Allelic Relationship between Lr9 and the Leaf Rust Resistance Gene in Kharchia Local Mutant of Wheat
Copyright © 2011 SciRes. AJPS
690
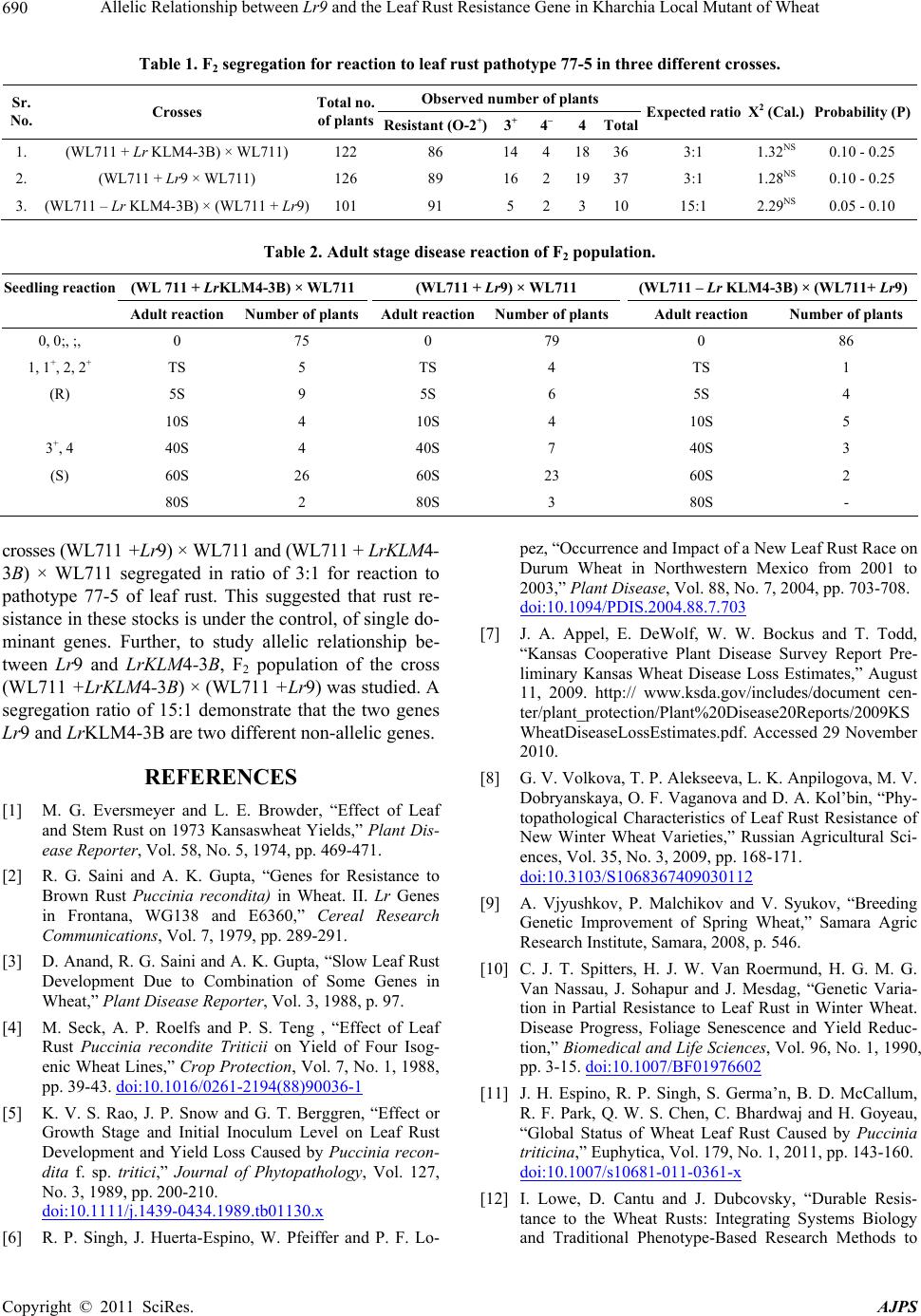
Table 1. F2 segregation for reaction to leaf rust pathotype 77-5 in three different crosses.
Observed number of plants
Sr.
No. Crosses Total no.
of plantsResistant (O-2+)3
+ 4– 4Total Expected ratio X2 (Cal.) Probability (P)
1. (WL711 + Lr KLM4-3B) × WL711) 122 86 14418363:1 1.32NS 0.10 - 0.25
2. (WL711 + Lr9 × WL711) 126 89 16219373:1 1.28NS 0.10 - 0.25
3. (WL711 – Lr KLM4-3B) × (WL711 + Lr9) 101 91 5231015:1 2.29NS 0.05 - 0.10
Table 2. Adult stage disease reaction of F2 population.
Seedling reaction (WL 711 + LrKLM4-3B) × WL711 (WL711 + Lr9) × WL711 (WL711 – Lr KLM4-3B) × (WL711+ Lr9)
Adult reaction Number of plants Adult reactionNumber of plantsAdult reaction Number of plants
0, 0;, ;, 0 75 0 79 0 86
1, 1+, 2, 2+ TS 5 TS 4 TS 1
(R) 5S 9 5S 6 5S 4
10S 4 10S 4 10S 5
3+, 4 40S 4 40S 7 40S 3
(S) 60S 26 60S 23 60S 2
80S 2 80S 3 80S -
crosses (WL711 +Lr9) × WL711 and (WL711 + LrKLM4-
3B) × WL711 segregated in ratio of 3:1 for reaction to
pathotype 77-5 of leaf rust. This suggested that rust re-
sistance in these stocks is under the control, of single do-
minant genes. Further, to study allelic relationship be-
tween Lr9 and LrKLM4-3B, F2 population of the cross
(WL711 +LrKLM4-3B) × (WL711 +Lr9) was studied. A
segregation ratio of 15:1 demonstrate that the two genes
Lr9 and LrKLM4-3B are two different non-allelic genes.
REFERENCES
[1] M. G. Eversmeyer and L. E. Browder, “Effect of Leaf
and Stem Rust on 1973 Kansaswheat Yields,” Plant Dis-
ease Reporter, Vol. 58, No. 5, 1974, pp. 469-471.
[2] R. G. Saini and A. K. Gupta, “Genes for Resistance to
Brown Rust Puccinia recondita) in Wheat. II. Lr Genes
in Frontana, WG138 and E6360,” Cereal Research
Communications, Vol. 7, 1979, pp. 289-291.
[3] D. Anand, R. G. Saini and A. K. Gupta, “Slow Leaf Rust
Development Due to Combination of Some Genes in
Wheat,” Plant Disease Reporter, Vol. 3, 1988, p. 97.
[4] M. Seck, A. P. Roelfs and P. S. Teng , “Effect of Leaf
Rust Puccinia recondite Triticii on Yield of Four Isog-
enic Wheat Lines,” Crop Protection, Vol. 7, No. 1, 1988,
pp. 39-43. doi:10.1016/0261-2194(88)90036-1
[5] K. V. S. Rao, J. P. Snow and G. T. Berggren, “Effect or
Growth Stage and Initial Inoculum Level on Leaf Rust
Development and Yield Loss Caused by Puccinia recon-
dita f. sp. tritici,” Journal of Phytopathology, Vol. 127,
No. 3, 1989, pp. 200-210.
doi:10.1111/j.1439-0434.1989.tb01130.x
[6] R. P. Singh, J. Huerta-Espino, W. Pfeiffer and P. F. Lo-
pez, “Occurrence and Impact of a New Leaf Rust Race on
Durum Wheat in Northwestern Mexico from 2001 to
2003,” Plant Disease, Vol. 88, No. 7, 2004, pp. 703-708.
doi:10.1094/PDIS.2004.88.7.703
[7] J. A. Appel, E. DeWolf, W. W. Bockus and T. Todd,
“Kansas Cooperative Plant Disease Survey Report Pre-
liminary Kansas Wheat Disease Loss Estimates,” August
11, 2009. http:// www.ksda.gov/includes/document cen-
ter/plant_protection/Plant%20Disease20Reports/2009KS
WheatDiseaseLossEstimates.pdf. Accessed 29 November
2010.
[8] G. V. Volkova, T. P. Alekseeva, L. K. Anpilogova, M. V.
Dobryanskaya, O. F. Vaganova and D. A. Kol’bin, “Phy-
topathological Characteristics of Leaf Rust Resistance of
New Winter Wheat Varieties,” Russian Agricultural Sci-
ences, Vol. 35, No. 3, 2009, pp. 168-171.
doi:10.3103/S1068367409030112
[9] A. Vjyushkov, P. Malchikov and V. Syukov, “Breeding
Genetic Improvement of Spring Wheat,” Samara Agric
Research Institute, Samara, 2008, p. 546.
[10] C. J. T. Spitters, H. J. W. Van Roermund, H. G. M. G.
Van Nassau, J. Sohapur and J. Mesdag, “Genetic Varia-
tion in Partial Resistance to Leaf Rust in Winter Wheat.
Disease Progress, Foliage Senescence and Yield Reduc-
tion,” Biomedical and Life Sciences, Vol. 96, No. 1, 1990,
pp. 3-15. doi:10.1007/BF01976602
[11] J. H. Espino, R. P. Singh, S. Germa’n, B. D. McCallum,
R. F. Park, Q. W. S. Chen, C. Bhardwaj and H. Goyeau,
“Global Status of Wheat Leaf Rust Caused by Puccinia
triticina,” Euphytica, Vol. 179, No. 1, 2011, pp. 143-160.
doi:10.1007/s10681-011-0361-x
[12] I. Lowe, D. Cantu and J. Dubcovsky, “Durable Resis-
tance to the Wheat Rusts: Integrating Systems Biology
and Traditional Phenotype-Based Research Methods to