Paper Menu >>
Journal Menu >>
 J. Biomedical Science and Engineering, 2009, 2, 526-531 doi: 10.4236/jbise.2009.27076 Published Online November 2009 (http://www.SciRP.org/journal/jbise/ JBiSE ). Published Online November 2009 in SciRes. http://www.scirp.org/journal/jbise Enzymatic pretreatment and microwave extraction of asiaticoside from Centella asiatica Cheng-Xiao Wang, Wei Han*, Liang Fan, Chun-Li Wang Engineering Center for Traditional Chinese Medicine Modernization, East China University of Science and Technology, Shanghai, China. E-mail: *whan@ecust.edu.cn Received 3 July 2009; revised 13 July 2009; accepted 19 July 2009. ABSTRACT The extraction of asiaticoside from Centella asiatica by enzymatic pretreatment and microwave extraction (EPME) was studied in this article. The effects of several important factors such as temperature of en- zymatic pretreatment, liquid to solid ratio and mi- crowave radiation time were investigated by quadric regression orthogonal design experiment and were analyzed by response surface. An extraction model with well forecast performance was then established. The results indicate that the optimum extraction condition was as follows: liquid to solid ratio was 36mL/g, temperature of enzymatic pretreatment was 45℃, enzymatic time was 30min, and microwave ra- diation time was 110s. On such conditions the yield was 27.10%. Keywords: Asiaticoside; Enzymolysis; Microwave Ex- traction; Centella Asiatica 1. INTRODUCTION Centella asiatica (L.) Urban, a perennial herb belonging to the umbelliferae family, is well known as a traditional Chinese herbal medicine. Its conventional efficacy is clearing away heat and toxic, inducing diuresis and re- ducing edema. The major active constituents of Centella asiatica are asiaticoside and madecassoside. It has been used for the treatment of hot and humid jaundice, trau- matic injuries, infectious hepatitis and dermatosis [1]. Currently, some conventional extraction methods are mostly adopted for the extraction of asiaticoside [2,3,4], such as aqueous extraction and ethanol extraction. In recent years, new kinds of extraction techniques appeared, including enzymolysis and microwave extrac- tion. The former has impressive effects with characteris- tics of high catalytic efficiency, high specificity, mild reactive conditions and preserving the original efficacy of active compounds to the maximum [5]. The later method has many advantages, such as shorter time, less solvent, higher extraction rate and better products with lower cost [6,7]. However, the application of the combi- nation of these two methods on plant materials was rare- ly reported. For the purpose of improving the efficient of asiatico- side extraction, reducing the cost, the EPME method is employed in this article, quadric regression orthogonal design is adopted to investigate the effects of three main extraction parameters including temperature of enzyma- tic pretreatment, liquid to solid ratio and microwave ra- diation time on the yield of asiaticoside, and optimum extraction process is worked out. 2. MATERIALS AND METHODS 2.1. Equipments and Reagents An ER-692 microwave oven (as shown in Figure 1) with a power output of 650W, operating at 2450MHz, was mechanically modified to fit a reflux system that enables extraction performed under atmospheric settings and permits solvent containment. The extraction vessel was a 250-mL three-necked round-bottomed flask con- nected to a water condenser. Thermostat with magnetic stirrer (Model DF-101S, Yuhua Experimental Apparatus Co., Shanghai) was used as enzymolysis device. UV spectrophotometer (Model 1900PC, Yayan Electronic Co., Shanghai) was used for determination. Dry Centella asiatica (fitted for the Chinese Pharma- copoeia); Asiaticoside standard (Provided by National Institute for the Control of Pharmaceutical and Biologi- cal Products, Beijing); Cellulase (≥400U/mg); Anhy- drous ethanol and concentrated sulfuric acid used in the experiment were all of analytical grade. 2.2. Analytic Method of Asiaticoside The concentration of the asiaticoside in this process was determined by ultraviolet spectrophotometry [8] and the result was expressed as extraction yield, i.e. unit extrac- 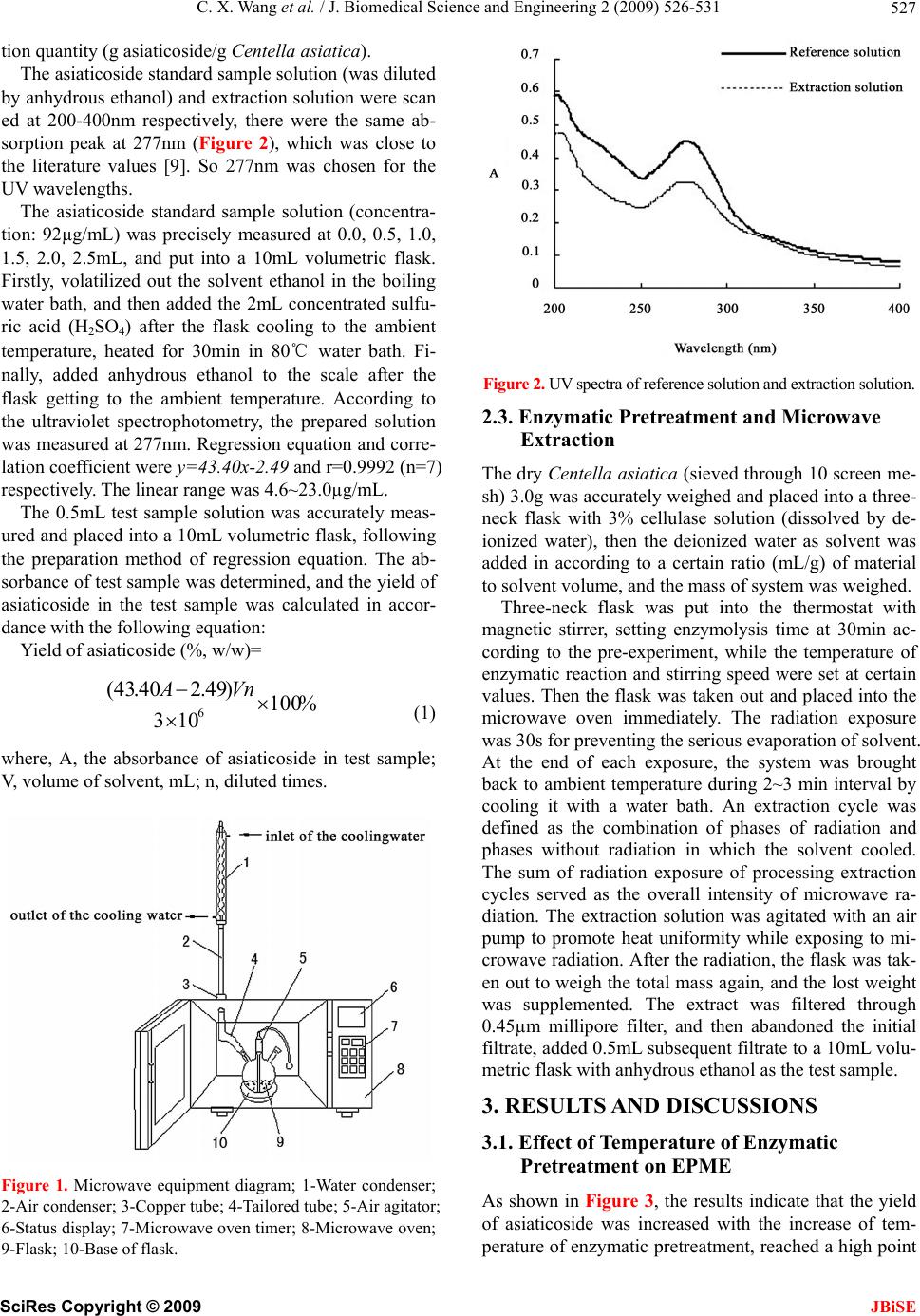 C. X. Wang et al. / J. Biomedical Science and Engineering 2 (2009) 526-531 527 tion quantity (g asiaticoside/g Centella asiatica). The asiaticoside standard sample solution (was diluted by anhydrous ethanol) and extraction solution were scan ed at 200-400nm respectively, there were the same ab- sorption peak at 277nm (Figure 2), which was close to the literature values [9]. So 277nm was chosen for the UV wavelengths. The asiaticoside standard sample solution (concentra- tion: 92µg/mL) was precisely measured at 0.0, 0.5, 1.0, 1.5, 2.0, 2.5mL, and put into a 10mL volumetric flask. Firstly, volatilized out the solvent ethanol in the boiling water bath, and then added the 2mL concentrated sulfu- ric acid (H2SO4) after the flask cooling to the ambient temperature, heated for 30min in 80℃ water bath. Fi- nally, added anhydrous ethanol to the scale after the flask getting to the ambient temperature. According to the ultraviolet spectrophotometry, the prepared solution was measured at 277nm. Regression equation and corre- lation coefficient were y=43.40x-2.49 and r=0.9992 (n=7) respectively. The linear range was 4.6~23.0µg/mL. The 0.5mL test sample solution was accurately meas- ured and placed into a 10mL volumetric flask, following the preparation method of regression equation. The ab- sorbance of test sample was determined, and the yield of asiaticoside in the test sample was calculated in accor- dance with the following equation: Yield of asiaticoside (%, w/w)= %100 103 )49.240.43( 6 VnA (1) where, A, the absorbance of asiaticoside in test sample; V, volume of solvent, mL; n, diluted times. Figure 1. Microwave equipment diagram; 1-Water condenser; 2-Air condenser; 3-Copper tube; 4-Tailored tube; 5-Air agitator; 6-Status display; 7-Microwave oven timer; 8-Microwave oven; 9-Flask; 10-Base of flask. Figur e 2 . UV spectra of reference solution and extraction solution. 2.3. Enzymatic Pretreatment and Microwave Extraction The dry Centella as iatica (sieved through 10 screen me- sh) 3.0g was accurately weighed and placed into a three- neck flask with 3% cellulase solution (dissolved by de- ionized water), then the deionized water as solvent was added in according to a certain ratio (mL/g) of material to solvent volume, and the mass of system was weighed. Three-neck flask was put into the thermostat with magnetic stirrer, setting enzymolysis time at 30min ac- cording to the pre-experiment, while the temperature of enzymatic reaction and stirring speed were set at certain values. Then the flask was taken out and placed into the microwave oven immediately. The radiation exposure was 30s for preventing the serious evaporation of solvent. At the end of each exposure, the system was brought back to ambient temperature during 2~3 min interval by cooling it with a water bath. An extraction cycle was defined as the combination of phases of radiation and phases without radiation in which the solvent cooled. The sum of radiation exposure of processing extraction cycles served as the overall intensity of microwave ra- diation. The extraction solution was agitated with an air pump to promote heat uniformity while exposing to mi- crowave radiation. After the radiation, the flask was tak- en out to weigh the total mass again, and the lost weight was supplemented. The extract was filtered through 0.45µm millipore filter, and then abandoned the initial filtrate, added 0.5mL subsequent filtrate to a 10mL volu- metric flask with anhydrous ethanol as the test sample. 3. RESULTS AND DISCUSSIONS 3.1. Effect of Temperature of Enzymatic Pretreatment on EPME As shown in Figure 3, the results indicate that the yield of asiaticoside was increased with the increase of tem- perature of enzymatic pretreatment, reached a high point SciRes Copyright © 2009 JBiSE 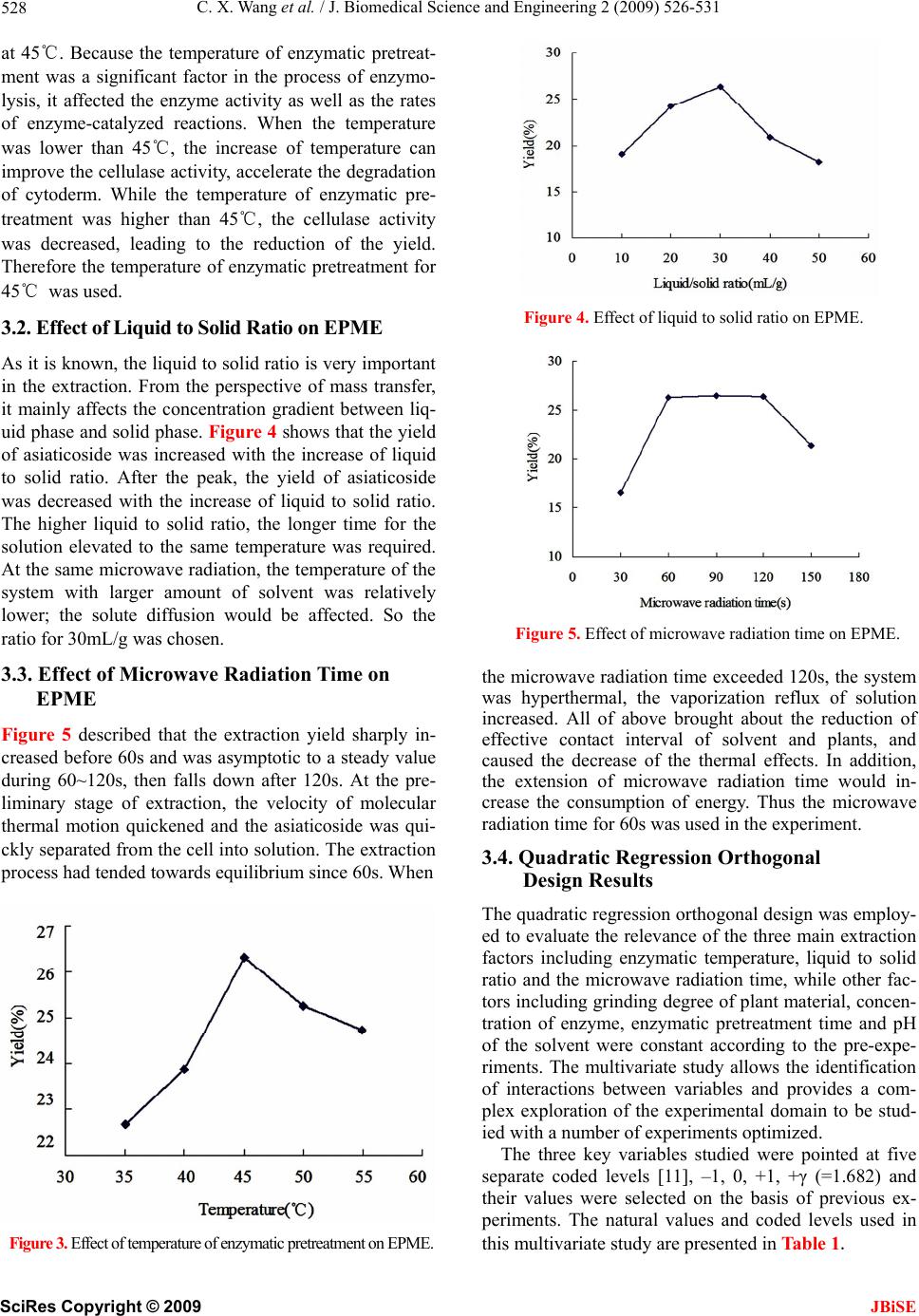 C. X. Wang et al. / J. Biomedical Science and Engineering 2 (2009) 526-531 528 at 45℃. Because the temperature of enzymatic pretreat- ment was a significant factor in the process of enzymo- lysis, it affected the enzyme activity as well as the rates of enzyme-catalyzed reactions. When the temperature was lower than 45℃, the increase of temperature can improve the cellulase activity, accelerate the degradation of cytoderm. While the temperature of enzymatic pre- treatment was higher than 45℃, the cellulase activity was decreased, leading to the reduction of the yield. Therefore the temperature of enzymatic pretreatment for 45℃ was used. 3.2. Effect of Liqu id to Solid Rati o on EPME As it is known, the liquid to solid ratio is very important in the extraction. From the perspective of mass transfer, it mainly affects the concentration gradient between liq- uid phase and solid phase. Figure 4 shows that the yield of asiaticoside was increased with the increase of liquid to solid ratio. After the peak, the yield of asiaticoside was decreased with the increase of liquid to solid ratio. The higher liquid to solid ratio, the longer time for the solution elevated to the same temperature was required. At the same microwave radiation, the temperature of the system with larger amount of solvent was relatively lower; the solute diffusion would be affected. So the ratio for 30mL/g was chosen. 3.3. Effect of Microwave Radiation Time on EPME Figure 5 described that the extraction yield sharply in- creased before 60s and was asymptotic to a steady value during 60~120s, then falls down after 120s. At the pre- liminary stage of extraction, the velocity of molecular thermal motion quickened and the asiaticoside was qui- ckly separated from the cell into solution. The extraction process had tended towards equilibrium since 60s. When Figure 3. Effect of temperature of enzymatic pretreatment on EPME. Figure 4. Effect of liquid to solid ratio on EPME. Figure 5. Effect of microwave radiation time on EPME. the microwave radiation time exceeded 120s, the system was hyperthermal, the vaporization reflux of solution increased. All of above brought about the reduction of effective contact interval of solvent and plants, and caused the decrease of the thermal effects. In addition, the extension of microwave radiation time would in- crease the consumption of energy. Thus the microwave radiation time for 60s was used in the experiment. 3.4. Quadratic Regression Orthogonal Design Results The quadratic regression orthogonal design was employ- ed to evaluate the relevance of the three main extraction factors including enzymatic temperature, liquid to solid ratio and the microwave radiation time, while other fac- tors including grinding degree of plant material, concen- tration of enzyme, enzymatic pretreatment time and pH of the solvent were constant according to the pre-expe- riments. The multivariate study allows the identification of interactions between variables and provides a com- plex exploration of the experimental domain to be stud- ied with a number of experiments optimized. The three key variables studied were pointed at five separate coded levels [11], –1, 0, +1, +γ (=1.682) and their values were selected on the basis of previous ex- periments. The natural values and coded levels used in this multivariate study are presented in Table 1. SciRes Copyright © 2009 JBiSE  C. X. Wang et al. / J. Biomedical Science and Engineering 2 (2009) 526-531 529 Table 1. Factors of orthogonal experiment. X1-Microwave X2-Liquid to X3-Enzymatic -γ 9 13 28 -1 30 20 35 0 60 30 45 1 90 40 55 γ 111 47 62 Δ 30 10 10 The statistical analysis software was applied in this experiment to establish a regression Eq.2, in this equa- tion, the terms of Y, X1, X2, and X3 respectively repre- sent the yield of asiaticoside, microwave radiation time, liquid to solid ratio and enzymatic temperature: Y=25.8963+1.8150X1-0.7646X2+0.346 4X3 +0.9363X1X2+0.4 688X 1X3+0.1363X2X3 (2) -0.7065X1 2-0.6852X2 2-1.7547X3 2 Analysis of variance was carried out in order to test the signification of the regression model. Thus, various statistical data such as sum of squares (SS), mean sq- uares (MS), F-ratio were given in Table 2. The F-ratio in Table 2 was the ratio of the meansq- uared error to the pure error obtained from the replicates at the design center. The significance of the F-ratio de- pends on the number of degrees of freedom (d. f.) in the model. Thus, the effects lower than 0.05 in this column were significant. As shown in Table 2, FRegression=10.866>F0.05 (9, 8)=3.388, the regression of (2) was significant, while FLack of fit=3.478<F0.05 (5, 8) =3.687, the lack of fit was non-significant. Therefore, (2) had well predictivity un- der the experimental condition. In the test of regression coefficient, F-ratio for terms X3, X1X3, X2X3 (1.253, 1.344, 0.114) were lower than F0.05 (1, 8) =5.318, so these terms were not significant. On the contrary, the F-ratio for other terms (X1, X2, X1X2, X1 2, X2 2 and X3 2) which were higher than F0.05, indicated the significance of these terms. As the orthogonality of this experiment, the insignificant terms were cut out to simplify the (3): Y=25.8963+1.8150X1-0.7646X2+0.936 3X1X2 -0.7065X1 2-0.6852X2 2-1.7547X3 2 (3) Table 2. Variance analysis of test results. Source d.f. SS MS F F0.05 X1 1 44.995 44.995 34.409 5.318 X2 1 7.985 7.985 6.106 X3 1 1.639 1.639 1.253 X1X2 1 7.012 7.012 5.363 X1X3 1 1.758 1.758 1.344 X2X3 1 0.148 0.148 0.114 X12 1 7.935 7.935 6.068 X22 1 7.463 7.463 5.707 X32 1 48.949 48.949 37.432 Regression 9 127.885 14.209 10.866 3.388 Lack of fit 5 22.740 4.548 3.478 3.687 Pure error 8 10.461 1.308 Total 22 161.087 7.322 Eq.3 indicated that the microwave radiation time and liquid to solid ratio were the main factors that influence the yield because of the significance of the terms X1 and X2 .The significance of X1X2 suggested the obvious in- teraction between microwave radiation time and liquid to solid ratio. And the significance of all the quadratic terms demonstrated the nonlinear relationship between the three factors and the yield of extraction. 3.5. Analysis of Response Surface The 3D surface curves were drawn to illustrate the main and interactive effects of the three factors on the yield. The response surfaces are shown in Figures 6,7,8 with one variable kept at optimum level and the other two varied within the experimental range. Figure 6 shows the effect of liquid to solid ratio(X2) and extraction time(X1) on the yield. A quadratic effect for both factors on the response can be observed. At a low level of X2 (-2), the system was readily hyperther- mal and vaporization of the solvent could reduced the yield with the increasing of microwave time. And at a high level of X2 (2), the yield displayed an increasing curve in the experimental range of X1. It is due to the distinctly interaction between X1 and X2, which was implied in the Eq.3. The maximum yield was predicted when X1 was in the range of 1.5 to 1.7 and X2 varied from 0.4 to 0.6. Figure 7 depicts the effect of enzymatic tempera- ture(X3) and extraction time(X1),as both them exerting a quadratic effect. As shown in the Figure 7, an increase in yield resulted when X3 was increased in the level range from –2 to 0, then the curve started to go down, which may indicate that a level of X1 at approximate 0 is required to achieve maximum yield. Likewise, an in- crease in yield resulted when X1 was increased in the code range from –2 to 1.5, and then the yield was slightly reduced. In the response surface, X1 exerted a more significant effect on yield than X3, and no obvious interaction between X1 and X3 was observed, which was well in agreement with Eq. 3. In Figure 8, yield showed quadratic curve depending upon the liquid to solid ratio(X2), whereas no significant effect was observed in the enzymatic temperature(X3). Because the ratio was a key factor which influences the impetus in mass transfer of both enzymatic pretreatment and microwave extraction processes, it exerted a more significant effect on yield than the factor of enzymatic temperature. According to the response surface, there was no obviously interaction between X2 and X3, and it was also supported by the Eq.3. 3.6. Optimization of EPME Condition Yield of extraction was employed as the evaluation ob- jective in the optimization of the parameter to ensure it reaches the peak under constraint conditions. According SciRes Copyright © 2009 JBiSE 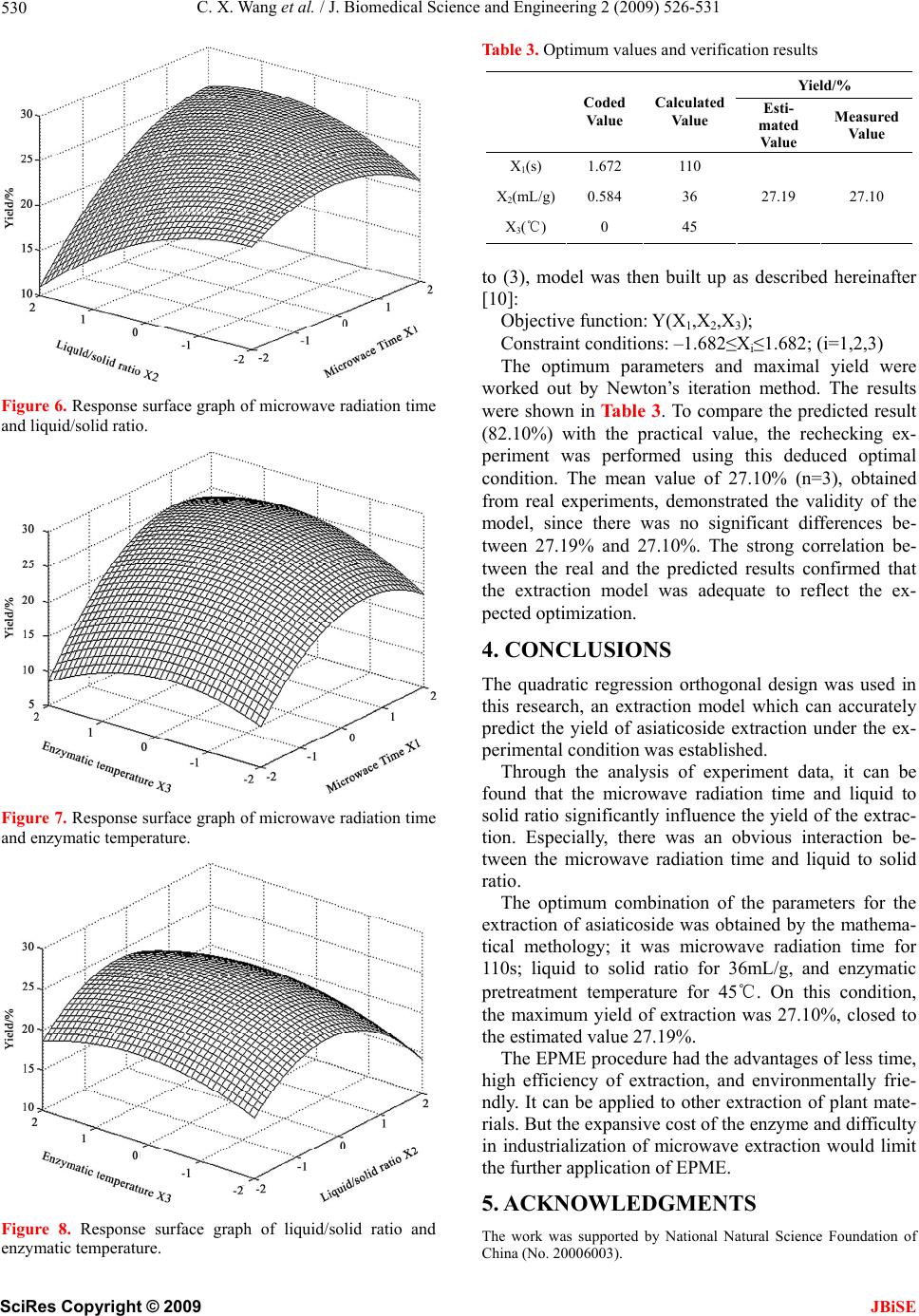 C. X. Wang et al. / J. Biomedical Science and Engineering 2 (2009) 526-531 530 Figure 6. Response surface graph of microwave radiation time and liquid/solid ratio. Figure 7. Response surface graph of microwave radiation time and enzymatic temperature. Figure 8. Response surface graph of liquid/solid ratio and enzymatic temperature. Table 3. Optimum values and verification results Yield/% Coded Value Calculated Value Esti- mated Value Measured Value X1(s) 1.672 110 X2(mL/g)0.584 36 X3(℃) 0 45 27.19 27.10 to (3), model was then built up as described hereinafter [10]: Objective function: Y(X1,X2,X3); Constraint conditions: –1.682≤Xi≤1.682; (i=1,2,3) The optimum parameters and maximal yield were worked out by Newton’s iteration method. The results were shown in Ta bl e 3 . To compare the predicted result (82.10%) with the practical value, the rechecking ex- periment was performed using this deduced optimal condition. The mean value of 27.10% (n=3), obtained from real experiments, demonstrated the validity of the model, since there was no significant differences be- tween 27.19% and 27.10%. The strong correlation be- tween the real and the predicted results confirmed that the extraction model was adequate to reflect the ex- pected optimization. 4. CONCLUSIONS The quadratic regression orthogonal design was used in this research, an extraction model which can accurately predict the yield of asiaticoside extraction under the ex- perimental condition was established. Through the analysis of experiment data, it can be found that the microwave radiation time and liquid to solid ratio significantly influence the yield of the extrac- tion. Especially, there was an obvious interaction be- tween the microwave radiation time and liquid to solid ratio. The optimum combination of the parameters for the extraction of asiaticoside was obtained by the mathema- tical methology; it was microwave radiation time for 110s; liquid to solid ratio for 36mL/g, and enzymatic pretreatment temperature for 45℃. On this condition, the maximum yield of extraction was 27.10%, closed to the estimated value 27.19%. The EPME procedure had the advantages of less time, high efficiency of extraction, and environmentally frie- ndly. It can be applied to other extraction of plant mate- rials. But the expansive cost of the enzyme and difficulty in industrialization of microwave extraction would limit the further application of EPME. 5. ACKNOWLEDGMENTS The work was supported by National Natural Science Foundation of China (No. 20006003). SciRes Copyright © 2009 JBiSE  C. X. Wang et al. / J. Biomedical Science and Engineering 2 (2009) 526-531 SciRes Copyright © 2009 531 REFERENCES [6] Bayramoglu, B., Sahin, S., and Sumnu, G., (2008) Sol- ventfree microwave extraction of essential oil from oregano, J. Food Eng., 88, 535−540. [1] Randriamampionona, D., Diallo, B., and Rakotoniriana, F., (2007) Comparative analysis of active constituents in Centella asiatica samples from Madagascar: Application for ex situ conservation and clonal propagation, Fitotera- pia, 78, 482–489. [7] Proestos, C. and Komaitis, M., (2008) Application of microwave-assisted extraction to the fast extraction of plant phenolic compounds, Food Sci. Technol-LEB, 41, 652−659. [2] Verma, K., Bhartariya, G., and Gupta, M., (1999) Rever- sephase high performance liquid chromatography of asi- aticoside in Centella asiatica, Phytochem. Anal., 10, 191–193. [8] Pan, X. J., Niu, G. G., and Liu, H. Z., (2003) Micro- wave-assisted extraction of tea polyphenols and tea caf- feine from green tea leaves, Chem. Eng. Process, 42, 129− 133. [3] Sarma, K., Khosa, L., and Chansauria, N., (1995) An- tistress activity of Tinospora cordfolia and Centella asi- atica extracts, Phytother. Res., 10, 181−183. [9] Lu, X. Y., “Determination of asiaticosides by UV- spec- trophotography. (2005) J Modern Food Drug, 16, 15−16. [10] Ma, S. F., Wang, L. Q., and Hu, Z. C., (2005) Enzymatic extraction of the submerged mycelium polysaccharide from Pleurotus nebrodensis, T CSAE, 22, 198−201. [4] Flora, S. and Gupta, R., (2007) Beneficial effects of Cen- tella asiatica aqueous extract against arsenic-induced oxidative stress and essential metal status in rats, Phyto- ther. Res., 21, 980−988. [11] Virot, M., Tomao, V., and Colnagui, G., (2007) New mi- crowave-integrated Soxhlet extraction an advantageous tool for the extraction of lipids from food products, J. Chromatogr. A, 1174, 138−144. [5] Li, B. B., Smith, B., and Hossain, M., (2006) Extraction of phenolics from citrus peels II, Enzyme-assisted ex- traction method, Sep. Purif. Technol., 48, 189−196. JBiSE |

