Open Journal of Obstetrics and Gynecology
Vol.09 No.05(2019), Article ID:92393,12 pages
10.4236/ojog.2019.95062
Change in Vaginal Flora as Indicated by Pap Smear (Schröder’s Classification) in Women Using Levonorgestrel-Releasing Intrauterine System “Mirena”—Prospective Cohort Study
Malames M. Faisal1*, Magdy M. Abdel-Gawad1, Ragia A. Fahmy2, Mona S. Nour3, Marwa M. Shakweer4, Ahmed H. Abdel-Hakim1
1Department of Obstetrics and Gynecology, Faculty of Medicine, Ain Shams University, Cairo, Egypt
2Early Cancer Detection Unit, Faculty of Medicine, Ain Shams University, Cairo, Egypt
3Department of Microbiology, Faculty of Medicine, Ain Shams University, Cairo, Egypt
4Department of Pathology, Faculty of Medicine, Ain Shams University, Cairo, Egypt

Copyright © 2019 by author(s) and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: April 13, 2019; Accepted: May 12, 2019; Published: May 15, 2019
ABSTRACT
Background: The use of intrauterine device alters the normal balance between vaginal flora. It might change cervicovaginal environment, which was suggested to increase the risk for vulvovaginitis. It was found that up to 85% of levonorgestrel-releasing intrauterine system users become amenorrheic 4 months after insertion, or bleed only sporadically. It was hypothesized that this would have a beneficial effect on vaginal flora maintaining normal balance. Objectives: The aim of this study was to detect the effect of hormonal intrauterine system (“Mirena”: 20 microgram/24 hours intrauterine delivery system Levonorgestrel) on vaginal flora by using pap smear before and 6 months after insertion. Study design: A prospective cohort study was carried out in a university maternity hospital. 210 subjects were included with pap smear taken at time of insertion of IUS “Mirena” and another one taken 6 months later to detect percentage of incidence of infection in IUS users. Vaginal flora was studied, and lactobacilli were graded according to Schröder’s classification. A comparison was made to allow detection of changes in vaginal flora caused by presence of hormonal IUS “Mirena”. Results: Over half of cases (57.2%) showed worse vaginal flora after hormonal IUS “Mirena” insertion than before. There was a highly statistically significant difference between Schröder’s Grades before and after insertion (P value < 0.001) as 44.8% of cases presented by Grade I, 44.4% of cases presented by grade IIa, 38% of cases presented by grade IIb before insertion developed Grade III after insertion. Conclusion: Insertion of hormonal IUS “Mirena” results in significant change in microbial flora towards pathological bacteria.
Keywords:
IUD, Bacterial Vaginosis, Lactobacilli, Vulvovaginitis, Mirena, Contraception

1. Introduction
Vaginal flora is the microorganisms that normally colonize the vagina. It is a dynamic and complex example of microbial colonization, the regulation of which is markedly affected by many factors [1]. One of the most important organisms maintaining this eco system is “Lactobacilli”. Lactobacilli have been shown to inhibit in vitro growth of pathogenic microorganisms, through the action of lactic acid [2]. Moreover, they normally help to prevent long-term colonization of the vagina by adhering to vaginal epithelial cells. As well they produce antimicrobial peptides [3].
Normal vaginal flora includes a wide variety of microorganisms with varying types of both aerobic and anaerobic bacteria up to yeast as well. In this work, vaginal flora is classified according to Schröder’s classification ranging from grade I flora corresponding to predominantly lactobacillary morphotypes with very few coccoid bacteria up to grade III flora consisting of numerous other pathogenic microorganisms with no lactobacilli present and causing infection. This work is mainly focusing on three types of infections which are aerobic vaginitis, bacterial vaginosis and candidiasis as those infections arise from normally preexisting microorganisms due to disturbance of normal balance between vaginal flora [4].
In the last few years, a great advance in the world of contraception was achieved by the development of long acting reversible contraception, especially the intrauterine system (IUS) which is a hormonal intrauterine device (IUD). The most famous hormonal IUD is called Mirena and it is a levonorgestrel-releasing device. Reviewing literature, it was found that IUD is safe for all females seeking contraception even at high risk for sexually transmitted infections [5].
However, it is not settled yet whether hormonal IUD changes vaginal flora or not. Thus, this study targets those females using hormonal IUD “Mirena” to study the changes in vaginal flora influenced by the presence of the hormonal IUD [6].
The LNG-IUS is an ideal contraceptive method for women with underlying medical troubles as diabetes mellitus, or in women at increased risk of bleeding. In addition, use of LNG-IUS is not associated with increased risk of breast cancer [7].
The effect of LNG-IUS on the vaginal flora hasn’t been studied widely in different publications with different methodologies. A prospective study on cervical cytology and vaginal flora in long-term LNG-IUS users has shown no increase in cytopathological abnormalities, bacterial vaginosis or Trichomonas vaginalis incidence [8].
Another Prospective comparison of vaginal smears of LNG-IUS users and copper IUD users has shown that Copper-releasing IUD users were more likely than LNG-IUS users to have abnormal vaginal flora and bacterial vaginosis. Women usually develop an abnormal vaginal discharge 4 - 6 weeks after insertion of a copper IUD compared to IUS, however, this trend was not significant at the six-month follow-up. The lower prevalence of bacterial vaginosis found in the IUS users may also have been due to the mechanical influence of oligomenorrhoea rather than the direct hormonal effect. This may also justify the normal or low prevalence of bacterial vaginosis in users of DMPA despite their hypo-estrogenic state [9]. Conversely, another study stated that Pap smears suggested more vaginal infections after 1 year of LNG-IUS use than prior to insertion of the device [10].
Therefore, this work was carried out in normal women seeking hormonal IUS “Mirena” insertion for contraception; by performing Pap smear before and 6 months following insertion of the device to determine whether insertion of Levonorgestrel-releasing IUS would change the probability of occurrence of bacterial vaginosis, aerobic vaginitis and Moniliasis; due to impaired equilibrium between normally occurring vaginal flora.
2. Materials and Methods
2.1. Study Setting
A university maternity hospital, those attending family planning clinic for hormonal IUS insertion.
2.2. Study Design
A prospective cohort study studying change in vaginal flora following insertion of hormonal IUS, thus it is considered a safety trial (clinical trial) study. At the initial visit; the participants were counseled and told about the study and Pap smear was taken prior to insertion of the hormonal IUS “Mirena”. Vaginal flora was studied, and lactobacilli were graded according to Schröder’s original classification, further refined by Donders (4). After 6 months another Pap smear was taken with study of vaginal flora and lactobacillary grading was done with comparison to the Pap smear taken prior to insertion. This comparison allowed detection of changes in vaginal flora caused by presence of hormonal IUS “Mirena”.
2.3. Inclusion Criteria
Women in child bearing period without any comorbidity and with regular menses with average amount of menses with average built with body mass index ranging from 27 to 42.
2.4. Exclusion Criteria
1) Uterine or cervical fibroid.
2) Current or history of sexually transmitted disease.
3) Vulvovaginal infection of any type
4) Unexplained vaginal bleeding.
5) Structural abnormalities of the genital tract.
6) Sexual intercourse or vaginal douching during the last 48 hours.
7) Antibiotics taken through the last week.
8) Diabetics.
9) Contraindications to Progestogen e.g.: progestin-sensitive cancer and liver disease.
10) Hypersensitivity to any component of this hormonal intrauterine system e.g.: polyethylene and silicone.
11) Acute or chronic pelvic inflammatory disease.
2.5. Interventions
All included women were subjected to the following:
1) Proper history taking with emphasis on menstrual, obstetric and past medical history.
2) Detailed demographic, environmental and lifestyle information
3) General, abdominal and vaginal examination.
4) Pap smear was collected prior to Mirena insertion with proper preparation and fixation with alcohol. Pap smear was studied, and vaginal flora was stated.
5) Another Pap smear was collected 6 months following insertion of Mirena.
6) A comparison was made between Pap smears taken prior to insertion of IUS and the one taken 6 months after insertion as regards lactobacillary grading and vaginal flora changes.
2.6. Method
Microscopy procedures were performed on all Pap smears by two investigators who were blinded from any knowledge of both the timing of sampling and of the identity of the individual.
Study of vaginal flora changes by pap smear under microscopy was carried out as the following:
1) According to Schröder’s original classification, further refined by Donders (4), lactobacillary grading was as the following:
- Grade 0: Smears devoid of microflora
- Grade I: predominant lactobacilli with very few coccoid.
- Grade II: diminished lactobacilli mixed with other bacteria. This group is further subdivided into slightly disturbed, fairly normal IIa and moderately disturbed, rather abnormal IIb.
- Grade III: numerous other bacteria with no lactobacilli present.
2) Smears deficient in lactobacilli and positive for clue cells were considered to indicate a diagnosis of bacterial vaginosis. Aerobic vaginitis was diagnosed if smears were deficient in lactobacilli, positive for cocci (background flora), positive for parabasal epithelial cells, positive for vaginal leucocytes and presence of toxic leucocytes [11]. Severity of aerobic vaginitis was categorized according to a composite score (AV score), which assessed the four previous variables [11]. AV score ≤ 2 represents normality and corresponds to lactobacillary grade I and Grade II. AV score ≥ 3 represents infection. AV score of 8 to 10 is usually identical to so called “desquamative vaginitis” [12].
The study protocol was designed in agreement to the declaration of Helsinki for ethical medical research. The protocol was approved by the local Ethical Committee.
The study purpose and procedures were explained to all approached and eligible women. Women had to sign an informed written consent before participating in the study.
Any participating woman was informed that she has the right to withdraw from the study at any phase without any adverse impact on the medical service she receives.
Statistical methods:
Qualitative data were presented as number and proportion. The sum of number of cases of the three most common infections: aerobic vaginitis, bacterial vaginosis and moniliasis were used as an indicator of change in vaginal flora. Relative risk and attributable relative risk were measured. The statistical analysis was done with x2 test and odds ratio.
3. Results
To get accurate results 210 subjects were included in this study with Pap smear of each taken at time of insertion of IUS and another one taken 6 months later to detect percentage of incidence of infection in IUS users. In this study candidates having Pap smear with lactobacillary grade I or IIa were considered normal healthy individuals with normal vaginal flora and those with lactobacillary grade IIb or III were considered infected and this was supposed to be attributed to changes in vaginal flora occurring after insertion of hormonal IUD.
The mean age for included subjects was 28.8 ± 8.8 years. The findings of Pap smear before and after insertion are listed in (Table 1 & Table 2).
57.2% of the cases worsened after insertion of hormonal IUS “Mirena” (Graph 1).
There was significant worsening in Pap smear grades after insertion of IUS “Mirena” (Table 3, Figures 1-5).
4. Discussion
Although IUD is considered safer than COC in many aspects, many studies had found that both of them increase the risk of recurrent vulvovaginitis [13].
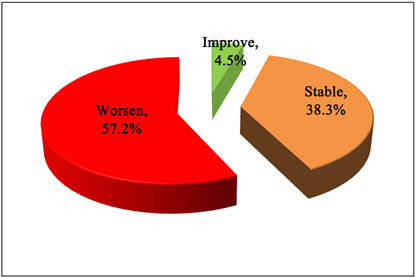
Graph 1. Pap change after insertion of hormonal IUS.
Figure 1. Inflammatory cells with degenerative changes with some bacilli.
Figure 2. Inflammatory cells with degenerative changes with some bacilli.
Figure 3. Inflammatory cells with degenerative changes with mixture of cocci and bacilli.
Figure 4. Diminished lactobacilli, with some cocci.
Figure 5. Bacteria cover squamous cells with no bacilli (clue cells).
Table 1. Detailed pap smear finding before and after insertion.
Total = 201.
Table 2. Brief of the pap smear finding before and after insertion. It shows that: The frequencies of abnormal grades increased after insertion.
Total = 201.
Table 3. Pap smear grades before and after insertion.
Percentages taken from before levels (rows), McNemar test, *Significant.
The use of intrauterine device was found to alter the normal balance between the different types of vaginal flora, changing the cervico-vaginal environment, so increasing the risk of vulvovaginal infection [14].
Recent studies proved that LNG-IUS is the safest methods of reversible contraception. This is attributed to 85% of females on LNG-IUS becoming amenorrhoeic 4 months after insertion. This helps to maintain the vaginal microbiological environment decreasing the risk of infection [15] [16] [17] [18].
However, a retrospective study done by (Donders and his colleagues 2011) had found that females using LNG-IUS were not completely safe from vaginal infection. It was found that they were at risk of vaginal infection after one year from its insertion [10].
This prospective study was carried out on 234 females in child bearing age attending the family planning clinic of university maternity hospital seeking levonorgestrel releasing IUS insertion. Nineteen of them were excluded, because they didn’t fulfill the inclusion criteria and five of them were missed in the follow-up, so the study was finally conducted on 210 cases.
Participants were subjected to full history taking, examination as well as pap smear taken prior to LNG-IUS insertion and six month after the IUS insertion to detect any changes in the vaginal flora.
The mean age of the participants was 28.8 years. Their mean BMI was 32.9 and they have a mean parity of 2.2.
As regards the last method of contraception used before attending family planning clinic, 30.3% didn’t use any methods of contraception before, while 22.9% were on COC which represents the most commonly previously used contraceptive method in the study group.
As regards the Cu IUD, 18.4% of participants were using it. This represents the second most common previously used contraceptive method. The POP was used by 10.4% of participants, the Implanon by 8% of participants and Depot Provera by 6.5% of participants. As regards Mirena, it was the least used method: by 3.5% of participants only.
All these different previously used contraceptive methods were shown to have no effect on the vaginal flora before and after LNG-IUS insertion as well as not to contribute to any pathological condition.
This study showed that 7% of cases had abnormal Pap smear findings before LNG-IUS insertion and it became 51.7% six months after insertion. Most of these abnormal smears were of grade III (38.8%) followed by grade IIb (13.9%).
As regards, the percentage of changes in Pap smear which reflect the change of vaginal microflora towards the pathological one, this study showed that more than half (57.2%) of the studied cases developed worse changes. Thus, this study revealed a significant worsening in the studied cases before and after LNG-IUS “Mirena” insertion. As regards the different Pap smear grades a significant worsening was found in the different grades. Our results were in agreement with Donders et al. [19] who found a temporary worsening in lactobacillary grades and increased rates of bacterial vaginosis and aerobic vaginosis after 3 months of LNG-IUS insertion. Candida increased significantly after long-term carriage of LNG-IUS compared to the period before insertion [OR 2.0 (CL 951.1 - 3.5), P = 0.017].
This study also goes in agreement with the retrospective study done by (Donders and his colleagues 2011) on 286 female that attended the gynecology outpatient clinic of the General Hospital of Heilig Hart Tienen in Belgium from June 1997 to September 2008 seeking LNG-IUS insertion as a method of contraception where 2 Pap smears were taken; one before IUS insertion in an average duration of 1 month to 2 year and the other one was obtained 1 to 2 years after insertion [10].
Regarding, the grade of lactobacilli the microflora was classified according to Schröder’s original classification that was further refined by Donders in 1999 into four grades from grade 0 to III. It was found that there was a significant change in microbial flora after IUS insertion towards the pathological bacteria [4].
However, this study disagrees with the Prospective study done by (Lessard and his colleagues in 2008) on 187 female attending the family planning clinic at the University of Estadual de Campinas that used LNG-IUS for long term as a contraceptive method. It was found by cervical cytology analysis that neither an increase in cytopathological abnormalities nor an increase in the incidence of vaginitis has occurred [8].
It also disagrees with that done by (Neale and his colleagues in 2009) that were done on 172 females using both types of IUD: Cu and hormonal IUS. It was found that only Cu IUD not hormonal IUS was associated with high incidence of vaginitis following its insertion, however, this disappeared within six months after insertion [9].
Few studies had focused on the effect of IUS on the vaginal flora. So further studies are needed in this field.
The current study concluded that there was a significant change in microbial flora towards the pathological bacteria six months after IUS insertion which might predisposes to vulvovaginitis. Further studies are needed with longer follow up.
Acknowledgements
None.
Funding Sources
None.
Conflicts of Interest
The authors declare no conflicts of interest regarding the publication of this paper.
Cite this paper
Faisal, M.M., Abdel-Gawad, M.M., Fahmy, R.A., Nour, M.S., Shakweer, M.M. and Abdel-Hakim, A.H. (2019) Change in Vaginal Flora as Indicated by Pap Smear (Schröder’s Classification) in Women Using Levonorgestrel-Releasing Intrauterine System “Mirena”—Prospective Cohort Study. Open Journal of Obstetrics and Gynecology, 9, 631-642. https://doi.org/10.4236/ojog.2019.95062
References
- 1. Larsen, B. and Monif, G.R. (2001) Understanding the Bacterial Flora of the Female Genital Tract. Clinical Infectious Diseases, 32, e69-e77. https://doi.org/10.1086/318710
- 2. Tachedjian, G., Aldunate, M., Bradshaw, C.S. and Cone, R.A. (2017) The Role of Lactic Acid Production by Probiotic Lactobacillus Species in Vaginal Health. Research in Microbiology, 168, 782-792.https://doi.org/10.1016/j.resmic.2017.04.001
- 3. Aroutcheva, A., Gariti, D., Simon, M., Shott, S., Faro, J., Simoes, J.A., Faro, S., et al. (2001) Defense Factors of Vaginal Lactobacilli. American Journal of Obstetrics and Gynecology, 185, 375-379.https://doi.org/10.1067/mob.2001.115867
- 4. Donders, G.G. (1999) Erratum: Microscopy of the Bacterial Flora on Fresh Vaginal Smears. Infectious Diseases in Obstetrics and Gynecology, 7, 177-179. https://doi.org/10.1155/S1064744999000290
- 5. Birgisson, N.E., Zhao, Q., Secura, G.M., Madden, T. and Peipert, J.F. (2015) Preventing Unintended Pregnancy: The Contraceptive CHOICE Project in Review. Journal of Women’s Health, 24, 349-353. https://doi.org/10.1089/jwh.2015.5191
- 6. Johnson, B.A. (2005) Insertion and Removal of Intrauterine Devices. American Family Physician, 71, 95-102.
- 7. Gemzell-Danielsson, K., Inki, P. and Heikinheimo, O. (2013) Safety and Efficacy of the Levonorgestrel-Releasing Intrauterine System: Recent Insights. Expert Review of Obstetrics & Gynecology, 8, 235-247. https://doi.org/10.1586/eog.13.18
- 8. Lessard, T., Simoes, J.A., Discacciati, M.G., Hidalgo, M. and Bahamondes, L. (2008) Cytological Evaluation and Investigation of the Vaginal Flora of Long-Term Users of the Levonorgestrel-Releasing Intrauterine System (LNG-IUS). Contraception, 77, 30-33. https://doi.org/10.1016/j.contraception.2007.09.009
- 9. Neale, R., Knight, I. and Keane, F. (2009) Do Users of the Intrauterine System (Mirena) Have Different Genital Symptoms and Vaginal Flora than Users of the Intrauterine Contraceptive Device? International Journal of STD & AIDS, 20, 423-424. https://doi.org/10.1258/ijsa.2008.008391
- 10. Donders, G.G., Berger, J., Heuninckx, H., Bellen, G. and Cornelis, A. (2011) Vaginal Flora Changes on Pap Smears after Insertion of Levonorgestrel-Releasing Intrauterine Device. Contraception, 83, 352-356. https://doi.org/10.1016/j.contraception.2010.08.007
- 11. Donders, G.G., Vereecken, A., Bosmans, E., Dekeersmaecker, A., Salembier, G. and Spitz, B. (2002) Definition of a Type of Abnormal Vaginal Flora That Is Distinct from Bacterial Vaginosis: Aerobic Vaginitis. BJOG: An International Journal of Obstetrics & Gynaecology, 109, 34-43. https://doi.org/10.1111/j.1471-0528.2002.00432.x
- 12. Gardner, H.L. (1968) Desquamative Inflammatory Vaginitis: A Newly Defined Entity. American Journal of Obstetrics & Gynecology, 102, 1102-1105. https://doi.org/10.1016/0002-9378(68)90399-2
- 13. Donders, G.G., Van Calsteren, K., Bellen, G., Reybrouck, R., Van den Bosch, T., Riphagen, I. and Van Lierde, S. (2009) Predictive Value for Preterm Birth of Abnormal Vaginal Flora, Bacterial Vaginosis and Aerobic Vaginitis during the First Trimester of Pregnancy. BJOG: An International Journal of Obstetrics & Gynaecology, 116, 1315-1324. https://doi.org/10.1111/j.1471-0528.2009.02237.x
- 14. Ocak, S., Cetin, M., Hakverdi, S., Dolapcioglu, K., Gungoren, A. and Hakverdi, A.U. (2007) Effects of Intrauterine Device and Oral Contraceptive on Vaginal Flora and Epithelium. Saudi Medical Journal, 28, 727-731.
- 15. Guttinger, A. and Critchley, H.O. (2007) Endometrial Effects of Intrauterine Levonorgestrel. Contraception, 75, S93-S98. https://doi.org/10.1016/j.contraception.2007.01.015
- 16. Prileszky, G., Kai, J. and Gupta, J. (2008) Mirena Coil for Heavy Menstrual Bleeding. British Journal of General Practice, 58, 886-887. https://doi.org/10.3399/bjgp08X376212
- 17. Romer, T. and Linsberger, D. (2009) User Satisfaction with a Levonorgestrel-Releasing Intrauterine System (LNG-IUS): Data from an International Survey. The European Journal of Contraception & Reproductive Health Care, 14, 391-398. https://doi.org/10.3109/13625180903203154
- 18. Pillai, M., O’Brien, K. and Hill, E. (2010) The Levonorgestrel Intrauterine System (Mirena) for the Treatment of Menstrual Problems in Adolescents with Medical Disorders, or Physical or Learning Disabilities. British Journal of Obstetrics and Gynaecology, 117, 216-221. https://doi.org/10.1111/j.1471-0528.2009.02372.x
- 19. Donders, G.G.G., Bellen, G., Ruban, K. and Van Bulck, B. (2018) Short- and Long-Term Influence of the Levonorgestrel-Releasing Intrauterine System (Mirena®) on Vaginal Microbiota and Candida. Journal of Medical Microbiology, 67, 308-313.https://doi.org/10.1099/jmm.0.000657







