 American Journal of Plant Sciences, 2011, 2, 660-668 doi:10.4236/ajps.2011.25079 Published Online November 2011 (http://www.SciRP.org/journal/ajps) Copyright © 2011 SciRes. AJPS Physico-Chemical and Microbial Quality of Locally Composted and Imported Green Waste Composts in Oman Saifeldin A. F. El-Nagerabi1*, Abdulkadir E. Elshafie2, Saif N. Al-Bahry 2, Hasina S. AlRawahi2, Huda AlBurashdi2 1Department of Biological Sciences and Chemistry, College of Arts and Sciences, University of Nizwa, Birkat Al Mouz, Nizwa, Sultanate of Oman; 2Department of Biology, College of Science, Sultan Qaboos University, AlKhoudh, Sultanate of Oman. Email: nagerabi@unizwa.edu.om Received August 7th, 2011; revised September 20th, 2011; accepted October 20th, 2011. ABSTRACT In this work the physical, chemical and microbial properties of four locally composted green waste composts (GWCs) namely Almukhasib, Grower s, Plantex, and Super along with four imported GWC (Florabella, Mikskaar, Potgrond, and Shamrock) were studied to evaluate the quality of these composts with the acceptable standards. All composts showed normal physical properties, except the bad smell from sulfur reducing bacteria in Almukhasib, light brown color Plantex and one viable weed seed in Shamrock compost. The germination indexes of the composts comparable to the standard (90%) were 100% for Mikskaar, followed by Shamrock (92%), Florabella (97), Potgrond (95%), Plantex (98%), Growers (77%), and 5% for both Super and Almukhasib. The physical and chemical properties vary considera- bly as follows: pH 3 - 10.5, 5.1 - 6.5 (standard 5 - 8), electrical conductivity (EC) 0.4 - 10.2 mS·cm–1, 0.8 - 1.8 mS·cm–1 (standard 0.0 - 4.0 mS·cm–1), moisture content (MC%) 29% - 43.7%, 64% - 74% (standard 35% - 60%) and water holding capacity (WHC%) 92% - 200% and 400% - 800% for the locally produced and imported composts, respectively. Wide ranges in the chemical properties were expressed as ammonia concentration 512.4 - 1640.1 mg·kg–1, 459.4 - 656.5 mg·kg–1 (standard < 500 mg·kg–1), organic matter 17% - 67.6%, and 53.3% - 66.2% (standard 35%) for the locally composted and imported composts, respectively. The concentrations of the heavy metals (Zn, Ni, Pb, Hg, As, Cd, and Cr) were lower than the recommended levels. The average of the bacterial colony forming unit per gram of locally pro- duced and imported composts rang ed between 260 - 1740 CF U /g and 330 - 2870 CFU/g, whereas the fungal CFU were 10 - 2800 CFU/g and 27 - 1800 CFU/g, respectively. The most probable number (MPN) for coliform ba cteria was 43 - 1100 CFU/g for loca lly produced composts, and 23 - 480 CFU/g for the impo rted comp osts. Therefore, these com posts can not be used directly without effective treatment as substrate for plant growth, soil amendment and as biofertilizer. Keywords: Aspergillus Niger, Coliform Bacteria, Physico-Chemical Properties, Green Waste Compost, Heavy Metals, Oman 1. Introduction Composting is a natural decaying of the organic matter to stable compost by aerobic and anaerobic actions of bac- teria, fungi and other organisms [1-3]. This process has the potential of managing organic materials in the waste stream such as leaves, farm wastes, animal manure, pa- per products, sewage sludge and domestic wastes [1,4]. Green waste compost (GWC) is a biodegradable waste that originates from pure plant materials of garden trim- mings or garbage collected from vegetable and fruits markets [5]. It provides benefits for soil biological acti- vity, and nutritive value to plant growth [6-8]. It im- proves the physical and chemical properties of the soil [9], enhances plant growth [10,11], remediates contami- nated soil [7,12-14], and suppresses some of these soil borne disease [15,16]. Quality control of the compost significantly promotes the recycling of the organic wastes [17]. The compost must comply with certain national and international stan - dards and quality grading [5,18]. The qualities of the compost include physical, chemical and biological pro- perties such as moisture content, odor, carbon and nitro- gen contents, phytotoxic substances, harmful elements,  Physico-Chemical and Microbial Quality of Locally Composted and Imported Green Waste Composts in Oman661 weeds, nutrient contents, plant pathogens and effective- ness to plant growth and soil amendment [19]. These can be maintained by the maturation of the compost and va- ried with the degree of transformation achieved by the organic materials [17]. Nonetheless, there is no single method that can be adopted universally to all compost types due to the wide range of feedstock, composting processes [20-22], and widely different chemical charac- teristics of organic wastes [23,24]. On the other hand, pathogens are commonly present in sewage and household wastes, both of which are commonly composted [25]. Therefore, composting is an efficient method for destruc- tion of pathogens to safe and acceptable level for human, animal and plant uses [21,26]. In Oman, land is the major non-renewable resource facing the challeng ing threat of soil d egrad ation. Sustain- able agriculture must be environmentally safe and must produce adequate amounts of quality foods with mini- mum purchased fertilizers and rely, as much as possible, on the renewable resources of the farm itself [27,28]. This is especially important in 90% of the farms that exist in the third world, where agricultural inputs are often not available or affordable [27]. Thus, compost is one of the important lo w cost inputs for meeting nutrient requirements of crops [29]. In Oman, green waste com- posts are imported from other countries at high cost and economic burden, therefore, compost industry was lo- cally developed for production of high quality composts with affordable prices to the small village farmers. The present study was conducted to evaluate the physical, chemical and microbial properties of locally produced and imported green waste composts in order to determine their ability to meet the acceptable standards. 2. Materials and Methods 2.1. Compost Samples Four locally produced green waste composts (Almuk- hasib, Growers, Plantex, Super), and four imported com- posts (Florabella, Mikskaar, Potgrond, Shamrock) were selected. The samples were collected according to the Gulf standard number GS0901/1997 [5]. Five samples of 1 kg each were collected from the compost bags, mixed to form composite samples and were then divided into four working samples. The samples were kept 5˚C for further analysis. 2.2. Hydrophysical Characterization Immediately after collection, the samples were visually inspected for free flowing, hard lumps, objectionable odor, and color. The particle size of the composts was determined according to the Gulf standard number GS01167/2002 using three replicates of 100 g oven dried samples [18]. The samples were placed on 12 mm sieves and shaken for 5 min at 100 shakes per min [30]. The percentage of the particles greater than 12 mm was calculated as per- centage by mass of the remaining materials on the top of the sieve to the mass of the test sample [31]. For testing the phytotoxicity of the compost samples and the presence of the viable seeds and plant parts, six plastic pots (10 × 15 cm) were filled with the compost samples. Three pots were seeded with 100 seeds of Phaseolus mungo (mungbean) and the remaining three pots were kept without seeds. As a control, another 100 seeds of P. mungo were inoculated into plastic trays with moistened cotton and incubated in the green house of the Biology Department, Sultan Qaboos University and were moistened daily for 17 days. The pots were examined regularly for seed germination. The hydrogen ion concentration (pH), moisture con- tents (MC%), electrical conductivity (EC), and water holding capacity (WHC %) of the composts were mea- sured using basic standard procedures and techniques [32]. The pH was determined in triplicate with the pH meter. For calculation of the moisture content, immedi- ately after the collection of the samples, moisture content was determined by the oven method [33]. Replicates of 10 g were placed in glass Petri dishes; soft lumps were crushed with a spatula and dried at 105˚C in an electric oven for 16 hours. The moisture content was determined as a percentage to the initial weig ht. For detection of electrical conductivity (EC) of the composts samples, replicates of 2 g from each sample were mixed in 5 ml of distilled water and the mixture was filtered through filtration unit with regu lar Whatman filter paper No. 42 (Whatman International Ltd, Maid- stone, UK). The electrical conductivity of each filtrate was measured by electrical conductivity meter. For determination of the water holding capacity (WHC%) of the composts, 500 g from each sample were added to pre-weighed dry sieve and pressed evenly. The samples were saturated with water, kept covered over-night, and then the dripped water was wiped off the sieve with fine tissues. The sie ve with the moistened sample was weig hed, placed in desiccators, allowed to dry and then reweighed to calculate the amount of water held by the samples. The WHC was calculated as percentage mass of the ab- sorbed water to the mass of the dried sample according to the gulf standard No. GS01/2002 [18]. 2.3. Chemical Analysis The organic matter (OM) of dried ground samples was determined by measuring the loss of mass through igni- tion at 550˚C according to the modified combustion me- thod suggested by many authors [21,34,35] and adopted by the Gulf standard NO. GSO1167/2002 [18]. From each sample, 10 g were used instead of 5 g in order to in- Copyright © 2011 SciRes. AJPS  Physico-Chemical and Microbial Quality of Locally Composted and Imported Green Waste Composts in Oman 662 crease the degree of the method accuracy. The samples were dried to constant mass in an oven at 105˚C and cooled in desiccators to avoid moisture absorption from the atmosphere. Ten grams from each sample were put into an oven-dried porcelain dish, placed in the furnace and the temperature was increased to 550˚C to convert the sample into ash. The percentage of the organic matter was calculated in triplicates as percentage loss of mass to the mass of the original test sample as a result of ignition. The ammonia-nitrogen contents of the compost sam- ples were determined in triplicate using Kjeld ahl method (Kjeltec Foss, Tecator AB, Hogana, Sweden, N-analy- zer). For this, 0.5 g from each sample and one keltab catalyst (SeK2SO4) were added to a digestion tube and mixed with 10 ml of sulfuric acid. The tubes were di- gested for 3 hour s, allowed to cool and the concentr ation of the ammonia was measured. For the heavy metal concentrations in the compost samples, 5 g from each sample were mixed with 25 ml of distilled water and the mixture was filtered with Milli- pore filter papers. Ten ml from the filtrate were analyzed with Inductive Couple Plasma (ICP-MS OPTIMA, 3100 RL Spectrometer, Perkin Elmer and Norwalk, USA). 2.4. Enumeration of Microorganisms The microorganisms including both fungi and bacteria were isolated from the compost using agar plate method. One gram from each sample was added to a test tube containing 9 ml sterile distilled water, vortexed, and se- rial dilutions were prepared. One ml was aseptically in- oculated on Potato Dextrose Agar (PDA) for fungal growth, and similarly Nutrient Agar (NA) was inoculated for bacterial growth. The inoculated PDA plates were incubated at 28˚C for 7 days, and the NA plates were incubated at 37˚C for 48 hours. At the end of th e incuba- tion period, the number of colony forming units (CFU) per gram of the compost was calculated. The isolated fungi were identified using different taxonomic books and monographs. The presence of coliform bacteria in the compost samples was screened using the standard table of the most probable number (MPN) . 2.5. Statistical Analysis Duncan’s multiple range and one way ANOVA were used for comparison between the co mpost types with p = 0.05. The analysis was carried out using statistical pack- age software SPSS (version 11.0). 3. Results and Discussion 3.1. Physical Properties Four locally produced green waste composts (Almukha- sib, Growers, Plantex, and Super), and four imported composts (Florabella, Mikskaar, Potgrond, and Sham- rock) were examined for their physical, chemical and microbial properties. The visual inspection showed that all the samples were physically uniform, free flowing, no hard lumps, dark brown to black in color, free from ob- jectionable odor, absent of foreign seeds and particle size less than 12 mm, except the bad smell of Almukhasib, light bro wn color of Pl an t ex, and one v i a ble weed seed i n shamrock (Table 1). These characteristics indicate the good quality of th e composts, the completion of the d eg- radation process and compost maturity as suggested by many authors [23,36]. The bad smell of the composts is due to production of hydrogen sulfide by sulfur reducing bacteria or faecal coliforms present in animal dropping mixed with plant materials without any pretreatment [23]. These odorous fumes contained hydrogen sulfide, me- thylmercaptan, and methylsulfide, and were present in large quantities at the initial stage of composting which decreased rapidly with maturation [19]. The presence of viable weed seeds and brown color of the composts was associated with compost immaturity [36]. Seed germination indexes in compost or compost ex- tracts are common biological methods to evaluate the degree of the maturity of the composted materials (the decomposition of phototoxic substance) and acids pro- duced during the early active composting stages [24,37]. In the present study, the germination percentages of the mungbean seeds in the tested composts were reported (Table 1). The germination percentages (in parenthesis) of the mungbean seeds in each of the corresponding compost were as follows: Mikskaar (100%), Shamrock (92%), Florab ella (97% ), Potgr ond (95 %), Plantex (9 8%), Growers (77%), and 5% for Super and Almukhasib. The germination levels in locally produced Super (5%), Al- mukhasib (5%) and Growers (77%) which were lower than the acceptable index (>90%) can be attributed to the phytotoxic effects of the organic acid and ammonia toxi- city produced during the active composting process [37, 38]. Therefore, these composts were not suitable for many potential uses. In the present study, the ammonia con- centration of the imported composts were: Mikskaar (617.9 mg·kg–1), Shamrock (656.5 mg·kg–1), Florabella (570.5 mg·kg–1), Potgrond (459.4 mg·kg–1), whereas the locally produced composts showed relatively higher ammonia concentration for Super (1640.1 mg·kg–1), Growers (1156.4 mg·kg–1), Almukhasib (804.2 mg·kg–1), and Plantex (712.4 mg·kg–1) (Table 2). Similarly, the electrical conductively (EC) which indicates the salt con- tents of the compost is injurious to plant roots and pre- vents their growth [39]. Therefore, the low level of ger- mination of the bean seeds in locally produced composts may be associated with the high electric conductivity of Super (10.2 mS·cm–1), Almukhasib (5.4 mS·cm–1) and Growers (7.5 mS·cm–1) which were higher than the upper standard limit (4 mS·cm–1) (Table 3). The imported Mik- Copyright © 2011 SciRes. AJPS 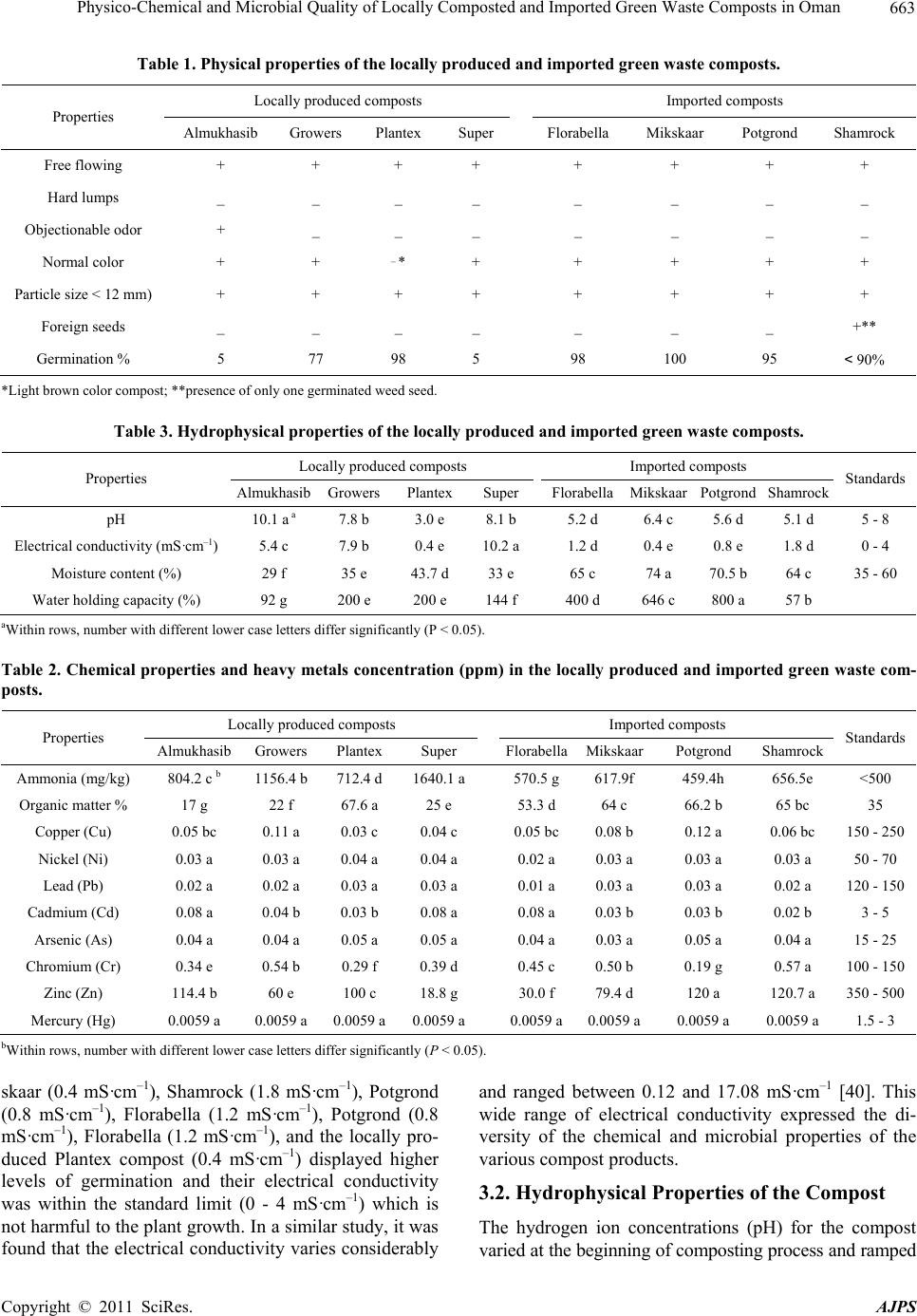 Physico-Chemical and Microbial Quality of Locally Composted and Imported Green Waste Composts in Oman Copyright © 2011 SciRes. AJPS 663 Table 1. Physical properties of the locally produced and imported green waste composts. Locally produced composts Imported c omposts Properties Almukhasib Growers Plantex Super FlorabellaMikskaar Potgrond Shamrock Free flowing + + + + + + + + Hard lumps _ _ _ _ _ _ _ _ Objectionable odor + _ _ _ _ _ _ _ Normal color + + _ * + + + + + Particle size < 12 mm) + + + + + + + + Foreign seeds _ _ _ _ _ _ _ +** Germination % 5 77 98 5 98 100 95 < 90% *Light brown color compost; **pre s ence of only one germ i nated weed seed. Table 3. Hydrophysical properties of the locally produced and imported green waste composts. Locally produced composts Imported c omposts Properties Almukhasib GrowersPlantexSuperFlorabellaMikskaarPotgrond ShamrockStandards pH 10.1 a a 7.8 b 3.0 e 8.1 b 5.2 d 6.4 c 5.6 d 5.1 d 5 - 8 Electrical conductivity (mS·cm–1) 5.4 c 7.9 b 0.4 e 10.2 a1.2 d 0.4 e 0.8 e 1.8 d 0 - 4 Moisture content (%) 29 f 35 e 43.7 d 33 e 65 c 74 a 70.5 b 64 c 35 - 60 Water holding capac it y (%) 92 g 200 e 200 e 144 f 400 d 646 c 80 0 a 57 b aWithin rows, number with different lower case letters differ significantly (P < 0.05). Table 2. Chemical properties and heavy metals concentration (ppm) in the locally produced and imported green waste com- posts. Locally produced composts Imported c omposts Properties AlmukhasibGrowers Plantex Super FlorabellaMikskaarPotgrond Shamrock Standards Ammonia (mg/kg) 804.2 c b 1156.4 b 712.4 d 1640.1 a570.5 g617.9f 459.4h 656.5e <500 Organic matter % 17 g 22 f 67.6 a 25 e 53.3 d 64 c 66.2 b 65 bc 35 Copper (Cu) 0.05 bc 0.11 a 0.03 c 0.04 c 0.05 bc0.08 b 0.12 a 0.06 bc 150 - 250 Nickel (Ni) 0.03 a 0.03 a 0.04 a 0.04 a 0.02 a 0.03 a 0.03 a 0.03 a 50 - 70 Lead (Pb) 0.02 a 0 .02 a 0.03 a 0.03 a 0.01 a 0.03 a 0.03 a 0.02 a 120 - 150 Cadmium (Cd) 0.08 a 0.04 b 0.03 b 0.08 a 0.08 a 0.03 b 0.03 b 0.02 b 3 - 5 Arsenic (As) 0.04 a 0.04 a 0.05 a 0.05 a 0.04 a 0.03 a 0.05 a 0.04 a 15 - 25 Chromium (Cr) 0.34 e 0.54 b 0.29 f 0.39 d 0.45 c 0.50 b 0.19 g 0.57 a 100 - 150 Zinc (Zn) 114.4 b 60 e 100 c 18.8 g 30.0 f 79.4 d 120 a 120.7 a 350 - 500 Mercury (Hg) 0.0059 a 0.0059 a 0.0059 a0.0059 a0.0059 a0.0059 a 0.0059 a 0.0059 a 1.5 - 3 bWithin rows, number with different lower case letters differ significantly (P < 0.05). skaar (0.4 mS·cm–1), Shamrock (1.8 mS·cm–1), Potgrond (0.8 mS·cm–1), Florabella (1.2 mS·cm–1), Potgrond (0.8 mS· c m–1), Florabella (1.2 mS·cm–1), and the locally pro- duced Plantex compost (0.4 mS·cm–1) displayed higher levels of germination and their electrical conductivity was within the standard limit (0 - 4 mS·cm–1) which is not harmful to the plant growth. In a similar study, it was found that the electrical conductivity varies considerably and ranged between 0.12 and 17.08 mS·cm–1 [40]. This wide range of electrical conductivity expressed the di- versity of the chemical and microbial properties of the various compost products. 3.2. Hydrophysical Properties of the Compost The hydrogen ion concentrations (pH) for the compost varied at the beginning of composting process and ramped  Physico-Chemical and Microbial Quality of Locally Composted and Imported Green Waste Composts in Oman 664 from 7.3 to 7.7 as the composting proceeded up to 8.8 - 9.6 [2]. All the screened composts, except the locally processed Almukhasib, showed acceptable pH value (5 - 8.0) (Table 3 ). However, the highly acidic Plantex (pH 3) may be due to production of phytotoxic organic acids during immature composting process which causes im- mediate growth injuries [38]. Therefore, the addition of this compost to soil may modify the pH of the final mix and buffer the soil pH [1]. The moisture contents of the composts ranged between 3.1% - 82.7% and varied considerably with the variation in the composted materials [40]. The moisture content values for the compost was considerably high in the first 3 weeks of composting after which it increased signifi- cantly in the later weeks [1]. Therefore, the addition of compost provides excellent drought resistance and great efficient water retention. In the present research (Table 3), the moisture contacts of the imported Shamrock is 64%, followed by Florabella (65%), Potgrond (70.5%) and Mikskaar (74%), which were higher than the ac- ceptable limits (35% - 60%), comparable to and locally produced Plantex (43%), Growers (35%), Super (33%), and Almukhasib (29%). The moisture content ranged between 50% - 60% and was considered as the optimal level for further composting [41]. Therefore, the compost with higher moisture content will inhibit aerobic degra- dation and enhance the unpleasant odor from the growth of anaerobic sulfate reducing bacteria. Yet, the ideal moisture content depends on how one plans to use the compost. Water retention cap acity of substrate is generally con- sidered as the quality determining factor [30,42]. The highest saturation of the compost is 75% and the good compost must have high water holding capacity and low filtration rate for supporting the plant growth. In the pre- sent study (Table 3), the water holding capacity (W HC% ) of the tested composts was found to be more than their actual weight. The water holding capacity of the imported composts ranged between 400% and 800% (Florabella 400%, Mikskaar 646%, shamrock 757%, Potgrond 800%) which is significantly higher than the locally processed Almukhasib (92%), Super (144%), Growers and Plantex compost (200%). Therefore, these composts can be used separately or mixed with sandy soil of low water holding capacity if they satisfy the other quality control parame- ters and the essential requirements for the plant growth. 3.3. Chemical Properties of the Compost Wide range in the values of the chemical properties of the compost expressed the diversity of various compost products and the raw materials used [40]. The total car- bon contents (TC) for various composts were in the range of between 16.9% - 51.0%. A approximately 11% - 27% of the total carton was lost during the 7 days of active composting, and 62% - 66% during the whole compost- ing time [43]. In the present results (Table 2), the total organic matter for the imported Potgrond (66.2%), Shamrock (65%), Mikskaar (64%), Florabella (53.5%), and locally produced Plantex (67.6%), were relatively higher than the standard set by the Gulf countries (35% and optimum 40% - 60%) [5]. On the other hand, the organic contents of the locally produced Super (25%), Growers (22%), and Almukhasib (17%) were below the standard limit. The high organic matter contents of the compost indicate the presence of uncomposted organic materials that can be degraded slowly by microorganisms and eventually used by higher pl ant s [44]. Heavy metals, as harmful elements, are one of the de- terminant factors for compost quality [19]. They may come from sewage water, addition of manure from chic- ken and other animal dung, and from soil added to the composted materials. They are released from compost and negatively affect the plant during the slow degrada- tion process. On the contrary, compost reduces the mo- bility of some toxic metals to the plants through forma- tion of some complexes. In this study, although ther e are significant variations in the heavy metals concentrations (Zn, Ni, Pb, Hg, As, Cd, Cr) between the screened com- posts, the concentration levels of heavy metals in the compost samples were lower than the acceptable limits recommended by the Gulf countries [5]. Similar standard limits were adopted in Germany [45], and Canada [46]. Nonetheless, the high contents of heavy metals may be due to addition of these metals to animal feeds [19] or contamination during the composting process [47]. 3.4. Microbial Estimates of the Compost It is natural to have large numbers of bacteria and fungi in the compost during composting process and they are essential for slow degradation of partially decomposed organic materials [1]. The pathogenic fungi and bacteria were normally detected in composted household wastes, and sewage sludge [25]. Nonetheless, composting is an efficient method for destruction of pathogenic microor- ganisms in the compost to a safer level for humans, ani- mals and plants [21,26]. Reasonable amounts of micro- organisms are still present in the compost at maturity [1]. In the present study, the average of the bacterial colony forming unit per gram of the compost were as follows: Plantex (260 cfu/g), Almukhasib (280 cfu/g), Shamrock (330 cfu/g), Growers (1490 cfu/g), Potgrond (1720 cfu/g), Super (1740 cfu/g), Mikskaar (2580 cfu/g), and Flora- bella compost (2870 cfu/g), whereas the colony forming unit of fungi in the compost were Almukhasib (10 cfu/g), Shamrock (30 cfu/g), Super (190 cfu/g), Florabella (200 cfu/g), Mikskaar (270 cfu/g), Plantex (360 cfu/g), Pot- Copyright © 2011 SciRes. AJPS  Physico-Chemical and Microbial Quality of Locally Composted and Imported Green Waste Composts in Oman Copyright © 2011 SciRes. AJPS 665 grond (1800 cfu/g), and Growers (2800 cfu/g) (Table 4). It is evident that the imported Florabella, Mikskaar, and the locally produced Super composts have the highest bacterial colonies, whereas the imported Potgrond, and the locally produced Growers and Plantex contain rela- tively high numbers of fungal colonies. These large num- bers of bacterial and fungal colonies were responsible for the slow degradation of the organic matter as suggested by several authors [21,23,48]. Most of the fungi involved in the slow degr adation of the screened composts be long to the thermophilic genus and moisture tolerant Asper- gillus. In the present study (Table 4), A. niger was the predominant species recovered from all compost types (100%) at the later stage as reported by many authors [1, 48,49]. This fungus was followed by A. fumigatus (75%), A. sparsus (50%), yeasts (50%), A. flavus (37.5%), where the remaining fungi such as A. restrictus, A. ver- sicolor, Cladosporium spp., and Penicillium spp. were recovered from 50% of the compost types, and to a lesser extent Acremonium sp. and A. ochraceous (12.5%). In similar studies, different species of Aspergillus and Peni- cllium were isolated from the compost [1,8,48]. Various pathogenic bacteria were isolated from diffe- rent composts and composted materials [1,21]. The ma- jor faecal coliforms found in the raw materials composed of Escherichia coli, where in the finished compost the majority of the faecal coliforms were probably of non- faecal origin [21]. Therefore, the species composition of the faecal coliforms can vary considerably depending on the composted materials and composting system. In the present results, the most probable number (MPN) was used to determine the faecal contamination of the com- posts. Our findings (Table 4) showed that locally pro- duced Almukhasib was highly contaminated with coli- forms (1100 cfu/g), followed by the imported Florabella (480 cfu/g), Potgrond (240 cfu/g), and to a lesser extent by locally produced Plantex (150 cfu/g), Growers (93 cfu/g), Super (43 cfu/g), and the imported Mikskaar (43 cfu/g) and Shamrock (23 cfu/g). The presence of higher number of coliforms in the locally processed Almukhasib, followed by imported Florabella and Potgrond indicates the possible contamination of these composts with sew- age water or other animal products during composting process, which was confirmed by isolation of coliform bacteria as suggested by many researchers [1,21]. There- fore, there is a high possibility of transmission of serious diseases during handling and usage of these composts in addition of expected infestation of the cultivated plants with serious pathogenic bacteria. 4. Conclusions It is apparent that all the investigated composts were free from most of the physical constraints, except the light brown color of Plantex, bad smell of Almukhasib and viable seed in Shamrock which indicates immaturity of the composts. The locally processed composts contain phytotoxic acids, ammonia, and with high electrical con- ductivity which affect the seed germination. The mois- ture contents of the imported Florabella, Mikskaar, Pot- grond, and Shamrock were higher than the locally pro- Table 4. Microbial properties of the locally produced and imported green waste composts. Locally produced composts Imported c omposts Properties Almukhasib GrowersPlantexSuper FlorabellaMikskaar Potgrond Shamrock Bacteria (CFU/g) 280h *c 1490 e 260 g 1740 c2870 a 2580 b 1720 d 330 f Fungi (CFU/g) 10 h 2800 a 360 c 190 f 200 e 270 d 1800 b 30 g MPN (CFU/g) 1100 a 93 e 15 0 d 43 f 480 b 43 f 240 c 23 g Acremonium sp. - - - - - - + - Aspergillus flavus - - + + - - - + A. fumigatus - + + + - + + + A. niger + + + + + + + + A. ochraceous - - + - - - - - A. sparsus + - - + - + - + A. restrictus - - - + - + - - A. versicolor - - - - + - - + Cladosporium sp. - + - - - - + - Penicillium sp. - - - + + - - - Yeasts + + - - - - + + cWithin rows, number with different lower case letters differ significantly (P <0.05).  Physico-Chemical and Microbial Quality of Locally Composted and Imported Green Waste Composts in Oman 666 duced Almukhasib, Growers, Plantex, and Super com- posts. The water holding capacity was significantly higher in all composts and meets the standard limit. The total organic matters were higher in the imported composts in comparison with the locally composted materials. The heavy metals contents of all composts were below the acceptable limits. The composts were contaminated with variable levels of saprophytic fungi and colifor m bacteria. It is evident the imported composts were relatively better than the locally produced composts, however, none of them meet most of the recommended characteristics [50]. Therefore, they can not be used directly and without any treatment as media for plant growth, soil biofertilizer and for soil amendment. Therefore, there is a high necessity for setting detailed legislation, regu lation policies, proper testing methods, quality control measurements, and strong quarantine regulations for exportimport and local produ- ction of green waste composts. Attention should be given to the local production of high quality composts which serve the environment, waste management, recycling in- dustry and satisfaction of the local markets. 5. Acknowledgements We thank the Department of Biology, College of Science, Sultan Qaboos University for providing space and facul- ties to carry out this research. Dr. Tom Hughes of Uni- versity of Nizwa Writing Center proof read the English of this manuscript. Dr. Peter Cowan of the Department of Biological Sciences and Chemistry, University of Nizwa, improved the scientific content of the article. REFERENCES [1] D. V. Adegunloye, F. C. Adetuyi, F. A. Akinyosoye and M. O. Doyeni, “Microbial Analysis of Compost Using Cow Dung as Booster,” Pakistan Journal of Nutrition, Vol. 6, 2007, pp. 506-610. doi:10.3923/pjn.2007.506.510 [2] R. Briancesco, A. M. Coccia, G. Chairetti, S. Della, Libera, M. Semproni and L. Bonadonna, “Assessment of Microbiological and Parasitological Quality of Composted Wastes: Health Implications,” Waste Management and Re- search, Vol. 26, No. 2, 2008, pp. 196-202. doi:10.1177/0734242X07085064 [3] K. Salvator and W. E. Sabee, “Evaluation of Fertilizer Value and Nutrient Release from Corn and Soybean Residue under Laboratory and Greenhouse Conditions,” Communication in Soil Science and Plant Analysis, Vol. 26, No. 3-4, 1995, pp. 469-484. doi:10.1080/00103629509369312 [4] J. A. Adeniran, L. B. Taiwo and R. A. Sobulo, “Effects of Organic Wastes and Method of Composting on Compost Maturity, Nutrient Composition of Compost and Yield of Two Vegetable Crops,” Journal of Sustainable Agricul- ture, Vol. 22, No. 4, 2003, pp. 95-101. doi:10.1300/J064v22n04_08 [5] Anonymous, “Municipal Solid Waste Compost,” Stan- dardization Organization for G. C. C. (GSO) 901, 1997. [6] R. Barberis and P. Nappi, “Evaluation of Compost Stabi- lity,” In: M. de Bertoldi, et al., Eds., The Science of com- posting, Blackie Academic and Professional, Glasgow, 1996, pp. 175-184. [7] W. Hartley, N. M. Dickison, P. Riby and N. W. Lepp, “Arsenic Mobility in Brownfield Soils Amended with Green Waste Compost or Biochar and Planted with Mis- canthus,” Environmental Pollution, Vol. 157, No. 10, 2009, pp. 2654-2662. doi:10.1016/j.envpol.2009.05.011 [8] G. Straatsma, R. A. Samson, T. W. Olijnsma, H. J. M. O. P. Den Camp, J. P. G. Gerrits and L. J. L. D. van Griens- ven, “Ecology of Thermophilic Fungi in Mushroom Com- post, with Emphasis on Scytalidium thermophilum and Growth Stimulation of Agaricus biosporus Mycelium,” Applied and Environmental Microbiology, Vol. 60, 1994, pp. 454-458. [9] R. C. Dalal, I. Gibson, D. E. Allen and N. W. Menzies, “Green Waste Compost Reduces Nitrous Oxide Emissions from Feedlot Manure Applied to Soil,” Agriculture, Eco- systems and Environment, Vol. 10, 2009, pp. 1-9. [10] M. Ali, A. J. Griffith, K. P. Williams and D. L Jones, “Evaluating the Growth Characteristics of Lettuce in Ver- micompost and Green Waste Compost,” European Jour- nal of Soil Biology, Vol. 43, 2007, pp. 316-319. doi:10.1016/j.ejsobi.2007.08.045 [11] A. A. Keeling, K. R. McCallum and C. P. Beckwith, “Mature Green Waste Compost Enhances Growth and Nitrogen Uptake in Wheat (Triticum aestivum L.) and Oil Seed Rape (Brassica napus L.) through the Action of Water-Extractable Factors,” Bioresource Technology, Vol. 90, No. 2, 2003, pp. 127-132. doi:10.1016/S0960-8524(03)00125-1 [12] P. Alvarenga, A. P. Goncalves, R. M. Fernandes, A. de Varennes, E. Duarte, A. C. Cunha-Queda and G. Vallin, “Reclamation of a Mine Contaminated Soil Using Bio- logically Reactive Organic Matrices,” Waste Management and Research, Vol. 27, No. 2, 2009, pp. 101-111. doi:10.1177/0734242X08091556 [13] R. Van Herwijnen, T. R. Hutchings, A. Al-Tabbaa, A. J. Moffat, M. L. Johns and S. K. Ouki, “Remediation of Metal Contaminated Soil with Mineral-Amended Com- posts,” Environmental Pollution, Vol. 150, No. 3, 2007, pp. 347-354. doi:10.1016/j.envpol.2007.01.023 [14] R. van Herwijnen, S. K. Ouki, A. Al-Tabbaa, A. J. Moffat, M. L. Johns and T. R. Hutchings, “The Effect of Two Different Composts on the Performance and Metal Up- take of Poplar Growing on Heavy Metal Contaminated Soil,” SEESOIL, Vol. 17, 2008, pp. 39-48. [15] J. Lozano, W. J. Blok and A. J. Termorshuizen, “Effect of Compost Particle Size on Suppression of Plant Diseases,” Environmental Engineering Science, Vol. 26, No. 3, 2009, pp. 601-607. doi:10.1089/ees.2008.0002 [16] D. J. Van deer Gaag, F. R. van Noort, L. H. M. Stapel- Cuijpers, C. de Kreij, A. J. Termorshuizen, E. van Rijn, S Zmora-Nahum and Y. Chen, “The Use of Green Waste Copyright © 2011 SciRes. AJPS  Physico-Chemical and Microbial Quality of Locally Composted and Imported Green Waste Composts in Oman667 Compost in Peat-Based Potting Mixtures: Fertilization and Suppressiveness against Soil Borne Diseases,” Scien- tia Horticulturae, Vol. 114, No. 4, 2007, pp. 289-297. doi:10.1016/j.scienta.2007.06.018 [17] M. T. C. Mondini,. L Dell’Abate, Leita and A. Benedetti, “An Integrated Chemical, Thermal, and Microbiological Approach to Compost Stability Evaluation,” Journal of Environmental Quality, Vol. 32, No. 6, 2003, pp. 2379- 2386. doi:10.2134/jeq2003.2379 [18] Anonymous, “Test Method of Potting Mixes (Potting Soil),” Standardization Organization for G. C. C. (GSO) 1169, 2002. [19] Y. Harada, K. Haga, T. Osada and M. Koshino, “Quality of Compost Produced from Animal Wastes,” JARQ, Vol. 26, No. 4, 1993, pp. 238-246. [20] Y. Chen, Y. Inbar, B. Chefetz and Y. Hadar, “Compost and Recycling of Organic Wastes P.341-362,” In: D Rosen, et al., Eds., Modern Agriculture and Environment, Kulwer Academic Publishers, Dordrecht, 1996. [21] K. K. Christensen, M. Carisbaek and E. Kron, “Strategies for Evaluating the Sanitary Quality of Composting,” Journal of Applied Microbiology, Vol. 92, 2002, pp. 1143-1158. doi:10.1046/j.1365-2672.2002.01648.x [22] M. Itävaara, O. Venelampi, M. Vikman and A. Kapanen, “Compost Maturity-Problem Associated with Testing,” In: H Insam, et al., Eds., Microbiology of Composting, Springer Verlag, Heidelberg, 2002, pp. 373-382 [23] M. Benito, A. Masaguer, A. Moliner, N. Arrigo and R. M. Palma, “Chemical and Microbial Parameters for the Characterization of the Stability and Maturity of Pruning Waste Compost,” Biology of Fertile Soils, Vol. 37, No. 3, 2003, pp. 184-189. [24] T. J. He, X. T. Logan and S. J. Traine, “Physical and Chemical Characteristics of Selected U.S. Municipal Waste Composts,” Journal of Environmental Quality, Vol. 24, No. 3, 1995, pp. 543-552. doi:10.2134/jeq1995.00472425002400030022x [25] H. Pahren, “Microorganisms in Municipal Solid Waste and Public Health Applications,” CRC Critical Reviews in Environmental Control, Vol. 17, 1987, pp. 187-228. doi:10.1080/10643388709388334 [26] S. Dumontet, H. Dinel and S. B. Baloda, “Pathogen Re- duction in Sewage Sludge by Composting and Other Biological Treatments. A review,” Biological Agriculture and Horticulture, Vol. 16, No. 4, 1999, pp. 409-430. [27] C. Chitravadivu, V. Balakrishnan, J. Manikandan, T. Elavazhagan and S. Jayakumar, “Application of Food Waste Compost on Soil Microbial Population in Groundnut Cultivated Soil, India,” Middle-East Journal of Scientific Research, Vol. 4, No. 2, 2009, pp. 90-93. [28] G. Sarwar, H. Schme isky, N. Hussain, S. Muhammad, M. Ibrahim and E. Safdar, “Improvement of Soil Physical and Chemical Properties with Compost Application in Rice-Wheat Cropping System,” Pakistan Journal of Bot- any, Vol. 40, No. 1, 2008, pp. 275-282. [29] F. Zameer, S. Meghashri, S. Copal and B. R. Rao, “Chemical and Microbial Dynamics during Composting of Herbal Pharmaceutical Industrial Waste,” E-Journal of Chemistry, Vol. 7, No. 1, 2010, pp. 143-148. [30] M. Abd El-Hady and E. I. El-Dirdiry, “Improving Hy- drophysical Properties of Compost,” Journal of Applied Sciences Research, Vol. 2, No. 12, 2006, pp. 1137-1141. [31] R. Orozco, S. Gschwander and O. Marfa, “Substrate Clas- sification from Particle Size Analysis,” Acta Horticulturae, Vol. 450, 1997, pp. 397-403. [32] G. C. S. Wilson, “The Physio-Chemical and Physical Properties of Horticultural Substrates,” Acta Horticul- turae, Vol. 150, 1983, pp. 19-32. [33] S. A. F. El-Nagerabi and A. E Elshafie, “Incidence of See-Borne Fungi and Aflatoxins in Sudanese Lentil Seeds,” Mycopathologia, Vol. 149, 2000, pp. 151-156. doi:10.1023/A:1007241125586 [34] Y. Inbar, Y. Chen and Y. Hadar, “Humic Substances Formed during the Composting of Organic Matter,” Soil Science Society of America Journal, Vol. 54, No. 5, 1990, pp. 1316-1323. doi:10.2136/sssaj1990.03615995005400050019x [35] A. C. Petrus, O. H. Ahmed, A. N. Muhamed, H. M. Nas- sir, M. Jiwan and M. G Banta, “Chemical Characteristic of Compost and Humic Acid from Sago Waste (Metroxylon sagu) ,” American Journal of Applied Sci- ences, Vol. 6, No. 11, 2009, pp. 1880-1884. doi:10.3844/ajassp.2009.1880.1884 [36] E. Iglesias-Jiménez and V. Pérez-Garcia, “Determination of Maturity Indices for City Refuses Composts,” Agricul- ture, Ecosystems and Environment, Vol. 38, No. 4, 1992, pp. 331-343. doi:10.1016/0167-8809(92)90154-4 [37] G. A. Wu, L. Ma and L. Q. Martinez, “Comparison of Methods for Evaluating Stability and Maturity of Biosol- ids Compost,” Journal of Environmental Quality, Vol. 29, No. 2, 2000, pp. 424-429. doi:10.2134/jeq2000.00472425002900020008x [38] M. H. Wong, “Phytotoxicity of Refuse Compost during the Process of Maturation,” EA, Ecological and Biolo- gical, Vol. 37, 1985, pp. 159-174. [39] H. Cai, T. Chen, H. Liu, D. Gao, G. Z heng and J. Zhang, “The Effect of Salinity and Porosity of Sewage Sludge Compost on the Growth of Vegetable Seedlings,” Scien- tia Horticulturae, Vol. 124, No. 3, 2010, pp. 381-386. doi:10.1016/j.scienta.2010.01.009 [40] J. C. Tang, “Chemical and Microbial Properties of Various Compost Products,” Soil Science and Plant Nutrition, Vol. 49, 2003, pp. 273-280. [41] S. M. Tiquia, N. F. Y T.am and I. J. Hodgkiss, “Microbial Activity during Composting of Spent Pig-Manure Saw- dust Litter at Different Moisture Contents,” Bioresource Technology, Vol. 55, 1996, pp. 2010-206. doi:10.1016/0960-8524(95)00195-6 [42] J. Caron and V. K. N. Nkongollo, “Aeration in Growing Media: Recent Developments,” Acta Horticulturae, Vol. 481, 1999, pp. 545-551. [43] A. H. Vuorienin and M. H. Saharinen, “Evaluation of Microbial and Chemical Parameters during Manure and Copyright © 2011 SciRes. AJPS  Physico-Chemical and Microbial Quality of Locally Composted and Imported Green Waste Composts in Oman Copyright © 2011 SciRes. AJPS 668 Straw Composting in a Drum Composting System,” Agri- culture, Ecosystems and Environment, Vol. 66, No. 1, 1997, pp. 19-29. doi:10.1016/S0167-8809(97)00069-8 [44] A. Bary, C. Cogger and D. Sullivan, “What Does Com- post Analysis Tell You about Your Compost?” Poster at Biologically Intensive and Organic Farming Research Conference, Yakima, 2002. [45] W. F. Brinton, “Compost Quality Standards and Guide- lines,” Wood and Research Laboratory, West Lafayette, 2000, pp.1-41. [46] Canadian Food Inspection Agency, “Standards for Metals in Fertilizers and Supplement,” Trade Memorandum, T-4-93, 1997. [47] G. R. E. M. Van Roosmallen, J. W. A. Lustenhouwer, J. Oosthoek and M. M. G. Senden, “Heavy Metal Sources and Contamination Mechanisms in Compost Production,” Resources and Conservation, Vol. 14, 1987, pp. 321-334. doi:10.1016/0166-3097(87)90032-0 [48] E. S. Dias, S. E. Guimaraes, F. G. de Siqueira, R. da Silva and L. R Batista, “Allergenic and Toxigenic Fungi in the Compost of Cultivation of Agaricus brasiliensis,” Scien- tia Agrarian, Vol. 10, No. 6, 2009, pp. 507-511. [49] P. D. Millner, P. B. Marsh, R. B. Snowden and J. F. Parr, “Occurrence of Aspergillus fumigatus during Composting Sewage Sludge,” Applied and Environmental Microbiology, Vol. 34, No. 6, 1977, pp. 765-772. [50] Anonymous, “Method for Testing Municipal Solid Waste Compost,” Standardization Organization for G. C. C. (GSO) 1167, 2002.
|