American Journal of Plant Sciences
Vol.3 No.10(2012), Article ID:24118,4 pages DOI:10.4236/ajps.2012.310170
Antinutritive Compounds in Twelve Camelina sativa Genotypes
![]()
Istituto di Biologia e Biotecnologia Agraria, CNR, Milano, Italy.
Email: reggiani@ibba.cnr.it
Received May 4th, 2012; revised June 15th, 2012; accepted July 18th, 2012
Keywords: Camelina sativa; Glucosinolate; Sinapine; Phytic Acid; Condensed Tannins
ABSTRACT
Camelina sativa is an oilseed crop becoming important in North America and Europe for biodiesel production. The use of Camelina flours in animal diet may be limited by antinutritive compounds. The content of glucosinolates, phytic acid, sinapine and condensed tannins was evaluated in twelve accessions of Camelina sativa. All compounds showed significant differences among genotypes. Only the concentration of glucosinolates in the flour deserves attention, while the content of phytic acid, sinapine and condensed tannins are to acceptable levels. Camelina showed the presence of three different glucosinolates (GSL1, GSL2 and GSL3) in the flour, with differences among genotypes regarding the relative abundance of each glucosinolate. The content of glucosinolates is inversely correlated with that of sinapine. The glucosinolate content in Camelina flour has to be reduced to increase the use of this flour in animal diet, but avoiding altering the sinapine content.
1. Introduction
Camelina sativa (L) Crantz. (CS) offers a solution to reach biodiesel production goals by providing a sustainable low-input biofuel feedstock option that does not interfere with food production. CS can be harvested and crushed for oil and the remaining parts used to produce high quality omega 3 rich animal feed, flour, fiberboard and glycerin [1]. An economical exploitation of CS oil requires usage of flours or cakes as well. The use of press cake as protein rich ingredient in fodder for animals and in human nutrition was legalized by Commission Directive 2008/76/EC [2]. The American Food and Drug Administration has recently raised the inclusion level of CS meal in feedlot beef cattle rations to 10% [3]. CS meal was evaluated on its acceptability and potential to replace soybean meal in finishing beef cattle [4]. In chickens, there were not toxic effects on animal at low concentration (5%) but it would optimize the fatty acid content in eggs and meat [5]. Series of experiments were carried out to assess the exploitation of CS-cake in human nutrition as ingredient in bread. The final products with a proportion of CS flour of 5% - 10% were characterized by a distinct smell and taste [6]. For the use of this by-product is important the knowledge about nature and quantity of the antinutritive compounds present in CS seed. Especially plant secondary metabolites such as glucosinolates (GSLs), sinapine, phytic acid and condensed tannins belong to widespread antinutritive compounds generally present in oilseed like CS. GSLs and sinapine usually have been associated with members of the Brassiacaceae whereas phytic acid and condensed tannins are more generally distributed in flora.
GSLs are β-thioglucoside N-hydroxysulfates with a side chain and sulfur linked β-D-glucopyranose. Their breakdown products isothiocyanates, thiocyanates, nitriles and epithionitriles are responsible for toxicity of GSLs like impairment of thyroid, growth, fertility and reproduction, irritation of gastro-intestinal mucosa followed by local necroses [7].
Sinapine is a choline ester of sinapic acid. Sinapine has a several undesirable properties as a constituent in animal feeds. It is a bitter tasting compound, making it less palatable to animals, while its presence in the diet of certain brown egg-laying hens at levels exceeding 1 g·kg–1 leads to a fishy odour or taste in the eggs [8].
Phytic acid (inositol exaphosphate) is the main organic form of phosphorus present in plant seed. Recently, nutritional observations have renewed interest in phytic acid. In fact, it complexes with many mineral elements (Ca, Mg, Zn, Fe), in some case rendering them insoluble and biologically unavailable [9].
Condensed tannins (flavan-3-ol based biopolymers) act as an antinutrient compounds of plant origin because they precipitate proteins, inhibit digestive enzyme and decrease the utilization of vitamins and minerals. These compounds tend to complexing proteins and enzymes, thus rendering the food protein indigestible directly linking them or indirectly interfering with the action of digestive enzymes such as trypsin and chymotrypsin. Tannins can create complex with vitamin B12, thus leading to a decrease of its absorption. Yet, they have also been considered a health-promoting component in plant derived foods and beverage. For example, tannins have been shown to have anticarcinogenic and antimutagenic potential and antimicrobial properties [10].
In the present study, the content of antinutritive compounds was evaluated in twelve CS accessions in order to better understand the limit of application of CS flour as feed ingredient.
2. Materials and Methods
2.1. Reagents and Plant Material
DEAE-Sephadex A-25, sinigrin and sulfatase Type H1 were purchased from Sigma-Aldrich (Milano, Italy). All organic solvents were analytical grade.
CS seeds were sown in spring in fields near Firenze, Italy. Twelve CS genotypes different by origin were used: Calena (Germany), Ligena (Germany), Cam120 (Poland), Cam180 (Germany), D11851 (Italy), FF006 (Austria), Ames28372 (USA), Cam31 (Poland), Cam37 (Russia), Cam76 (Russia), Cam173 (Russia), D9952 (Unknown).
2.2. Extraction and Separation of GSLs
GSLs were extracted and assayed according to Russo and Reggiani [11]. CS seeds were defatted with hexane and GSLs were extracted with 70% hot ethanol for 3 h. The sample were centrifuged (15 min, 15,000 rpm). Five hundred µL of ethanol extract were adsorbed onto a small DEAE-Sephadex A-25 column in formate form (100 mg). The column was then washed twice with 1 mL of sodium acetate buffer (20 mM, pH 4.0). Desulfation of GSLs was obtained by 50 µL of sulfatase (500 U) at 37˚C overnight. Desulfo-GSLs were eluted from the column with 1.5 mL of water and dried at 65˚C. The samples were resuspended in ethanol before GSL analysis. Alcoholic samples were loaded with a Linomat IV (Camag, Switzerland) on a HPTLC RP18W plate (Merck, Germany). The plate run into an HPTLC Developmental Chamber (Camag, Switzerland) using acetonitrile/water (4:6, v/v) as solvent (15 mL per plate). After the run, the absorbance (229 nm) of each lane was read into a densitometer TLC Scanner II (Camag, Switzerland) using desulfosinigrin as a standard. The data of GSLs were quantitated by a D-2000 integrator (Hitachi-Merck, Germany).
2.3. Extraction and Separation of Sinapine
The extraction of sinapine on defatted flours was achieved with 70% methanol as described by Cai and Arntfield [12]. The HPLC analysis for sinapine was performed according to Clausen et al. [13].
2.4. Extraction and Separation of Phytic Acid
Phytic acid was isolated from defatted flour using a modified acid extraction-iron precipitation method by deBoland et al. [14]. The phosphorus content of the precipitate was determined, after acid digestion with sulfuric acid, colorimetrically according to Chen et al. [15]. Phytic acid was calculated by multiplying phytic acid phosphorus values by 3.55 [16].
2.5. Extraction and Separation of Condensed Tannins
The determination of condensed tannins was carried out as described by Butler et al. [8]. Tannins were extracted from defatted flour with 70% acetone, the samples evaporated to dryness and then resuspended in methanol. Condensed tannins were determined by the vanillin method using catechin as a standard [17].
2.6. Statistical Analysis
All statistical analysis were performed by SPSS version 11.5 software. Analysis of Variance (ANOVA) was applied to establish significant differences (P < 0.01) between Camelina genotypes in the levels of antinutritive compounds. Mean separation was performed using Duncan’s test and referring to P ≤ 0.05 probability level. Pearson’s correlations between antinutritive compounds were also calculated.
3. Results and Discussion
In Table 1 is shown ANOVA for GSLs, phytic acid, sinapine and condensed tannins for twelve genotypes of CS. ANOVA showed highly significant genotypic variation (P < 0.01) for all the antinutritive compounds considered.
In Table 2 are shown the levels of antinutritive compounds for the twelve CS accessions. As can be seen, total GSL content ranged from 15.2 to 24.6 mmol·Kg–1 dry matter (DM), with most genotypes with less than 20

Table 1. Mean square and F value from Analysis of Variance (ANOVA) for antinutritive compound contents in flours of twelve genotypes of Camelina sativa.
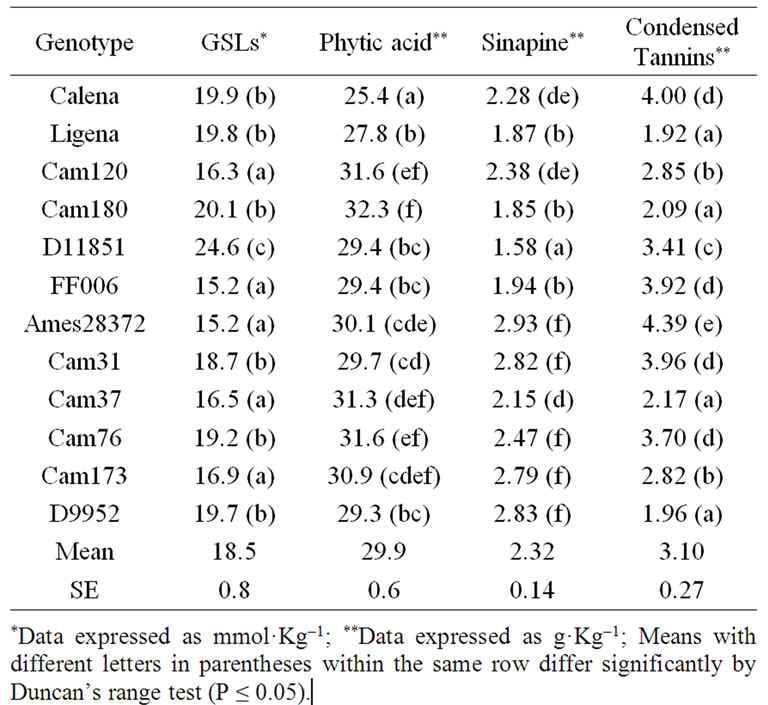
Table 2. Antinutritive compounds in different genotypes of Camelina sativa.
mmol·Kg–1 DM. Significant differences between genotypes were put in evidence by the Duncan’s range test, with the group marked with the letter (a) exhibiting GSL contents below the general mean. Differences among genotypes were also observed on the GSL pattern (Figure 1). In CS, three main GSLs were identified named GSL1 (9-methyl-sulfinyl-nonyl-GSL), GSL2 (glucocamelinin, 10-methyl-sulfinyl-decyl-GSL) and GSL3 (11-methylsulfinyl-undecyl-GSL) (Russo and Reggiani, 2012). In all genotypes, GSL2 represented the most abundant GSL being between 50% - 60% (Figure 1). The content of the other 2 species, GSL1 and GSL3, exhibited differences among genotypes. Six genotypes showed levels of GSL1 higher than that of GSL3 (Calena, Ligena, Cam120, Cam180, D11851 and FF006). In 5 genotypes, GSL1 and GSL3 levels were similar (Cam31, Cam37, Cam76, Cam- 173 and D9952). This different GSL pattern was suggested to be associated to winter genotypes or wild Camelina like C. microcarpa or C. pilosa [18]. In AMES- 28372, GSL3 was higher than GSL1. The climatic and soil conditions affects the content of GSLs [17,18]. Expecially sulfur in soil is a determinant of the concentration of total GSLs in plant organs [19]. In this work, 12 genotypes of different origin were grown in the same location in order to avoid variations due to the environment or soil. In these conditions, 15.2 mmol·Kg–1 DM was the lowest concentration of GSLs observed among genotypes (Table 2).
Although CS has a relatively low concentration of GSLs in flour, this is higher than that required by EFSA [2]. Breeding in rapeseed reduced GSL content in meal from 50 - 150 mmol·Kg–1 DM to less than 2 mmol·Kg–1 DM [20,21]. In CS, the objective of low GSLs would be

Figure 1. Distribution (%) of GSL1 (black bar), GSL2 (light gray bar) and GSL3 (dark gray bar) on total GSL content in twelve Camelina sativa genotypes.
closer to reach and the different GSL pattern among genotypes might help (Figure 1). Nevertheless, some studies showed the use of CS meal in animal diet as it is [22- 24].
The content of phytic acid ranged from 25.4 to 32.3 mg·Kg–1 DM (Table 2). Significant differences between genotypes were observed and, in particular, Calena genotype resulted different and lower in phytic acid than the other genotypes. The content in this genotype is just higher than the content of phytic acid in soybeans [25], which flour is the main ingredient used in animal diet.
The sinapine content ranged from 1.58 g·Kg–1 (D11851) to 2.93 g·Kg–1 (Ames28372), with a general mean of 2.32 ± 0.14 g·Kg–1 DM (Table 2). This sinapine content is similar to that reported for CS by Matthäus [26] and can be considered low in comparison with other members of Brassicaceae (rapeseed, mustard).
The variation of the condensed tannin content in different CS genotypes was quite high. In Table 2, the contents varied from 1.92 g·Kg–1 to 4.39 g·Kg–1 of defatted flour. Matthäus [26] found that CS genotypes contained from 1.5 to 3 g condensed tannins per·Kg–1 of seed. Considering that defatting seeds results in a concentration of condensed tannins, our data can be considered similar to that previously reported. These levels are no toxic in animal diets since they are below 1% DM [27].
In Table 3 is shown the correlation matrix calculated for the different antinutritional compounds. In this table, Pearson correlation coefficients are given as a measure of linearity between two class of compounds. From Table 3, it becomes clear that there is a significant inverse correlation (P < 0.0.1) between GSLs and sinapine.
For its bitter taste, the role of sinapine in plants is probably to make them less palatable to animals. GSL degradation by animal metabolism instead affects animal growth, reproductive performance as well as intake and
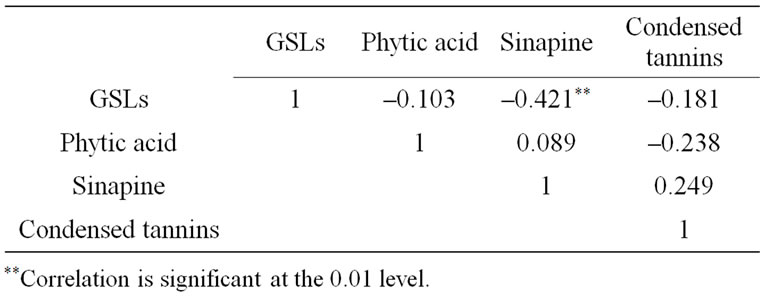
Table 3. Pearson correlation coefficients (r) between the different antinutritive compounds in twelve genotypes of Camelina sativa.
palatability of fodder [28]. It might be that sinapine and GSLs are part of the same defense mechanism against herbivores. This would explain the inverse correlation here observed between these compounds so that the decrease of one is compensated by the increase of the other.
4. Conclusion
The exploitation of CS oil for biofuels will lead to availability of CS flour to use in animal feed. This flour cannot be considered complete food because some lipophilic vitamins are lost during the chemical extraction of oil. CS flour must therefore be administered in combination with other ingredients. The mixing process has the advantage of reducing the content of antinutritionals. The use of CS flour is currently limited both in Europe and USA by the content of GSLs. So it is desirable that genotypes of CS low in GSLs are selected as was the case in the 1970s-1980s for rapeseed. In this specie, GSLs were reduced to less than 2 mmol·Kg–1 by traditional breeding [21]. From CS genotypes low in GSLs (Table 2, letter a) which contain just 15 - 16 mmol·Kg–1 and exploiting the different patterns of GSLs (Figure 1), the selection of genotypes low in GSLs by traditional breeding seems a result close to reach. However, it is to be determined whether a decrease in the content of GSLs would lead to an increase in the content of sinapine.
5. Acknowledgements
This work was supported by Regione Lombardia, agreement Regione/CNR, project 2 “Risorse biologiche e tecnologie innovative per lo sviluppo sostenibile del sistema agro-alimentare”.
REFERENCES
- R. Dulmaine, “Camelina: A Better Source of Biofuel?” 2008. http://www.current.com/1nlbu4c
- European Commission, “Directive 2008/76/CE”, 2008. http://www.eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2008:198:0037:01:IT:HTML
- S. R. Schill, “Camelina Meal Approved for Feedlot Cattle,” Biodiesel Magazine, 2010. http://www.biodieselmagazine.com/articles/3837/camelina-meal-approved-for-feedlot-cattle
- J. Zubr, “Qualitative Variation of Camelina sativa Seed from Different Locations,” Industrial Crops and Products, Vol. 17, No. 3, 2003, pp. 161-169. doi:10.1016/S0926-6690(02)00091-2
- E. L. Ryhänen, S. Perttilä, T. Tupasela, J. Valaja, C. Eriksson and K. Larkka, “Effect of Camelina sativa Expeller Cake on Performance and Meat Quality Broilers,” Journal of the Science of Food and Agriculture, Vol. 87, No. 8, 2007, pp. 1489-1494. doi:10.1002/jsfa.2864
- J. Zubr, “Carbohydrates, Vitamins and Minerals of Camelina sativa Seed,” Nutrition & Food Science, Vol. 40, No. 5, 2010, pp. 523-531. doi:10.1108/00346651011077036
- European Food Safety Authority, “Glucosinolates as Undesirable Substances in Animal Feed,” EFSA Journal, Vol. 590, 2008, pp. 1-76.
- E. J. Butler, A. W. Pearson and G. R. Fenwick, “Problems Which Limit the Use of Rapeseed Meal as a Protein Source in Poultry Diets,” Journal of the Science of Food and Agriculture, Vol. 33, No. 9, 1982, pp. 866-875. doi:10.1002/jsfa.2740330909
- U. Schlemmer, W. Frǿlich, R. M. Prieto and F. Grases, “Phytate in Foods and Significance for Humans: Food Sources, Intake, Processing, Bioavailability, Protective Role and Analysis,” Molecular Nutrition & Food Research, Vol. 53, No. 2, 2009, pp. 330-375. doi:10.1002/mnfr.200900099
- R. Amarowicz, I. Estrella, T. Hernández, S. Robredo, A. Troszynska, A. Kosinska and R. B. Pegg, “Free Radical-Scavenging Capacity, Antioxidant Activity, and Phenolic Composition of Green Lentil (Lens culinaris)”, Food Chemistry, Vol. 121, No. 3, 2010, pp. 705-711. doi:10.1016/j.foodchem.2010.01.009
- R. Russo and R. Reggiani, “Rapid Separation of Seed Glucosinolates from Camelina sativa by Thin Layer Chromatography,” International Journal of Plant Biology, 2012, in press.
- R. Cai and S. D. Arntfield, “A Rapid High-Performance Liquid Chromatographic Method for the Determination of Sinapine and Sinapic Acid in Canola Seed and Meal,” Journal of the American Oil Chemists Society, Vol. 78, No. 9, 2001, pp. 902-910. doi:10.1007/s11746-001-0362-4
- S. Clausen, L. M. Larsen and A. Plöger, “Aromatic Choline Esters in Rapeseed,” In: H. Sorensen, Ed., Martinus Nijhoff, Martinus Nijhoff/Dr. W. Junk Publishers, Dordrecht, 1985, pp. 61-71.
- A .R. deBoland, G. B. Garner and B. L. O’Dell, “Identification and Properties of Phytate in Cereal Grains and Oilseed Products,” Journal of Agricultural and Food Chemistry, Vol. 23, No. 6, 1975, pp. 1186-1189. doi:10.1021/jf60202a038
- P. S. Chen, T. Y. Toribara and H. Warner, “Microdetermination of Phosphorus,” Analytical Chemistry, Vol. 28, No. 11, 1956, pp. 1756-1758. doi:10.1021/ac60119a033
- V. Raboy and D. B. Dickinson, “Effect of Phosphorus and Zinc Nutrition on Soybean Seed Phytic Acid and Zinc,” Plant Physiology, Vol. 75, No. 4, 1984, pp. 1094- 1098. doi:10.1104/pp.75.4.1094
- B. Matthäus and J. Zubr, “Variability of Specific Components in Camelina sativa Oilseed Cakes,” Industrial Crops and Products, Vol. 12, No. 1, 2000, pp. 9-18. doi:10.1016/S0926-6690(99)00040-0
- A. Schuster and W. Friedt, “Glucosinolate Content and Composition as Parameters of Quality of Camelina Seed,” Industrial Crops and Products, Vol. 7, No. 2-3, 1998, pp. 297-302. doi:10.1016/S0926-6690(97)00061-7
- M. D. Omirou, K. K. Papadopoulou, I. Papastylianou, M. Constantinou, D. G. Karpouzas, I. Asimakopoulos and C. Ehaliotis, “Inpact of Nitrogen and Sulfur Fertilization on the Composition of Glucosinolates in Relation to Sulfur Assimilation in Different Plant Organs of Broccoli,” Journal of Agricultural and Food Chemistry, Vol. 57, No 20, 2009, pp. 9408-9417. doi:10.1021/jf901440n
- J. P. Sang and P. A. Salisbury, “Glucosinolate Profiles of International Rapeseed Lines (Brassica napus and Brassica campestris),” Journal of the Science of Food and Agriculture, Vol. 45, 1988, pp. 255-261. doi:10.1002/jsfa.2740450308
- J. M. Bell, M. O. Keith and D. S. Hutcheson, “Nutritional Evaluation of Very Low Glucosinolate Canola Meal,” Canadian Journal of Animal Science, Vol. 71, No. 2, 1991, pp. 497-506. doi:10.4141/cjas91-059
- C. Hurthaud and J. L. Peyraud, “Effects of Feeding Camelina (Seeds or Meal) on Milk Fatty Acid Composition and Butter Spreadability,” Journal of Dairy Science, Vol. 90, No. 11, 2007, pp. 5134-5145. doi:10.3168/jds.2007-0031
- A. Y. Pekel, P. H. Patterson, R. M. Hulet, N. Acar, T. L. Cravener, D. B. Dowler and J. M. Hunter, “Dietary Camelina Meal versus Flaxseed with and without Supplemental Copper,” Poultry Science, Vol. 88, No. 11, 2009, pp. 2392-2398. doi:10.3382/ps.2009-00051
- A. E. Aziza, N. Quezada and G. Cherian, “Feeding Camelina sativa Meal to Meat-Type Chickens: Effect on Production Performance and Tissue Fatty Acid Composition,” The Journal of Applied Poultry Research, Vol. 19, No. 2, 2010, pp. 157-168. doi:10.3382/japr.2009-00100
- P. Kwanyuen and J. W. Burton, “A Simple and Rapid Procedure for Phytate Determination in Soybeans and Soy Products,” Journal of the American Oil Chemists Society, Vol. 82, No. 2, 2005, pp. 81-85. doi:10.1007/s11746-005-1046-9
- B. Matthäus, “Antinutritive Compounds in Different Oilseeds,” Fett/Lipid, Vol. 99, No. 5, 1997, pp. 170-174.
- V. L. Singleton, “Naturally Occurring Food Toxicants: Phenolic Substances of Plant Origin Common in Foods,” Advances in Food Research, Vol. 27, 1981, pp. 149-242. doi:10.1016/S0065-2628(08)60299-2
- B. Matthäus and L. G. Angelini, “Anti-Nutritive Constituents in Oilseed Crops from Italy,” Industrial Crops and Products, Vol. 21, No. 1, 2005, pp. 89-99. doi:10.1016/j.indcrop.2003.12.021

