Journal of Water Resource and Protection
Vol.5 No.8A(2013), Article ID:36215,5 pages DOI:10.4236/jwarp.2013.58A008
Validation of 226Ra and 228Ra Measurements in Water Samples Using Gamma Spectrometric Analysis
Nuclear and Radiological Regulatory Authority, Cairo, Egypt
Email: *hnndiab@yahoo.co.uk
Copyright © 2013 Hanan M. Diab, Wanan M. Abdellah. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received April 17, 2013; revised May 20, 2013; accepted June 17, 2013
Keywords: Radium isotopes; Gamma-ray spectrometry; Water analysis
ABSTRACT
Radium isotopes can be analyzed by different analytical methods based on gamma spectrometric measurements or alpha spectrometry. An improved method was developed to determine radium isotopes from water using gamma spectrometry after radiochemical separation. The Radium was selectively extracted from acidified samples using co-precipitation procedure with iron hydroxide and followed by precipitation of radium as radium sulphate Ba(Ra)SO4. The precipitate Ba(Ra)SO4 was filtered through the Millipore filter paper, dried and weighed to calculate chemical yield. 226Ra and 228Ra activities were measured using low-background gamma spectrometry in water samples. Radium was pre-concentrated from environmental samples by co-precipitation with BaSO4. The amounts of 226Ra and 228Ra on the sample were obtained by gamma-ray spectrometry for the 351 keVγ-ray from 214Pb and for the 911 keV γ-ray from 228Ac, both in radioactive equilibrium with precursors, respectively. The accuracy, selectivity, traceability, applicability and Minimum Detectable Activity (MDA) of the technique were discussed. Also, the effect of physical and chemical characteristics of the water samples such as TDS, pH, soluble species, sulphate and bicarbonate that effect on the radium determination were taking into consideration. The method has been validated with a certified reference material supplied by the International Atomic Energy Agency and reliable results were obtained. The radiochemical yields for radium were 70% - 90% and recovery was 97% and 80% for 226Ra and 228Ra, respectively.
1. Introduction
Radium (Ra) isotopes are important from the viewpoints of radiation protection and environmental protection. Their high toxicity has stimulated the continuing interest in methodology research for determination of Ra isotope in various media. Radium is a radioactive element for which no stable isotope is known. The mass numbers of the known isotopes range from 206 to 230. Among them, two radium isotopes, 226Ra and 228Ra are very significant from a radiological protection viewpoint due to their relatively long half-lives, presence in nature, and high dose conversion factors. Radium isotopes are important because they can be easily incorporated into bones due to having similar properties to other elements from Group II (i.e. calcium) and produce short lived radionuclides of high massic activity [1]. Gamma spectrometry is a useful non-destructive method that permits the simultaneous determination of many radionuclides in a bulk sample [2,3], without the need for complicated and time consuming radiochemical separations as undertaken for alpha spectrometry. However, it is limited by the weak emission probabilities of many potentially useful emission lines, the relatively poor efficiency of the High Purity Germanium (HPGe) detectors over a wide range of energy, the difficult task of precisely calibrating the efficiency of the detector, and the need to evaluate self-absorption effects [4].
Barium co-precipitation is used as a method of radium analysis due to the very similar chemical properties of barium and radium. The exploitation of the ability of barium to react with an excess of sulphat ions to produce a precipitate allows the quantitative analysis of environmental activity concentrations of radium in water [5].
In this study, rapid radiochemical separation technique for determination of isotopic 226Ra and 228Ra in water samples using co-precipitation with iron hydroxide is applied. The co-precipitation with iron hydroxide acts as a separator to remain radium and barium in the solution (as radium barium sulphate) and remove the interfering radionuclides such as 210Pb and 210Po and matrix constituents with additional co-precipitation. The activity of 226Ra and 228Ra on filter paper is determined by γ-ray spectrometry. Uncertainties associated with the 226Ra and 228Ra activities were determined.
2. Experimental
2.1. Instrumentation
All samples were prepared in the same size Marinilli beaker to obtain the similar counting geometry. Gammaspectrometry based on hyper-pure germanium detectors was used. The HPGe detector has a relative efficiency of 40% and full width at half maximum (FWHM) of 1.95 keV for 60Co gamma energy line at 1332 keV and operated with Canberra Genie 2000 software for gamma acquisition and analysis. 226Ra was then quantified by gamma counting for its 186-keV gamma emission, 351.9 (214Pb), and 609.3. For 228Ra 338.4 gamma emission, 911.1 and 583 keV was used. The gamma-spectrometers were calibrated using both 226Ra point source and potassium chloride standard solutions in the same geometry as the samples [6]. Analysis of the spectra with known amounts of 226Ra and 228Ra allowed determination of peak widths, peak resolution and counter efficiencies at different energies and it allowed determination of counter precision, accuracy and recovery, so, two samples spiked with known amounts of 226Ra and 228Ra were analyzed in this study. All samples (including standard) were counted directly in the system with a suitable counting time. Data was collected through counting different types of certified reference material and also from prepared standard source solution for 228Ra and 228Ra.
2.2. Analytical Procedures
Concentrate 5 L of water samples to 1 L, then added 1ml 133Ba tracer with activity (9.5 Bq/ml). Add 20 mg FeCl3 to water sample then stir and boil for 10 min, adjust the pH to 9 by ammonium hydroxide to separate 210Pb and 210Po. Add 5.0 ml of barium carrier (9 mg/ml) into the sample, stir with boiling to precipitate barium sulphat by adding 10 ml of 18N H2SO4 in a fine stream while stirring. Cool the solution for 30 minutes in an ice bath or allow over night settling in covered beaker [7,8], filter the solution which contains BaSO4 through a tared filter. Remove the dried filter with the precipitate. Weigh the tared filter on a tared planchet to the nearest 0.1 mg. Record the weight and time. Mount the precipitate in a holder such as a ring and disk with Mylar cover. The precipitated sample, collected on the filter, should be directly analyzed by gamma-ray spectroscopy to determine 228Ac. Then, the sample was sealed for about 4 weeks to reach secular equilibrium between radium and thorium and their progenies (222Rn). Radioactivity of 226Ra was measured by gamma spectrometry. Figure 1 illustrates the analytical procedure for the determination of radium isotopes.
2.3. Estimation of Ra Concentration in Watersamples and Uncertainty Calculation
The activity concentration of 226Ra can be calculated from the following equation for the 351.9 keVγ-ray from 214Pb and the 609.3 keVγ-ray from 214Bi, respectively. The activity concentration of 228Ra can also be calculated for 911.2 keV and 583 of the 228Ac progeny.
 (1)
(1)
where:
As: The concentration of 226Ra or 228Ra in pCi/LYs: Chemical yield determined for the sampleDs: Ingrowth factor (1 - e−λt), where λ = 0.693/t1/2; t = time interval in d between the separation of the radium from the water and the midpoint of the counting time, and t1/2 = half life of 222Rn of 3.82 dR: Net counts per s for the gamma-ray under consideration (background and Compton contributions have been subtracted; the uncertainty or error reported for that gamma ray should be noted)Fi: Branching ratio of γ-rays under consideration; 0.358 for 351.9, 0.46 for 609.3, 0.309 for 583 and 0.29 for 911.
ε: Counting efficiency of specific gamma ray and V: Sample volume L.
The procedure is based on Radium co-precipitation as Ba-RaSO4: The yield of the procedure is determined in each sample by adding a 133Ba radioactive tracer. The 356.0-keV gray is used for yield determinations. The highest energy γ-ray from 133Ba is 383.8 keV, which does not interfere with the energies 911 keV from 228Ac. The yield determined using133Ba has the advantage over the conventional gravimetric method (weighing barium sulphat BaSO4 precipitate), which could give a high yield if stable Ba was present in the water sample in an appreciable amount.
 (2)
(2)
Where:
Ys: Chemical yield determined for the sample, equation (2).
The error associated with any particular counting result is determined by the use of the following equation [9]:
 (3)
(3)
where:
r: The net count rate.
In this case we are interested in subtracting one count from another (gross counts minus background counts) and determining the resulting % error of the NCPS (Net Count Per Second) based on the standard deviation σ value. Counting instruments typically have a confidence interval of 95%. Thus equation (2) is written as:
 (4)
(4)
Where:
r0Y, rsY: The net count rate at the gamma line (Y) for the background and the sample respectivelyto, ts: The real counting time of the background and the sample respectively.
2.4. Method Validation
Comparison of the chemical recovery obtained through HPGe spectrometer provides an additional QA process. The calibration of high resolution gamma spectrometers allows for accurate determination of chemical recovery in samples for 226Ra analysis where226Ra activity levels are very high. The energy and efficiency calibration were validated by using the Certified Reference Materials (CRM), IAEA-423 and IAEA-426, present in the same counting geometry. All the samples (including standard)
were counted directly in the system with a suitable counting time. The precision and accuracy of the method were also determined. The precision achieved was 6.7% for Ra isotopes. Typical lower limits of detection for the gamma measurements were 1.6 mBq/l for 226Ra and 1.8 mBq/l for 228Ra.
3. Results and Discussion
Analysis of 226Ra and 228Ra via this developed technique showed suitability for relatively rapid, cheap and accurate analysis of drinking water for the assessment of drinking water. The filter samples were analyzed using HpGe and the results were obtained from the analysis reports of the different sample spectra. Background spectrum was determined and the intensities of the major peaks were recorded to subtract from the peaks recorded for filter samples. A number of spectra were analyzed taking the formation of secular equilibrium between 226Ra and itsdaughters214Pb and 214Bi for obtaining activity concentrations for 226Ra and secular equilibrium between 228Ra and its daughter 228Ac for obtaining activity concentrations for 228Ra.
Results for both 226Ra and 228Ra activity concentrations were presented in table1. The results indicated that the concentration of 226Ra ranged from 1.02 - 8.1 and 3.1 - 7.9 mBq·L−1 of analyzed water samples (spiked and reference samples).
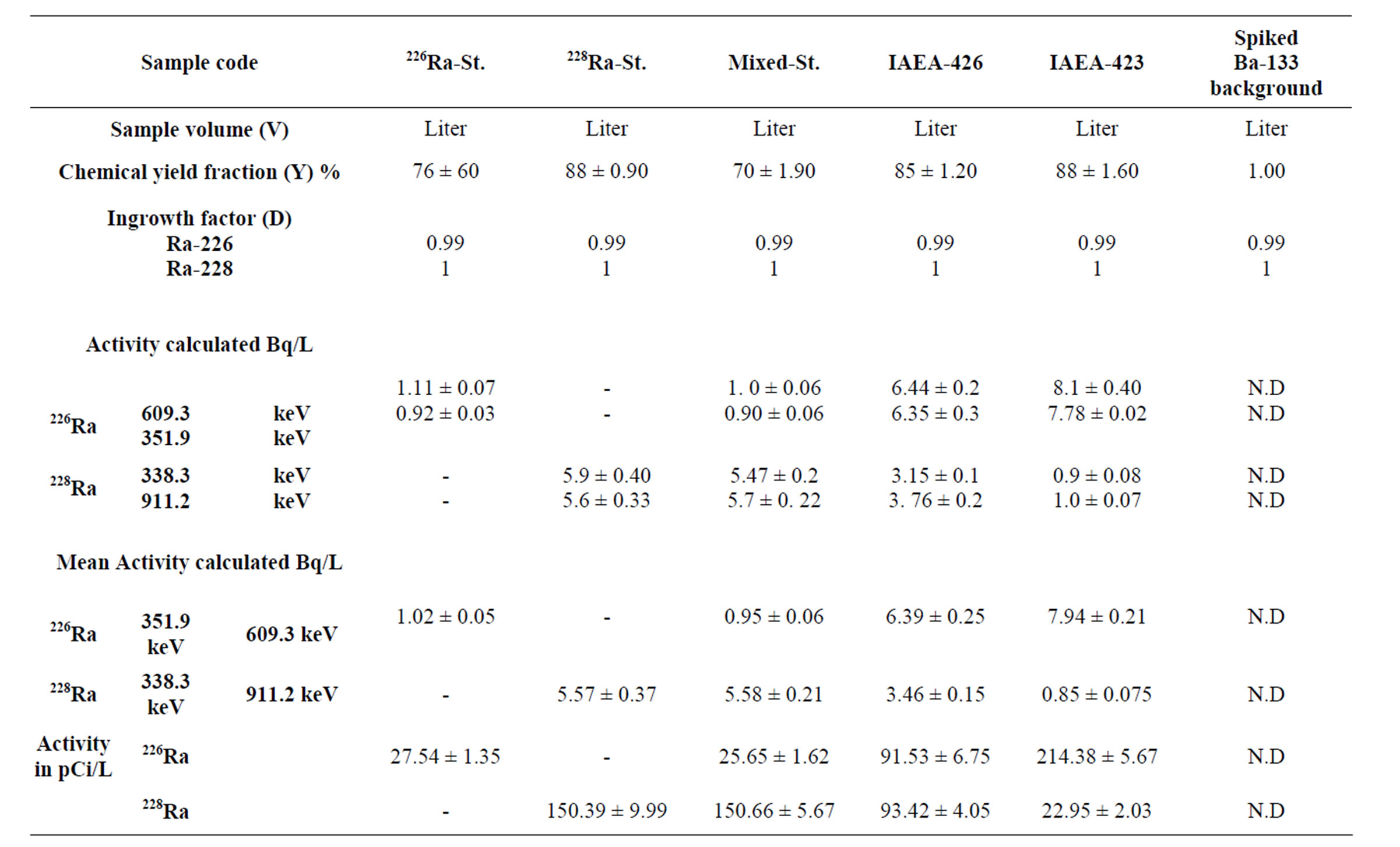
Table 1. Activity concentration of 226Ra and 228Ra in water samples.
The detection of these very low activities was made possible because we used the low background facility available at the laboratory and counting time were exceeding 3 days analyses. Results for both 226Ra and 228Ra activity concentrations determined by α-spectrometry were then compared with those obtained for the same nuclide by γ-ray spectrometry, as shown in Table 2.
Two ground water samples from different regions (Banha and Bilbies) were analyzed to determine Ra concentrations as shown in Table 2. Despite their low counting statistics, the data with very low counting rates did not rejected because these counts were obtained during long counting sessions (5 days). The mean radioactivity level of both 226Ra and 228Ra in water samples collected from Banha and Belbeis were 18.36 ± 1.08 pCi, 6.48 ± 0.68 pCi and 21.06 ± 2.16 pCi, 17.01 ± 2.57 pCi respectively. Dyck and Jonasson in 1986 study the geochemistry of radium in the environment depending on the environmental distribution of radium varies depending on its origin. Nevertheless, according to Dyck and Jonasson, the processes affecting distribution can be referring to the following main (oversimplified) equations [10].
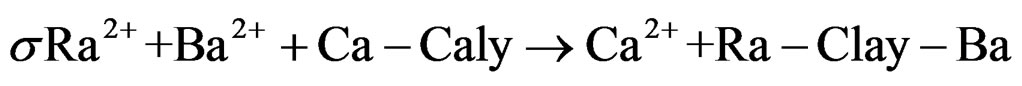
Adsorption,
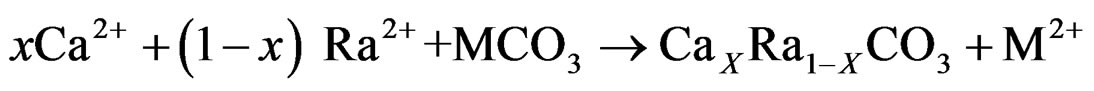
Co-precipitation,

Surface reaction
Soluble Species… etc.
Also, the physical and chemical characteristics of water mostly affected the radioactivity level of the radium in groundwater rather than the concentration of the parent naturally occurring nuclides in host rocks [11]. So, physical characteristics and major element compositions were determined (chemical contents, conductivities and different chemical compositions) as shown in Table 3 taking into consideration the effect of physical and chemical properties of the samples such as TDS, pH, soluble species, sulphate and bicarbonate. As shown in Table 3, the chemical composition of water in the first sample is Ca-Mg-HCO-type water, whereas the second one is Ca-Mg-Cl-type depending on the geological area and its composition [12].
Table 4 represents the comparison of the minimum detectable activity and efficiency obtained using the gamma spectrometry with other techniques [13] such as LSC and alpha spectroscopy for the determination of 226Ra and 228Ra. However, this method is the first trial and in the near future, more development will considered
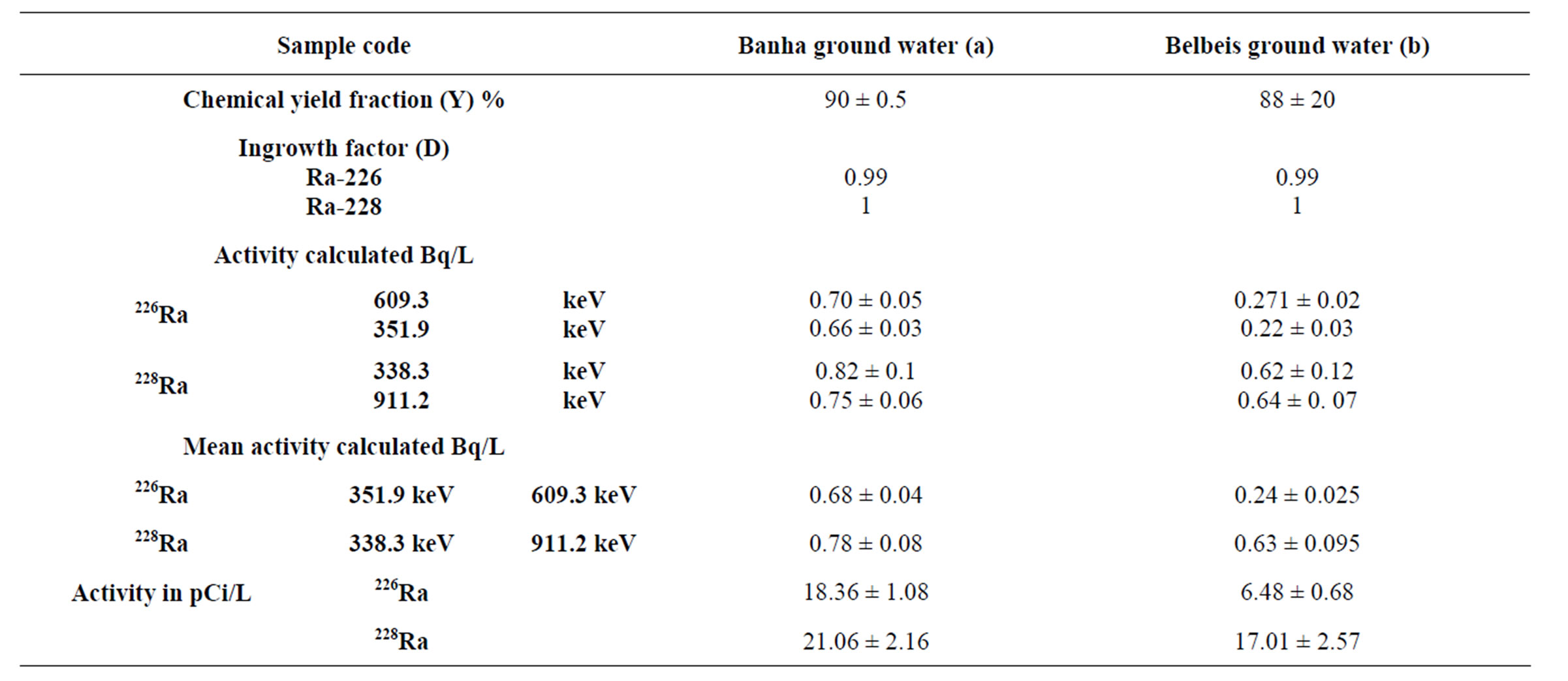
Table 2. The radioactivity levels of 226Ra and 228Ra in groundwater samples.
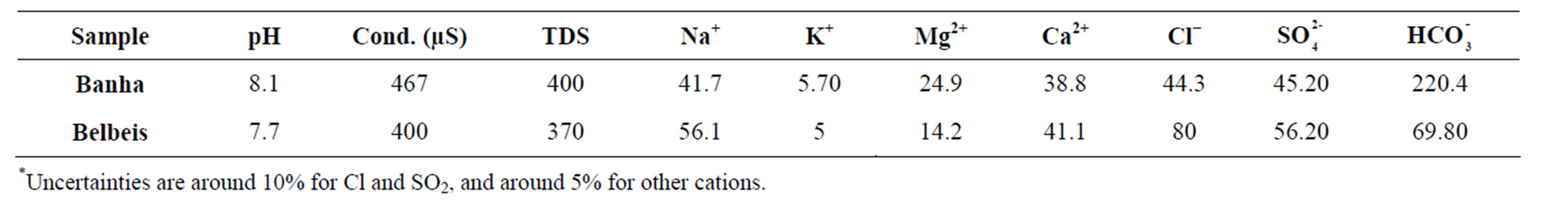
Table 3. Physical characteristics and major elements composition in the groundwater under this study.

Table 4. Minimum detectable activity and counting efficiency for alpha particle 226Ra using LSC, Alpha spectrometry and gamma spectrometry.
to optimize the measuring conditions.
4. Conclusion
Calibration of the gamma spectrometric method for 228Ra measurement via the 228Ac daughter, and development of a digestion procedure for radium sources prepared by BaSO4 co-precipitation to enable alpha spectrometric measurement of the 228Ra daughter, 228Th, were undertaken. This work is aimed to the validation of the methodology currently applied in our laboratory for the determination of 226Ra and 228Ra in environmental samples by γ-spectrometry. The methodology presented in this paper has been checked by means of its application to different water samples in which the 226Ra and 228Ra activity concentrations had been previously determine by a validated α-ray spectrometry procedure. This comparison was carried out through the application of both 226Ra and 228Ra methodologies (γ and α) to a total of 4 water samples (2 spiked and 2 references).
REFERENCES
- G. Wallner, E. Sther Herincs and S. Ayromlou, “Determination of Natural Radionuclides in Drinking Water from the Waldviertel, Austria,” Arizona Board of Regents on Behalf of the University of Arizona, USA, 2009, pp. 345-352.
- R. Rihs and M. Condomines, “An Improved Method for Ra Isotope (226Ra, 228Ra, 224Ra) Measurements by Gamma Spectrometry in Natural Waters: Application to CO2-Rich Thermal Waters from the French Massif Central,” Chemical Geology, Vol. 182, No. 2-4, 2002, pp. 409-421. doi:10.1016/S0009-2541(01)00332-1
- T. M. Semkow, P. P. Parekh, C. D. Schwenker, A. J. Khan, A. Bari, J. F. Colaresi, O. K. Tench, G. David and W. Guryn, “Low-Background Gamma Spectrometry for Environmental Radioactivity,” Applied Radiation and Isotopes, Vol. 57, No. 2, 2002, pp. 213-223. doi:10.1016/S0969-8043(02)00085-4
- P. Jodlowski, “Self-Absorption Correction in Gamma- Ray Spectrometry of Environmental Samples—An Overview of Methods and Correction Values Obtained for the Selected Geometries,” Nukleonika, Vol. 51, Suppl. 2, 2006, pp. S21-S25.
- W. C. Burnett and W. C. Tai, “Determination of Radium in Natural Waters by a Liquid Scintillation,” Analytical Chemistry, Vol. 64, No. 15, 1992, pp. 1691-1697. doi:10.1021/ac00039a012
- M. S. El-Tahawy, M. A. Farouk, F. H. Hammad and N. M. Ibrahim, “Natural Potassium as a Standard Source for the Absolute Efficiency Calibration of Germanium Detectors,” Journal of Nuclear Science, Vol. 29, No. 1, 1992, pp. 361-363.
- L. Zikovsky, “Determination of Alpha-Radioactivity in Ground Water by Precipitation of Alpha-Emitters with Sulphates and Hydroxides,” Journal of Radio-Analytical and Nuclear Chemistry, Vol. 251, No. 2, 2002, pp. 329- 331. doi:10.1023/A:1014845117078
- W. C. Lawrie, J. A. Desmond, D. Spence, S. Anderson and C. Edmondson, “Determination of Radium-226 in Environmental and Personal Monitoring Samples,” Applied Radiation and Isotopes, Vol. 53, No. 1, 2000, pp. 133-137. doi:10.1016/S0969-8043(00)00168-8
- K. Horiuchi, T. Ishii, and Y. Murakami, “Results on the Simultaneous Determination of 222Rn, 220Rn and 226Ra Contents in Mineral Springs of Izu Peninsula,” Journal of Balneological Society of Japan, Vol. 30, No. 30, 1979, pp. 84-89.
- W. Dyck and I. R. Jonasson, “Geochemistry of Radon and Its Application to Prospecting from Minerals, Internal Report, Geological Survey of Canada, Ottawa,” Internal Report in Geological Survey of Canada, 1986.
- IAEA, “The Environmental of Radium,” IAEA Technical Report Series 310, IAEA, Viena, 1990.
- L. Žarmand, “Hydrological Considerations on Some Mineral Sources Massif Central,” Annales de l’Institut d’Hydrologie et de Climatologie, Vol. 9, No. 1, 1934, pp. 221- 241.
- J. Floeckher, “High Throughput Screening of Samples Containing Alpha & Beta Radionuclides: An Overview of Methods,” 2011. http://ww2.packardinstrument.com
NOTES
*Corresponding author.

