Journal of Power and Energy Engineering
Vol.05 No.12(2017), Article ID:81118,13 pages
10.4236/jpee.2017.512011
Desulphurization of Transportation Fuels by Per-Formic Acid Oxidant Using MoOx Loaded on ZSM-5 Catalyst
Waqas Ahmad, Imtiaz Ahmad
Institute of Chemical Sciences, University of Peshawar, Peshawar, Pakistan




Received: September 13, 2017; Accepted: December 15, 2017; Published: December 18, 2017
ABSTRACT
Desulphurization of model and real oil samples was investigated using performic acid as oxidant assisted by air as co-oxidant. The catalysts used were Mo-oxide supported on ZSM-5 zeolite, which was synthesized in the laboratory and characterized by FT-IR, XRD, SEM and SSA analysis. In case of model oil, the optimum condition determined for complete oxidation of all the model compounds including thiophene, DBT and 4,6-DMDBT were; 60˚C, 60 min, ambient pressure and air flow rate of 100 mL/min. The oxidation reactivity decreased from 4,6-DMDBT to DBT and thiophene, which was found to follow pseudo first order kinetics. The real oil sample used in the study included untreated naphtha (NP), light gas oil (LGO), heavy gas oil (HGO) and Athabasca bitumen (Bit.). In case of NP and LGO the sulfur removal of above 78% was attained whereas in case of HGO and Bit. samples about 60% of desulfurization was achieved.
Keywords:
Molybdenum Oxide, NH4-ZSM-5, Oxidative Desulphurization, Air Oxidation, Performic Acid, Model Sulfur Compounds, Wet-Impregnation

1. Introduction
Because of the low cost and mild operating conditions required, oxidative desulfurization (ODS) is the most suitable alternative to the industrial hydrodesulfurization (HDS). The process has also the advantage of high efficiency in treating the refractory sulfur compounds, which are difficult to be treated by HDS under its normal operating conditions. In this process, sulfur compounds in petroleum are selectively oxidized, whereby their polarity increases. The oxidized sulfur compounds are the separated from the non-polar phase through extraction or adsorption. Various oxidizing agents are employed in the ODS process, such as nitric oxide, sodium perchlorate, Ozone, hydroperoxides and per-acids generated in situ by the reaction of H2O2 and organic acids like formic acid or acetic acid etc. [1]-[8]. Per-acids constitutes very effective oxidant for ODS process. In this oxidation system H2O2 and formic acid or acetic acid is used to in situ generate per-acids, which bear high selectivity towards oxidation of sulfur compounds. The process efficiency has been found to increase by using various types of catalysts, which include activated carbon [9], EDTA, Na or Mg-silicates [10], silica gel [11] molecular sieves loaded with metal oxides [12], sodium bicarbonate[13], transition metal salts [14] etc. These catalysts actually promote the formation of peracid radical from H2O2 and formic acid, which selectively oxidizes the sulfur compounds. Air or molecular oxygen O2 along with various types of initiators or catalyst is also as a cost-effective oxidant for ODS of liquid fuels. O2 with various catalysts and initiators such as aldehydes [15], n-Octanal and cobalt salts [16], Iron pthalocyanin [17] complexes and TBPH etc. has proved to be very efficient ODS systems by showing promising desulfurization yields.
Generally heavy petroleum distillates contain high amount of sulfur, the sulfur compounds also have complex nature. Besides, due to high viscosity and high density, the efficient ODS treatment of heavy fractions is very difficult. Therefore, more effective ODS system is required for heavy fractions in order to reduce their sulfur content. We have previously reported the catalytic ODS of model and real oil including heavy distillate fraction by per-formic acid oxidation along with air oxidation over amphiphilic polyoxometalates (POM) and Fe exchanged ZSM-5 catalyst [18] [19]. In both systems, the presence of air as co-oxidant efficiently enhanced the desulfurization yields in model oil and in real oil samples.
In the present work the ODS of model and real oil sample is investigated through air assisted performic acid oxidation system over oxides of Mo, V and W loaded on ZSM-5 as catalysts. The optimum process parameters are determined. The effect of catalysts on reactivity of model sulfur compounds is also investigated.
2. Experimental
2.1. Reagents
The model oil used was thiophene, DBT and 4,6-DMDBT dissolved in n-heptane (total S 1275 ppm). NH4-ZSM-5 zeolite purchased from Zeolyst International USA, was used as catalysts support which was transformed in to H-ZSM-5 by calcination in air for 2 - 3 h at 300˚C. The real oil samples used Naphtha (NP), LGO, HGO and Athabasca Bitumen (Bit.).
2.2. Preparation of Catalyst
The catalysts used were oxides of Mo, V and W loaded on H-ZSM-5 zeolites,
synthesized in the laboratory by wet impregnation technique [20]. For synthesis of Mo oxide supported on HZSM-5, stoichiometric amount of (NH4)6Mo7O24∙4H2O (for 2 wt% loading) was dissolved in about 100 mL of distilled water. 3 g of HZSM-5 was dispersed in this solution, the pH was adjusted to 4.5 with HNO3 solution and stirred for about 2 h at 60˚C. The suspension was filtered and residue was collected, dried and then calcined for 4 h under air at 500˚C. VOx/ZSM-5 was prepared using Na3VO4 and WOx/ZSM-5 by using Na2WO4 as precursors, through similar process. During synthesis of WOx/ZSM-5, pH was adjusted to 9, with NaOH solution. The synthesised catalysts were characterized by elemental analysis through XRF, surface properties, FT-IR and SEM analysis.
2.3. ODS of Model and Real Oil Samples
ODS experiments of model oil was conducted, by taking 20 ml of model oil a three-neck flask fitted with a condenser, placed in oil bath mounted on magnetic stirring hot plate. 2 ml of H2O2, 2.5 ml HCOOH and 0.05 g catalyst was added to the sample, and stirred for 60 min under constant air bubbling and temperature maintained at 80˚C. After oxidation, equal volume of extraction solution (80% methanol aq.) was mixed with the sample and transferred to the separating funnel, in which the oil layer was separated. Reaction temperature and reaction time were optimized to attain maximum sulfur removal.
Under optimized conditions the ODS of real oil samples i.e. NP, LGO, HGO and Bit. was carried about using same procedure as followed for model oil. In case of heavy oil samples i.e. HGO and Bit., 5 ml benzene as a diluent was added to reduce viscosity of the sample. After oxidation, the real oil sample was extracted with 20:80 mixture of acetonitrile and methanol.
2.4. Product Analyses
In oil samples, the concentration of total S was determined through sulfur analyzer (Antek PAL) equipped with chemiluminescent and vacuum UV detector. The extent of sulfur removed was calculated as % desulphurization, using the following relation,
(1)
where,
So = Concentration of sulfur in original oil.
St = Concentration of sulfur in treated oil.
In model oil, the concentration of model sulfur compounds was determined by gas GC-FID (Agilent 7890 A) equipped with PIONA capillary column. The oxidative conversion of model sulfur compounds was reported as % conversion, which was calculated from the difference in the initial concentration (Co) and the final concentration (Ct) after time T.
(2)
Kinetic investigation was conducted by applying first order kinetics equation (Equation (3)), the oxidation rate constant was determined from the plot of lnCo/Ct vs T.
(3)
Using Arrhenius equation, the activation energy for oxidation reactions was calculated from the plot of ln K vs 1/T.
(4)
3. Results and Discussions
3.1. Catalytic ODS of Model Oil
Oxidative desulfurization of model and real oil samples was conducted through oxidation using H2O2 and HCOOH as oxidants, and air as co-oxidant in the presence of MoOx, VOx and WOx loaded of ZSM-5 zeolite as catalyst. Initially the ODS of model oil was investigated in the presence of the catalysts with 2 ml H2O2 and 2 ml of HCOOH under 80˚C for 1 h reaction time, after oxidation, the model oil was extracted with 80% methanol (aq) solution. The results of total sulfur analysis are indicated in Table 1, which indicate that maximum level of sulphur removal was found to occur in the presence of MoOx-ZSM-5 catalyst, e.g. 96.60%. Whereas in the presence of VOx and WOx-ZSM-5, the level of desulfurization was lower, i.e. 90.87 and 90.33 respectively. In the presence of only HZSM-5 zeolite the desulfurization level was about 88%. It is clear from the data that the presence of oxides of Mo, V and W, the extent of desulfurization has been sufficiently increased.
Several studies show that Mo, V and W supported on various supports exhibit high efficiency in the ODS process, in the presence of different oxidation systems these have promisingly enhances the oxidative conversion of sulfur compounds [21] [22]. During the ODS reactions, these metals i.e. Mo, V and W leads to formation of peroxo species, which can oxidize the sulfur compounds conveniently. It has been shown that in the ODS of liquid fuels using MoOx/Al2O3 as catalysts and H2O2 as oxidizing agent in biphasic oil-acetonitrile system, the oxidation process occurs through the formation of hydroperoxymolybdate species, which is formed by electrophilic attack of H2O2 over octamolybdate and heptamolybdate species. The hydroxyperoxymolybdate species further
Table 1. Catalytic ODS of model oil in the presence of WoOx/VoOx/MoOx-ZSM-5.
oxidizes the aromatic sulfur compounds through electophilic attack on sulfur atom in the molecule and convert it to sulfoxide and sulfone, respectively. During this process the hydroperoxomolybdate species is reduced, which is again re-oxidized by the oxidant i.e. H2O2 [23]. In the current catalytic ODS system, it is assumed that per-formic acid supplies the active oxygen to form peroxomolybdate species, which further leads to oxidation of the sulfur compounds. This could be confirmed from the poor desulfurization yield shown when only H-ZSM-5 was used as a catalyst, where no Mo atoms are available to form peroxomolybdate species. A generalized mechanism for the reaction is given below:
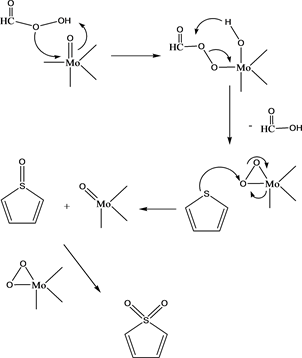
Mechanism of thiophene oxidation by per-formicacid using MoOx/ZSM5 catalyst
The catalysts synthesized in the laboratory were characterized, by SAA, SEM, FT-IR and EDX analysis. The data in Table 2 show that the surface area of the plain ZSM-5 is higher than the metal oxides supported ZSM-5. In other words, the surface area of the zeolite decreased with the incorporation of the metal oxides through impregnation. In case of ZSM-5, the BET surface area was 420 m2/g, whereas in case of MoOx, WOx and VOx-ZSM-5 the surface area was shown to be 251.29, 272.82 and 255.34 m2/g, respectively. It may be suggested that the BET surface area of the zeolite decreased with the impregnation of metal
Table 2. Surface area and concentration of active metal in catalysts.
oxides due to blockage of the micropores and the tunnel in the zeolites which leads to decrease in the void volume or surface area. It is further added that due to their large size, the metal oxides species may have occupied the larger dimension pores which led to blockage of meso and micropores, hence the large decrease in surface area is observed. The FT-IR spectra of MoOx, WOx and VOx -ZSM-5 catalysts are shown in Figure 1, which was recorded in the wave length raging between 500 to 1350 cm−1 in order to locate the characteristic absorption bands for metal oxides. The FT-IR spectra of all the metal oxide supported ZSM-5 catalyst exhibit similar prominent absorption bands centred at wave lengths of 1150 - 1170 cm−1 which can be attributed to asymmetric stretching of Si-O-Si bonds [24], 795 - 805 cm−1 show metal bridged by corner oxygen i.e. M-Oc-M configuration [25] and 545 - 540 cm−1 indicates symmetric stretching of MO2 [26].
It can be concluded from these results that the impregnated metals oxides exist as polymeric oxides which are adsorbed the surface of ZSM-5.
The SEM images of fresh and spent MoOx/ZSM-5 and H-ZSM-5 are shown in Figure 3. Figure 2 show SEM image of H-ZSM5 zeolite which exhibit uniformly fine dispersed granules, and each grain has smooth surface. The micrographs of
Figure 1. FT-IR spectra of (a) VOx; (b) WOx and (c) MoOx loaded on ZSM-5.
Figure 2. SEM images of (a) H-ZSM-5; (b) MoOx/ZSM-5; (c) Spent MoOx/ZSM-5.
MoOx/ZSM-5 shows fine granules which seems agglomerated together. The size of the granules is non-uniform and slightly larger than H-ZSM5. The increase in size may be due addition of the polymeric molybdates covering the grains surface after impregnation. The surface of the granules depicts uniform morphology with no plateaus or cracks, which suggests that MoOx is uniformly dispersed on the zeolite surface. The micrographs of spent catalyst i.e. MoOx/ZSM-5 recovered after reaction show more swollen granules with smooth surfaces but agglomerated together.
3.2. Reactivity of Sulfur Compounds
The catalytic ODS of model oil over MoOx/ZSM-5 was initially investigated at 80˚C for 60 min reaction time. Further the reactivity of model sulfur compounds in the model oil was investigated at different temperatures and reaction times. The conversion of model sulfur compounds was monitored at 25˚C, 40˚C, 60˚C and 80˚C reaction temperatures for 15, 30, 45, 60, 90 and 120 min reaction times. We have previously confirmed through GC-MS analysis that the catalytic oxidation of individual model sulfur compounds present in the model oil i.e. Thiophene, DBT and 4,6-DMDBT, leads to form respective sulfones in the present oxidation system [19]. In the current study, the extent of oxidation of model sulfur compounds was investigated by GC-FID. It can be observed from the results that the conversion of all model compounds linearly increased with increasing the reaction time and temperature. The conversion was slow at lower temperatures i.e. at 25˚C and 40˚C, even at longer reaction times and never reached to completion. However, at higher temperatures the conversion was rapid. In case of thiophene the at 60˚C highest conversion of 93% was achieved, but at 80˚C the conversion reached to about 95% in 60 min. The higher DBT conversion of 94% was achieved at 60˚C in 60 min, but at 80˚C same level of oxidation was observed in 45 min. similarly, in case of 4,6-DMDBT, 95% oxidation was observed in 60 min at 60˚C, but conversion reached to more than 97% at 80˚C in 60 min. Thus in the presence of MoOx/ZSM-5, about 92% to 96% conversion was observed for all the model compounds in 60 min at 60˚C.
From the conversion of model compounds under different condition of temperature and times, oxidation kinetics were investigated. Initially first order kinetics was applied, and ln(Co/Ct) was plotted against the time (t), which gave linear plot for all the model compounds. The first order kinetics plots for thiophene, DBT and 4,6-DMDBT are displayed as Figures 3-5, showing that their ODS follow first order kinetics, which is in well agreement published literature [27]. The rate constants for oxidation of model compounds were calculated for 25˚C, 40˚C and 60˚C, the data is displayed in Table 3. Results show that for each model compounds the rate constant uniformly increases with increase in reaction temperature. From the rate constants, it can be observed that among the model sulfur compounds the reactivity order decreased as;
4,6-DMDBT > DBT > Thiophene
Figure 3. Pseudo-first order kinetics of Thiophene oxidation.
Figure 4. Pseudo-first order kinetics of DBT oxidation.
Figure 5. Pseudo-first order kinetics of 4,6-DMDBT oxidation.
Table 3. Pseudo-first order rate constants for ODS of model S compounds.
The reactivity order is same as the order of electron densities on the sulfur atom among these model compounds.
This shows that in the current catalytic oxidation system, the oxidative conversion of the model compounds is governed by the electron density on the sulfur atoms and not by the steric effects posed by the alkyl substituent’s. The reason could be the large surface area of the MoOx/ZSM-5 catalyst which allow the organosulfur compounds to access the active metal oxide sites of catalysts, hence the stearic hindrance effects are eliminated. Gang et al. also reported similar results using [28] MoO3/Al2O3 catalyst and H2O2 as oxidant for the ODS of model oil.
The activation energies of model sulfur compounds were calculated using Arrhenius equation. The activation energies for 4,6-DMDBT, DBT and Thiophene were calculated to be 28.30, 29.73 and 31.46 kJ/mol respectively. These activation energies are lower than the reported in literature using different catalytic oxidation systems for ODS, which reveals that the present catalytic ODS system is more efficient. For example, Caero et al. [29] have shown that using H2O2/VOx oxidation system, that activation energies of different sulfur compounds including thiophene, BT and DBT ranges from 35.3 to 48.4 kJ/mol.
3.3. ODS of Real Oil Samples
The ODS of real oil samples was conducted under optimized conditions, i.e. 60˚C, 60 min and 2 ml of H2O2 and HCOOH for 20 ml oil. The ODS of real oil samples was studied using MoOx/ZSM-5 catalyst and without catalyst. After oxidation, the real oil samples were extracted in two steps, 1st step extraction with 80% acetonitrile, and second step extraction with 80% methanol. Initial experiments revealed that single step extraction was insufficient for complete removal of the oxidized sulfur compounds from real oil samples, since the 80% acetonitrile extraction was found effective for oxidized gas oil [30], hence 2 stage extraction was used. The oil samples used as feed were untreated naphtha (NP), light gas oil (LGO), heavy gas oil (HGO) and Athabasca bitumen (Bit.) with total sulfur concentrations of 2.30, 1.28, 4.20 and 4.90 wt%, respectively. The desulfurization results are indicated in Figure 6. Results show that in case of NP, LGO, HGO and Bit. the level of sulfur removal without catalyst was 61.74%, 63.36%, 49.22% and 47.54% respectively, whereas in the presence of MoOx/ZSM-5 catalyst it was 81.6%, 78.68%, 64.86% and 60.27% respectively. From these results,
Figure 6. Sulfur removal in real oil samples by ODS over MoOx/ZSM-5 catalyst.
the advantage of the catalyst is clear, which considerably enhanced the desulfurization yield in real oil samples. It is evident that although in the absence of the catalyst the desulfurization yield was sufficient, which means that air assisted per-formic acid oxidation system efficiently decreased the level of sulfur in real oil samples, however the addition of the catalyst further raised the sulfur removal. This means that like in case of model oil, the peroxomolybdate species produced from MoOx loaded on ZSM-5 is also very efficient in oxidation of complex organosulfur compounds in real oil.
It is also revealed from the results that during the catalytic ODS the sulfur removal gradually decreases in case of real oil samples as its density or boiling range increases i.e. the desulfurization yield for untreated naphtha was 81.6% whereas for Athabasca bitumen the sulfur was 60.27%. Since with increase in the boiling point range of the fraction, its density and viscosity increase. With the increase in the boiling points, the structural complexity of the prevailing sulfur compounds increases. In heavy oil the sulfur compounds contains multiple aromatic rings and several alkyl side chains, and because of steric hindrance their oxidation is more difficult [31]. Also in case of heavy oils, the high viscosity creates the mass transfer problems due to which the polar phase oxidants cannot access the nonpolar sulfur compounds. It is therefore the heavy fractions i.e. HGO and Athabasca bitumen show lower desulfurization yield as compared to untreated naphtha and LGO.
4. Conclusion
Catalytic ODS of model and real oil was studied over oxide of Mo, V and W supported on ZSM-5 zeolite, using air assisted performic acid oxidation. The catalysts were synthesized and characterized in the laboratory. Maximum sulfur removal of model oil was found to occur at 60˚C, 60 min, ambient pressure and air flow rate of 100 mL/min. The oxidation of model sulfur compounds were found to follow pseudo first order kinetics. In case of NP and LGO the sulfur removal of above 78% was attained whereas in case of HGO and Bit. samples about 60% of desulfurization was achieved.
Acknowledgements
The financial support from HEC Pakistan, and technical support from Prof. Dr. Arno De Klerk University of Alberta, Canada, is highly acknowledged.
Cite this paper
Ahmad, W. and Ahmad, I. (2017) Desulphurization of Transportation Fuels by Per-Formic Acid Oxidant Using MoOx Loaded on ZSM-5 Catalyst. Journal of Power and Energy Engineering, 5, 87-99. https://doi.org/10.4236/jpee.2017.512011
References
- 1. Wang, D., Qian, E.W., Amano, H., Okata, K., Ishihara, A. and Kabe, T. (2003) Oxidative Desulfurization of Fuel Oil: Part I. Oxidation of Dibenzothiophenes Using Tert-Butyl Hydroperoxide. Applied Catalysis A: General, 253, 91-99. https://doi.org/10.1016/S0926-860X(03)00528-3
- 2. Zhou, X., Gai, H., Wang, J., Zhang, S., Yang, J. and Zhang, S. (2009) Oxidation of Benzothiophenes Using Tert-amyl Hydroperoxide. Chinese Journal of Chemical Engineering, 17, 189-194. https://doi.org/10.1016/S1004-9541(08)60192-5
- 3. Otsuki, S., Nonaka, T., Takashima, N., Qian, W., Ishihara, A., Imai, T. and Kabe, T. (2000) Oxidative Desulfurization of Light Gas Oil and Vacuum Gas Oil by Oxidation and Solvent Extraction. Energy & Fuels, 14, 1232-1239. https://doi.org/10.1021/ef000096i
- 4. Ali, M.F., Al-Malki, A. and Ahmed, S. (2009) Chemical Desulfurization of Petroleum Fractions for Ultra-Low Sulfur Fuels. Fuel Processing Technology, 90, 536-544. https://doi.org/10.1016/j.fuproc.2009.01.005
- 5. Shakirullah, M., Ahmad, W., Ahmad, I. and Ishaq, M. (2010) Oxidative Desulphurization Study of Gasoline and Kerosene: Role of Some Organic and Inorganic Oxidants. Fuel Processing Technology, 91, 1736-1741. https://doi.org/10.1016/j.fuproc.2010.07.014
- 6. Paybarah, A., Bone, R.L. and Corcoran, W.H. (1982) Selective Oxidation of Dibenzothiophene by Peroxybenzoic Acid Formed In Situ. Industrial & Engineering Chemistry Process Design and Development, 21, 426-431. https://doi.org/10.1021/i200018a014
- 7. Tam, P.S., Kittrell, J.R. and Eldridge, J.W. (1990) Desulfurization of Fuel Oil by Oxidation and Extraction. 1. Enhancement of Extraction Oil Yield. Industrial & Engineering Chemistry Research, 29, 321-324. https://doi.org/10.1021/ie00099a002
- 8. Tam, P.S., Kittrell, J.R. and Eldridge, J.W. (1990) Desulfurization of fuel oil by oxidation and extraction. 2. Kinetic modeling of oxidation reaction, Industrial & Engineering Chemistry Research, 29, 324-329. https://doi.org/10.1021/ie00099a003
- 9. Yu, G., Lu, S., Chen, H. and Zhu, Z. (2005) Diesel Fuel Desulfurization with Hydrogen Peroxide Promoted by Formic Acid and Catalyzed by Activated Carbon. Carbon, 43, 2285-2294. https://doi.org/10.1016/j.carbon.2005.04.008
- 10. Hao, L., Benxian, S. and Zhou, X. (2005) An Improved Desulfurization Process Based on H2O2/Formic Acid Oxidation System Followed by Liquid-Liquid Extraction. Part 1. Coker Gas Oil Feedstocks. Petroleum Science and Technology, 23, 991-999. https://doi.org/10.1081/LFT-200034498
- 11. Chen, L.J., Guo, S.H. and Zhao, D.S. (2006) Oxidation of Thiophenes over Silicagel in Hydrogen Peroxide/Formicacid System. Chinese Journal of Chemical Engineering, 14, 835-838.
- 12. Chen, L.J., Guo, S.H. and Zhao, D.S. (2007) Oxidative Desulfurization of Simulated Gasoline over Metal Oxide-Loaded Molecular Sieve. Chinese Journal of Chemical Engineering, 15, 520-523. https://doi.org/10.1016/S1004-9541(07)60118-9
- 13. Deshpande, A., Bassi, A. and Prakash, A. (2004) Ultrasound-Assisted, Base-Catalyzed Oxidation of 4,6-Dimethyldibenzothiophene in a Biphasic Diesel-Acetonitrile System. Energy & Fuels, 19, 28-34. https://doi.org/10.1021/ef0340965
- 14. de Souza, W.F., Guimarães, I.R., Guerreiro, M.C. and Oliveira, L.C.A. (2009) Catalytic Oxidation of Sulfur and Nitrogen Compounds from Diesel Fuel. Applied Catalysis A: General, 360, 205-209. https://doi.org/10.1016/j.apcata.2009.03.023
- 15. Venkateshwar Rao, T., Sain, B., Kafola, S., Nautiyal, B.R., Sharma, Y.K., Nanoti, S.M. and Garg, M.O. (2007) Oxidative Desulfurization of HDS Diesel Using the Aldehyde/Molecular Oxygen Oxidation System. Energy & Fuels, 21, 3420-3424. https://doi.org/10.1021/ef700245g
- 16. Murata, S., Murata, K., Kidena, K. and Nomura, M. (2003) A Novel Oxidative Desulfurization System for Diesel Fuels with Molecular Oxygen in the Presence of Cobalt Catalysts and Aldehydes. Energy & Fuels, 18, 116-121. https://doi.org/10.1021/ef034001z
- 17. Zhou, X., Li, J., Wang, X., Jin, K. and Ma, W. (2009) Oxidative Desulfurization of Dibenzothiophene Based on Molecular Oxygen and Iron Phthalocyanine. Fuel Processing Technology, 90, 317-323. https://doi.org/10.1016/j.fuproc.2008.09.002
- 18. Imtiaz, A., Waqas, A. and Muhammad, I. (2013) Desulfurization of Liquid Fuels Using Air-Assisted Performic Acid Oxidation and Emulsion Catalyst. Chinese Journal of Catalysis, 34, 1839-1847. https://doi.org/10.1016/S1872-2067(12)60668-8
- 19. Ahmad, W., Ahmad, I. and Yaseen, M. (2016) Desulfurization of Liquid Fuels by Air Assisted Peracid Oxidation System in the Presence of Fe-ZSM-5 Catalyst. Korean Journal of Chemical Engineering, 33, 2530-2537. https://doi.org/10.1007/s11814-016-0099-1
- 20. Chen, K., Xie, S., Bell, A.T. and Iglesia, E. (2001) Structure and Properties of Oxidative Dehydrogenation Catalysts Based on MoO3/Al2O3. Journal of Catalysis, 198, 232-242. https://doi.org/10.1006/jcat.2000.3125
- 21. González-García, O. and Cedeño-Caero, L. (2009) V-Mo Based Catalysts for Oxidative Desulfurization of Diesel Fuel. Catalysis Today, 148, 42-48. https://doi.org/10.1016/j.cattod.2009.03.010
- 22. Maciuca, A.-L., Ciocan, C.-E., Dumitriu, E., Fajula, F. and Hulea, V. (2008) V-, Mo- and W-Containing Layered Double Hydroxides as Effective Catalysts for Mild Oxidation of Thioethers and Thiophenes with H2O2. Catalysis Today, 138, 33-37. https://doi.org/10.1016/j.cattod.2008.04.031
- 23. García-Gutiérrez, J.L., Fuentes, G.A., Hernández-Terán, M.E., Murrieta, F., Navarrete, J. and Jiménez-Cruz, F. (2006) Ultra-Deep Oxidative Desulfurization of Diesel Fuel with H2O2 Catalyzed under Mild Conditions by Polymolybdates Supported on Al2O3. Applied Catalysis A: General, 305, 15-20. https://doi.org/10.1016/j.apcata.2006.01.027
- 24. Yan, X.-M., Lei, J.-H., Liu, D., Wu, Y.-C. and Guo, L.-P. (2007) Oxidative Desulfurization of Diesel Oil Using Mesopo-rous Phosphotungstic Acid/SiO2 as Catalyst. Journal of the Chinese Chemical Society, 54, 911-916. https://doi.org/10.1002/jccs.200700131
- 25. Jiang, X., Li, H., Zhu, W., He, L., Shu, H. and Lu, J. (2009) Deep Desulfurization of Fuels Catalyzed by Surfactant-Type Decatungstates Using H2O2 as Oxidant. Fuel, 88, 431-436. https://doi.org/10.1016/j.fuel.2008.11.010
- 26. He, L., Li, H., Zhu, W., Guo, J., Jiang, X., Lu, J. and Yan, Y. (2008) Deep Oxidative Desulfurization of Fuels Using Peroxophosphomolybdate Catalysts in Ionic Liquids. Industrial & Engineering Chemistry Research, 47, 6890-6895. https://doi.org/10.1021/ie800857a
- 27. Ishihara, A., Wang, D., Dumeignil, F., Amano, H., Qian, E.W. and Kabe, T. (2005) Oxidative Desulfurization and Denitrogenation of a Light Gas Oil Using an Oxidation/Adsorption Continuous Flow Process. Applied Catalysis A: General, 279, 279-287. https://doi.org/10.1016/j.apcata.2004.10.037
- 28. Jia, Y., Li, G. and Ning, G. (2011) Efficient Oxidative Desulfurization (ODS) of Model Fuel with H2O2 Catalyzed by MoO3/γ-Al2O3 under Mild and Solvent Free Conditions. Fuel Processing Technology, 92, 106-111. https://doi.org/10.1016/j.fuproc.2010.09.011
- 29. Caero, L.C., Hernández, E., Pedraza, F. and Murrieta, F. (2005) Oxidative Desulfurization of Synthetic Diesel Using Supported Catalysts: Part I. Study of the Operation Conditions with a Vanadium Oxide Based Catalyst. Catalysis Today, 107-108, 564-569. https://doi.org/10.1016/j.cattod.2005.07.017
- 30. Sobati, M.A., Dehkordi, A.M. and Shahrokhi, M. (2010) Extraction of Oxidized Sulfur-Containing Compounds of Non-Hydrotreated Gas Oil. Chemical Engineering & Technology, 33, 1515-1524. https://doi.org/10.1002/ceat.200900622
- 31. Flores, R., Rodas, A. and Chavarria, W. (2004) Desulfurization of Fuel Oils Using an Advanced Oxidation Method. Preprints of Papers - American Chemical Society, Division of Fuel Chemistry, 49, 341-342.








