American Journal of Plant Sciences
Vol.06 No.15(2015), Article ID:60130,12 pages
10.4236/ajps.2015.615254
Microalgae Lipid and Biodiesel Production: A Brazilian Challenge
Carolina T. Miranda1, Roberta F. Pinto1,2, Daniel V. N. de Lima1, Carolina V. Viegas3, Simone M. da Costa1, Sandra M. F. O. Azevedo1
1Instituto de Biofísica Carlos Chagas Filho, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brasil
2Instituto Nacional de Metrologia, Qualidade e Tecnologia-Inmetro, Rio de Janeiro, Brasil
3Departamento de Engenharia Química, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brasil
Email: ctmiranda@biof.ufrj.br
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 4 July 2015; accepted 27 September 2015; published 30 September 2015
ABSTRACT
Global increases in atmospheric CO2 and climate change are drawing considerable attention to identify sources of energy with lower environmental impact than those currently in use. Biodiesel production from microalgae lipids can, in the future, occupy a prominent place in energy generation because it represents a sustainable alternative to petroleum-based fuels. Several species of microalgae produce large amounts of lipids per biomass unit. Triacylglycerol is the fatty acid used for biodiesel production and the main source of energy reserves in microalgae. The current literature indicates that nutrient limitations can lead to triacylglycerol accumulation in different species of microalgae. Further efforts in microalgae screening for biodiesel production are needed to discover a native microalgae that will be feasible for biodiesel production in terms of biomass productivity and oil. This revision focuses in the biotechnological potential and viability of biodiesel production from microalgae. Brazil is located in a tropical region with high light rates and adequate average temperatures for the growth of microalgae. The wide availability of bodies of water and land will allow the country to produce renewable energy from microalgae.
Keywords:
Biodiesel, Microalgae, Lipids, Cultivation

1. Introduction
The use of renewable energy sources and reduction of environmental impacts is the main theme of the global public policy for reducing the effects of greenhouse gas emissions on global climate change [1] . In 2013, the emissions associated with the Brazilian energetic matrix exceeded 400 million tons of carbon dioxide released into the atmosphere, most of which are generated by the transport sector. The Brazilian annual emission per capita for producing and consuming energy is estimated to be 2.3 tons, which is 8 times less than the American and 3 times less than the European or Chinese emissions per capita. Despite the fact that 41% of the internal Brazilian energy supply is originated from renewable sources, in transportation sectors only 16.5% of fuel sources have renewable origin. The main biofuels being used are ethylic alcohol (14.3%) from sugar cane; and biodiesel (2.4%) from vegetable oils, or recycling process of fatty sources as chicken fat, pork fat and frying oil [2] . Projections made by federal institutions and energy companies, indicate continuous increase in fuel consumption by the transportation sector, in this sense, biofuels play an important role in the future of clean energy generation and environmental security [3] [4] .
Following the worldwide movement, Brazil turned its attention to biodiesel research in the late 1990s. The initial goal was to introduce biodiesel into the Brazilian energy matrix, with a focus on social inclusion and regional development. However, it was the diffusion of the National Program for Production and Use of Biodiesel (PNPB) and definition of a legal and regulatory framework for biodiesel production, distribution and investment resources that made a significant progress in the use of biodiesel. In 2005, the addition of 2% biodiesel in diesel oil was mandated, which was successfully extended to 5% by the National Energy Policy Council (CNPE) in 2010, anticipating the three-year goal set by Law No. 11.097 on January 13, 2005. To suit these targets, there was an increase in biodiesel production in the country. In 2005, production was approximately 4.6 million petroleum barrel equivalent (BEP), while in 2014, it was driven by the increase in the diesel oil mix, and production reached 19.4 million BEP, which was 4.2 times more than in 2005. Brazil currently imported a more expensive diesel than the biodiesel produced internally because the expectation for the coming years was for the biodiesel content in diesel oil to increase to 40% [2] .
As of 2013, there were 65 authorized plants located in 16 Brazilian states with a nominal capacity for biodiesel production of 7.1 million m3/year. The highest concentration of these biodiesel plants was in the states of Mato Grosso (31%), Rio Grande do Sul (11.5%) and Goiás (9.8%) (ANP 2014). The raw materials for biodiesel production in Brazil are diverse, such as palm in the north; soybeans, sunflower and peanuts in the South, Southeast and Midwest; and castor beans, which are considered the best option in the northeast but have not shown significant results in other regions. Soybean has been prominent in the commercial production of biodiesel in Brazil, and the second most common raw material is bovine fat, which accounts for approximately 10%. Even the cultivation of oilseeds is a true alternative for biofuel production, and some relevant aspects need to be considered, including the competition for land that can be used for food production, increased market prices of derived products, growing and harvesting crop-based regimes, among others [5] [6] .
Research on biodiesel production has highlighted the use of additional renewable sources such as microalgae. These microorganisms are promising raw materials in the production of third-generation fuels, especially if they are associated with the use of other high-value compounds produced by microalgae [7] .
In the last decade, an increased of government and private initiatives related to financial support for projects focus on alternatives sources for biodiesel production were observed in Brazil. Those initiatives resulted in a growing number of scientific publications in the area and advances in order to optimize the cultivation and transpose laboratory scale to production scale. In 2010 it was created a Microalgae Network, which involves 10 Brazilian institutions from different geographical locations. Currently some pilot plants for algae cultivation in higher scale are found in the states of Santa Catarina, Rio de Janeiro, Pernambuco and Rio Grande do Norte [8] .
This work aims to present some fundamental aspects of microalgae screening, cultivation, lipid improvement, oil extraction and conversion related to advances and challenges in the generation of microalgae biodiesel in Brazil.
2. Biotechnological Potential of Microalgae
Microalgae is a generic term for organisms, mostly photosynthetic and predominantly aquatic, that are distributed in the water column guided by spatial and seasonal gradients according physical, chemical and biological parameters. The main taxonomic divisions are Cyanophyta, Chlorophyta, Charophyta, Euglenophyta, Heterokontophyta, Cryptophyta, Dinophyta and Prymnesiophyta. It is estimated that there are approximately 26,000 species of microalgae in the environment, of which only a few have been identified for successful commercial application [9] [10] . Within the US Department of Energy’s Aquatic Species Program (ASP) to develop microalgae as a source of biodiesel, more than 3000 strains of microalgae from ponds and oceans have been isolated. In the Brazilian territory, representatives of all classes of algae have been identified in inland waters, soil and/or sub aerial environments, particularly from the Chlorophyta class [11] .
Chlorophyta is a division of green algae that includes approximately 500 genera and 8000 species and is preferably found in environments with a great availability of nutrients [12] . The main representatives belong to the Chlorococcales and Volvocales orders and have wide morphological variety, including unicellular, colonial and filamentous forms. The photosynthetic pigments are chlorophyll a and b; xanthophylls; lutein and prasinoxantin; and carotenoids α, β, and γ, which can be synthesized and accumulated outside of the chloroplast under conditions of nitrogen limitation or other cellular stress [13] [14] .
Microalgae are recognized by their photosynthetic efficiency, high growth rates and proportionally high level of lipids. The commercial exploitation of these organisms began in the 1960s in Japan, when strains of Chlorella were used to produce high concentrations of polysaccharides and value-added fatty acids. In the last 30 years, industries in Japan, the United States, Mexico, Italy, Israel, France, Canada, Australia and India have enhanced techniques, such as combustion, gasification, pyrolysis, fermentation and oil extraction, from microalgae biomass. The use of pigments in aquaculture, animal feed and supplements for human consumption are widely reported. Recently, the bioenergy industry has experienced an expectation by the consolidation of the use of microalgae as a viable biomass source for methane, biodiesel and bio-hydrogen production. The commercial production of microalgae for these uses shows the potential of microalgae use as a feedstock for high value-added products. Indeed, for biofuel production, microalgae can be cultivated in places unsuitable for food cultivation, eliminating wasted space by making use of non-arable, nutrient-poor land that will not support conventional agriculture [15] - [18] .
3. Cultivation
Microalgae are usually cultivated at a large scale using two types of systems: open systems (open ponds) and closed systems (photobioreactors). Open ponds are generally shallow and constructed of concrete, fiberglass or polycarbonate; the cultures are maintained in constant circulation with natural lighting and temperature. These systems are suitable for crops that tolerate extreme conditions, such as high pH (Spirulina) and high salt concentrations (Dunaliella), or those with fast growth, for example, Chlorella, Chlamydomonas, Scenedesmus and Ankistrodesmus [19] [20] . Under optimal conditions, biomass production rates are estimated to be 50 g∙m−2∙day−1 and lipid production to be 12 L∙m−2∙a−1 [21] . Until recently, only production in open ponds was considered due to the high cost of the most advanced bioreactors. However, open ponds are more susceptible to contamination by indigenous species that have low lipid productivity, predators, extreme temperatures and low light-utilization efficiency. In this sense, it is common to use the combination of open ponds and closed bioreactors, i.e. a closed- tank bioreactor that grows algae inside a contained environment in which ideal growing conditions can be artificially maintained to ensure the growth of only the desired strains of microalgae [22] .
Photobioreactors could minimize contamination by other microorganisms and have the potential for higher lipid productivity scales, compensating, in theory, for the high associated cost. The most-used materials are plastic, glass and polycarbonate. A vertical photobioreactor such as a thin-plate photobioreactor is cited in the literature for the high yields reported, 24 g∙m−2∙day−1 and lipid production of 23 L∙m−2∙a−1 [23] . These systems reduce the light path by increasing the light availability for each cell. They also have advantages in continuous operation mode and the control of culture conditions. Therefore, the amount of nutrients, temperature, light and pH can be adjusted to obtain higher biomass production in a shorter time. Even though closed bioreactors solve some of the problems of open ponds, they come with their own set of challenges, such as higher costs due to more specialized equipment and more intensive energy needs [24] - [26] .
4. Light, CO2 and Nutrients
Light provides energy to transfer protons (H+) and electrons (e-) from water molecules to NADP+, forming NADPH (nicotinamide adenine dinucleotide phosphate) and generating ATP (adenosine tri-phosphate). The light saturation point of microalgae is approximately 600 foot-candles. Once the light energy is received by the microalgae, only a fraction of it is assimilated in the form of biomass. It is assumed that no efficiency can exceed 32% of the photosynthetically active radiation (PAR) available. However, even though light is the primary substrate for the photosynthetic energy conversion, the excess of light can reduce growth by photoinhibition or promote cell death by damaging the polypeptides in the PSII reaction center. The light regime and photoperiod are also critical components in determining the biomass production of culture. Optimal light/dark regimes have been found to vary from 12:12 to 16:8 hours for most cultures [27] [28] .
CO2 is the one of the most important nutrients for microalgal growth, and high concentrations of CO2 are responsible for lipid synthesis during microalgal growth. The availability and mechanisms of the CO2 concentration are closely linked to the photosynthetic efficiency, nutrient acquisition, assimilation, cell growth and lipid production. CO2 concentrations up to 5% promote an increase in cell growth, but further investigations are still required to determine the influence of higher CO2 concentrations on the physiology of microalgae, with special attention to growth, biomass and lipid production [29] - [31] .
The nutrient availability affects the chemical composition and growth rate of the algae. The nutritional pro- cess of microalgae is based on the uptake of dissolved nutrients, mainly in their inorganic form. Thus, some elements are required in low concentrations (on the order of nanograms or micrograms per liter), such as silica, iron, manganese, molybdenum, copper, cobalt, zinc, boron and vanadium. These micronutrients are incorporated into essential organic molecules that participate in the primary reactions for these organisms. Other elements are required in higher concentrations, on the order of milligrams per liter, such as carbon, hydrogen, nitrogen, oxygen, phosphorus, potassium, sulfur and sodium. These macronutrients form structural components of biomolecules and membranes, participate in energy processes, and regulate metabolic activities, and their absence or insufficiency can affect vital functions of these microorganisms. The most important nutrients are phosphorus and nitrogen, which are only directly available for the growth of microalgae in their dissolved form [32] - [35] .
Phosphorus comprises the molecules involved in vital processes such as nucleic acids and adenosine triphosphate (ATP), enzymatic phosphorylation, dephosphorylation modulations and composition of the phospholipids. The relatively small amount of bioavailable phosphorus makes it a limiting element of aquatic primary productivity. Although microalgae are capable of absorbing phosphate from different sources, orthophosphate is preferred. The phosphorous absorption is stimulated in the presence of light and the maximum absorption rate varies depending on the intracellular and extracellular concentrations of this element. In phosphorus deficiency conditions, microalgae can achieve higher absorption rate than cells in phosphate-saturating conditions, absorbing 8 - 16 times more than the minimum cell share of phosphate and converting the internal stock in the form of polyphosphate bodies. This mechanism sustains 3 - 4 generations of growth in phosphate-depleted conditions. However, the relationship between the reduction in phosphorus present in the medium and the increase in lipid synthesis is not clear, but it is physiologically interesting when considering that these organisms have compartments capable of storing phosphorous in the form of polyphosphate bodies [36] - [38] .
Nitrogen is another important nutrient for cell growth. The inorganic nitrogen source available for absorption is ammonium ions (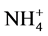 ), nitrite (
), nitrite (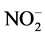 ) and nitrate (
) and nitrate (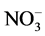 ). Nitrate is the most used form in culture medium because when using ammonia the pH could drop significantly during cell growth due to the release of H+ ions. A nitrogen limitation promotes a decrease in the content of the thylakoid membrane, the activation of acyl hydrolase, and the stimulation of phospholipid hydrolysis. On the other hand, limiting the concentrations of nitrogen promotes the activation of diacylglycerol acyltransferase, which converts acyl-CoA to triglyceride in microalgae. This indicates that the utilization of intracellular stores of nitrogen-rich compounds provides energy and carbon for the biosynthesis of triacylglycerols [39] [40] .
). Nitrate is the most used form in culture medium because when using ammonia the pH could drop significantly during cell growth due to the release of H+ ions. A nitrogen limitation promotes a decrease in the content of the thylakoid membrane, the activation of acyl hydrolase, and the stimulation of phospholipid hydrolysis. On the other hand, limiting the concentrations of nitrogen promotes the activation of diacylglycerol acyltransferase, which converts acyl-CoA to triglyceride in microalgae. This indicates that the utilization of intracellular stores of nitrogen-rich compounds provides energy and carbon for the biosynthesis of triacylglycerols [39] [40] .
Several factors can stress microalgae. For biotechnological purposes, the limitation of nutrients is the most studied stress condition because it leads to the accumulation of high value compounds. When light sources and CO2 are available and the photosynthetic mechanism is active, there is a decrease in cell division, photosynthetic rates, protein synthesis and the deviation of the photosynthetic energy waived for cell division for the accumulation of carbohydrates and lipid synthesis [41] [42] .
5. Lipids from Microalgae
Different types of microalgae have different lipid yields, i.e. lipid productivity largely depends on the microalgae species [43] . Using carbon, photosynthetically fixed microalgae synthesize fatty acids from sources of inorganic carbon and also directly from organic carbon sources such as glucose and acetate. The lipids are compounds of glycerol, sugar or base esterified with 12 - 22 carbon chains, saturated or unsaturated, primarily classified into polar lipids and neutral lipids. Polar lipids include phospholipids, galactolipids, sphingolipids, steroids and prostaglandins. The neutral lipids are divided into monoacylglycerol, diacylglycerol and triacylglycerol, along with the number of esterifications present in fatty acid chain. Fatty acids comprise the largest fraction of lipids, between 25% - 60% of the total lipids. Among the neutral lipids, triacylglycerols are targeted for biodiesel production [44] .
As observed for most photosynthetic organisms, the biosynthesis of fatty acids from microalgae is divided into three main stages: 1) acetyl coenzyme A (acetyl CoA) synthesis in the cytosol; 2) saturated fatty acids synthesis with 16 - 18 carbons, desaturation and carbon chain elongation; and 3) acyl glycerol (triglyceride) synthesis. Lipids are produced throughout the cell cycle, and initially the synthesis is directed to form structural lipids for primary metabolism and then to the triacylglycerol formation. Triacylglycerols are the main form of energy stored in microalgae, and the composition of this fatty acid class seems to be related to not only the genetics of species but also environmental conditions and cellular stress by which microalgae are submitted. Illustrations about metabolic pathways of fatty acid biosynthesis in microalgae can be accessed through the Kyoto Encyclopedia of Genes and Genomes (KEGG), as in [45] and [46] . Despite some progress has been made in order to unravel the genetic and biochemical processes of TAG accumulation in microalgae a unison explanation for this phenomenon is yet to be found.
The concentration and composition of oils in microalgae can vary significantly at different stages of growth and among species. For example, in the early stages of growth, green algae produce relatively high concentrations of polar lipids and polyunsaturated. While in the stationary phase, green algae produce predominantly neutral lipids [47] [48] . The major profile of lipids and fatty acids methyl are shown in Table 1 and Table 2, respectively.
Several studies support the hypothesis that microalgae fatty acids present taxonomic specificity and relative stability compared to vascular plants. It is interesting to determine a mono-specific marking between the dominant groups in the environment and to monitor the material transfers through marine or freshwater food chain.
Table 1. Composition of neutral and polar lipids of Isochrysis galbana, Phaeodactylum tricornutum and Porphyridium cruentum under continuous cultivation in tubular photobioreactor. Adapted from [49] - [51] .
Determination of the percentage of lipid via transesterification according to modification to the Lepage and Roy method [52] . In this process, the proportion of lipids refers to the unsaponifiable fraction of lipids. Using methyl esters, exclude the presence of the unsaponifiable fraction (chlorophylls and carotenoids). The value in brackets corresponds to the same cultures in the stationary growth phase.
Table 2. Fatty acids methyl esters (FAMEs) of soybean (Glycine max), cotton (Gossypium hirsutum) and microalgae. Adapted from [53] - [55] .
1Scenedesmus sp. (KCTC AG20831), 2Chlamydomonas reinhardtii (CC3491) and 3Chlorella vulgaris.
Saturated fatty acids C14:0 (myristic acid) and C16:0 (palmitic acid) are the major components of fatty acids of most classes of microalgae, while unsaturated fatty acids appear with a specific distribution between the algal groups. Green algae are characterized by a C18:3(n − 3)/∑C20 + C22 ratio and the predominance of C14:0, C16:0, C16:1, C16:4, C18:0, C18:1, C18:2, C18:2(n − 9), C18:2(n − 12) C18:3, C18:4(n − 3), C20:5(n − 3) e C22:6(n − 3) [56] - [59] .
The fatty acid profile for green algae is quite similar to other terrestrial plants, such as soy and cotton, and is being considered as a potential alternative to biofuel industries. For over 50 years, studies of microalgae for biofuel production have been described in the literature [60] . However, the microalgae fermentation processes for the production of methanol and ethanol and oil production are relatively new approaches and the studies are limited to a few strains [61] - [63] . In terms of lipid production, microalgae are divided into two categories: those with high lipid content and low growth rate and those with high growth rate and low lipid content (Table 3) [64] . Species such as Phaeodactylum tricornutum, Scenedesmus almeriensis, Chlorella vulgaris and Nannochloropsis oculata, under specific conditions, showed yields of approximately 90% of recovered methyl esters [65] [66] . However, in the Brazilian scope, the industrial potential of these organisms remains to be met, largely due to the incomplete knowledge surrounding the production of metabolites in strains isolated in Brazilian environments.
Table 3. Lipid content of microalgae. Adapted from [67] - [71] .
6. Conversion of Fatty Acids into Biodiesel
Biodiesel is a mixture of methyl or ethyl esters of long chain produced from oils by esterification and transesterification of fatty acids. The esterification is the reaction of fatty acids with an alcohol, such as methanol or ethanol, to form methyl or ethyl esters and water. Generally, mineral acids such as sulfuric acid catalyze the esterification reaction. Both reactions are reversible, and the displacement of the balance to obtain the products may occur by removing one of the products, preferably water, or using an excess of a reagent such as alcohol. The reactions start with the conversion of triglycerides to diglycerides and then diglycerides to monoglycerides; finally, the monoglycerides are converted into alkyl monoesters and glycerol [72] [73] .
The physical and chemical characteristics of the oil are determined by the nature of the fatty acids. Each oil has specific characteristics with respect to the density, viscosity, oxidation stability, solidification point and acid index. Table 4 presents data relating the properties found in microalgae biodiesel and conventional diesel [74] [75] .
The alkaline transesterification is the route most used industrially in oil matrices for economic reasons. However, it is recognized that basic catalysis has operational problems when the vegetable oil has a high content of free fatty acids, which, during the reaction, produce emulsions that hinder the separation of the glycerine esters at the end of the reaction [76] . Considering the high rate of free fatty acids in the oil from microalgae, the basic catalyst is not suitable for the process of transesterification [77] - [80] . It showed that the use of acid catalysts to produce methyl esters from microalgae resulted in higher yields than when catalysts were in the presence of base [81] .
The direct acid-catalyzed transesterification has also proven to be a promising technology for the production of biodiesel from a feedstock that contains large amounts of free fatty acids, such as seed oil of Jatropha curcas [82] . Therefore, acid transesterification biomass may prove to be an important technology for the production of biodiesel, not only from microalgae oils. Recently, hydroesterification has been viewed as an alternative to microalgae biodiesel production due to its promising results to obtain fatty acids with above 80% recovery from the hydrolysis of wet biomass [83] [84] .
7. Challenges, Prospect and Conclusion
One of the challenges for the commercialization of biodiesel from microalgae is the economic viability. Another barriers still need to be overcome since the energy demanded for cultivation, harvesting, oil extraction and conversion could be higher than the energy produced. The demand for water, nutrients and carbon are also aspects that have been considered. From a technical point of view biodiesel production from microalgae is not yet feasible but with possibilities for improvements [85] [86] .
In the course of fatty acid production, there are other useable products such as proteins, carbohydrates, essential fatty acids and other nutrient contents. The encouragement of the total use of biomass improves the industrial value of microalgae biomass and becomes economically attractive microalgae biodiesel production [86] [87] .
Table 4. Comparison of properties between diesel with microalgae biodiesel and with the standard of the American Society for Testing and Materials (ATSM). Adapted from [76] .
As part of the integrated biorefineries concept, it is recommended the efficient use of natural resources and recovery of materials, energy and nutrients contained in the by-products or generated from other processes. The utilization of domestic wastewater in microalgae ponds has demonstrated a possibility for the integrated process to produce oil for biodiesel. These ponds can be used in secondary or tertiary treatment and are advantages in terms of cost, energy requirements and greenhouse gas mitigation [88] .
Some countries, such as China, Taiwan, Israel, India, Germany, Canada and the United States, have announced initiatives for the commercial production of microalgae for biodiesel production purposes. It is clear the advance of knowledge about this theme. However, it is also observed that this effort takes place in an isolated form, with few cross-actions and exchange of know-how [89] . The Brazilian market is faced with the prospect of a significant increase in biodiesel demand by the evolution of biodiesel addition to the diesel blend. A lot of work is still needed before the potential offered by microalgae as source of biodiesel is fully exploited.
The literature cited throughout this review shows that most of these studies were performed with strains isolated from temperate regions and therefore environmental conditions different than those observed in Brazil. Another point is that these studies are not effective in elucidating what would be the best growing conditions in both laboratory and scale production, which enables the use of microalgae as the raw material for the production of biodiesel. It reiterates the need to obtain Brazilian strains that can withstand the climatic conditions to which they will be exposed to in our country, as well as to enhance the design and development of technologies that can reduce costs while increasing yields.
Acknowledgements
The authors would like to thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and the Instituto Nacional de Metrologia, Qualidade e Tecnologia (Inmetro) for the scholarships.
Cite this paper
Carolina T. Miranda,Roberta F. Pinto,1 1,Daniel V. N. de Lima,Carolina V. Viegas,Simone M. da Costa,Sandra M. F. O. Azevedo, (2015) Microalgae Lipid and Biodiesel Production: A Brazilian Challenge. American Journal of Plant Sciences,06,2522-2533. doi: 10.4236/ajps.2015.615254
References
- 1. Schenk, P.M., Thomas-Hall, S.R., Stephens, E., Marx, U.C., Mussgnung, J.H., Posten, C., Kruse, O. and Hankamer, B. (2008) Second Generation Biofuels: High-Efficiency Microalgae for Biodiesel Production. Bioenergy Research, 1, 20-43.
http://dx.doi.org/10.1007/s12155-008-9008-8 - 2. EPE (2014) Brazilian Energy Balance 2014 Year 2013. Empresa de Pesquisa Energética (EPE), Rio de Janeiro.
- 3. Kan, S.A., Rashmi, Hussain, M.Z., Prasad, S. and Banerjee, U.C. (2009) Prospects of Biodiesel Production from Microalgae in India. Renewable & Sustainable Energy Reviews, 13, 2361-2372.
http://dx.doi.org/10.1016/j.rser.2009.04.005 - 4. Pant, D., Bogaret, G.V., Diels, L. and Vanbroekhoven, K. (2010) A Review of the Substrates Used in Microbial Fuel Cells (MFCs) for Sustainable Energy Production. Bioresource Technology, 101, 1533-1543.
http://dx.doi.org/10.1016/j.biortech.2009.10.017 - 5. Chisti, Y. (2007) Biodiesel from Microalgal. Biotechnology Advances, 25, 294-306.
http://dx.doi.org/10.1016/j.biotechadv.2007.02.001 - 6. Greenwell, H.C., Laurens, L.M.L., Shields, R.J., Lovitt, R.W. and Flynn, K.J. (2010) Placing Microalgae on the Biofuels Priority List: A Review of the Technological Challenges. Journal of the Royal Society Interface, 7, 703-726.
http://dx.doi.org/10.1098/rsif.2009.0322 - 7. Harun, R., Singh, M., Forde, G.M. and Danquah, M.K. (2010) Bioprocess Engineering of Microalgae to Produce a Variety of Consumer Products. Renewable & Sustainable Energy Reviews, 14, 1037-1047.
http://dx.doi.org/10.1016/j.rser.2009.11.004 - 8. Rajendran, R., Kanimozhi, B., Prabhavathi, P., Dinesh Kumar, S., Santhanam, P., Abirami, M., Karthlk Sundaram, S. and Manikandan, A. (2015) A Method of Central Composite Design (CCD) for Optimization of Biodiesel Production fom Chlorella vulgaris. Journal of Petroleum & Environmental Biotechnology, 6, 3.
- 9. Guschina, I.A. and Harwood, J.L. (2006) Lipids and Lipid Metabolism in Eukaryotic Algae. Progress in Lipid Research, 45,160-186.
http://dx.doi.org/10.1016/j.plipres.2006.01.001 - 10. Bicudo, C.E. and Menezes, M. (2006) Gêneros de algas de águas continentais do Brasil. Chave para identificação e descrições. Editora Rima, Brazil.
- 11. Singh, A., Nigam, P.S. and Murphy, J.D. (2011) Renewable Fuels from Microalgae: An Answer to Debatable Land Based Fuels. Bioresource Thechnology, 102, 10-16.
http://dx.doi.org/10.1016/j.biortech.2010.06.032 - 12. Becker, E.W. (2004) Microalgae in Human and Animal Nutrition. In: Richmond, A., Ed., Handbook of Microalgal Culture, Blackwell, Oxford, 312-351.
- 13. Wiltshire, K.H., Boersma, M., Molle, A. and Buhtz, H. (2000) Extraction of Pigments and Fatty Acids from the Green Alga Scenedesmus obliquus (Chlorophyceae). Aquatic Ecology, 34, 119-126.
http://dx.doi.org/10.1023/A:1009911418606 - 14. Work, V.H., D’Adamo, S., Radakovits, R., Jinkerson, R.E. and Posewitz, M.C. (2012) Improving Photosynthesis and Metabolic Networks for the Competitive Production of Phototroph-Derived Biofuels. Current Opinion in Biotechnology, 23, 290-297.
http://dx.doi.org/10.1016/j.copbio.2011.11.022 - 15. Borowitzka, M.A. and Borowitzka, L.J. (1988) Microalgal Biotechnology. Cambridge University Press, Cambridge.
- 16. Ben-Amotz, A., Saish, A. and Avron, M. (1989) Mode of Action of the Massively Accumulated β-Carotene of Du- naliella bardawil in Protecting the Algae against Damage by Excess Irradiation. Plant Phisiology, 91, 1040-1043.
http://dx.doi.org/10.1104/pp.91.3.1040 - 17. Benemann, J.R. (2000) Hydrogen Production by Microalgae. Journal of Applied Phycology, 12, 291-300.
http://dx.doi.org/10.1023/A:1008175112704 - 18. Raposo, M.F.J., Morais, R.M.S.C. and Morais, A.M.M.B. (2014) Health Apllications of Bioactive Compounds from Microalgae. Life Sciences, 93, 479-486.
http://dx.doi.org/10.1016/j.lfs.2013.08.002 - 19. Richmond, A. (1988) Handbook of Microalgal Mass Culture. CRC Press, Boca Raton.
- 20. Ben-Amotz, A. (1995) New Mode of Dunaliella Biotechology: Two-Phase Growth for β-Carotene Production. Journal of Applied Phycology, 7, 65-68.
http://dx.doi.org/10.1007/BF00003552 - 21. Sheehan, J., Dunahay, T., Benemann, J. and Roessler, P. (1998) A Look Back at the US Department of Energy’s Aquatic Species Program—Biodiesel from Algae. National Renewable Energy Laboratory, Report NREL/TP-580-24190.
- 22. Koller, M., Salerno, A., Tuffner, P., Koinigg, M., Bochzelt, H., Schober, S., Pieber, S., Schnitzer, H., Mittelbach, M. and Braunegg, G. (2012) Characteristics and Potential of Microalgal Cultivation Strategies: A Review. Journal of Cleaner Production, 37, 377-388.
http://dx.doi.org/10.1016/j.jclepro.2012.07.044 - 23. Ho, S.H., Chen, C.Y. and Chang, J.S. (2012) Effect of Light Intensity and Nitrogen Starvation on CO2 Fixation and Lipid/Carbohydrate Production of an Indigenous Microalga Scenedesmus obliquus CNW-N. Bioresource Technology, 113, 244-252.
http://dx.doi.org/10.1016/j.biortech.2011.11.133 - 24. Lv, J.-M., Cheng, L.H., Xu, X.H., Zhang, L. and Chen, H.L. (2010) Enhanced Lipid Productions of Chlorella vulgaris by Adjustment of Cultivation Conditions. Bioresource Technology, 101, 6797-6804.
http://dx.doi.org/10.1016/j.biortech.2010.03.120 - 25. Liu, J., Huang, J., Sun, Z., Zhong, Y., Jiang, Y. and Chen, F. (2010) Differential Lipid and Fatty Acid Profiles of Pho- toautothrophic and Heterotrofic Chlorella zofingiensis: Assessment of Algal Oils for Biodiesel Production. Bioresource Technology, 102, 106-110.
http://dx.doi.org/10.1016/j.biortech.2010.06.017 - 26. Rodolfi, L., Zittelli, G.C., Bassi, N., Padovani, G., Biondi, N., Bonini, G. and Tredici, M.R. (2009) Microalgae for Oil: Strain Selection, Induction of Lipid Synthesis and Outdoor Mass Cultivation in a Low-Cost Photobiorreactor. Bio- technology and Bioengeneering, 102, 100-112.
http://dx.doi.org/10.1002/bit.22033 - 27. Parmar, A., Singh, N.K., Pandey, A., Gnansounou, E. and Madamwar, D. (2011) Cyanobacteria and Microalgae: A Positive Prospect for Biofuels. Bioresource Technology, 102, 10163-10172.
http://dx.doi.org/10.1016/j.biortech.2011.08.030 - 28. Wahidin, S., Idris, A. and Shaleh, S.R.M. (2013) The Influence of Light Intensity and Photoperiod on the Growth and Lipid Content of Microalgae Nannochloropsis sp. Bioresource Technology, 129, 7-11.
http://dx.doi.org/10.1016/j.biortech.2012.11.032 - 29. Giordano, M., Beardall, J. and Raven, J.A. (2005) CO2 Concentrating Mechanisms in Algae: Mechanisms, Environmental Modulation and Evolution. Annual Review of Plant Biology, 56, 99-131.
http://dx.doi.org/10.1146/annurev.arplant.56.032604.144052 - 30. Gardner, R.D., Lohman, E., Gerlach, R., Cooksey, K.F. and Peyton, B.M. (2012) Comparison of CO2 and Bicarbonate as Inorganic Carbon Sources for Triacylglycerol and Starch Accumulation in Chlamydomonas reinhardtii. Biotechnology and Bioengeneering, 110, 87-96.
http://dx.doi.org/10.1002/bit.24592 - 31. Huang, G., Chen, F., Wei, D., Zhang, X. and Chen, G. (2010) Biodiesel Production by Microalgal Biotechnology. Applied Energy, 87, 38-46.
http://dx.doi.org/10.1016/j.apenergy.2009.06.016 - 32. Fogg, G.E. and Thake, B. (1987) Algae Cultures and Phytoplankton Ecology. 3rd Edition, The University of Winsconsins Press Ltd., London.
- 33. Dowhan, W. (1997) Molecular Basis for Membrane Phospolipd Diversity: Why Are There So Many Lipids? Annual Review of Biochemistry, 66,199-232.
http://dx.doi.org/10.1146/annurev.biochem.66.1.199 - 34. Reynolds, C.S. (2006) Ecology of Phytoplankton: Ecology, Biodiversity and Conservation. Cambridge University Press, Cambridge.
http://dx.doi.org/10.1017/CBO9780511542145 - 35. Andersen, R.A., Ed. (2005) Algal Culturing Techniques. Elsevier Academic Press, New York, 578 p.
- 36. Harold, F.M. (1963) Enzymic and Genetic Control of Polyphosphate Accumulation in Aerobacter aerogenes. Journal of General Microbiology, 35, 81-90.
http://dx.doi.org/10.1099/00221287-35-1-81 - 37. Hutchinson, G.E. (1973) Eutrophication. American Scientist, 61, 269-279.
- 38. Yao, B., Xi, B.D., Hu, C.M., Huo, S.L., Su, J. and Liu, H.L. (2011) A Model and Experimental Study of Phosphate Uptake Kinetics in Algae: Considering Surface Adsorption and P-Stress. Journal of Environmental Sciences, 23, 189- 198.
http://dx.doi.org/10.1016/S1001-0742(10)60392-0 - 39. Piorreck, M., Baasch, K.H. and Pohl, P. (1984) Biomass Production, Total Protein, Chlorophylls, Lipids and Fatty Acids of Freshwater Green and Blue-Green Algae under Different Nitrogen Regimes. Phytochemistry, 23, 207-216.
http://dx.doi.org/10.1016/S0031-9422(00)80304-0 - 40. Msanne, J., Xu, D., Konda, A.R., Casas-Mollano, A.J., Awada, T., Cahoon, E.B. and Cerutti, H. (2012) Metabolic and Gene Expression Changes Triggered by Nitrogen Deprivation in the Photoautotrophically Grown Microalgae Chalmydomonas reinhardtii and Coccomyxa sp. Phytochemistry, 75, 50-59.
http://dx.doi.org/10.1016/j.phytochem.2011.12.007 - 41. Courchesne, N.M.D., Parisien, A., Wang, B. and Lan, C.Q. (2009) Enhancement of Lipid Production Using Biochemical, Genetic and Transcription Factor Engineering Approaches. Journal of Biotechnology, 141, 31-41.
http://dx.doi.org/10.1016/j.jbiotec.2009.02.018 - 42. Pancha, I., Chokshi, K., George, B., Ghosh, T., Paliwal, C., Maurya, R. and Mishara, S. (2014) Nitrogen Srtess Triggered Biochemical and Morphological Changes in the Microalgae Scenedesmus sp. CCNM1077. Bioresourource Technology, 156, 146-154.
http://dx.doi.org/10.1016/j.biortech.2014.01.025 - 43. Nacimento, I.A., Marques, S.S.I., Cabanelas, I.T.D., Pereira, S.A., Duzian, J.I., Souza, C.O., Vich, D.V., Carvalho, G.C. and Nascimento, M.A. (2013) Screening Microalgae Strais for Biodiesel Production: Lipid Productivity and Estimation of Fuel Quality Based on Fatty Acids Profiles as Selective Criteria. Bioenergy Research, 6, 1-13.
http://dx.doi.org/10.1007/s12155-012-9222-2 - 44. Derner, R.B., Ohse, S., Vilela, M., Carvalho, S.M. and Efett, R. (2006) Microalgas, produtos e aplicações. Ciência Rural, 36, 1959-1967.
http://dx.doi.org/10.1590/S0103-84782006000600050 - 45. Chiu, S.Y., Kao, C.Y., Tsai, M.T., Ong, S.C., Chen, C.H. and Lin, C.S. (2008) Lipid Accumulation and CO2 Utilization of Nannochloropsis oculata in Response to CO2 Aeration. Bioresource Technology, 100, 833-838.
http://dx.doi.org/10.1016/j.biortech.2008.06.061 - 46. Pereira, C.M.P., Hobuss, C.B., Maciel, J.V., Ferreira, L.R., Del Pino, F.B. and Mesko, M.F. (2013) Biodiesel renovável derivado de microalgas: Avanço e perspectivas tecnológicas. Quimica Nova, 35, 2013-2018.
- 47. Li, S., Xu, J.L., Chen, J., Chen, J.J., Zhou, C.X. and Yan, X.J. (2014) The Major Lipid Changes of Some Important Diet Microalgae during the Entire Growth Phase. Aquaculture, 428, 104-110.
http://dx.doi.org/10.1016/j.aquaculture.2014.02.032 - 48. Sánchez-Saavedra, M.P. and Votolina, D. (2005) The Growth Rate, Biomass Production and Composition of Chaetoceros sp. Grown with Different Light Sources. Aquacultural Engineering, 35, 161-165.
http://dx.doi.org/10.1016/j.aquaeng.2005.12.001 - 49. Cartens, M., Molina, E., Robles, A., Giménez, A. and Ibénez, M.J. (1996) Eicosapentaenoic Acid (20:4n-3) from the Marine Microalga Phaeodactylum tricornutum. Journal of the American Oil Chemists Society, 73, 1025-1031.
http://dx.doi.org/10.1007/BF02523411 - 50. Molina Grima, E., Belarbi, E.-H., Acién Fernández, F.G., Robles Medina, A. and Chisti, Y. (2003) Recovery of Microalgal Biomass and Metabolites: Process Options and Economics. Biotechnology Advances, 20, 491-515.
http://dx.doi.org/10.1016/S0734-9750(02)00050-2 - 51. Giménez Giménez, A., Ibáñez, M.J., Robles, A., Molina, E., García, S. and Esteban, L. (1998) Downstream Processing and Purification of Eicosapentaenoic (20:5n-3) and Arachidonic Acids (20:4n-6) from the Microalga Porphyridium cruentum. Bioseparation, 7, 89-99.
http://dx.doi.org/10.1023/A:1008021330785 - 52. Rodriguez-Ruiz, J., Belarbi, E.H., Sánchez, J.L.G. and Alonso, D.L. (1998) Rapid Simultaneous Lipid Extraction and Transesterification for Fatty Acid Analyses. Biotechnology Techniques, 12, 689-691.
http://dx.doi.org/10.1023/A:1008812904017 - 53. Ma, F. and Hanna, M.A. (1999) Biodiesel Production: A Review. Bioresource Technology, 70, 1-15.
http://dx.doi.org/10.1016/S0960-8524(99)00025-5 - 54. Yoo, C., Jun, S.Y., Lee, J.Y., Ahn, C.Y. and Oh, H.M. (2010) Selection of Microalgae for Lipid Production under High Levels Carbon Dioxide. Bioresource Technology, 101, S71-S74.
http://dx.doi.org/10.1016/j.biortech.2009.03.030 - 55. La Russa, M., Bogen, C., Uhmeyer, A., Doebbe, A., Filippone, E., Kruse, O. and Mussgnug, J.H. (2012) Functional Analysis of Three Type-2 DGAT Homologue Genes for Triacylglycerol Production in the Green Microalga Chla- mydomonas Lacustrian Phytoplankton. Journal of Plankton Research, 19, 469-490.
- 56. Léveillé, J.C., Amblard, C. and Bourdier, G. (1997) Fatty Acids as Specific Algal Markers in a Natural Lacustrian Phytoplankton. Journal of Plankton Research, 19, 469-490.
http://dx.doi.org/10.1093/plankt/19.4.469 - 57. Reuss, N. and Poulsen, L.K. (2002) Evaluation of Fatty Acids as Biomarkers for a Natural Plankton Community: A Field Study of a Spring Bloom and Post-Bloom Period off West Greenland. Marine Biology, 141, 423-434.
http://dx.doi.org/10.1007/s00227-002-0841-6 - 58. Sushchik, N.N., Gladyshev, M.I., Ivanova, E.A. and Kravchuk, E.S. (2010) Seasonal Distribution and Fatty Acid Compositon of Littoral Microalgae in the Yenisei River. Journal of Applied Phycology, 22, 11-24.
http://dx.doi.org/10.1007/s10811-009-9418-9 - 59. Lang, I., Hodac, L. and Friedl Feussner, T. (2011) Fatty Acid Profiles and Their Distribution Patterns in Microalgae: A Comprehensive Analysis of More than 2000 Strains from the SAG Culture Collection. Plant Biology, 11, 124.
http://dx.doi.org/10.1186/1471-2229-11-124 - 60. Uziel, M., Oswald, W.J. and Gouleke, C.G. (1975) Solar Energy Fixation and Conversion with Algal Bacterial Systems. NSF/RANN/SE/GI-39216/FR/75Q5 Sanitary Engineering Research Laboratory, University of California, Berkeley, CA.
- 61. Mata, T.M., Martins, A.A. and Caetano, N.S. (2010) Microalgae for Biodiesel Production and Other Applications: A Review. Renewable and Sustainable Energy Reviews, 14, 217-232.
http://dx.doi.org/10.1016/j.rser.2009.07.020 - 62. Pulz, O. and Gross, W. (2008) Valuable Products from Biotechnology of Microalgae. Applied Microbiology and Biotechnology, 65, 635-648.
http://dx.doi.org/10.1007/s00253-004-1647-x - 63. Ho, S.-H., Huang, S.-W., Chen, C.-Y., Hasunuma, T., Kondo, A. and Chang, J.-S. (2013) Bioethanol Production Using Carbohydrate-Rich Microalgae Biomass as Feedstock. Bioresource Technology, 135, 191-198.
http://dx.doi.org/10.1016/j.biortech.2012.10.015 - 64. Dayananda, C., Sarada, R., Usha-Rani, M., Shamala, T.R. and Ravishankar, G.A. (2007) Autotrophic Cultivation of Botryococcus braunii for the Production of Hydrocarbons and Exopolysaccharides in Various Media. Biomass and Bioenergy, 31, 87-93.
http://dx.doi.org/10.1016/j.biombioe.2006.05.001 - 65. Musharraf, S.G., Ahmed, M.A., Zehra, N., Kabir, N., Choudhary, M. and Rahman, A. (2012) Biodiesel Production from Microalgal Isolates Southern Pakistan and Quantification of FAMEs by GC-MS/MS Analysis. Chemistry Central Journal, 6, 149.
http://dx.doi.org/10.1186/1752-153X-6-149 - 66. Rawat, I., Kumar, R., Mutanda, T. and Bux, F. (2013) Biodiesel from Microalgae: A Critical Evaluation from Laboratory to Large Scale Production. Applied Energy, 103, 444-467.
http://dx.doi.org/10.1016/j.apenergy.2012.10.004 - 67. Becker, E.W. (1994) Microalgae: Biotechnology and Microbiology. Cambridge University Press, Cambridge.
- 68. Chisti, Y. (2008) Biodiesel from Microalgae Beats Bioethanol. Trends in Biotechnology, 26, 126-131.
http://dx.doi.org/10.1016/j.tibtech.2007.12.002 - 69. Su, C.H., Chien, L.J., Gomes, J., Lin, Y.S., Yu, Y.K., Liou, J.S. and Syu, R.J. (2010) Factors Affecting Lipid Accumulation by Nannochloropsis oculata in a Two Stage Cultivation Process. Journal of Applied Phycology, 23, 903- 908. http://dx.doi.org/10.1007/s10811-010-9609-4
- 70. Miranda, C.T. (2011). Avaliação dos efeitos da intensidade luminosa no crescimento e produção de lipídeos por Ankistrodesmus sp. (Chlorophyceae) visando a produção de biodiesel. Dissertation, Rio de Janeiro Federal University, Rio de Janeiro.
- 71. Pereira, C.M.P., Hobuss, C.B., Maciel, J.V., Ferreira, L.R., Del Pino, F.B., Mesko, M.F., Jacob-Lopes, E. and Neto, P.C. (2012) Biodiesel renovável derivado de microalgas: Avanços e perspectivas tecnológicas. Quimica Nova, 35, 2013-2018.
http://dx.doi.org/10.1590/s0100-40422012001000022 - 72. Fukuda, H., Kondo, A. and Noda, H. (2001) Biodiesel Fuel Production by Transesterification of Oils. Journal of Bioscience and Bioengineering, 92, 405-416.
http://dx.doi.org/10.1016/S1389-1723(01)80288-7 - 73. Marchetti, J.M., Miguel, V.U. and Errazu, A.F. (2007) Possible Methods for Biodiesel Production. Renewable and Sustainable Energy Reviews, 11, 1300-1311.
http://dx.doi.org/10.1016/j.rser.2005.08.006 - 74. Monyem, A. and Van Gerpen, J.H. (2001) The Effect of Biodiesel Oxidation on Regime Permance and Emissions. Biomass and Bioenergy, 20, 317-325.
http://dx.doi.org/10.1016/S0961-9534(00)00095-7 - 75. Boehman, A.L. (2005) Biodiesel Production and Processing. Fuel Processing Technology, 86, 1057-1058.
http://dx.doi.org/10.1016/j.fuproc.2004.11.001 - 76. Khalil, C.N. (2006) As Tecnologias de Produção de Biodiesel. Coletanea de artigos—O futuro da indústria: Biodiesel. Ministério do Desenvolvimento, Indústria e Comércio Exterior, Brazil.
- 77. Miao, X. and Wu, Q. (2006) Biodiesel Production from Heterotrophic Microalgal Oil. Bioresource Technology, 97, 841-846.
http://dx.doi.org/10.1016/j.biortech.2005.04.008 - 78. Griffiths, M.J., van Hille, R.P. and Harrison, S.T.L. (2010) Selection of Direct Tansesterification as the Preferred Method for Assay of Fatty Acid Content of Microalgae. Lipids, 45, 1053-1060.
http://dx.doi.org/10.1007/s11745-010-3468-2 - 79. Liu, B. and Zhao, Z.B. (2007) Biodiesel Production by Direct Methanolysis of Oleaginous Microbial Biomass. Journal of Chemical Technology and Biotechnology, 82, 775-780.
- 80. Suarez, P.A.Z., Santos, A.L.F., Rodrigues, J.P. and Alves, M.B. (2009) Biocombustíveis a partir de óleos e gorduras: desafios tecnológicos para viabilizá-los. Quimica Nova, 32, 768-775.
http://dx.doi.org/10.1590/S0100-40422009000300020 - 81. Nagle, N. and Lemke, P. (1990) Production of Methyl Ester from Microalgae. Biotechnology and Applied Biochemistry, 25, 355-361.
http://dx.doi.org/10.1007/BF02920259 - 82. Shuit, S., Lee, K., Kamaruddin, A. and Yusup, S. (2010) Reactive Extraction and in Situ Esterification of Jatropha curcas Seeds for the Production of Biodiesel. Fuel, 89, 527-530.
http://dx.doi.org/10.1016/j.fuel.2009.07.011 - 83. Almarales, A., Chenard, G., Abdala, R., Gomes, D.A., Reyes, Y. and Tapanes, N.O. (2012) Hydroesterification of Nannochloropsis oculata Microalga’s Biomass to Biodiesel on Al2O3 Supported Nb2O5 Catalyst. Natural Science, 4, 204-210.
http://dx.doi.org/10.4236/ns.2012.44031 - 84. Reyes, Y., Chenard, G., Aranda, D.G., Mesquita, C., Fortes, M., João, R. and Bacellar, L. (2012) Biodiesel Production by Hydroesterification of Microalgal Biomass Using Heterogeneous Catalyst. Natural Science, 4, 778-783.
http://dx.doi.org/10.4236/ns.2012.410102 - 85. Lardon, L., Hélias, A., Sialve, B., Steyer, J.P. and Bernard, O. (2009) Life-Cycle Assessment of Biodiesel Production Froma Microalgae. Environmental Science & Technology, 43, 6475-6481.
- 86. Carioca, J.O.B., Hiluy Filho, J.J., Leal, M.R.L.V. and Macambira, F.S. (2009) The Hard Choice for Alternative Biofuels to Diesel in Brazil. Biotechnology Advances, 27, 1043-1060.
http://dx.doi.org/10.1016/j.biotechadv.2009.05.012 - 87. Stuart, A.S., Atthew, P.D., John, S.D., Irmtraud, H., Christopher, J.H., David, J.L.S. and Alison, G.S. (2010) Biodiesel from Microalgae: Challenges and Prospects. Current Opinion in Biotechnology, 21, 277-286.
http://dx.doi.org/10.1016/j.copbio.2010.03.005 - 88. Sydney, E.B., da Silva, T.E., Tokasrki, A., Novak, A.C., de Carvalho, J.C., Woiciecohwski, A.L., Larroche, C. and Soccol, C.R. (2011) Screening of Microalgae with Potential for Biodiesel Production and Nutrient Removal from Trated Domestic Sewage. Applied Energy, 88, 3291-3294.
http://dx.doi.org/10.1016/j.apenergy.2010.11.024 - 89. Franco, A.L.C., Lôbo, I.P. and Cruz, R.S. (2013) Biodiesel de microalgas: Avanços e desafios. Quimica Nova, 36, 437-448.
http://dx.doi.org/10.1590/S0100-40422013000300015


