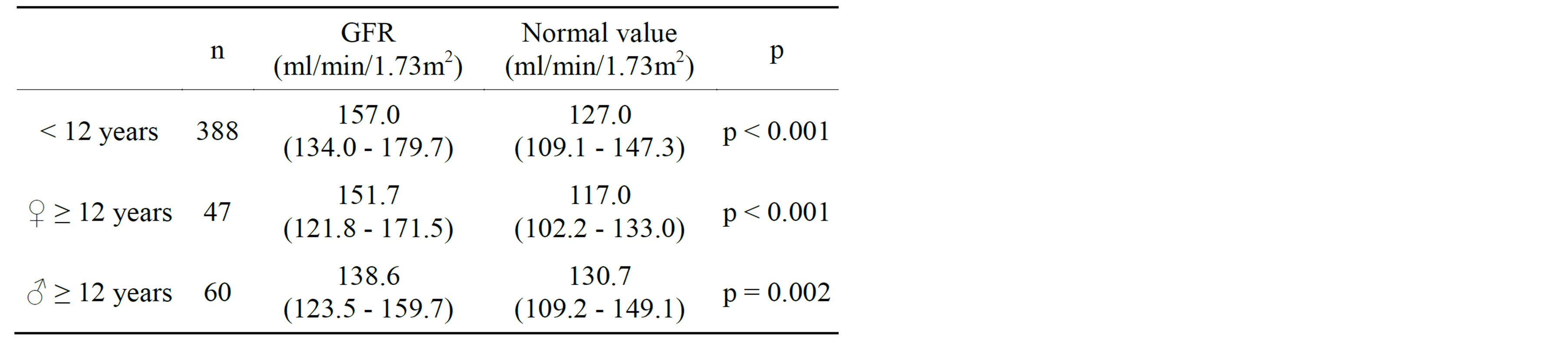
Table 2. Glomerular filtration rate.
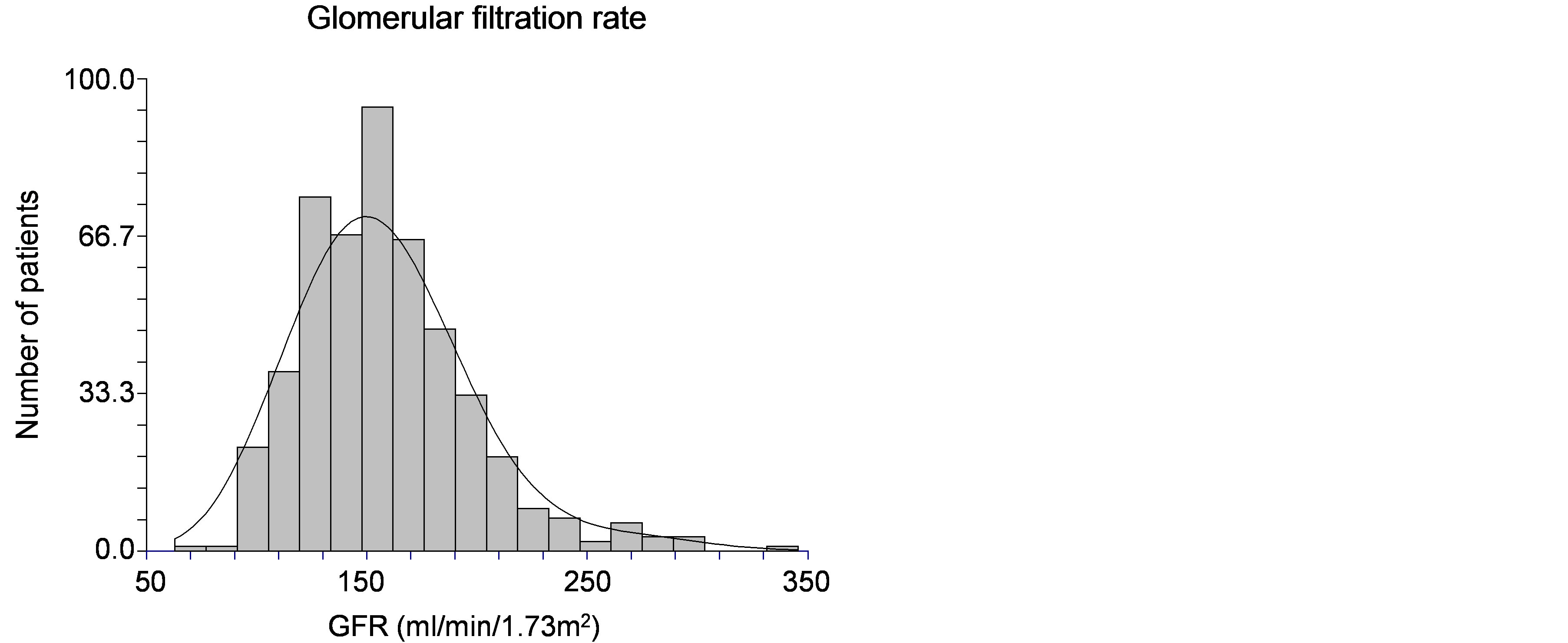
Figure 1. Glomerular filtration rate in the study population: GFR was superior to upper normal limit in 36% of the patients.
4. DISCUSSION
This study focused on the relationship between Cpeptide level and GFR at diagnosis of Type 1 diabetes in a large population. We confirm the glomerular hyperfiltration in young Type 1 diabetic patients. Higher the GFR, younger the age and lower the C-peptide level are. This hyperfiltration is independent of gender, BMI and HbA1c.
Young age and low C-peptide level are bound to a faster course of β cell destruction [10]. Therefore, hyperglycemia could be more important which could explain
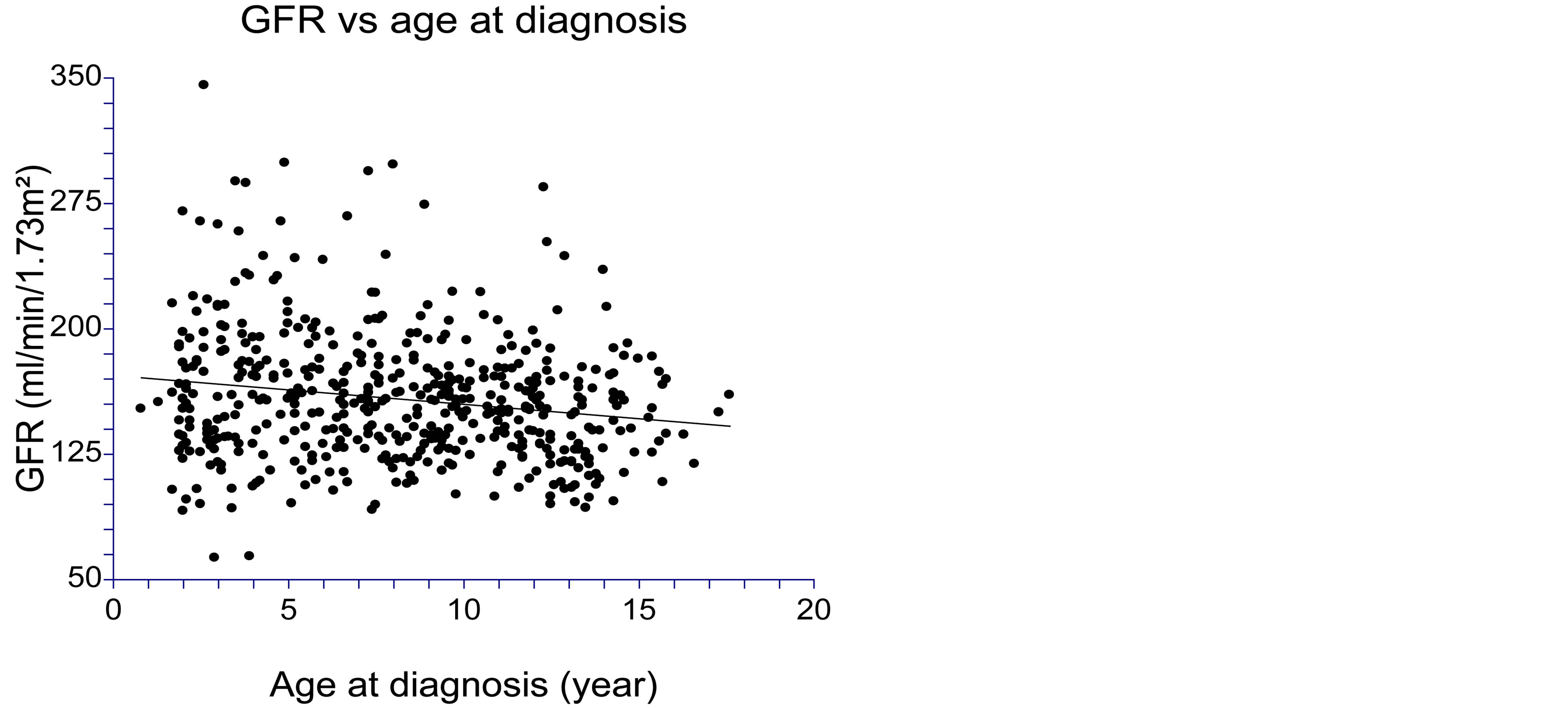
Figure 2. Glomerular filtration rate (GFR) was significantly negatively correlated with age at diagnosis at diagnosis (p < 0.001).
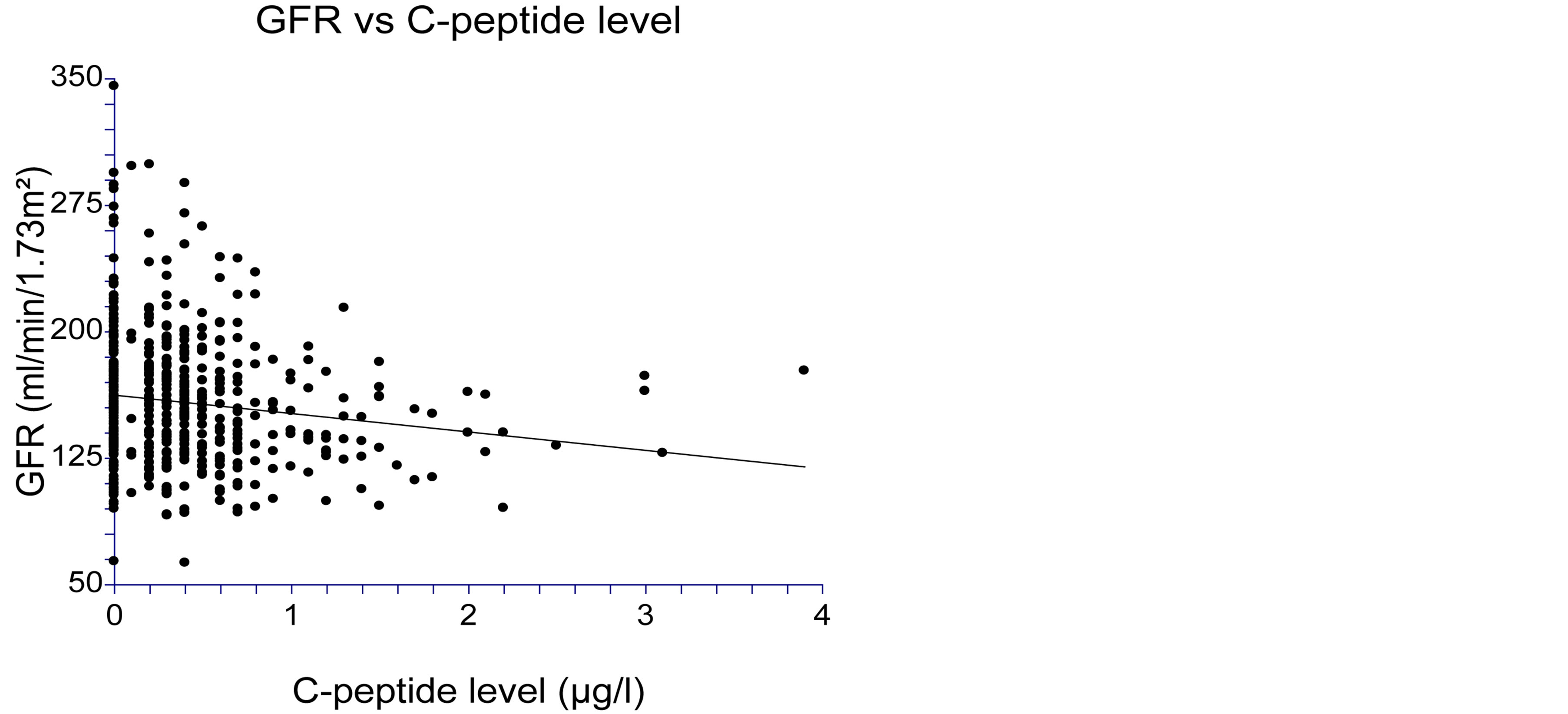
Figure 3. Glomerular filtration rate (GFR) was significantly negatively correlated with C-peptide level at diagnosis (p = 0.001).
part of the increased GFR. The lack of relationship with HbA1c could reflect that HbA1c is an index of mean blood glucose over more or less 120 days.
Acute hyperglycemia is known to increase GFR in normal and Type 1 diabetic patients [11,12]. It has been largely shown that the initial hyperfiltration normalized with good glycemic control [13,14]. Nevertheless, published studies reported that individuals with hyperfiltration were at increased risk of progression to diabetic nephropathy [15]. GFR at diagnosis could be a predictor of overt diabetic nephropathy, even in children. Glomerular hyperfiltration may play a crucial role in the subsequent development of diabetic nephropathy and thus represent potential therapeutic targets in the prevention of diabetic nephropathy.
We demonstrated that this hyperfiltration is associated with low C-peptide in children and adolescents. Studies have demonstrated a series of physiological effects of the C-peptide itself [16,17]: although the mechanisms are not fully understood, a hormonal therapeutic role of C-peptide as an active protective factor for the diabetic kidney should be considered. For example, beneficial effects of C-peptide on glomerular hyperfiltration and urinary albumin excretion have been shown [18,19]. C-peptide administrations could become a preventive treatment for the diabetic nephropathy.
This study has a solid base of 495 young patients. However, there were no healthy controls. Moreover, this cross-sectional study does not allow clarifying whether hyperfiltration at diagnosis predicts the development of later nephropathy.
A longitudinal long-term follow-up, during many years, should be necessary to analyze the eventual role of glomerular hyperfiltration at onset of Type 1 diabetes, associated with lowest C-peptide level in the development of later nephropathy. The early increased glomerular filtration may play a crucial role in the development of diabetic nephropathy and inhibition of the diabetesinduced hyperfiltration has been shown to have beneficial effects, delaying the progression of kidney damage [20].
Several studies have shown that administration of physiologically relevant doses of C-peptide reduces diabetes-induced glomerular hyperfiltration, decreases albuminuria, reduces renal hypertrophy and normalizes glomerular volume [7,18,19]. However, the effects of C-peptide administrations on kidney function need to be evaluated.
REFERENCES
- Cambier, P. (1934) Application de la théorie de Rehberg à l’étude clinique des affections rénales et du diabète. Annals of Medicine, 35, 273-299.
- Dorchy, H. (2004) Screening for subclinical complications in young Type 1 diabetic patients: Experience acquired in Brussels. Pediatric Endocrinology Reviews, 1, 380-403.
- Morgensen, C.E. (1986) Early glomerular hyperfiltration in insulin-dependent diabetes and late nephropathy. Scandinavian Journal of Clinical & Laboratory Investigation, 46, 201-206. http://dx.doi.org/10.3109/00365518609083660
- Rudberg, S., Persson, B. and Dahlquist, G. (1992) Increased glomerular filtration rate as a predictor of diabetic nephropathy-an 8-year prospective study. Kidney International, 41, 822-828. http://dx.doi.org/10.1038/ki.1992.126
- Morgensen, C. (1994) Glomerular hyperfiltration in human diabetes. Diabetes Care, 17, 770-775.
- Wojcikowski, C., Maier, V., Dominiak, K., Fussganger, R. and Pfeiffer, E. (1983) Effects of synthetic rat C-peptide in normal and diabetic rats. Diabetologia, 25, 288-290. http://dx.doi.org/10.1007/BF00279945
- Nordquist, L., Brown, R., Fasching, A., Persson, P. and Palm, F. (2009) Proinsulin C-peptide reduces diabetesinduced glomerular hyperfiltration via efferent arteriole dilation and inhibition of tubular sodium reabsorption. Renal Physiology—American Journal of Physiology, 297, F1265-F1272. http://dx.doi.org/10.1152/ajprenal.00228.2009
- Russell, C., Bischoff, P., Kontzen, F., Rowel, K., Yester, M., Lloyd, L., Tauxe, W. and Dubovsky, E. (1985) Measurement of glomerular filtration rate: Single injection plasma clearance method without urine collection. Journal of Nuclear Medicine, 26, 1243-1247.
- Cole, T., Freeman, J. and Preece, M. (1990) Body mass index reference curves for the UK, 1990. Archives of Disease in Childhood, 73, 17-24.
- Couper, J., Hudson, I., Werther, G., Warne, G., Court, J. and Harrison, L. (1991) Factors predicting residual beta-cell function in the first year after diagnosis of childhood Type 1 diabetes. Diabetes Research and Clinical Practice, 11, 9-16. http://dx.doi.org/10.1016/0168-8227(91)90135-Z
- Christiansen, J., Frandsen, M. and Parving, H. (1981) Effect of intravenous glucose infusion on renal fuction in normal man and in insulin-dependent diabetics. Diabetologia, 21, 368-373. http://dx.doi.org/10.1007/BF00252683
- Skott, P., Vaag, A., Hother-Nielsen, O., Andersen, P., Bruun, N., Giese, J., Beck-Nielsen, H. and Parving, H. (1991) Effects of hyperglycemia on kidney function, atrial natriuretic factor and plasma rennin in patients with insulin-dependent diabetes mellitus. Scandinavian Journal of Clinical & Laboratory Investigation, 51, 715-727. http://dx.doi.org/10.3109/00365519109104586
- Morgensen, C. and Anderson, M. (1975) Increased kidney size and glomerular filtration rate in untreated juvenile diabetes: Normalization by insulin treatment. Diabetologia, 11, 221-224. http://dx.doi.org/10.1007/BF00422325
- The DCCT/EDIC Research Group (2011) Intensive diabetes therapy and glomerular filtration rate in Type 1 diabetes. The New England Journal of Medicine, 365, 2366-2376. http://dx.doi.org/10.1056/NEJMoa1111732
- Magee, G., Bilous, R., Cardwell, C., Hunter, S., Kee, F. and Fogarty, D. (2009) Is hyperfiltration associated with the future risk of developing diabetic nephropathy? A meta-analysis. Diabetologia, 52, 691-697. http://dx.doi.org/10.1007/s00125-009-1268-0
- Johansson, B., Sjoberg, S. and Wahren, J. (1992) The influence of human C-peptide on renal function and glucose utilization in Type 1 (insulin-dependent) diabetic patients. Diabetologia, 35, 121-128. http://dx.doi.org/10.1007/BF00402543
- Nordquist, L. and Wahren, J. (2009) C-peptide: The missing link in diabetic nephropathy? The Review of Diabetic Studies, 6, 203-210. http://dx.doi.org/10.1900/RDS.2009.6.203
- Johansson, B., Borg, K., Fernqvist-Forbes, E., Kernell, A., Odergren, T. and Wahren, J. (2000) Benefical effects of C-peptide on incipient nephropathy and neuropathy in patients with Type 1 diabetes mellitus. Diabetic Medicine, 17, 181-189. http://dx.doi.org/10.1046/j.1464-5491.2000.00274.x
- Samnegard, B., Jacobson, S., Jaremko, G., Johansson, B. and Sjoquist, M. (2001) Effects of C-peptide on glomerular and renal size and renal function in diabetic rats. Kidney International, 60, 1258-1265. http://dx.doi.org/10.1046/j.1523-1755.2001.00964.x
- Nath, K., Kren, S. and Hostetter, T. (1986) Selective role of glomerular capillary pressure in progressive glomerular dysfunction. The Journal of Clinical Investigation, 78, 1199-1205. http://dx.doi.org/10.1046/j.1523-1755.2001.00964.x
ABBREVIATIONS
BMI—Body mass index, GFR—Glomerular filtration rate, HbA1c—Glycated hemoglobin, SDS—Standard deviation score

