 Materials Science s a nd Applications, 2011, 2, 1644-1653 doi:10.4236/msa.2011.211219 Published Online November 2011 (http://www.SciRP.org/journal/msa) Copyright © 2011 SciRes. MSA Dispersibility, Shape and Magnetic Properties of Nano-Fe3O4 Particles Xiaojuan Liang1, Haowei Shi2, Xiangchen Jia2, Yuxiang Yang2*, Xiangnong Liu3 1College of Chemistry and Materials Engineering, Wenzhou University, Wenzhou, China; 2Department of Chemistry, East China University of Science & Technology, Nanjing, China; 3Analysis Test Center, Yangzhou University, Yangzhou, China. Email: *yxyang@ecust.edu.cn Received July 25th, 2010; revised November 19th, 2010; accepted September 8th, 2011. ABSTRACT Nano-Fe3O4 particles were prepared by a two-step microemulsion method, the influence of molar ratio of water to NP-5 (R), alkali concentration and temperature on dispersibility and shape of the nanoparticles were discussed. Mag- netic studies were also carried out using VSM in this paper. It was found that the optimum preparation parameters are R = 6.0, alkali concentration = 2.5 mol·L–1, initial total iron concentration as 0.88 mol·L–1, and the temperature being 30˚C, the prepared nano magnetite particles have uniform size and good dispersibility with a crystal structure belong- ing to cubic Fe3O4 and lattice parameters of a = 8.273 Å. The results of magnetic studies show, magnetic properties of particles are influenced by dispersibility of nanoparticles which depends on size of clusters. The better dispersibility of nanoparticles leads to more ordered inner magnetic vector, and so the stronger magnetic behavior of nano-Fe3O4 par- ticles. Keywords: Nano-Fe3O4 Particle, Dispersibility of Nanoparticles, Magnetic Properties, Saturation Magnetization 1. Introduction Magnetic nanoparticles have been widely studied be- cause of their fascinating properties and wide range of potential applications in ferrofluids, information storage, pigment, medicine, biomedical and bioengineering, etc. [1-3] The nano magnetite particles can be synthesized by precipitation from the solution of mixed Fe(II)/Fe(III) salts in alkaline medium. Besides this, many techniques have been used to synthesize magnetite such as thermal decomposition [4] hydrothermal synthesis [5], coprecipi- tation of an aqueous solution of ferrous and ferric ions by a base [6-8],oxidation of the ferrous hydroxide gels using KNO3 [9], γ-ray irradiation [10], microwave plasma syn- thesis [11] sol-gel [12], nonaqueous route [13-15] etc. Many applications depend on their size and stoichiome- try of particles, however the dispersibility and shape of prepared particles varies with the hydrolysis condition, the additive surfactant[4] and organic compound [5], most of them exhibit nano spherical particles [16-18]. Many reports have not systematically studied various factors affecting dispersibility and shape of nano-Fe3O4 particles, especially the relationship between dispersibil- ity and their magnetic behavior. So in this paper, we re- port the preparation process of nano-Fe3O4 particles us- ing two-step microemulsion method. The different fac- tors affecting dispersibility of nano-Fe3O4 particles, in- cluding NaOH concentration, reaction temperature, wa- ter-surfactant ratio, and initial total iron concentration, are discussed. The relationship between the magnetic properties and dispersion behavior of nano-Fe3O4 parti- cles is also studied. 2. Experimental 2.1. Synthesis of Nanoparticles Fe3O4 Surfactant NP-5, cosurfactant butanol, and kerosene were mixed in the ratio of 1:1:10, the muddy mixed solution was then stirred until a clear and transparent solution was obtained. The previous solution was divided into two parts of (1) and (2). Firstly, the boiled deionized water was added to the solution of (1) with a ratio R (R is the molar ratio of wa- ter to NP-5), the microemulsion became clearer and clearer through stirring. Then certain amount of FeCl2·4H2O and FeCl3·6H2O were added into the microemulsion system with the molar ratio of Fe2+ to Fe3+ as 3:2 while vigor- ously stirring. The microemulsion labeled as (1) became brown-yellow and transparent. Secondly, the boiled NaOH solution with the concen- 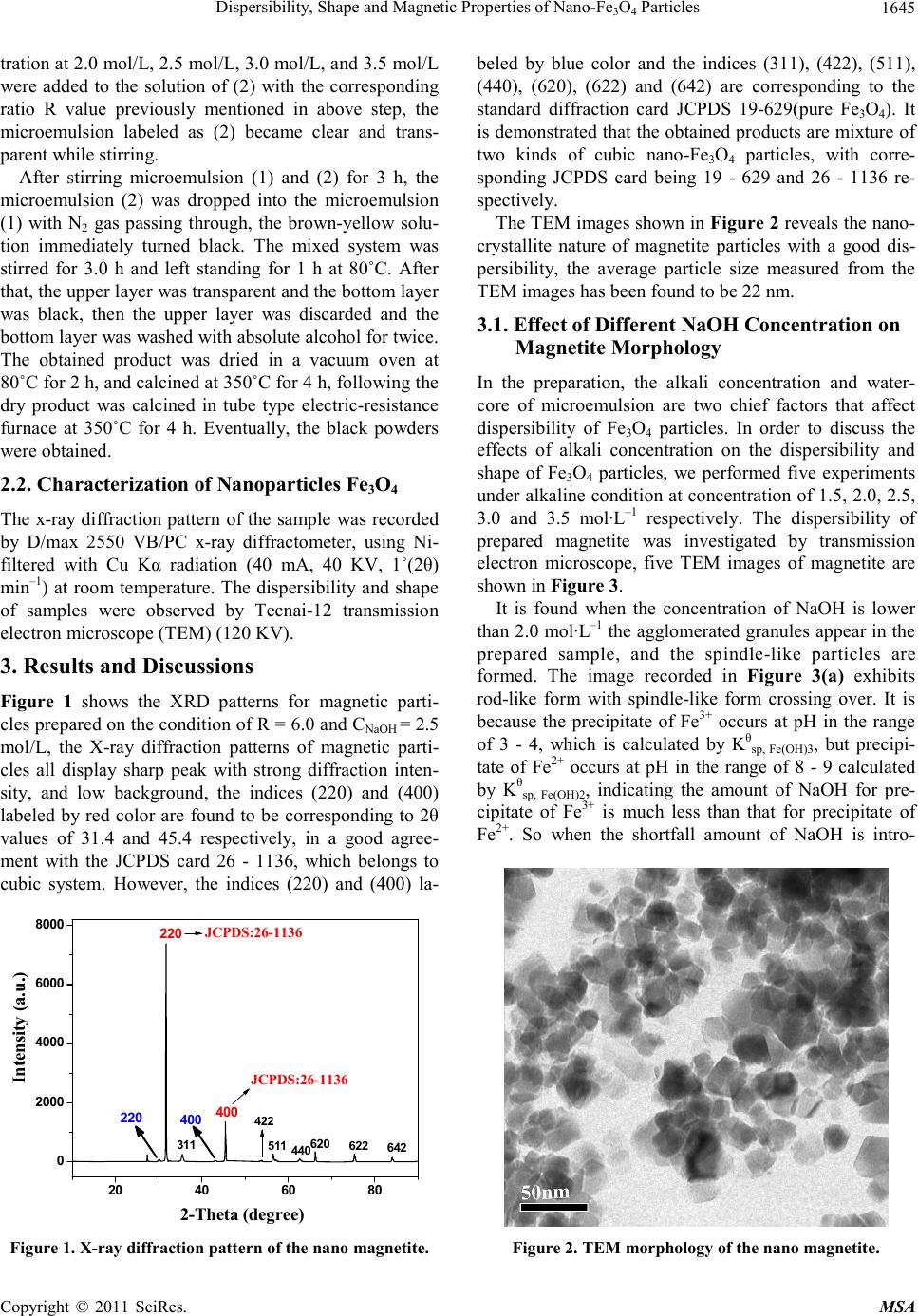 Dispersibility, Shape and Magnetic Properties of Nano-FeO Particles1645 3 4 tration at 2.0 mol/L, 2.5 mol/L, 3.0 mol/L, and 3.5 mol/L were added to the solution of (2) with the corresponding ratio R value previously mentioned in above step, the microemulsion labeled as (2) became clear and trans- parent while stirring. After stirring microemulsion (1) and (2) for 3 h, the microemulsion (2) was dropped into the microemulsion (1) with N2 gas passing through, the brown-yellow solu- tion immediately turned black. The mixed system was stirred for 3.0 h and left standing for 1 h at 80˚C. After that, the upper layer was transparent and the bottom layer was black, then the upper layer was discarded and the bottom layer was washed with absolute alcohol for twice. The obtained product was dried in a vacuum oven at 80˚C for 2 h, and calcined at 350˚C for 4 h, following the dry product was calcined in tube type electric-resistance furnace at 350˚C for 4 h. Eventually, the black powders were obtained. 2.2. Characterization of Nanoparticles Fe3O4 The x-ray diffraction pattern of the sample was recorded by D/max 2550 VB/PC x-ray diffractometer, using Ni- filtered with Cu Kα radiation (40 mA, 40 KV, 1˚(2θ) min–1) at room temperature. The dispersibility and shape of samples were observed by Tecnai-12 transmission electron microscope (TEM) (120 KV). 3. Results and Discussions Figure 1 shows the XRD patterns for magnetic parti- cles prepared on the condition of R = 6.0 and CNaOH = 2.5 mol/L, the X-ray diffraction patterns of magnetic parti- cles all display sharp peak with strong diffraction inten- sity, and low background, the indices (220) and (400) labeled by red color are found to be corresponding to 2θ values of 31.4 and 45.4 respectively, in a good agree- ment with the JCPDS card 26 - 1136, which belongs to cubic system. However, the indices (220) and (400) la- 20 40 60 80 0 2000 4000 6000 8000 JCPDS:26-1136 220 400 642 620 440 511 622 Intensity (a.u.) 2-Theta (degree) 311 422 400 220 JCPDS:26-1136 Figure 1. X-ray diffraction pattern of the nano magnetite. beled by blue color and the indices (311), (422), (511), (440), (620), (622) and (642) are corresponding to the standard diffraction card JCPDS 19-629(pure Fe3O4). It is demonstrated that the obtained products are mixture of two kinds of cubic nano-Fe3O4 particles, with corre- sponding JCPDS card being 19 - 629 and 26 - 1136 re- spectively. The TEM images shown in Figure 2 reveals the nano- crystallite nature of magnetite particles with a good dis- persibility, the average particle size measured from the TEM images has been found to be 22 nm. 3.1. Effect of Different NaOH Concentration on Magnetite Morphology In the preparation, the alkali concentration and water- core of microemulsion are two chief factors that affect dispersibility of Fe3O4 particles. In order to discuss the effects of alkali concentration on the dispersibility and shape of Fe3O4 particles, we performed five experiments under alkaline condition at concentration of 1.5, 2.0, 2.5, 3.0 and 3.5 mol·L–1 respectively. The dispersibility of prepared magnetite was investigated by transmission electron microscope, five TEM images of magnetite are shown in Figure 3. It is found when the concentration of NaOH is lower than 2.0 mol·L–1 the agglomerated granules appear in the prepared sample, and the spindle-like particles are formed. The image recorded in Figure 3(a) exhibits rod-like form with spindle-like form crossing over. It is because the precipitate of Fe3+ occurs at pH in the range of 3 - 4, which is calculated by Kθsp, Fe(OH)3, but precipi- tate of Fe2+ occurs at pH in the range of 8 - 9 calculated by Kθsp, Fe(OH)2, indicating the amount of NaOH for pre- cipitate of Fe3+ is much less than that for precipitate of Fe2+. So when the shortfall amount of NaOH is intro- Figure 2. TEM morphology of the nano magnetite. Copyright © 2011 SciRes. MSA 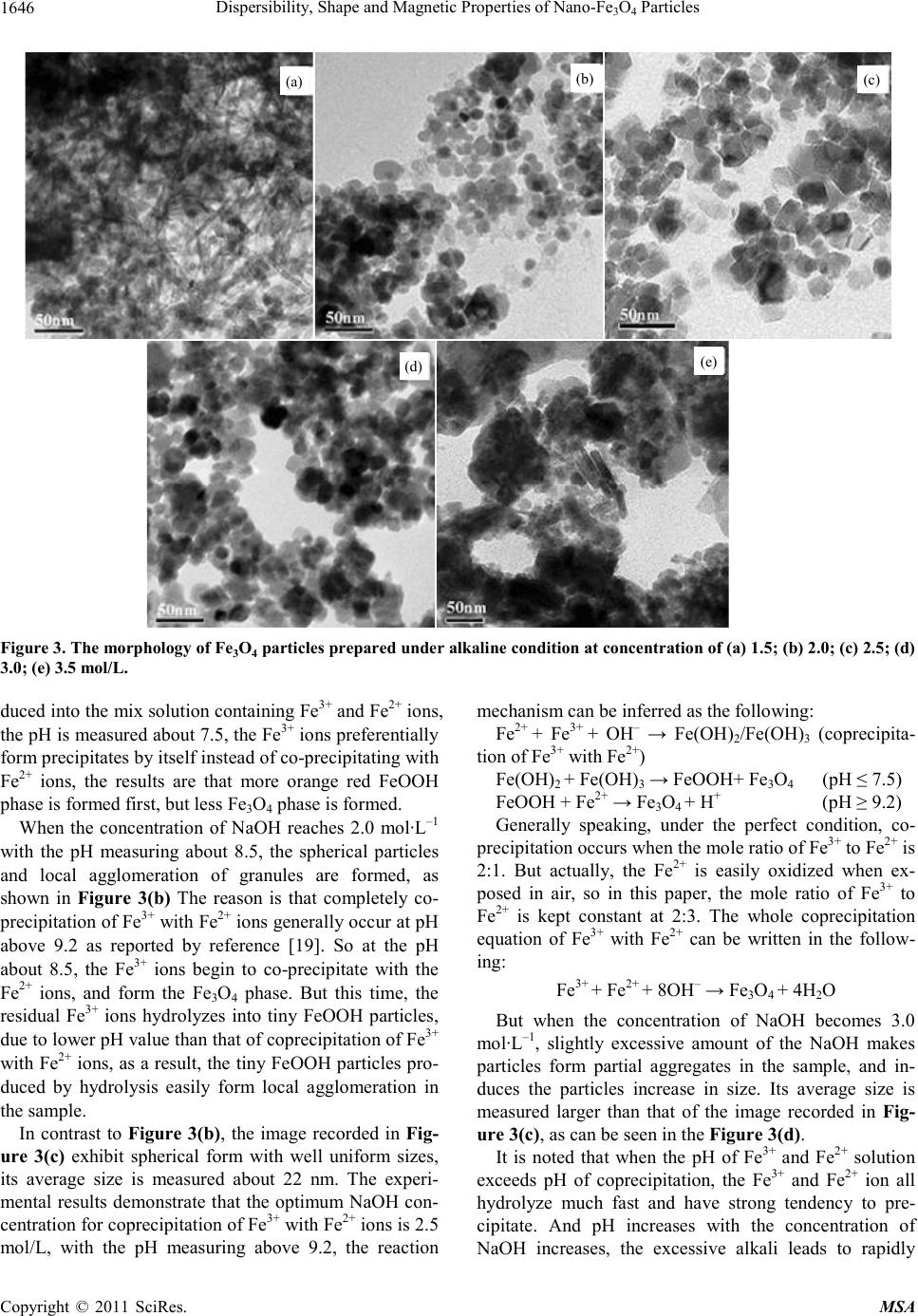 Dispersibility, Shape and Magnetic Properties of Nano-FeO Particles 1646 3 4 (a) (b) (c) (d) (e) Figure 3. The morphology of Fe3O4 parti cl es prepar e d un de r al kal i ne c on di ti o n at co nc e ntr at io n of (a) 1 .5 ; (b) 2 .0 ; (c ) 2. 5 ; (d) 3.0; (e) 3.5 mol/L. duced into the mix solution containing Fe3+ and Fe2+ ions, the pH is measured about 7.5, the Fe3+ ions preferentially form precipitates by itself instead of co-precipitating with Fe2+ ions, the results are that more orange red FeOOH phase is formed first, but less Fe3O4 phase is formed. When the concentration of NaOH reaches 2.0 mol·L–1 with the pH measuring about 8.5, the spherical particles and local agglomeration of granules are formed, as shown in Figure 3(b) The reason is that completely co- precipitation of Fe3+ with Fe2+ ions generally occur at pH above 9.2 as reported by reference [19]. So at the pH about 8.5, the Fe3+ ions begin to co-precipitate with the Fe2+ ions, and form the Fe3O4 phase. But this time, the residual Fe3+ ions hydrolyzes into tiny FeOOH particles, due to lower pH value than that of coprecipitation of Fe3+ with Fe2+ ions, as a result, the tiny FeOOH particles pro- duced by hydrolysis easily form local agglomeration in the sample. In contrast to Figure 3(b), the image recorded in Fig- ure 3(c) exhibit spherical form with well uniform sizes, its average size is measured about 22 nm. The experi- mental results demonstrate that the optimum NaOH con- centration for coprecipitation of Fe3+ with Fe2+ ions is 2.5 mol/L, with the pH measuring above 9.2, the reaction mechanism can be inferred as the following: Fe2+ + Fe3+ + OH– → Fe(OH)2/Fe(OH)3 (coprecipita- tion of Fe3+ with Fe2+) Fe(OH)2 + Fe(OH)3 → FeOOH+ Fe3O4 (pH ≤ 7.5) FeOOH + Fe2+ → Fe3O4 + H+ (pH ≥ 9.2) Generally speaking, under the perfect condition, co- precipitation occurs when the mole ratio of Fe3+ to Fe2+ is 2:1. But actually, the Fe2+ is easily oxidized when ex- posed in air, so in this paper, the mole ratio of Fe3+ to Fe2+ is kept constant at 2:3. The whole coprecipitation equation of Fe3+ with Fe2+ can be written in the follow- ing: Fe3+ + Fe2+ + 8OH– → Fe3O4 + 4H2O But when the concentration of NaOH becomes 3.0 mol·L–1, slightly excessive amount of the NaOH makes particles form partial aggregates in the sample, and in- duces the particles increase in size. Its average size is measured larger than that of the image recorded in Fig- ure 3(c), as can be seen in the Figure 3(d). It is noted that when the pH of Fe3+ and Fe2+ solution exceeds pH of coprecipitation, the Fe3+ and Fe2+ ion all hydrolyze much fast and have strong tendency to pre- cipitate. And pH increases with the concentration of NaOH increases, the excessive alkali leads to rapidly Copyright © 2011 SciRes. MSA 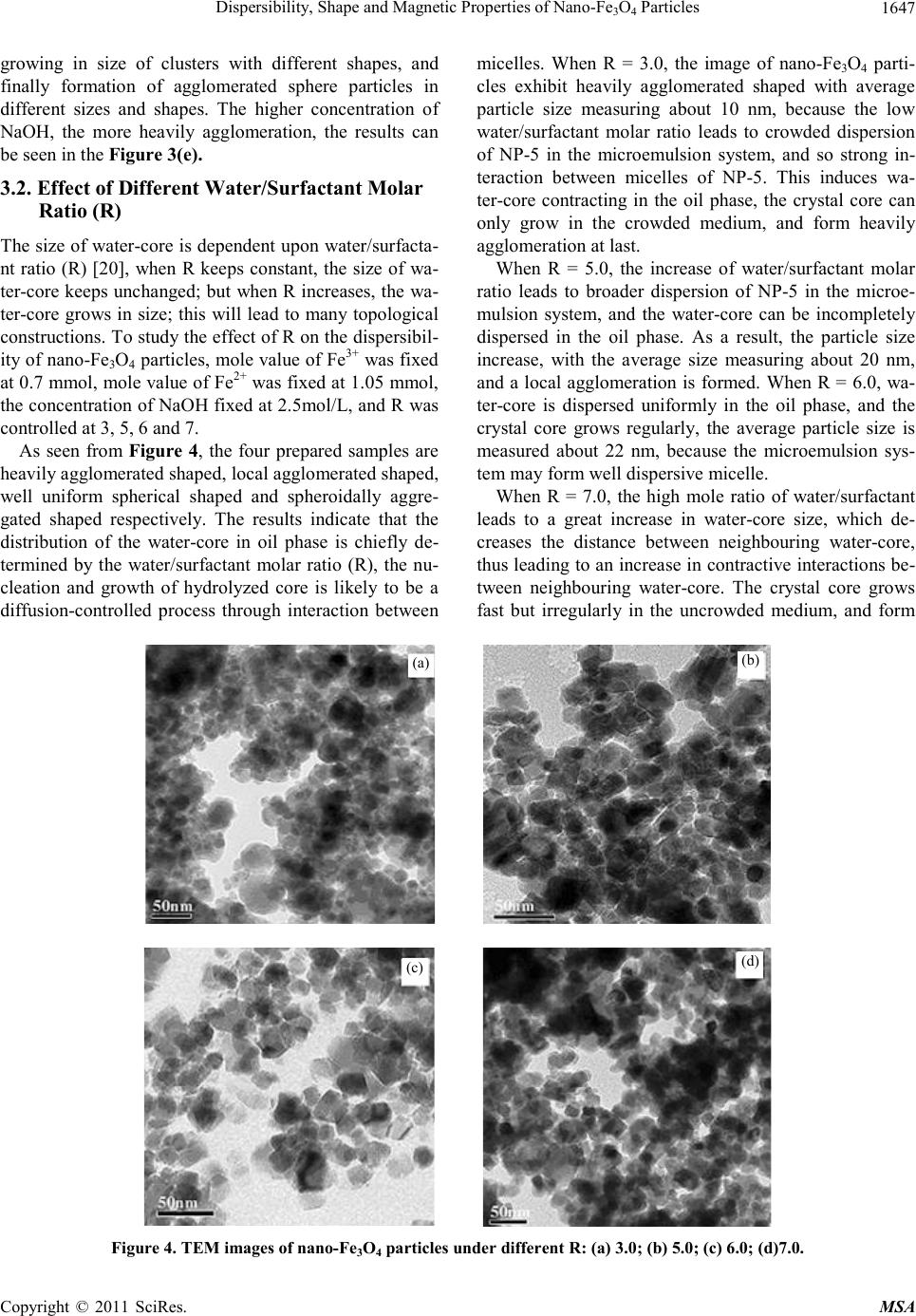 Dispersibility, Shape and Magnetic Properties of Nano-FeO Particles1647 3 4 growing in size of clusters with different shapes, and finally formation of agglomerated sphere particles in different sizes and shapes. The higher concentration of NaOH, the more heavily agglomeration, the results can be seen in the Figure 3(e). 3.2. Effect of Different Water/Surfactant Molar Ratio ( R ) The size of water-core is dependent upon water/surfacta- nt ratio (R) [20], when R keeps constant, the size of wa- ter-core keeps unchanged; but when R increases, the wa- ter-core grows in size; this will lead to many topological constructions. To study the effect of R on the dispersibil- ity of nano-Fe3O4 particles, mole value of Fe3+ was fixed at 0.7 mmol, mole value of Fe2+ was fixed at 1.05 mmol, the concentration of NaOH fixed at 2.5mol/L, and R was controlled at 3, 5, 6 and 7. As seen from Figure 4, the four prepared samples are heavily agglomerated shaped, local agglomerated shaped, well uniform spherical shaped and spheroidally aggre- gated shaped respectively. The results indicate that the distribution of the water-core in oil phase is chiefly de- termined by the water/surfactant molar ratio (R), the nu- cleation and growth of hydrolyzed core is likely to be a diffusion-controlled process through interaction between micelles. When R = 3.0, the image of nano-Fe3O4 parti- cles exhibit heavily agglomerated shaped with average particle size measuring about 10 nm, because the low water/surfactant molar ratio leads to crowded dispersion of NP-5 in the microemulsion system, and so strong in- teraction between micelles of NP-5. This induces wa- ter-core contracting in the oil phase, the crystal core can only grow in the crowded medium, and form heavily agglomeration at last. When R = 5.0, the increase of water/surfactant molar ratio leads to broader dispersion of NP-5 in the microe- mulsion system, and the water-core can be incompletely dispersed in the oil phase. As a result, the particle size increase, with the average size measuring about 20 nm, and a local agglomeration is formed. When R = 6.0, wa- ter-core is dispersed uniformly in the oil phase, and the crystal core grows regularly, the average particle size is measured about 22 nm, because the microemulsion sys- tem may form well dispersive micelle. When R = 7.0, the high mole ratio of water/surfactant leads to a great increase in water-core size, which de- creases the distance between neighbouring water-core, thus leading to an increase in contractive interactions be- tween neighbouring water-core. The crystal core grows fast but irregularly in the uncrowded medium, and form (a) (b) (c) (d) Figure 4. TEM images of nano-Fe3O4 particles under different R: (a) 3.0; (b) 5.0; (c) 6.0; (d)7.0. Copyright © 2011 SciRes. MSA 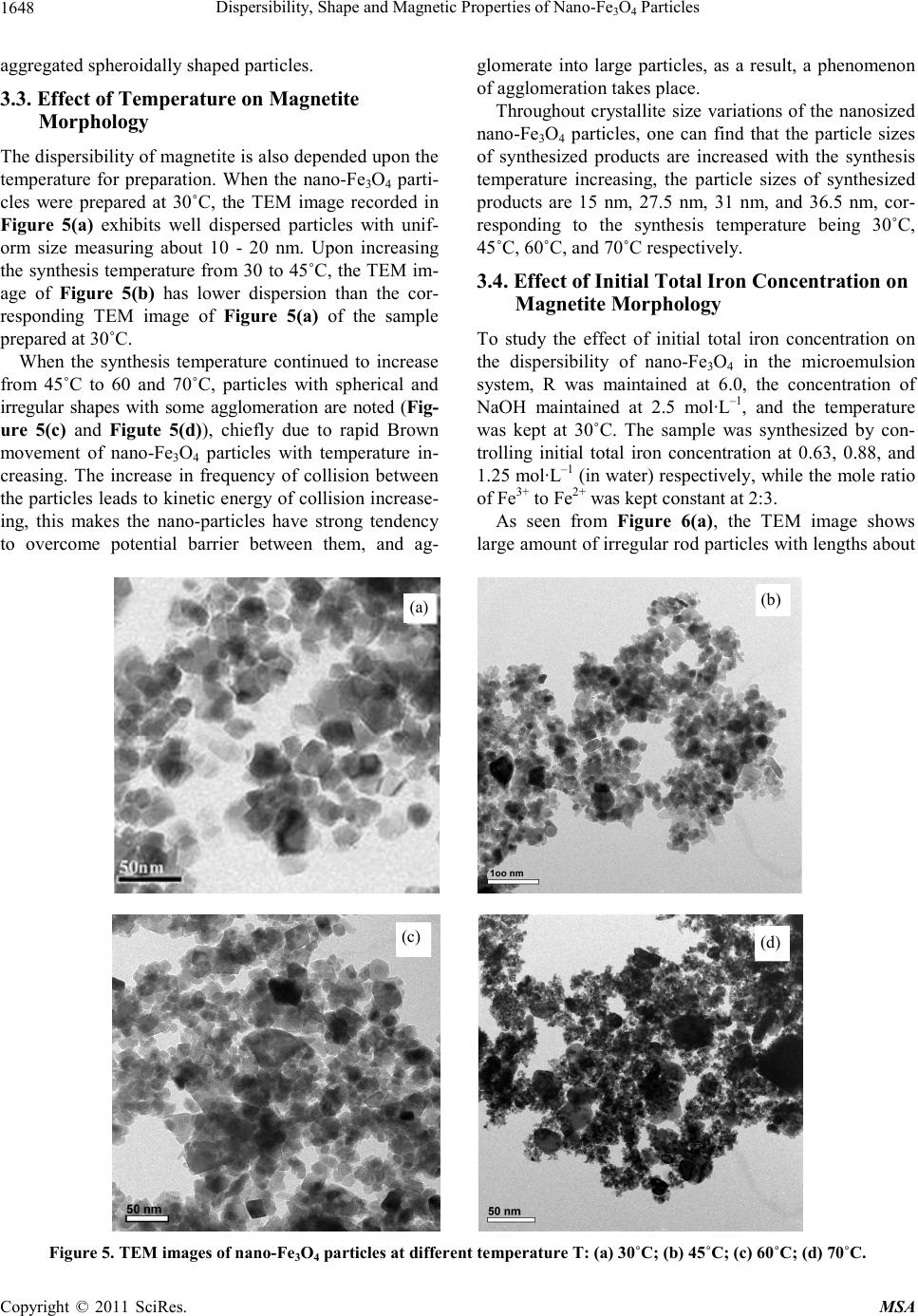 Dispersibility, Shape and Magnetic Properties of Nano-FeO Particles 1648 3 4 aggregated spheroidally shaped particles. 3.3. Effect of Temperature on Magnetite Morphology The dispersibility of magnetite is also depended upon the temperature for preparation. When the nano-Fe3O4 parti- cles were prepared at 30˚C, the TEM image recorded in Figure 5(a) exhibits well dispersed particles with unif- orm size measuring about 10 - 20 nm. Upon increasing the synthesis temperature from 30 to 45˚C, the TEM im- age of Figure 5(b) has lower dispersion than the cor- responding TEM image of Figure 5(a) of the sample prepared at 30˚C. When the synthesis temperature continued to increase from 45˚C to 60 and 70˚C, particles with spherical and irregular shapes with some agglomeration are noted (Fig- ure 5(c) and Figute 5(d)), chiefly due to rapid Brown movement of nano-Fe3O4 particles with temperature in- creasing. The increase in frequency of collision between the particles leads to kinetic energy of collision increase- ing, this makes the nano-particles have strong tendency to overcome potential barrier between them, and ag- glomerate into large particles, as a result, a phenomenon of agglomeration takes place. Throughout crystallite size variations of the nanosized nano -Fe 3O4 particles, one can find that the particle sizes of synthesized products are increased with the synthesis temperature increasing, the particle sizes of synthesized products are 15 nm, 27.5 nm, 31 nm, and 36.5 nm, cor- responding to the synthesis temperature being 30˚C, 45˚C, 60˚C, and 70˚C respectively. 3.4. Effect of Initial Total Iron Concentration on Magnetite Morphology To study the effect of initial total iron concentration on the dispersibility of nano-Fe3O4 in the microemulsion system, R was maintained at 6.0, the concentration of NaOH maintained at 2.5 mol·L–1, and the temperature was kept at 30˚C. The sample was synthesized by con- trolling initial total iron concentration at 0.63, 0.88, and 1.25 mol·L–1 (in water) respectively, while the mole ratio of Fe3+ to Fe2+ was kept constant at 2:3. As seen from Figure 6(a), the TEM image shows large amount of irregular rod particles with lengths about (a) (b) (c) (d) Figure 5. TEM images of nano-Fe3O4 particles at different temperature T: (a) 30˚C; (b) 45˚C; (c) 60˚C; (d) 70˚C. Copyright © 2011 SciRes. MSA 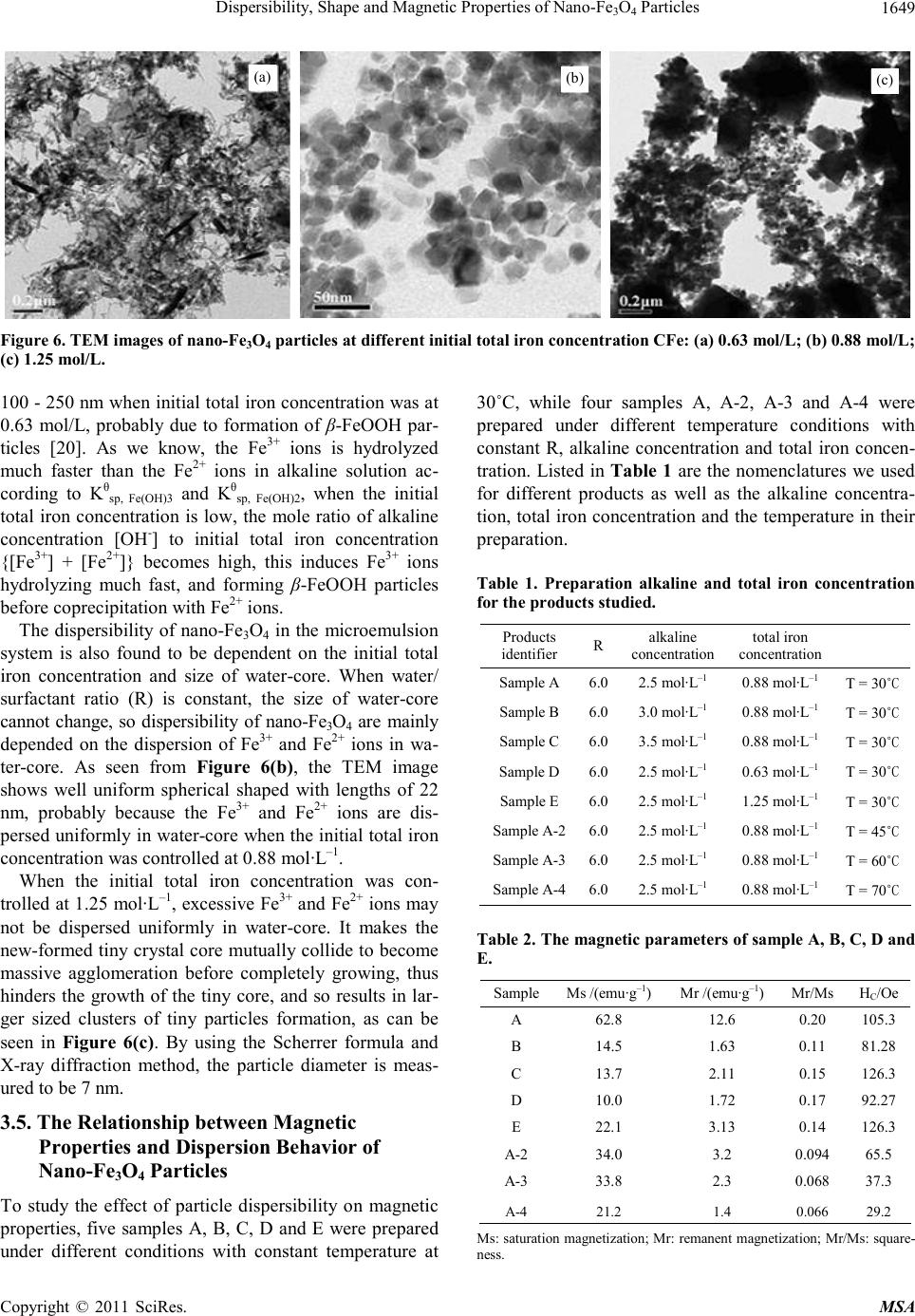 Dispersibility, Shape and Magnetic Properties of Nano-FeO Particles1649 3 4 (a) (b) (c) Figure 6. TEM images of nano-Fe3O4 pticles at different initial total iron concenation CFe: (a) 0.63 mol/L; (b) 0.88 mol/L 00 - 250 nm when initial total iron concentration was at e microemulsion sy as con- tro onship between Magnetic f To sdispersibility on magnetic 30˚C, while four samples A, A-2, A-3 and A-4 were aration alkaline and total iron concentration r the prod uc ts studied. ntration concentration artr; (c) 1.25 mol/L. 1 0.63 mol/L, probably due to formation of β-FeOOH par- ticles [20]. As we know, the Fe3+ ions is hydrolyzed much faster than the Fe2+ ions in alkaline solution ac- cording to Kθsp, Fe(OH)3 and Kθsp, Fe(OH)2, when the initial total iron concentration is low, the mole ratio of alkaline concentration [OH-] to initial total iron concentration {[Fe3+] + [Fe2+]} becomes high, this induces Fe3+ ions hydrolyzing much fast, and forming β-FeOOH particles before coprecipitation with Fe2+ ions. The dispersibility of nano-Fe3O4 in th stem is also found to be dependent on the initial total iron concentration and size of water-core. When water/ surfactant ratio (R) is constant, the size of water-core cannot change, so dispersibility of nano-Fe3O4 are mainly depended on the dispersion of Fe3+ and Fe2+ ions in wa- ter-core. As seen from Figure 6(b), the TEM image shows well uniform spherical shaped with lengths of 22 nm, probably because the Fe3+ and Fe2+ ions are dis- persed uniformly in water-core when the initial total iron concentration was controlled at 0.88 mol·L–1. When the initial total iron concentration w lled at 1.25 mol·L–1, excessive Fe3+ and Fe2+ ions may not be dispersed uniformly in water-core. It makes the new-formed tiny crystal core mutually collide to become massive agglomeration before completely growing, thus hinders the growth of the tiny core, and so results in lar- ger sized clusters of tiny particles formation, as can be seen in Figure 6(c). By using the Scherrer formula and X-ray diffraction method, the particle diameter is meas- ured to be 7 nm. 3.5. The Relati Properties and Dispersion Behavior o Nano-Fe3O4 Particl es tudy the effect of particle properties, five samples A, B, C, D and E were prepared under different conditions with constant temperature at prepared under different temperature conditions with constant R, alkaline concentration and total iron concen- tration. Listed in Table 1 are the nomenclatures we used for different products as well as the alkaline concentra- tion, total iron concentration and the temperature in their preparation. Table 1. Prep fo Products identifier R alkaline conce total iron Sample A 6.02.5 mol·L–1 0.88 mol·L–1 T =˚C 30 Sample B 6.03.0 mol·L 0. –1 88 mol·L –1 T = 30˚C –1–1 C –1–1 C –1 –1 S–1–1 C –1–1 C –1–1 C Sample C 6.03.5 mol·L 0.88 mol·L T = 30˚ Sample D 6.02.5 mol·L 0.63 mol·L T = 30˚ Sample E 6.02.5 mol·L 1.25 mol·L T = 30˚C ample A-26.02.5 mol·L 0.88 mol·L T = 45˚ Sample A-36.02.5 mol·L 0.88 mol·L T = 60˚ Sample A-46.02.5 mol·L 0.88 mol·L T = 70˚ T metis od . C able 2. Theagnc parameterf sample A, B, C, D an E SampleMs /(emu·g–1)Mr /(emu·g –1) Mr/MsH/Oe A 62.8 12.6 0.20 105.3 B 14.5 1.63 0.11 81.28 C 13.7 2.11 0.15 126.3 D 10.0 1.72 0.17 92.27 E 22.1 3.13 0.14 126.3 A-2 34.0 3.2 0.094 65.5 A0.-3 33.8 2.3 068 37.3 A-4 21.2 1.4 0.066 29.2 Ms: saation magnion; Mr: remanemagnetizar/Ms:re- ness. turetizatnt tion; M squa Copyright © 2011 SciRes. MSA  Dispersibility, Shape and Magnetic Properties of Nano-FeO Particles 1650 3 4 Tdispersi of nanopcles wsery ansmission electron microscope operated at 120KV, as mperature. The magnetic curves of th etization curves of sam- pl igure 7 display magnetic pr li induces the Fe and Fe ions all hydro- ly th increase of total iro ngest sa properties and their shapes. The better dis- pe and corresponding sters. Covercivity which depends on m he bilityartias obved b transmission electron microscopy (TEM) with a Tecnai- 12 tr can be seen in the Figures 3(c)-(e), Figures 5(a)-(d) and Figures 6(a)-(c). It was shown that cluster size and cluster distribution can be influenced in a wide range (some order of magnitude) by the variation of the prepa- ration parameters. Magnetic studies were carried out using a vibrating sample magnetometer (VSM BHS-55) with fields up to 30 k Gauss at room te e eight samples can be seen in Figure 7, all magnetic parameters are listed in Table 2. Magnetic properties such as saturation magnetization, coercivity and square- ness are discussed as following. 1) Saturation magnetization Saturation magnetization represents magnetic intensity of magnetic materials. The magn e A, B and C shown in the F operties of nano-Fe3O4 particles prepared with differ- ent alkaline concentration of 2.5, 3.0 and 3.5 mol·L–1 respectively. Whereas the magnetization curves of sam- ple A, D and E shown in the Figure 7 display magnetic properties of nano-Fe3O4 particles prepared with differ- ent initial total iron concentration at 0.88, 0.63 and 1.25 mol·L–1 respectively. The magnetization curves of sam- ple A, A-2, A-3 and A-4 shown in the Figure 7 display magnetic properties of nano-Fe3O4 particles prepared with different temperature at 30˚C, 45˚C, 60˚C and 70˚C respectively. By comparing magnetic parameters of sam- ples listed in Table 2 with their dispersibility shown in Figures 3(c)-(e), we obtain that sample A shows the strongest saturation magnetization, chiefly due to its uniform size and best dispersion behavior. It is under alkaline condition of concentration at 2.5 mol·L–1, that coprecipitation of Fe3+ with Fe2+ ions can occur. The saturation magnetization decreases to some extent with gradual increase of NaOH concentration from 2.5 mol·L–1 to 3.5 mol·L–1. Because the nano-Fe3O4 particles change from well-dispersed into partial aggregate, and finally heavily agglomerate, with an increase of alkaline con- centration. The better dispersibility of nanoparticles can be ex- plained by the size of clusters with uniform shape. Ex- cessive alka3+ 2+ zing much fast, leads to nanoparticles rapidly changing into larger sized clusters with arbitrary smaller shaped particles. The better dispersibility of nanoparticles, the stronger magnetic behavior of them [21]. This means inner magnetic vector becomes more ordered, with an in- crease of dispersion of nanoparticles, and thus the nano- particles display stronger magnetism, leading to an in- crease of saturation magnetizationσs. When different initial total iron concentration was adopted during preparation, the saturation magnetization of particles is found to be stronger wi n concentration. For sample D, the total iron concen- tration is so low that Fe3+ ions hydrolyzed faster than Fe2+ ions and formed β-FeOOH particles before copre- cipitation with Fe2+ ions. This would lead to decrease of its saturation magnetization. In the opposite side, when the total iron concentration was higher than 0.88 mol·L–1, coprecipitation of Fe3+ with Fe2+ ions occurs, this pre- vents β-FeOOH particles from generating, causing an increase of saturation magnetization σs of sample E. But mutually collision between new-formed tiny crystal cores hinders the growth of the tiny core, leading to formation of larger sized clusters with different tiny shapes. So dispersibility of nanoparticles of sample E is worse than that of sample A, inner magnetic vector becomes less ordered also, as a result, the saturation magnetization σs of sample E becomes lower than that of sample A. By comparing magnetic parameters of samples listed in Table 2 with their dispersibility shown in Figures 5 (a)-(d), we obtain that sample A shows the stro turation magnetization, also chiefly due to its uniform size and best dispersion behavior. When the nano-Fe3O4 particles were prepared at 30˚C, the sample A was ob- served to have the best dispersibility, when the tempera- ture increased from 30˚C to 70˚C, the dispersibility of the sample A-2, sample A-3 and ample A-4 decreased stead- ily, resulting in inner magnetic vector getting less or- dered [21], and thus a progressive decrease in the satura- tion magnetization σs for sample A-2, sample A-3 and sample A-4. The above results demonstrate that saturation mag- netization of nano-Fe3O4 particles is greatly influenced by dispersion rsed nano-Fe3O4 particles with more uniform and dis- persed shapes will have higher saturation magnetization, and thus display stronger magnetism. 2) Coercivity Coercivity of magnetic matericals mainly depends on anisotropy, saturation magnetization ructure paramet agneto-crystalline anisotropy can be expressed by the following formula according to Zhou.[22]: 0 2 cK Ms In this formula, K value depends only on property of material itself, but not related to particle size and shape, it is inversely proportional to saturation magnetization. As seen from Table 2, coercivity of sample A is higher than that of sample B and D, but lower than that of sam- ple C and sample E. Copyright © 2011 SciRes. MSA 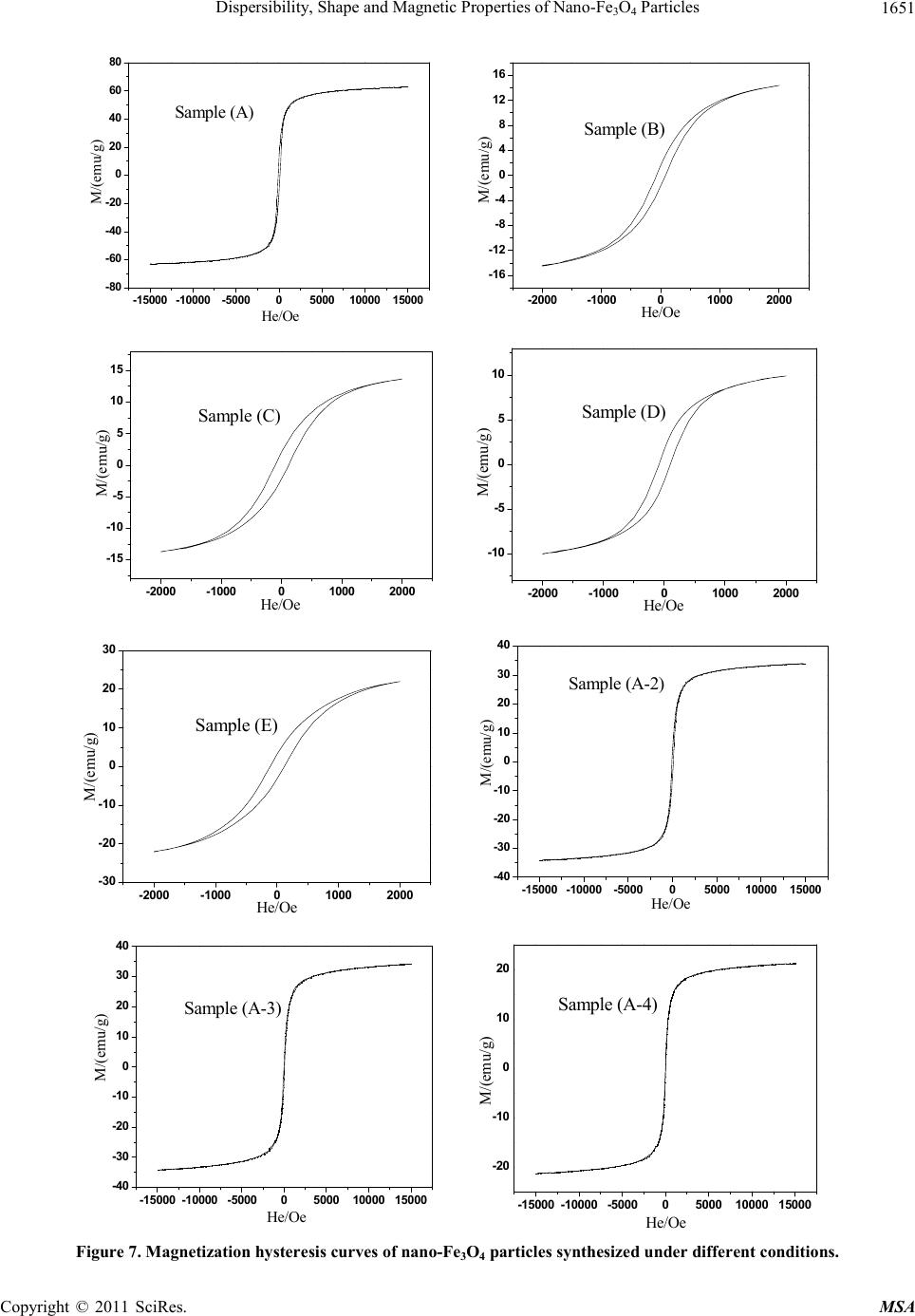 Dispersibility, Shape and Magnetic Properties of Nano-Fe3O4 Particles Copyright © 2011 SciRes. MSA 1651 60 80 -15000 -10000-5000050001000015000 -80 -60 -40 -20 0 20 40 M/(emu/g) He/Oe Sample (A) 16 -2000 -1000010002000 -16 -12 -8 -4 0 4 8 12 Sample (B) M/(emu g) He/Oe -2000 -1000010002000 -10 -5 0 5 10 He/Oe M/(emu/g) Sample (D) -2000 -1000010002000 -15 -10 -5 0 5 10 15 Sample (C) M/(emu/g) He/Oe -15000 -10000-5000050001000015000 -40 -30 -20 -10 0 10 20 30 40 He/Oe M/(emu/g) Sample (A-2) -2000 -1000010002000 -30 -20 -10 0 10 20 30 Sample (E) M/(emu/g) He/ Oe -15000 -10000-5000050001000015000 -40 -30 -20 -10 0 10 20 30 40 Sample (A-3) M (emu/g) He/Oe -15000 -10000-5000050001000015000 -20 -10 0 10 20 Sample (A-4) He/Oe M/(emu/g) Figure 7. Magnetization hysteresis curves of nano-Fe3O4 particles synthesized under differe nt conditions.  Dispersibility, Shape and Magnetic Properties of Nano-FeO Particles 1652 3 4 According to the formula, sample A should have the lowest coercivity compared with other samples because of its highest saturation magnetization. However, it is found that there is no such obvious linear relationship between coercivity and saturation magnetization. The coercivity of particles is related to their size of clusters and shapes. It is noted that coercivity decreased with reduction of particle size below 40nm [23]. As shown in Figures 3(d)-(e) and Figures 6(a)-(c), sample A shows well uniformly spherical shaped with lengths of 22 nm, but both of samples C and E display large size of clusters, but consisting of smaller size particles than sample A. So both of samples C and E show larger coercivity than mple A a sa l . On the other hand, average size of sample B is rger than that of sample A, and sample D is 100 - 250 nm length irregular rod particles, these all lead to sample A having a larger coercivity than the sample B and D. As shown in Figures 5(a)-(d), with an increase of temperature from 30˚C to 70˚C, the dispersibility of the sample decreased progressively, resulting in the coer- civeity of sample decreasing. A plot of coercivity versus the temperature is shown in Figure 8, the Figure 8 shows linear relationship between coercivity and tem- perature. It demonstrates that the temperature has signi- ficant influence on the anisotropy and dispersibility of the sample, with an increase of temperature, the anisot- ropy and dispersibility of the sample decreases, leading to coercivity decreasing. 3.6. Ind e xing t he Powder X - R ay Di f f r acti o n Pattern The results of indexing the powder X-ray diffraction pat- tern are listed in Table 3 . Table 3 shows that all the dif- fraction peaks in the pattern can be readily indexed by one set of lattice parameters. The largest relative devia- tion between the calculated Dcal and experimental Dexp is less than 0.347%, which indicates that the synthesized products are a single phase with cubic structure. The 100 120 30 40 50 60 70 20 40 60 80 Coer Temperature oC civity Oe Temperature ˚C Coerci vi Figure 8. Plot of coercivity versus the temperature. Table 3. The experimental data and the calculated results for powder X-ray diffraction pattern of the nano-Fe3O4 particles cubic system: a = 8.273 Å. Dexp (Å) Dcal (Å)h k lI(%)Dexp (Å) Dcal (Å) (nm)h k lI(%) 2.84302.92492201001.4846 1.4625 4 4 01.89 2.52662.49443113.711.4113 1.3081 6 2 04.33 1.99772.06824 0018.911.2622 1.2472 6 2 23.08 1.70081.68874 2 21.271.1522 1.1055 6 4 21.89 1.62871.59215 1 13.71 ty O e crystal structure of synthesized product belong cubic system. s to the 4. Conclusions In summary, the cubic nano-Fe3O4 particles have been successfully synthesized by using two-step microemul- sion method. The dispersibility, shape and anisotropy of nano -Fe 3O4 particles vary with R value, alkali concentra- tion, and the temperature. The prepared nano-Fe3O4 par- ticles all exhibits dispersive spherical form with well uniform sizes measured about 22 nm under condition of R = 6.0, alkali concentration at 2.5 mol/L, with the tem- perature at 30˚C. Magnetic properties are influenced greatly by dispersibility and shape of particles. The crys- tal structure of the nano-Fe3O4 particles belongs to cic syst ence Foundation of Ch- ina (No: 20971043), and the financial support from Wen- z e) RE S [1 aMtic Gu: s m Pg r. [2 K., “Preparation Thermosensi- etheres and their Alication to Bio- processes,” Colloids and Surfaces A: Physicochemical ub em with lattice parameters (a = 8.273 Å). 5. Acknowledgements We thank the National Natural Sci hou scince and technology project (G2010071. REFENCE ] V. E and . Fertm Applic n, “ ation agne s,” He Fluids isphere ide-Book ublishin Prop Co., ertie New Yok, 1990 ] A. tiv ondo a Magne nd H ic Mi Fukuda crosp of pp and Engineering, Vol. 153, 1999, pp. 435-438. doi:10.1016/S0927-7757(98)00465-8 [3] D. G. Shchukin, “Micron-Scale I. L. Radtchenko and G. B. Sukhorukov, Hollow Polyelectrolyte Capsules with Nano-Sized Magnetic Fe3O4 Inside,” Materials Letters, Vol. 57, 2003, pp. 1743-1747. doi:10.1016/S0167-577X(02)01061-3 [4] B. Gržeta, M. Ristić, I. Nowik and S. Musić, “Formation of Nanocrystalline Magnetite by Thermal Decomposition of Iron Choline Citrate,” Journal of Alloys and Com- pounds, Vol. 334, 2002, pp. 304-312. doi:10.1016/S0925-8388(01)01792-3 Copyright © 2011 SciRes. MSA  Dispersibility, Shape and Magnetic Properties of Nano-FeO Particles1653 3 4 [5] T. J. Daou, G. Pourroy, S. Bégin-Colin, J. M. Grenèche, C. Ulhaq-Bouillet, P. Legaré, P. Bernhardt, C. Leuvrey and G. Rogez, “Hydrothermal Synthesis of Monodisperse Magnetite Nanoparticles,” Chemistry Materials, Vol. 18, 2006, pp. 4399-4404. doi:10.1021/cm060805r [6] T. Sugimoto and E. Matijevic, “Formation of Uniform Spherical Magnetite Particles by Crystallization from Fer- nal of Colloid and Interface . 227-243. rous Hydroxide Gels,” Jour Science, Vol. 74, 1980, pp do i :10.10 16/0021-9797(80) 90187-3 [7] Y. S. Kang, S. Risbud, J. F. Rabolt and P. Stroeve, “Syn- thesis and Characterization of Nanometer-Size Fe3O4 and γ-Fe2O3 Particles,” Chemistry Materials, Vol. 8, 1996, pp. 2209-2211. do i:10.1021/cm960157j [8] T. Fried, G. Shemer, G. Markovich, “Ordered Two-Di- mensional Arrays of Ferrite Nanoparticles,” Advanced Materials, Vol. 13, 2001, pp. 1158-1161. doi:10.1002/1521-4095(200108)13:15<1158::AID-ADM A1158>3.0.CO;2-6 [9] G. Visalakshi, G. Venkataswaran, S. K. Kulshreshtha and P. N. Moorthy, “Compositional Characteristics of Mag- netite Synthesised from Aqueous Solutions at T tures Upto 523K,” Materials Research em Bulletin, Vo pera- l. 28, 1993, pp. 829-836. doi:10.1016/0025-5408(93)90024-8 [10] S. Wang, H. Xin and Y. Qian, “Preparation of Nanocrys- talline Fe3O4 by γ-ray Radiation,” Materials Letters, Vol. 33, 1997, pp. 113-116. doi:10.1016/S0167-577X(97)00077-3 [11] D. Vollath and D. V. Szabo, “Synthesis and Magnetic Properties of Nanostructured Maghemite,” Journal of Materials Research, Vol. 12, 1997, pp. 2175-2182. doi:10.1557/JMR.1997.0291 [12] C. Feldmann and H. O. Jungk, “Polyol-Mediated Prepara- tion of Nanoscale Oxide Particles,” Angewandte Chemie International Edition, Vol. 40, 2001, pp. 359-362. doi:10.1002/1521-3773(20010119)40:2<359::AID-ANIE 359>3.3.CO;2-2 [13] S. Sun and H. J. Zeng, “Size-Controlled Synthesis of Magnetite Nanoparticles,” Journal of the American Chemical Society, Vol. 124, 2002, pp. 8204-8205. do i :10.10 21/ja026501 x [14] N. Pinna, S. Grancharov, P. Beato and P. Bonville, “Mag- 021/cm050060+ netite Nanocrystals: Nonaqueous Synthesis, Characteriza- tion, and Solubility,” Chemistry Materials, Vol. 17, 2005, pp. 3044-3049. doi:10.1 ne-Pot Reaction to [15] Z. Li, H. Chen, H. Bao and M. Gao, “O Synthesize Water-Soluble Magnetite Nanocrystals,” Chemistry Materials, Vol. 16, 2004, p. 1391. doi:10.1021/cm035346y [16] Y. H. Zhu and Q. F. Wu, “Synthesis of Magnetite Nano- particles by Precipitation with Forced Mixing,” Journal of Nanoparticle Research, Vol. 1, 1999, pp. 393-396. doi:10.1023/A:1010091625981 [17] Z. L. Liu and X. Wang, “Synthesis of Magnetite Na- noparticles in W/O Microemulsion,” Journal of Materials Science, Vol. 39, 2004, pp. 2633-2636. doi:10.1023/B:JMSC.0000020046.68106.22 [18] M. D. Alcalá, J. M. Criado and C. Real, “Synth Nanocrystalline Magnetite esis of by Mechanical Alloying of Iron and Hematite,” Journal of Materials Science, Vol. 39, 2004, pp. 2365-2370. doi:10.1023/B:JMSC.0000019998.78644.74 [19] L. X. Song, Z. Y. Lu, D. C. Liu, S. B. Cui and X. Q. Xiao, ess, Vol. . 6, “Preparation of Magnetic Nanometer Fe3O4 with the Method of Complex Compound-Hydrolyzation Deposi- tion,” Chemical industry and engineering progr 25, No. 1, 2006, pp. 54-55. [20] Y. X. Yang, X. N. Liu, H. Zhu, Z. Huang, Y. R. Chen and Z. Y. Zhou, “Synthesis of β-FeOOH with Microemul- sion,” Materials Science and Technology, Vol. 23, No 2007, pp. 641-645. doi:10.1179/174328407X179665 [21] X. J. Liang, H. Zhu, Y. X. Yang, X. N. Liu, Y. R. Chen and H. P. Ying, “A Study on Synthesis of γ-Fe2O3 Pow- der and Magnetism Enhancement,” Acta Vol. 64, No. 23, 2006, pp. 2351-2356. Chimaca Sinica, gnetism and 2. [22] Z. G. Zhou, “The Ferrite,” Science Press, Beijing, 1981. [23] A. Manaf, R. A. Buckley, H. A. Davies, M. Leonowicz, “Enhanced Magnetic Properties in Rapidly Solidified Nd-Fe-B Based Alloys,” Journal of Ma Magnetic Materials, Vol. 101, 1991, pp. 360-36 doi:10.1016/ 0304-8853(91)90779-A [24] L. L. Wang, “Study of Magnetic Properties of Ni-Fe-P and Ni-Fe-P-B Chemical Films,” Journal of Materials Review, Vol. 15, No. 3, 2001, pp. 65-67. Copyright © 2011 SciRes. MSA
|