Q. CHEN ET AL.
6
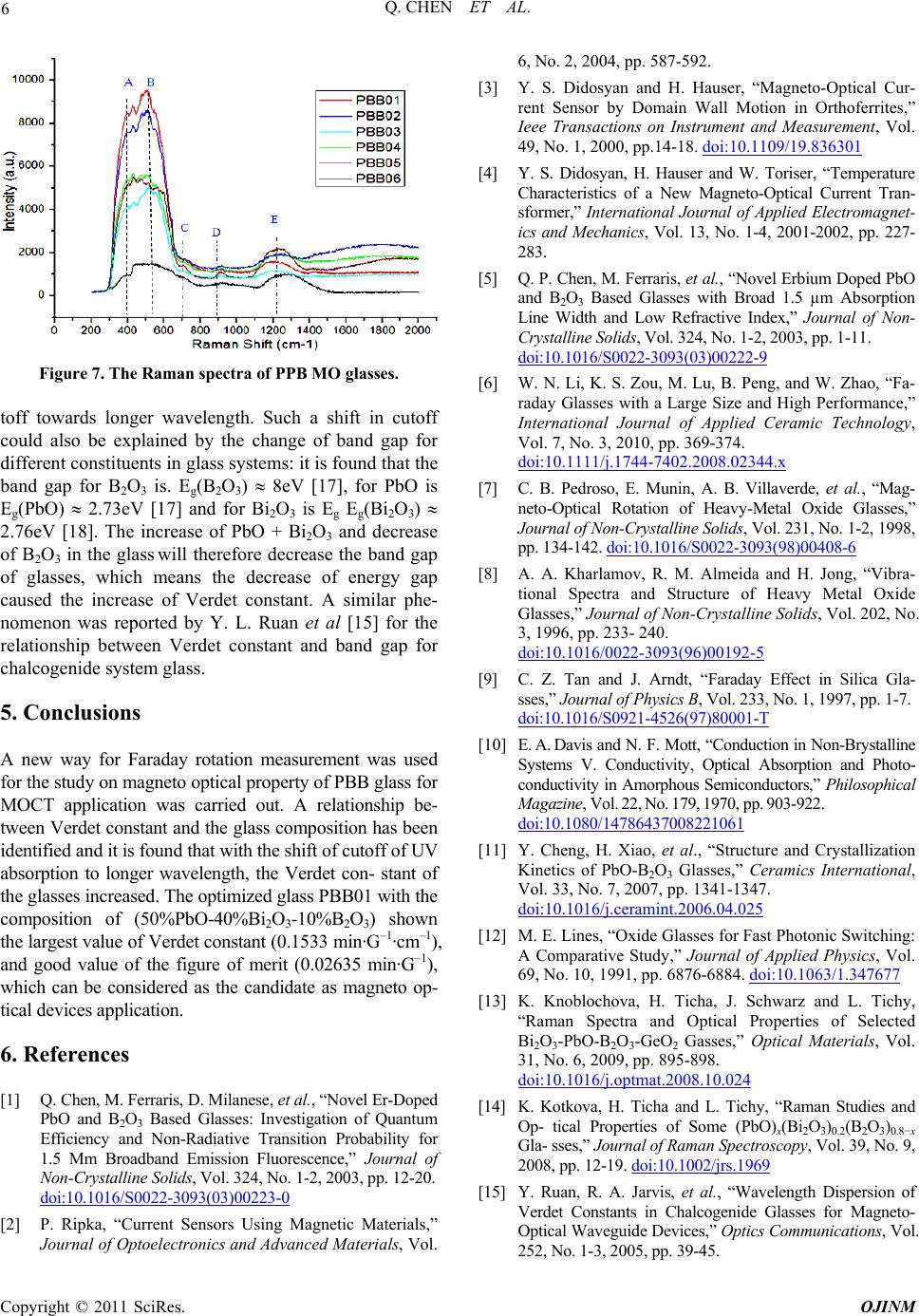
Figure 7. The Raman spectra of PPB MO glasses.
toff towards longer wavelength. Such a shift in cutoff
could also be explained by the change of band gap for
different constituents in glass systems: it is found that the
band gap for B
2
O
3
is. E
g
(B
2
O
3
) 8eV [17], for PbO is
E
g
(PbO) 2.73eV [17] and for Bi
2
O
3
is E
g
E
g
(Bi
2
O
3
)
2.76eV [18]. The increase of PbO + Bi
2
O
3
and decrease
of B
2
O
3
in the glass
will therefore decrease the band gap
of glasses, which means the decrease of energy gap
caused the increase of Verdet constant. A similar phe-
nomenon was reported by Y. L. Ruan et al [15] for the
relationship between Verdet constant and band gap for
chalcogenide system glass.
5. Conclusions
A new way for Faraday rotation measurement was used
for the study on magneto optical property of PBB glass for
MOCT application was carried out. A relationship be-
tween Verdet constant and the glass composition has been
identified and it is found that with the shift of cutoff of UV
absorption to longer wavelength, the Verdet con- stant of
the glasses increased. The optimized glass PBB01 with the
composition of (50%PbO-40%Bi
2
O
3
-10%B
2
O
3
) shown
the largest value of Verdet constant (0.1533 min·G
–1
·cm
–1
),
and good value of the figure of merit (0.02635 min·G
–1
),
which can be considered as the candidate as magneto op-
tical devices application.
6. References
[1] Q. Chen, M. Ferraris, D. Milanese, et al., “Novel Er-Doped
PbO and B
2
O
3
Based Glasses: Investigation of Quantum
Efficiency and Non-Radiative Transition Probability for
1.5 Μm Broadband Emission Fluorescence,”
Journal of
Non-Crystalline Solids, Vol. 324, No. 1-2, 2003, pp. 12-20.
doi:10.1016/S0022-3093 (03)002 23-0
[2] P. Ripka, “Current Sensors Using Magnetic Materials,”
Journal of Optoelectronics and Advanced Materials, Vol.
6, No. 2, 2004, pp. 587-592.
[3] Y. S. Didosyan and H. Hauser, “Magneto-Optical Cur-
rent Sensor by Domain Wall Motion in Orthoferrites,”
Ieee Transactions on Instrument and Measurement, Vol.
49, No. 1, 2000, pp.14-18. doi:10.1109/19.836301
[4] Y. S. Didosyan, H. Hauser and W. Toriser, “Temperature
Characteristics of a New Magneto-Optical Current Tran-
sformer,” International Journal of Applied Electromagnet-
ics and Mechanics, Vol. 13, No. 1-4, 2001-2002, pp. 227-
283.
[5] Q. P. Chen, M. Ferraris, et al., “Novel Erbium Doped PbO
and B
2
O
3
Based Glasses with Broad 1.5 µm Absorption
Line Width and Low Refractive Index,” Journal of Non-
Crystalline Solids, Vol. 324, No. 1-2, 2003, pp. 1-11.
doi:10.1016/S0022-3093 (03)002 22-9
[6] W. N. Li, K. S. Zou, M. Lu, B. Peng, and W. Zhao, “Fa-
raday Glasses with a Large Size and High Performance,”
International Journal of Applied Ceramic Technology,
Vol. 7, No. 3, 2010, pp. 369-374.
doi:10.1111/j.1744-7402.2008.02344.x
[7] C. B. Pedroso, E. Munin, A. B. Villaverde, et al., “Mag-
neto-Optical Rotation of Heavy-Metal Oxide Glasses,”
Journal of Non-Crystalline Solids, Vol. 231, No. 1-2, 1998,
pp. 134-142. doi:10.1016/ S0022-3 093(98) 00408-6
[8] A. A. Kharlamov, R. M. Almeida and H. Jong, “Vibra-
tional Spectra and Structure of Heavy Metal Oxide
Glasses,” Journal of Non-Crystalline Solids, Vol. 202, No.
3, 1996, pp. 233- 240.
doi:10.1016/0022-3093(96)00192-5
[9] C. Z. Tan and J. Arndt, “Faraday Effect in Silica Gla-
sses,” Journal of Physics B, Vol. 233, No. 1, 1997, pp. 1-7.
doi:10.1016/S0921-4526 (97)800 01-T
[10] E.
A.
Davis and N. F. Mott, “Conduction in Non-Brystalline
Systems V. Conductivity, Optical Absorption and Photo-
conductivity in Amorphous Semiconductors,” Philosophical
Magazine, Vol.
22,
No.
179,
1970, pp.
903-922.
doi:10.1080/1478643700 822106 1
[11] Y. Cheng, H. Xiao, et al., “Structure and Crystallization
Kinetics of PbO-B
2
O
3
Glasses,” Ceramics International,
Vol. 33, No. 7, 2007, pp. 1341-1347.
doi:10.1016/j.ceramint.2006.04.025
[12] M. E. Lines, “Oxide Glasses for Fast Photonic Switching:
A Comparative Study,” Journal of Applied Physics, Vol.
69, No. 10, 1991, pp. 6876-6884. doi:10.1063/1.347677
[13] K. Knoblochova, H. Ticha, J. Schwarz and L. Tichy,
“Raman Spectra and Optical Properties of Selected
Bi
2
O
3
-PbO-B
2
O
3
-GeO
2
Gasses,”
Optical Materials, Vol.
31, No. 6, 2009, pp. 895-898.
doi:10.1016/j.optmat.2008.10.024
[14] K. Kotkova, H. Ticha and L. Tichy, “Raman Studies and
Op- tical Properties of Some (PbO)
x
(Bi
2
O
3
)
0.2
(B
2
O
3
)
0.8−x
Gla- sses,” Journal of Raman Spectroscopy, Vol. 39, No. 9,
2008, pp. 12-19. doi:10.1002/jrs. 1969
[15] Y. Ruan, R. A. Jarvis, et al., “Wavelength Dispersion of
Verdet Constants in Chalcogenide Glasses for Magneto-
Optical Waveguide Devices,” Optics Communications, Vol.
252, No. 1-3, 2005, pp. 39-45.
Copyright © 2011 SciRes. OJINM