Journal of Water Resource and Protection
Vol. 1 No. 4 (2009) , Article ID: 762 , 4 pages DOI:10.4236/jwarp.2009.14035
Simultaneous Determination of Chemical Oxygen Demand (COD) and Biological Oxygen Demand (BOD5) in Wastewater by Near-Infrared Spectrometry
1School of Chemistry and Chemical EngineerWing, Southwest China University, Chongqing, China
2School of Chemistry and Chemical Engineering, Yangtze Normal University, Chongqing, China
E-mail: flyjd6400@sina.com
Received August 1, 2009; revised September 2, 2009; accepted September 8, 2009
Keywords: Near-Infrared Spectrometry, Wastewater, Biological Oxygen Demand, Chemical Oxygen Demand
ABSTRACT
To rapidly determine the pollution extent of wastewater, the calibration models were established for determination of Chemical Oxygen Demand and Biological Oxygen Demand in wastewater by partial least squares and near infrared spectrometry of 120 samples. Spectral data preprocessing and outliers’ diagnosis were also discussed. Correlation coefficients of the models were 0.9542 and 0.9652, and the root mean square error of prediction (RMSEP) were 25.24 mg·L-1 and 12.13 mg·L-1 in the predicted range of 28.40~528.0 mg·L-1 and 16.0~305.2 mg·L-1 for Chemical Oxygen Demand and Biological Oxygen Demand, respectively. By statistical significance test, the results of determination were compared with those of standard methods with no significant difference at 0.05 level. The method has been applied to simultaneous determination of Chemical Oxygen Demand and Biological Oxygen Demand in wastewater with satisfactory results.
1. Introduction
Chemical Oxygen Demand (COD) is the amount of oxygen consumed by the organic compounds and inorganic matter which were oxidized in water. Biological Oxygen Demand (BOD5) is the amount of oxygen consumed by the organic and inorganic compounds which were oxidized by biological-oxidation effect in a certain condition. Both of them reflect the pollution degree of the water, and are the comprehensive index of the relative content of organics. As the main comprehensive index of the organic pollution, COD and BOD5 are important in the control of the total content of pollution and the management of water environment. So it is significant to farther research and develop the simple and rapid method for the determination of COD and BOD5.
At present, potassium dichromate method is generally used to determine the value of COD [1] at home and abroad and other methods like spectrophotometry [2] and coulometric method [3] were reported. The method of BOD5 determination contains routine measurementbiosensor measurement [4] and spectral analysis [5]. Some of these methods of determination COD and BOD5 has secondary pollution as added toxical reagents for COD determination, some method need pretreatment for BOD5 determination, most of them need long time as five days at least. While the NIRS has many advantages, such as without pretreatment and destroy the sample, no pollution, convenient and fast, on-line detection, simultaneity multi-component, good reproducibility, etc. Since 1990s, NIR spectrum was developed fast and became the most conspicuous spectral analysis technique. It has been the official authorized method in many fields such as agriculture and food. It has been widely used in textile, polymer, drugs, petrochemical industry, biochemistry and environmental protection [6]. COD and BOD5 of wastewater was determined with the method in this experiment, chemical value of BOD5 and COD on the basis of national standard method was determined as the base data. Calibration model of BOD5 and COD was built with PLS1 and Unscrambler software. The pretreatment such as smoothing processing, first derivative and second derivative was compared, outliers diagnosis was also discussed. Further, the model was externally inspected. By statistical significance test, the results of determination were compared with those of standard methods with no significant difference. The method has been applied to simultaneously determine of Chemical Oxygen Demand and Biological Oxygen Demand in wastewater with satisfactory results.
2. Experiment Part
2.1. Instrument
Hitachi U-4100, detector type: PbS; 1cm quartz sample cell; biochemical constant temperature incubator; The Unscrambler 9.7 of CAMO Company.
2.2. Collection of Wastewater
The sample of wastewater collected from domestic sewage in which the Organics consuming oxygen, degradable organics, reducible inorganics, ammonia nitrogen, etc are the main pollutants in Fuling (Chong Qing). 120 samples were collected with glassware in different place. The method was offered by environmental monitoring.
2.3. Experiment Method
2.3.1. Determination Chemical Value of BOD5 and COD
120 samples (110 for calibration set and 10 for prediction set ) were deposited for 15 min. The value of BOD5 and COD was detected separately, and then the NIR spectrum was collected. Determination of COD value was 16.0~305.2 mg·L-1 was got by the method of reference [1], and Determination of BOD5 value was 28.4~528.0 mg·L-1 was got by the method of reference [7].
2.3.2. Collection of Spectrum
The spectrum was collected in the condition of 1cm quartz sample cell with air as reference, wavelength range was 800~1800 nm, wavelength spacing was 2 nm, slit width is 2 nm. scanning speed is 1500 nm/s, every sample was scanned 3 times and got average value. NIR Spectra of wastewater is shown in Figure 1.
2.3.3. PLS Mode’s Establishment
The NIR spectrum was transformed into JCAMP-DX format, and introduced into analyze software of the Unscrambler 9.7; then the spectrum was pretreated. Taking the chemical value of BOD5 and COD into the software. Calibration model of BOD5 and COD was built with PLS1 and Cross-validation.
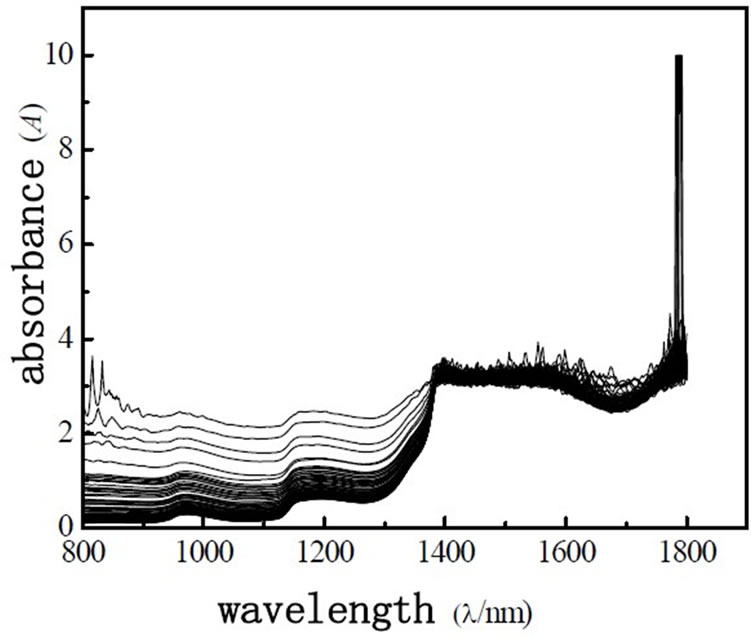
Figure 1. Near infrared spectra of 120 wastewater samples.
3. Results and Discussion
3.1. Choice of Model Algorithm
In the analysis of NIR spectrum, model algorithm includes MLR, PLS, PCA, ANN, TP, etc. The basic idea of model algorithm is that make use of total spectrum information of NIR spectrum, to eliminate influences of spectrum peak’s overlap and complex background. PLS has been widely used in NIR spectrum analysis, and has been a common standard method. PLS method contains PLS1 and PLS2. To the complex system, PLS1 method optimizes each components, so the prediction results of PLS1 is generally better than the prediction results of PLS2 [8] through Cross-validation experiment shown. So, PLS1 method is used in this experiment.
3.2. Choice of Spectrum Pretreatment Method
Spectral signal that is determined by detector contains spectral information and several noises. Smoothing processing, first derivative and second derivative was compared in this experiment. Parameters of model are shown in the Table 1. From Table 1, the smoothing processing is best to BOD5 and COD in the spectrum pretreatment.
3.3. Optimization of Model
As shown in Figure 1, spectra of wastewater is not concentrating, it shows that there are anomalous points in the spectra. So the anomalous points should be rejected when we build the model. This experiment use interactive check method, optimize the model step by step, determine the PCs, as far as get the best model. There are two methods for judging and rejecting anomalous points or influential point. The first is mahalanobis distance method, and the second method is that the influence of model is investigated by Leverage [9], it is also called “reject one” cross-check method. It is combined diagnosed by Leverage and studentized residual, the influential point rejected step by step. This experiment use Leverage and Residual to reject the anomalous points of spectrum and chemical value. Leverage is set as 3.0, error of chemical value is 6.0%, optimize the model by rejecting anomalous points gradually, get the best PLS1 regression model of determining BOD5 and COD. The correlation coefficient of best model about COD and BOD5 are 0.9542 and 0.9652, respectively. The predicting relative deviation are 25.24 mg·L-1 and 12.13 mg·L-1. The scatter plot of COD and BOD5 between NIR predicted and measured values are shown in Figure 2 and Figure 3, the changes of residual variance with PCs are shown in Figure 4 and Figure 5.
3.4. Verification of Model
The estimated method is not only parameters’ evaluating method of model itself, but also external check evaluating method of model by actual prediction. The 10 wastewater samples of prediction set were predicted about BOD5 and COD, the results are shown in Table 2. Matched bilateral t test was made between prediction result and standard method by origin 6.0 software. The results are tCOD=0.6437, tBOD5= 0.5297, pCOD= 0.6092, pBOD5= 0.6092, pCOD and pBOD5 are much larger than 0.05. When significance level is larger than 0.05, two kinds of determination methods have not significant difference, which shows that there is no system error.
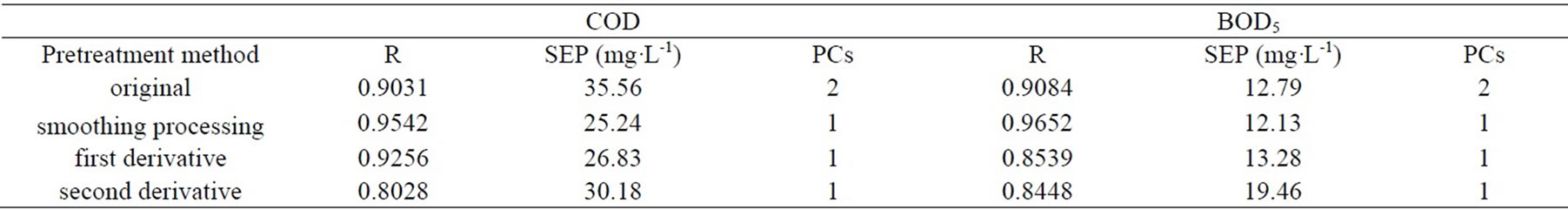
Table 1. Mathematical statistics results for calibration models of BOD5 and COD.
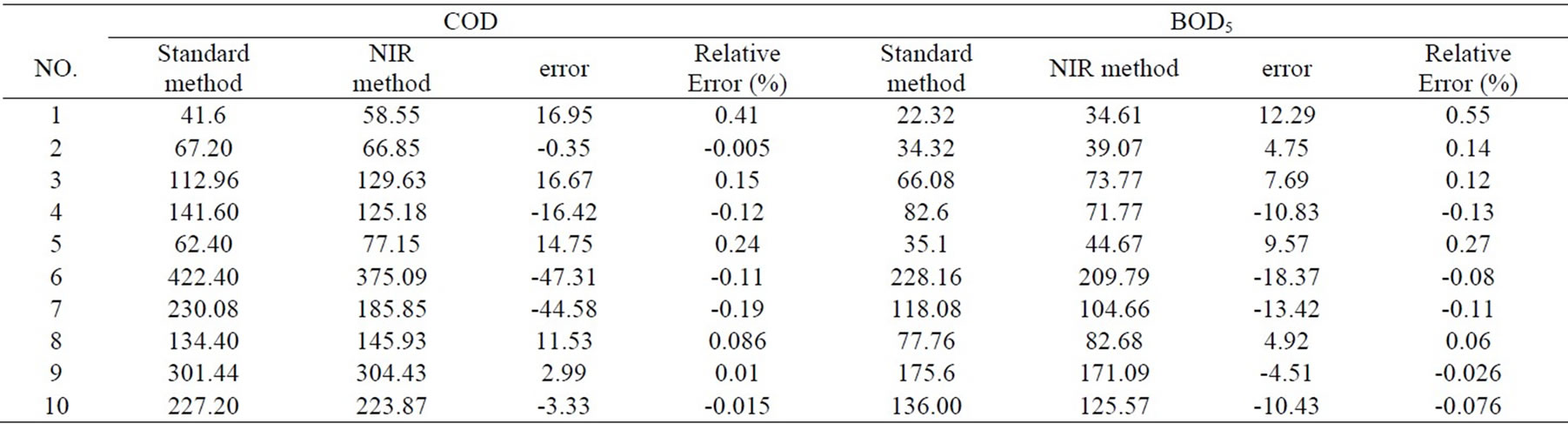
Table 2. Predicted results of the prediction samples.
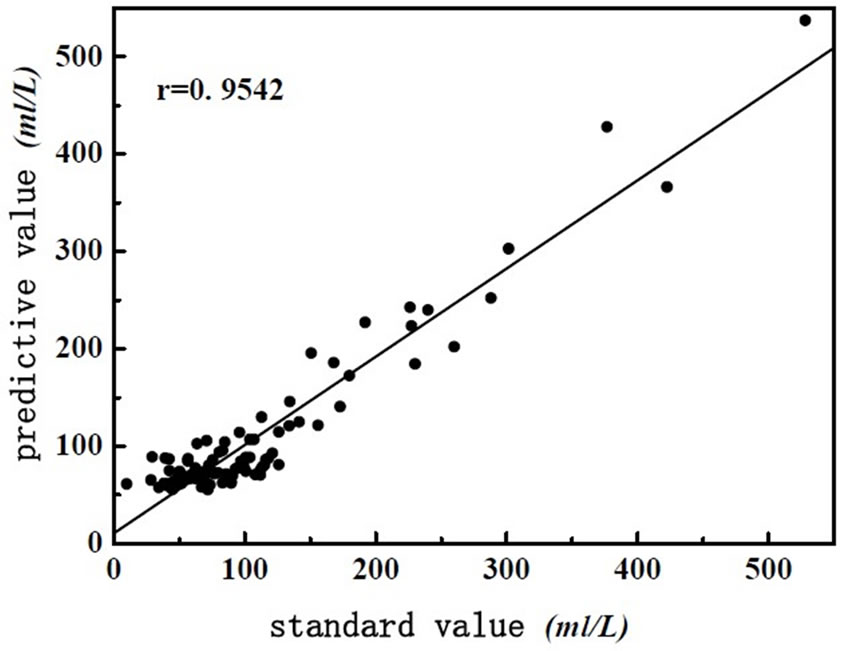
Figure 2. The scatter plot of COD between NIR predicted and measured values.
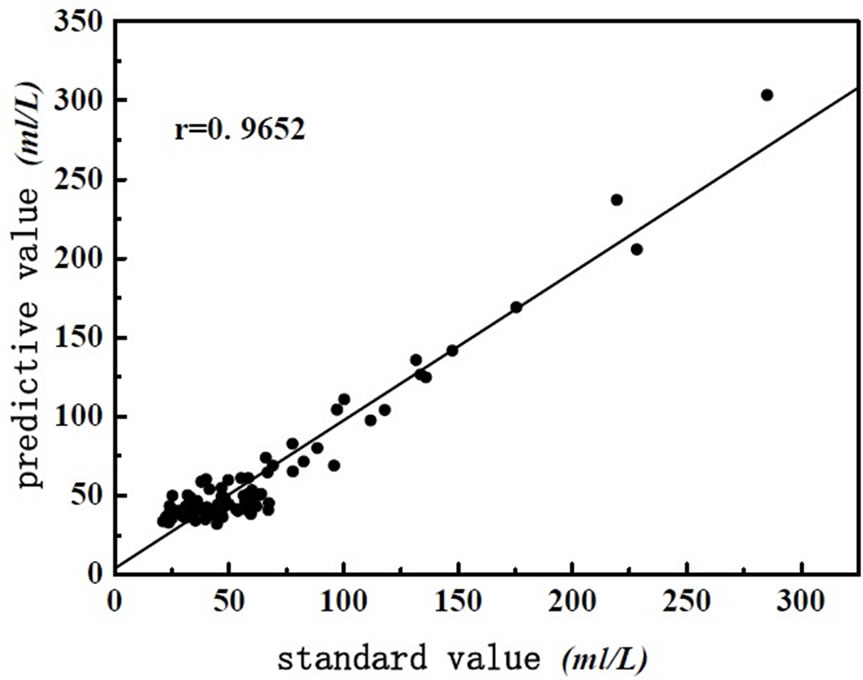
Figure 3. The scatter plot of BOD5 between NIR predicted and measured values.
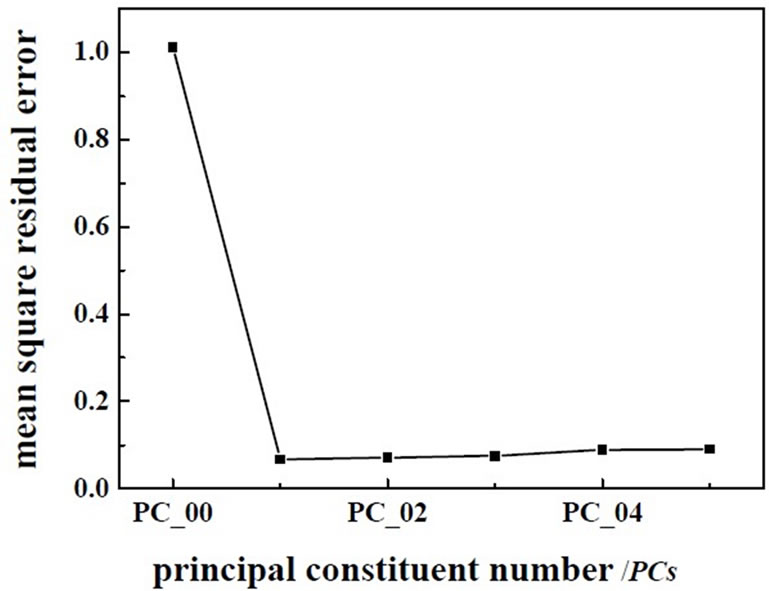
Figure 4. The changes of residual variance with PCs.
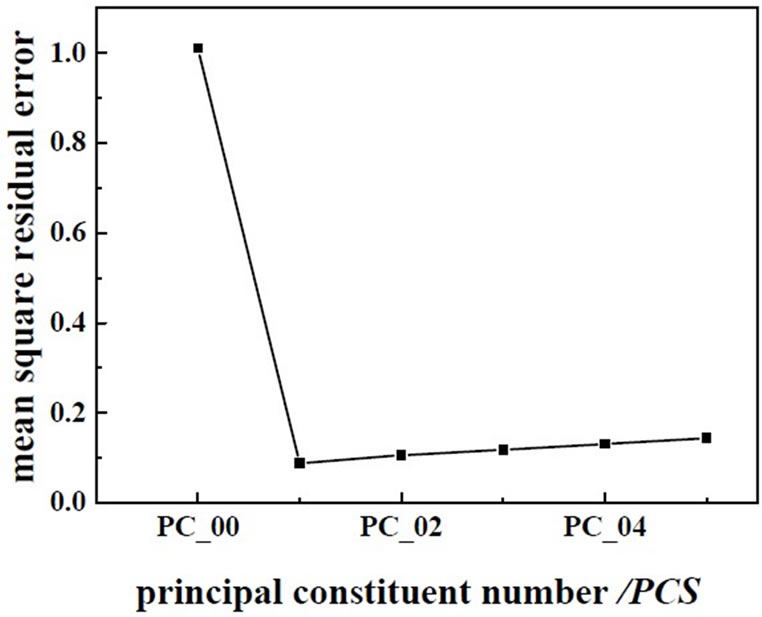
Figure 5. The changes of residual variance with PCs.
3.5. Precision Test
Precision test has been made in this experiment for checking the stability of instrument and repeatability of samples’ results. One sample was scanned 10 times, So, 10 NIR spectra were got, and then these 10 spectra were predicted by the model built already. RSD (n=10) of prediction (BOD5 and COD) are 1.48% and 1.66%, respectively, the precision is good enough. The result indicates that the repeatability of samples’ determination is very good.
The result indicates that the prediction effect of simultaneous determination about BOD5 and COD by calibration model is very good. But temperature and turbidity maybe influence the prediction result in the actual experiment. Nonlinear problem should be noticed, only the linear correction method as PLS is hard to set a model that it meet requirements of analysis index. We could try some nonlinear correction method as artificial neural network method to compare with linear correction method as PLS. All the works above need further study.
REFERENCES
- Chinese government standard GB11914-1989, “Determination of COD in water by dichromate titration method [S].”
- J. G. Zhan, G. Wei, and R. C Xiong, “Quick evaluation of chemical oxygen demand based on spectrophotometric and multi-linear analysis [J],” Journal of Beijing University of Chemical Technology (Natural Science Edition), Vol. 34, No. 7, pp. 389–392, 2007. (In Chinese).
- H. B. Yu, H. Wang, et al., “Amperometric determination of chemical oxygen demand using boron-doped diamond (BDD) sensor [J],” Electrochemistry Communications, Vol. 9, No. 9, pp. 2280–2285, 2007.
- Kwok Nga-Yan, S. J. Dong, and W. H. Lo, “An optical biosensor for multi-sample determination of biochemical oxygen demand (BOD) [J],” Sensors and Actuators B: Chemical, Vol. 110, No. 2, pp. 289–298, 2005.
- D. M. Reynolds and S. R. Ahmad, “Rapid and direct determination of wastewater BOD values using a fluorescence technique [J],” Water Research, Vol. 31, No. 8, pp. 2012–2018, 1997.
- Y. L. Yan, “Basic and application of Near Infrared Spectra [M],” Beijing: China light industry publishing company, Vol. 3, 2005.
- Chinese government standard GB7488-1987, “Determination of BOD5 in water by dilute and inoculation method [S].”
- L. Xu and X. G. Shao, “Chemomtrics [M],” Beijing: Science publishing company, Vol. 258, 2004.
- S. Weisberg, “Applied Linear Regression [M],” New York: John Wiley & Sons. Vol. 168, 1985.

