Surgical Science
Vol. 3 No. 6 (2012) , Article ID: 19988 , 7 pages DOI:10.4236/ss.2012.36065
Post Cholecystectomy Diarrhoea—A Systematic Review
1Department of Surgery, Barts and the London School of Medicine and Dentistry, Queen Mary University of London, Barts and Royal London Hospitals, London, UK
2Imperial College London, Hammersmith Hospital, London, UK
Email: mr_farahmand80@yahoo.co.uk
Received March 15, 2012; revised April 22, 2012; accepted May 16, 2012
Keywords: Diarrh*; Cholecystectomy; Bile Acid Malabsorption
ABSTRACT
Introduction: Post-cholecystectomy diarrhoea (PCD) is probably the most distressing postoperative non-pain symptom. Its nature is not fully understood. The aim of this systematic review was to assess the prevalence, aetiology, predisposing factors and management of PCD from the past study reports. Methods: We conducted a wide ranged review of published literature on PubMed, Web of Knowledge, and the Cochrane library without any time limitation. Results: Twenty five studies were included. The prevalence of PCD was 9.1% (302/3306) with no significant difference between genders. The prevalence of bile acid malabsorption (BAM), the most important suggested aetiological factor, was seen in 65.5% (36/55) patients with PCD. There were no obvious predisposing factors. We found 92% (23/25) of patients with PCD responded to cholestyramine therapy. The cure rate for cholecystectomy on preoperative cholegenic diarrhoea was 54.5% (121/222). The prevalence of post-cholecystectomy new onset constipation was 7.9% (78/987). Conclusion: The aetiology of PCD is unknown and appears to be multifactorial. The BAM prevalence was only 65.5%, however 92% of PCD patients responded to cholestyramine therapy suggesting that cholestyramine therapy could also have curative effect on other unknown aetiological factors related to bile metabolism. The complexity of the aetiology of PCD is more projected by considering the curative effect of cholecystectomy on preoperative cholegenic diarrhoea, and the occurrence of post-cholecystectomy new onset constipation. However, the relationship between cholecystectomy and PCD is an undeniable fact.
1. Introduction
Cholecystectomy is one of the most common operations in the world. It is appropriate to assess its outcomes from the patient’s perspective, taking into account the symptomspecific endpoints. Although the majority of patients after a cholecystectomy are satisfied with their outcomes, there are nonetheless a significant number of patients with bothersome postoperative non-pain symptoms [1], among which diarrhoea is probably the most distressing [2].
Even though most patients adapt to the change in the enterohepatic circulation after cholecystectomy and do not develop diarrhoea [3], diarrhoea evolving after cholecystectomy is a well-known clinical problem. The frequency of post-cholecystectomy diarrhoea (PCD) however is a matter of debate [4,5] and its nature, aetiology, and predisposing factors are not fully understood. Thus objective information regarding prevalence, mechanisms and predisposing factors for PCD is important for clarification of patient expectations, informed consent and postoperative management of these patients.
Underlining the importance of post-cholecystectomy diarrhoea Fort et al. [6] reported that patients with postcholecystectomy diarrhoea syndrome had markedly shortened total colonic transit times, which were remarkably in the range of transit times measured in patients with bouts of acute infectious diarrhoea. However, after removal of their gallbladder, some patients experienced a diarrheogenic trend strong enough to set them aside from the norm and make them regard themselves as anywhere from annoyed to socially disabled [6].
This systematic review of observational studies, detailing complications related to changes of bowel habit after cholecystectomy, would help surgeons modulate postoperative expectations about diarrhoea as a complication of cholecystectomy and advice on its possible persistence.
The aim of this study was to assess the prevalence, aetiology, predisposing factors and management of postcholecystectomy diarrhoea from the past study reports.
2. Methods and Materials
We conducted a systematic review of published literature using standard electronic databases, PubMed (MEDLINE), ISI Web of Knowledge, and Cochrane library. In order to identify the maximum number of articles related to our subject our search strategy had no time limitation. Key words, such as, “diarrh*”, “cholecystectomy”, “bile acid malabsorption” were used in the following combinations; [Cholecystectomy AND diarrh*] or [cholecystectomy AND bile acid malabsorption]. Reference lists of included articles were further analysed for additional studies.
2.1. Inclusion Criteria
Following the initial search, identified articles were screened using predetermined inclusion criteria. All included studies met our outlined parameters; articles about post-cholecystectomy diarrhoea, articles dedicated to post-cholecystectomy complications which included diarrhoea, studies describing both laparoscopic and open approaches, non-English articles which had an English language abstract with enough information, abstract only published papers containing relevant and usable information.
2.2. Exclusion Criteria
Rejected studies were; letters and comments or notes, case reports, articles which evaluated diarrhoea as an outcome of cholecystectomy in tandem with another procedure or included factors other than post-cholecystectomy which could play a role in the pathogenesis of the diarrhoea, thereby lacking a clear link between the outcomes and cholecystectomy, foreign language articles without English abstracts, English abstracts of foreign articles without enough information.
2.3. Data Extraction
Extracted data included; Trial design, number of included patients, demographics (age, gender), duration of follow up after operation, method of follow up, number of patients with PCD, the criterion for diagnosing diarrhoea, gastrointestinal transit time before and after surgery, faecal consistency assessment, patients with bile acid malabsorption, treatment for PCD, other abdominal nonpain symptoms pre and post cholecystectomy.
2.4. Statistics
Benefit ratios were calculated using the following formula: the number of patients with a particular symptom before operation minus those who developed it post operatively (including persistent and de novo), divided by the number of patients exhibiting the symptom preoperatively. Cure rate for diarrhoea was calculated fitting the extracted data into the following formula: patients with preoperative cholegenic diarrhoea minus the patients that still had diarrhoea after cholecystectomy divided by the patients with preoperative diarrhoea. Chi-squared test was used to calculate the P-value where applicable.
3. Results
The search of literature identified 64 articles, 39 of which including; non-related studies (n = 19), foreign language articles (n = 7) studies with cholecystectomy performed with another procedure or on patients with factors that could play a role in PCD (n = 6), Case reports (n = 3), and letter to the editor or commentaries (n = 4), were excluded. The 25 publications that met the inclusion criteria included 3388 cholecystectomised patients with a mean age of 53.40 years (12-88) and mean follow up of 32.4 months.
There were a total of 480 patients with diarrhoea after cholecystectomy in the included studies. However, 103 of these patients had diarrhoea prior to their cholecystectomy and were excluded for calculating the incidence of PCD as any information concerning progression of their symptoms was lacking. Finally, there were a total of 377 patients with PCD.
3.1. Incidence of Post Cholecystectomy Diarrhoea
Data on patients who developed diarrhoea following cholecystectomy was available in 21 studies [1,2,4,6-23]. Out of 3306 included patients, 302 developed PCD with a prevalence rate of 9.1%. The prevalence of PCD in male and female patients available in two studies [2,11] is 11.8% and 8.8% respectively (P-value = 0.86).
3.2. Prevalence of Bile Acid Malabsorption in Patients with Post Cholecystectomy Diarrhoea
Data regarding bile acid malabsorption (BAM) in patients with PCD was reported for 55/377 of patients [3, 5,24]. The prevalence of bile acid malabsorption among these patients was 65.5% (36/55).
3.3. Changes in the Number of Patients Who Reported More Than One Bowel Movement per Day after Cholecystectomy
Information regarding the number of patients who reported more than one bowel movement per day after cholecystectomy was available in three studies [4,7,8] with a total of 124/3388 cholecystectomised patients.
The number of patients with more than one bowel movement per day increased by 21.7% from 30/124 to 57/124 before and after cholecystectomy respectively. (P-value = 0.0005).
3.4. Intestinal Transit Time before and after Cholecystectomy
There was just one study that measured colonic transit time [6]. This trial reported a significant acceleration in colonic transit time after cholecystectomy (38 ± 5 hours), compared with before surgery (51 ± 5 hours) [P-value < 0.05] in 24 patients. In their study Fort et al. showed that patients after cholecystectomy had markedly shortened colonic transit times (by 19 ± 3 hours). There was also a delay of orocaecal transit time from 80 ± 4 minutes before operation versus 103 ± 8 minutes after (Table 1).
3.5. Changes in the Number of Patients Who Reported Formed Stools after Cholecystectomy
Two articles included information regarding the exact number of patients with changes in faecal consistency after cholecystectomy.
Moussavian et al. [7] and Sauter et al. [8] mentioned that the number of patients who reported formed stools decreased by 29% from 71/73 before cholecystectomy to 50/73 after operation within 12 weeks of surgery (P-value = 0.0001).
3.6. Treatment of Post Cholecystectomy Diarrhoea
We found information on the number of patients with PCD who had responded to treatment with cholestyramine in one study by Sciarretta et al. [5]. They reported 23 cases of complete PCD resolution (92%) out of the 25 patients who were treated with cholestyramine.
3.7. Predisposing Factors
We did not find any proof of clear predisposing factors for the development of PCD in the studies. Only two studies contained the information, which could be considered relevant. In one study, which was done by Fisher et al. [2], patients with post-operative diarrhoea were males with high BMIs, and were significantly younger (by a mean of 11.7 years) compared to patients without PCD. The authors concluded that the coexistence of age < 50 years with a high BMI and male gender are predictive for PCD. Another study suggested an increased risk of PCD in younger age group [9].
3.8. Benefit Ratio of Diarrhoea in Comparison with Benefit Ratio of the Other Abdominal Complications in Cholecystectomy
To evaluate the benefit ratio of diarrhoea and other symptoms we included all patients that complained of a particular symptom pre and post-operatively, comprising all present persistent and de novo post-cholecystectomy symptoms.
There were six studies [1,10-14] with information regarding diarrhoea in patients who underwent cholecystectomy. In total, there were 222 patients with preoperative diarrhoea, and 237 patients from the same samples who had diarrhoea after cholecystectomy. The benefit ratio for PCD was –0.07.
Nine articles [1,6,9-14,22] with information of pre and post-operative abdominal complications were analysed. Not each of the selected articles included data on all the considered abdominal symptoms (Table 2). We compared the benefit ratio of diarrhoea after cholecystectomy

Table 1. Intestinal transit time before and after cholecystectomy.
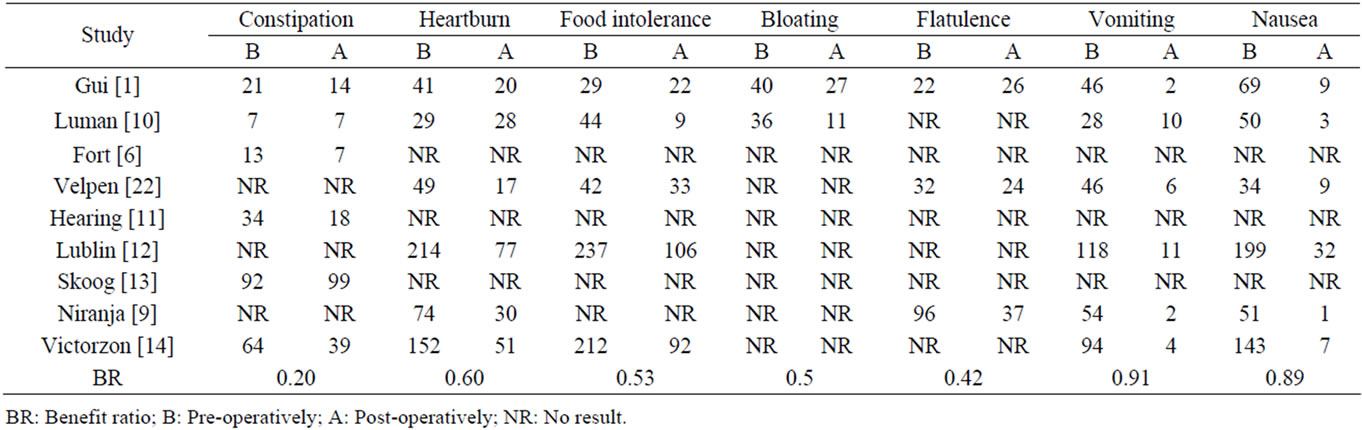
Table 2. Benefit ratio of the other abdominal complications in cholecystectomy.
with seven other abdominal symptoms (Figure 1).
3.9. Cure Rate of Cholegenic Diarrhoea
To assess the effect of cholecystectomy on those patients who had pre-operative diarrhoea we analysed six articles [1,10-14] containing data on patients with pre and postoperative diarrhoea.
The total number of patients with diarrhoea before surgery was 222, after deduction of patients who experienced persistent diarrhoea, there were 121/222 patients cured of this symptom after cholecystectomy, so the cure rate was 54.5% (Figure 2).
3.10. Prevalence of Post-Cholecystectomy New Onset Constipation
There were seven studies [1,6,10,11,13,16,21] with the data of new onset post-cholecystectomy constipation. 7.9% of patients (78/987) developed new onset constipation post-cholecystectomy.
3.11. Prevalence of Cholecystectomy in History of Patients with Chronic Diarrhoea
Through our electronic search, separately, we found four studies included patients with chronic diarrhoea [25-28]. Some of these patients had nothing notable in their history apart from having a cholecystectomy in the past. Out of the 280 patients with chronic diarrhoea, 48 just had cholecystectomy in their history without any other factors that could play a role in aetiology of diarrhoea (17.1%).
4. Discussion
Determining the incidence of post-cholecystectomy diarrhoea in cholecystectomised patients could play an important role in identifying whether cholecystectomy may be a cause for diarrhoea after surgery. Our systematic review showed that following cholecystectomy 9.1% of patients reported post-cholecystectomy diarrhoea as a troublesome problem. This figure is less than what has been reported in some studies, with figures ranging from 12% to 20% [2,6,9,13,16,20,22,29]. On the other hand several studies reported PCD prevalence to be between 2% - 8% [1,10,11,15,17-19,21,23] with two studies reporting rates as low as zero percent [7,8]. The differences in study groups, length of follow up, wording in assessment questionnaires, and definitions of diarrhoea could explain differences in the percentages of prevalence of post-cholecystectomy diarrhoea. In addition differences in views about diarrhoea of the people who were interviewed or participated in the studies, which reported bowel habits, are often not confirmed by objective records [18,30,31].
A larger overall sample size, included in our review, compared to individual studies does however lower the effect of the differences in each study giving a more accurate reflection of PCD’s true prevalence. The prevalence of post-cholecystectomy diarrhoea in males was 11.8% compared to 8.8% in females, however this difference was not statistically significant (P-value = 0.86).
The aetiology of PCD is not completely known but seems to be multifactorial. Cholecystectomy can alter the enterohepatic circulation in a number of ways. There is an increase in the number of enterohepatic cycles and also bile production after cholecystectomy [2-6,18], leading to higher bile acid concentration in the colon. In addition, lack of the reservoir of the gallbladder causes a continuous flux of bile after cholecystectomy [2,3,6,12]. Due to the individual limited bile reabsorption capacity of
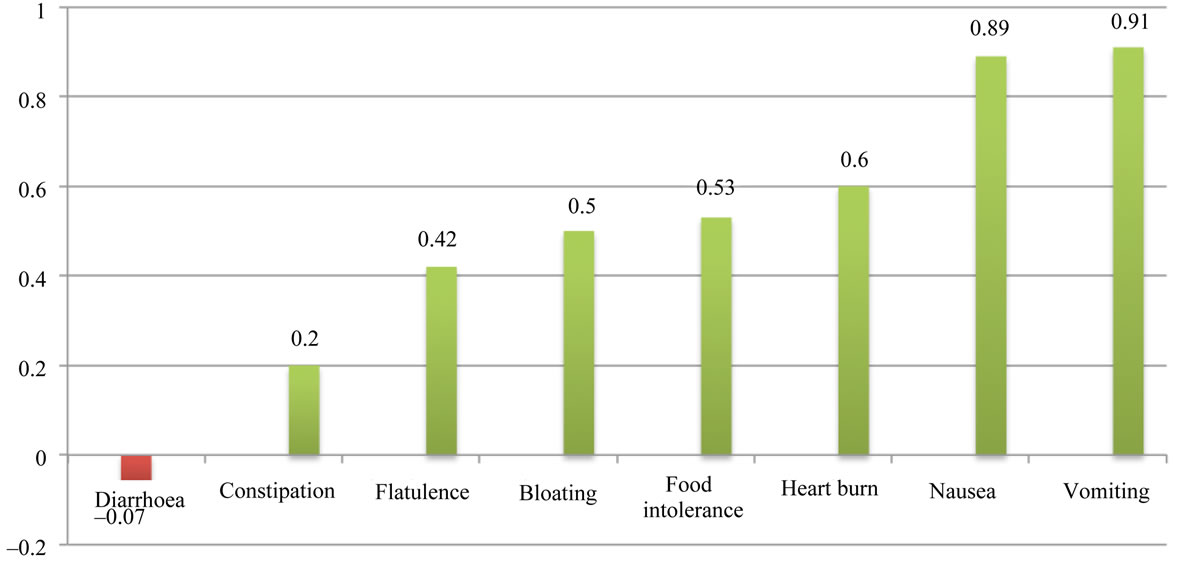
Figure 1. Benefit ratio of diarrhoea in comparison with benefit ratio of the other abdominal complications in cholecystectomy.
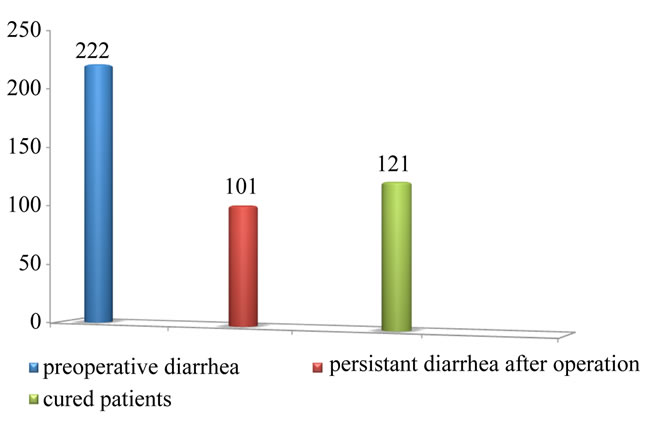
Figure 2. Cure rate of cholegenic diarrhoea after cholecystectomy.
terminal ileum some patients may develop diarrhoea. A shift in bile acid composition towards the diarrheogenic secondary bile acids [27], and even psychological and psychosomatic factors [2] are another suggested aetiologies.
Our results have shown that only 65.5% of patients with post-cholecystectomy diarrhoea have bile acid malabsorption, which suggest there are possibly additional so far unknown factors, which could play a role. An increase in orocaecal transit time and a significant acceleration of colonic transit time could be explained by bile acids slowing down small bowel transit, as a compensatory mechanism to facilitate additional bile acid reabsorption and to minimize spilling of bile acids into the colon. In a section of patients some amount of bile despite this adaptive capacity still spills into the colon, which could be responsible for colonic rate acceleration.
This is supported by our results, which indicated a statistically significant increase of 21.7% in the number of patients who reported more than one bowel movement per day after cholecystectomy.
92% of post-cholecystectomy diarrhoea patients clinically improved after being administered by cholestyramine while only 65.5% were diagnosed with bile acid malabsorption. The high response rate to the cholestyramine even in patients with diarrhoea and a normal 75- SeHCAT test value [5], suggests that cholestyramine may also have clinical effects through mechanisms other than pure bile acid binding. Although a good response to cholestyramine in patients with PCD could support the hypothesis that increased fecal bile acid may be the main reason for post-cholecystectomy diarrhoea.
Our review found no clear evidence supporting the existence of predisposing factors for PCD.
Cholecystectomy does not seem to have any positive effect on diarrhoea, with a benefit ratio of –0.07. However, we cannot ignore the role of cholecystectomy on improving the clinical picture of pre-operative diarrhoea in some patients. The cure rate of cholecystectomy on diarrhoea in our study was 54.5%. This indicates that while cholecystectomy cannot significantly improve preoperative diarrhoea, it may be the cause of new onset postoperative diarrhoea (post-cholecystectomy diarrhoea), which lends further support to the existence of postcholecystectomy diarrhoea as an entity.
The fact that 17.1% of patients with chronic diarrhoea presented only with a history of cholecystectomy could be considered as evidence in support of the relationship between diarrhoea and cholecystectomy even after many years of cholecystectomy.
5. Conclusion
The aetiology of PCD is unknown and appears to be multifactorial. There are some so far unknown factors, which can accelerate the colonic transit time significantly and lead to a significant decrease of faecal consistency after cholecystectomy. These changes in some cases are strong enough to develop PCD.
The good response up to 92% of patients to cholestyramine is in strong support that although an increase in the concentration of faecal bile acids is alleged to play an important role in aetiology of PCD, cholestyramine could also have a curative effect on the other unknown aetiologies.
The complexity of the aetiology of PCD is projected more when we consider the curative effect of cholecystectomy on pre-operative cholegenic diarrhoea, and the occurrence of post-cholecystectomy new onset constipation. However the relationship between cholecystectomy and chronic diarrhoea is an undeniable fact.
REFERENCES
- G. P. H. Gui, C. V. N. Cheruvu, N. West, K. Sivaniah and A. G. Fiennes, “Is Cholecystectomy Effective Treatment for symptomatic gallstones? Clinical outcome after Long- Term Follow-Up,” Annals of Royal of College of Surgeons of England, Vol. 80, No. 1, 1998, pp. 25-32.
- M. Fisher, D. C. Spilias and L. K. Tong, “Diarrhoea after Laparoscopic Cholecystectomy: Incidence and Main Determinants,” ANZ Journal of Surgery, Vol. 78, No. 6, 2008, pp. 482-486. doi:10.1111/j.1445-2197.2008.04539.x
- F. L. Arlow, A. A. Dekovich, R. J. Priest and W. T. Beher, “Bile Acid Mediated Postcholecystectomy Diarrhea,” Archives of Internal Medicine, Vol. 147, No. 7, 1987, pp. 1327-1329. doi:10.1001/archinte.1987.00370070139021
- G. H. Sauter, A. C. Moussavian, G. Meyer, H. O. Steitz, K. G. Parhofer and D. Jüngst. “Bowel Habits and Bile Acid Malabsorption in the Months after Cholecystectomy,” American Journal of Gastroenterology, Vol. 97, No. 7, 2002, pp. 1732-1735. doi:10.1111/j.1572-0241.2002.05779.x
- G. Sciarretta, A. Furno, M. Mazzoni and P. Malaguti, “Post Cholecystectomy Diarrhea: Evidence of Bile Acid Malabsorption Assessed by SeHCAT Test,” American Journal of Gastroenterology, Vol. 87, No. 12, 1992, pp. 1852-1854.
- J. M. Fort, F. Azpiroz, F. Casellas, J. Andreu and J. R. Malagelada, “Bowel Habit after Cholecystectomy: Physiological Changes and Clinical Implications,” Gastroenterology, Vol. 111, No. 3, 1996, pp. 617-622. doi:10.1053/gast.1996.v111.pm8780565
- C. A. Moussavian, K. G. Parhofer and D. Juengst, “Effect of Cholecystectomy on Bile Acid Metabolism and Serum Lipoprotein Profiles,” Gastroenterology, Vol. 118, No. 4, 2000, p. A884. doi:10.1016/S0016-5085(00)85675-9
- G. H. Sauter and C. A. Moussavian, “Occurrence of Bile Acid Malabsorption after Cholecystectomy,” Hepathology, Vol. 30, No. 4, 1999, p. 685A.
- B. Niranjan, S. Chumber and A. K. Kriplani, “Symptomatic Outcome after Laparoscopic Cholecystectomy,” Tropical gastroenterology, Vol. 21, No. 3, 2000, pp. 144-148.
- W. Luman, W. H. Adams, S. N. Nixon, I. M. Mcintyre, D. Hamer-Hodges, G. Wilson and K. R. Palmer, “Incidence of Persistent Symptoms after Laparoscopic Cholecystectomy: A Prospective Study,” Gut, Vol. 39, No. 6, 1996, pp. 863-866. doi:10.1136/gut.39.6.863
- S. D. Hearing, L. A. Thomas, K. W. Heaton and L. Hunt, “Effect of Cholecystectomy on Bowel Function: A Prospective, Controlled Study,” Gut, Vol. 45, No. 6, 1999, pp. 889-894. doi:10.1136/gut.45.6.889
- M. Lublin, D. L. Crawford, J. R. Hiatt and E. H. Phillips, “Symptoms before and after Laparoscopic Cholecystectomy for Gallstones,” American Surgeon, Vol. 70, No. 10, 2004, pp. 863-866.
- S. Skoog, J. Simonson, A. Arora, N. Diehl, W. S. Harmsen, G. R. Locke, G. Longstreth, Y. Romero, J. Thistle, A. Zinsmeister and A. E. Bharucha, “Post-Cholecystectomy Diarrhea: A Prospective Study of Incidence,” Gastroenterology, Vol. 126, No. 4, 2004, pp. A370.
- M. Victorzon, M. Lundin, C. Haglund, P. J. Roberts and I. Kellokumpu, “Short and Long Term Outcome after Laparoscopic Cholecystectomy,” Annales Chirurgiae et Gynaecologiae, Vol. 88, No. 4, 1999, pp. 259-263.
- E. Ros and D. Zambon, “Post Cholecystectomy Symptoms. A Prospective Study of Gall Stone Patients before and Two Years after Surgery,” Gut, Vol. 28, No. 11, 1987, pp. 1500-1504. doi:10.1136/gut.28.11.1500
- W. C. Watson and C. Marsh, “Post Cholecystectomy Bowel Function,” Gastroenterology, Vol. 96, No. 5, 1989, p. A538.
- K. W. Heaton, D. Parker and H. Cripps, “Bowel Function and Irritable Bowel Symptoms after Hysterectomy and Cholecystectomy a Population Based Study,” Gut, Vol. 34, No. 8, 1993, pp. 1108-1111. doi:10.1136/gut.34.8.1108
- R. G. Wilson and I. M. Macintyre, “Symptomatic Outcome after Laparoscopic Cholecystectomy,” British Journal of Surgery, Vol. 80, No. 4, 1993, pp. 439-441. doi:10.1002/bjs.1800800410
- C. S. Huang, F. C. Tai, M. Y. Shi, D. F. Chen and N. Y. Wang, “Complications of Laparoscopic Cholecystectomy: An Analysis of 200 Cases,” Journal of the Formosan Medical Association, Vol. 91, No. 8, 1992, pp. 785-792.
- D. Parker and K. W. Heaton, “Diarrhoea after Cholecystectomy: An Understimated Problem?” Gut, Vol. 32, No. 10, 1991, p. A1254.
- M. A. Mcnally, G. R. Locke, A. R. Zinsmeister, C. D. Schleck, J. Peterson and N. J. Talley, “Biliary Events and an Increased Risk of New Onset Irritable Bowel Syndrome: A Population-Based Cohort Study,” Alimentary Pharmacology and therapeutics, Vol. 28, No. 3, 2008, pp. 334-343. doi:10.1111/j.1365-2036.2008.03715.x
- G. C. Vander Velpen, S. M. Shimi and A. Cuschieri, “Outcome after Cholecystectomy for Symptomatic Gall Stone Disease and Effect of Surgical Access Laparoscopic v Open Approach,” Gut, Vol. 34, No. 10, 1993, pp. 1448-1451. doi:10.1136/gut.34.10.1448
- L. S. Schultz, M. Kamel, J. N. Graber and D. F. Hickok, “Four-Years Outcome Data for 400 Laparoscopic Cholecystectomy Patients: Recognition of Persistant Symptoms,” International Surgery, Vol. 79, No. 3, 1994, pp. 205-208.
- H. Fromm, C. A. Sherman, A. K. Tunuguntla and S. Ceryak, “Post Cholecystectomy (pcc) Diarrhea: Does Bile Acid Malabsorption Play a Role,” Gastroenterology, Vol. 86, No. 5, 1984, pp. 1082-1082.
- G. A. Ford, J. D. Preece, I. H. Davies and S. P. Wilkinson, “Use of the SeHCAT Test in the Investigation of Diarrhea,” Postgraduate Medical Journal, Vol. 68, No. 798, 1992, pp. 272-276. doi:10.1136/pgmj.68.798.272
- S. Eusufzai, M. Axelson, B. Angelin and K. Einarsson, “Serum 7 Alpha-hydroxy-4-cholesten-3-one Concentrations in the Evaluation of Bile Acid Malabsorption in Patients with Diarrhoea: Correlation to SeHCAT Test,” Gut, Vol. 34, No. 5, 1993, pp. 698-701. doi:10.1136/gut.34.5.698
- O. Suhr, A. Danielsson, H. Nyhlin and H. Truedsson, “Bile Acid Malabsorption Demonstrated by SeHCAT Test in Chronic Diarrhea, with Special Reference to the Impact of Cholecystectomy,” Scandinavian Journal of Gastroenterology, Vol. 23, No. 10, 1988, pp. 1187-1194. doi:10.3109/00365528809090189
- S. Eusufzai, “Bile Acid Malabsorption in Patients with Chronic Diarrhea,” Scandinavian Journal of Gastroenterology, Vol. 28, No. 10, 1993, pp. 865-868. doi:10.3109/00365529309103126
- J. Sand, S. Pakkala and I. Nordback. “Twenty to Thirty Year Follow-Up after Cholecystectomy,” Hepato-gastroenterology, Vol. 43, No. 9, 1996, pp. 534-537.
- A. P. Manning, J. B. Wyman and K. W. Heaton, “How Trustworthy Are Bowel Histories? Comparison of Recalled and Recorded Information,” British Medical Journal, Vol. 2, No. 6029, 1976, pp. 213-214. doi:10.1136/bmj.2.6029.213
- K. W. Heaton, J. Radvan, H. Cripps, R. A. Mountford, F. E. Braddon and A. O. Hughes, “Defecation Frequency and Timing, and Stool Form in the General Population: A Prospective Study,” Gut, Vol. 33, No. 6, 1992, pp. 818-824. doi:10.1136/gut.33.6.818

