Agricultural Sciences
Vol.5 No.8(2014), Article
ID:48085,6
pages
DOI:10.4236/as.2014.58075
Ethyl Methanesulfonate (EMS)-Mediated Mutagenesis of Cucumber (Cucumis sativus L.)
Lina Wang1,2, Bing Zhang1, Jinrui Li1,2, Xiaoyu Yang3, Zhonghai Ren1,2*
1College of Horticulture Science and Engineering, Shandong Agricultural University, Tai’an, China
2State Key Laboratory of Crop Biology, Tai’an, China
3School of Life Sciences, Faculty of Science, The Chinese University of Hong Kong, Hong Kong, China
Email: *zhren@sdau.edu.cn
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 15 April 2014; revised 18 May 2014; accepted 29 May 2014
ABSTRACT
Ethyl methanesulfonate (EMS) is a stable and effective chemical mutagen. In this study, cucumber (Cucumis sativus L. cv. “Shannong No. 5”) seeds were treated by 1% EMS for 12 h, 24 h and 48 h to optimize EMS mutagenesis and determined median lethal dose of EMS (1% EMS and 24 h) for “Shannong No. 5”. After treated by 1% EMS for 24 h, 541 M1 plants were grown in greenhouse for phenotype investigation. The fertility of M1 cucumbers was very low, and only 79 lines produced seeds after self crossing. 60 independent M2 families comprising 600 M2 plants were investigated for phenotypic alteration, and 11 individual mutant lines were isolated into six groups: short-fruit mutants, long-fruit mutants, small-flower mutants, big-flower mutants, opposite-tendril mutants and clustered-leaf mutants. The mutation frequency was 18.3%. Two selected representatives, short-fruit mutants and clustered-leaf mutants, showed 1:3 of segregation ratio in M2 populations. This ratio is consistent with classic Mendelian model, indicating that the two kinds of mutants may be controlled by a single recessive gene, respectively. Long-fruit phenotype was stably inherited and no segregation was observed in M3 generation, indicating that this mutant line may be homozygous.
Keywords:Cucumber, Ethyl Methanesulfonate, Mutagenesis

1. Introduction
In plant breeding, a great challenge is the collection or development of a large amount of germplasm resources. To this end, several strategies, such as T-DNA or transposon insertional mutagenesis [1] -[3] chemically-induced mutagenesis [4] [5] , have been used to develop different germplasms. However, the mutational spectrum of insertional mutagenesis with effect on gene function is mostly limited to gene knock-out disruptions, which often resulted in failure of functional dissection of mutants when mutations happen to lethal or highly pleiotropic genes [6] . Furthermore, the size of saturated populations is extremely large because each line carries only a rather small number of mutations [7] .
Compared with insertional mutation, chemically-induced mutation has been shown some advantages such as high efficiency because each individual line can bear single point missense and nonsense substitutions in hundreds of genes [8] . Therefore, an allelic series of induced mutations with different effects on gene function can be easily isolated by screening a relatively small population of mutated plants [7] . Among the chemical mutagens, ethyl methanesulfonate (EMS) is considered as an effective one because it can from adducts with nucleotides efficiently, resulting in mispairing among these nucleotides with their complementary bases and thus introducing base changes after replication [5] .
Cucumber is one of the most important horticultural crops and is now grown as either a fresh or processed vegetable in the world. Its fruits are rich sources of high quality protein and vitamins which provide an excellent supplement to the lower quality cereal or root and tuber protein consumed in much of the world [9] . Cucumber fruits vary markedly in size, shape and color, and thus always attract attentions from cucumber breeders. However, germplasm resources are still the big bottleneck for the present cucumber breeding. Here we determined the median lethal dose of EMS for cucumber cultivar “Shannong No. 5” and reported cucumber mutants of fruit, flower, tendril and plant size induced by EMS.
2. Materials and Methods
2.1. Plant Materials
Cucumber (Cucumis sativas L. cv. “Shannong No. 5”) seeds were kindly provided by Dr. Chenxing Cao in Shandong Agricultural University, China, and were used in this research.
2.2. Determination of Median Lethal Dose
One hundred of cucumber seeds were soaked in 20 ml of distilled water at low speed shaker for 30 min, and then EMS (Sigma, USA) was added to the distilled water at the final concentration of 1% (v/v). These seeds were treated in 1% EMS solution for 12 h (EMS-1), 24 h (EMS-2) and 48 h (EMS-3) at low speed shaker, respectively. The treated seeds were incubated at 28˚C for 15 h after being washed with 1 M Na2S2O3 solution, 100 mM Na2S2O3 solution and distilled water, respectively. Seeds not treated by EMS were incubated as control under the same conditions as EMS-treated seeds. The lethal dose (LD) was calculated as follow:
LD% = (1 − Germination rates of treated seeds/Germination rates of Control) × 100
2.3. EMS-Mediated Mutagenesis
Mutant populations were constructed based on the work flow in Figure 1. In brief, wild type (WT) seeds (M0) were treated with 1% EMS solution for 24 h. The treated seeds were planted in greenhouse of Shandong Agricultural University, China, according to randomized block design. Each M1 plant was harvested separately and the seeds were sown in the next season in the greenhouse to grow M2 generation in a randomized block design followed by M3 generation. Mutants were detected by observing the plants through the whole growth stage in all generations.
2.4. Statistical Analysis
The results were subjected to one-way analyses of variance (ANOVA) and LSD test with SAS software (Statistica version 6.1, StatSoft, St. Tulsa, OK, USA) and presented as means ± standard error (SE) of twenty replicates. Different letters indicate a significant difference from control at 0.01 probability level.
3. Results and Discussion
Median lethal dose (LD50) is a critical parameter for chemically induced mutagenesis. This value is determined by both mutagen concentration and treatment time and varies in different species and even different cultivars [10] -[13] . Thus we tried to find LD50 of EMS for “Shannong No. 5” at first. With the increase of treatment time by 1% EMS, the number of survival seeds was decreased (Table 1). When the seeds were treated with 1% EMS for 24 h, LD value was 54%, which is very close to LD50. Therefore, the combination of 1% EMS and 24 h was used for subsequent mutagenesis of cucumber.
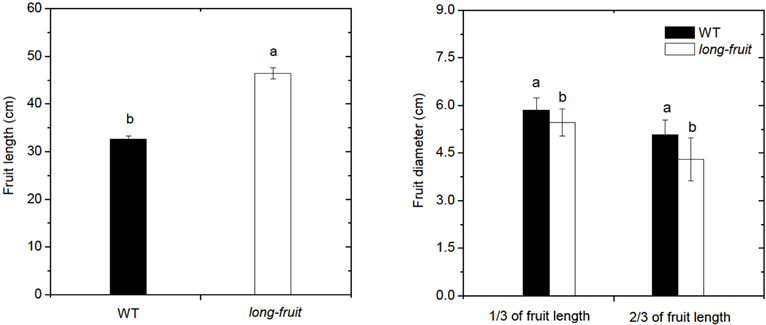
After induction by 1% EMS for 24 h, the treated seeds (M1) were planted in the greenhouse. Only 79 out of 541 M1 plants produced M2 seeds. Fertility rate of M1 plants was 14.6%. Low fertility is the common phenomenon observed in mutant plants by physical and chemical mutagenesis possibly due to severe damage of mutagens to plant genetic materials [5] [14] . The mutation frequency is one of the most dependable parameters for evaluating the genetic effects of mutagenic treatments [15] [16] . It was estimated by dividing the number of segregating M1 plant progeny with the determinate character by the total number of progeny. For the material grown in bulk, the mutant frequency was estimated by dividing the total number of mutants confirmed by the total number of M2 plants in the bulk population studied [5] . In this study, sixty lines (M2) from M1 cucumbers were raised for further investigation of phenotypes and calculation of mutation frequency. Eleven out of the sixty lines showed mutations including short-fruit mutants, long-fruit mutants, small-flower mutants, big-flower mutants, opposite-tendril mutants and clustered-leaf mutants compared with wild type plants (Figure 2). The mutation frequency was 18.3%. The main reason for this low mutation frequency is that most of the mutants bearing multi-mutational events may be lethal in the first generation [16] . The short-fruit mutants and clustered-leaf mutants showed 1:3 of segregation ratio in M2 populations (Table 2 and Table 3). This ratio is consistent with classic Mendelian model, indicating that these mutant phenotypes may be controlled by a single recessive gene, respectively. Long-fruit phenotype was inherited stably and no segregation was observed in M3 generation (Figure 3). This evidence suggests that this mutant line may be homozygous at M3 generation.
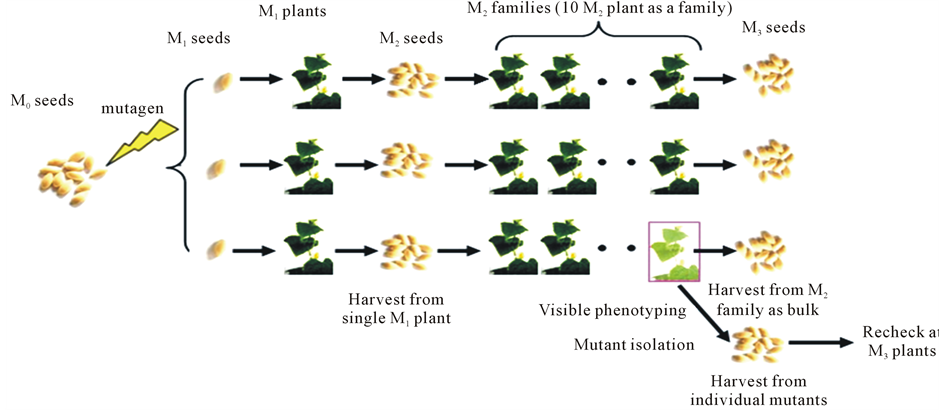
Figure 1. Flow chart for the construction of mutagenized populations.
Table 1. Effects of EMS treatments on the rate of survival in M1 plants and lethal dose.

Figure 2. Mutant lines of cucumber by EMS observed at M2 generation. Wide type (WT) and shortfruit (A1 and A2); wild type (WT) and long-fruit (B1 and B2); wild type (WT) and small-flower (C); wild type (WT) and big-flower (D); wild type (WT) and opposite-tendril (E1 and E2); and wild type (WT) and clustered-leaf (F). Scale bar represents 3 cm.
Table 2. χ2 of cucumber short-fruit mutant line in M2 population.
Note: ,
,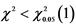 . The segregation ratio of short-fruit mutant line is consistent with classic Mendelian model.
. The segregation ratio of short-fruit mutant line is consistent with classic Mendelian model.
Table 3. χ2 of cucumber clustered-leaf mutant line in M2 population.
Note: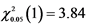 ,
,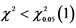 . The segregation ratio of clustered-leaf mutant line is consistent with classic Mendelian model.
. The segregation ratio of clustered-leaf mutant line is consistent with classic Mendelian model.
 (a) (b)
(a) (b)
Figure 3. Fruit length (a) and diameter (b) of wild-type cucumbers and M3 long-fruit mutants. Fruit diameter was measured at 1/3 of fruit length and 2/3 of fruit length from fruit tip. Vertical bars represent the standard errors (n = 20). Different letters indicate significant differences between wild type plants (WT) and long-fruit mutants at 0.01 level.
All these mutant lines are now still under further investigation and we believe that these mutants can contribute greatly to our future cucumber breeding.
Acknowledgements
This work was supported by Research Award Fund for Outstanding Middle-aged and Young Scientist of Shandong Province (NO. BS2011NY010), Research Fund for the Doctoral Program of Higher Education of China (20113702120008) and the China Agriculture Research System (CARS-25-D).
References
- Alonso, J.M., Stepanova, A.N., Leisse, T.J., Kim, C.J., Chen, H., Shinn, P., Stevenson, D.K., Zimmerman, J., Barajas, P., Cheuk, R., Gadrinab, G., Heller, C., Jeske, A., Koesema, E., Meyers, C.C., Parker, H., Prednis, L., Ansari, Y., Choy, N., Deen, H., Geralt, M., Hazari, N., Hom, E., Karnes, M., Mulholland, C., Ndubaku, R., Schmidt, I., Guzman, P., Aguilar-Henonin, L., Schmid, M., Weigel, D., Carter, D.E., Marchand, T., Risseeuw, E., Brogden, D., Zeko, A., Crosby, W.L., Berry, C.C. and Ecker, J.R. (2003) Genome-Wide Insertional Mutagenesis of Arabidopsis Thaliana. Science, 301, 653-657. http://dx.doi.org/10.1126/science.1086391
- Rosso, M.G., Li, Y., Strizhov, N., Reiss, B., Dekker, K. and Weisshaar, B. (2003) An Arabidopsis Thaliana T-DNA Mutagenized Population (GABI-Kat) for Flanking Sequence Tag-Based Reverse Genetics. Plant Molecular Biology, 53, 247-259.
- An, G., Jeong, D.H., Jung, K.H. and Lee, S. (2005) Reverse Genetic Approaches for Functional Genomics of Rice. Plant Molecular Biology, 59, 111-123. http://dx.doi.org/10.1007/s11103-004-4037-y
- Uchida, N., Sakamoto, T., Kurata, T. and Tasaka, M. (2011) Identification of Ems-Induced Causal Mutations in a Non-Reference Arabidopsis Thaliana Accession by Whole Genome Sequencing. Plant and Cell Physiology, 52, 716-722. http://dx.doi.org/10.1093/pcp/pcr029
- Girija, M., Dhanavel, D. and Gnanamurthy, S. (2013) Gamma Rays and EMS Induced Flower Color and Seed Mutants in Cowpea (Vigna unguiculata L. Walp). Advances in Applied Science Research, 4, 134-139. http://connection.ebscohost.com/c/articles/90260513/gamma-rays-ems-induced-flower-color-seed-mutants-cowpea-vigna-unguiculata-l-walp
- Martín, B., Ramiro, M., Martínez-Zapater, J.M. and Alonso-Blanco, C. (2009) A High-Density Collection of EMS-Induced Mutations for TILLING in Landsberg Erecta Genetic Background of Arabidopsis. BMC Plant Biology, 9, 147. http://dx.doi.org/10.1186/1471-2229-9-147
- Østergaard, L. and Yanofsky, M.F. (2004) Establishing Gene Function by Mutagenesis in Arabidopsis Thaliana. Plant Journal, 39, 682-696. http://dx.doi.org/10.1111/j.1365-313X.2004.02149.x
- Henikoff, S., Till, B.J. and Comai, L. (2004) TILLING. Traditional Mutagenesis Meets Functional Genomics. Plant Physiology, 135, 630-636. http://dx.doi.org/10.1104/pp.104.041061
- Vora, J.D., Rane, L. and Kumar, S.A. (2014) Biochemical, Anti-Microbial and Organoleptic Studies of Cucumber (Cucumis sativus). International Journal of Science and Research, 3, 662-664. http://www.ijsr.net/archive/v3i3/MDIwMTMxMjc0.pdf
- Garcia, D., Saingery, V., Chambrier, P., Mayer, U., Jürgens, G. and Berger, F. (2003) Arabidopsis Haiku Mutants Reveal New Controls of Seed Size by Endosperm. Plant Physiology, 131, 1661-1670. http://dx.doi.org/10.1104/pp.102.018762
- Qu, L.J. and Qin, G. (2014) Generation and Identification of Arabidopsis EMS Mutants. Methods in Molecular Biology, 1062, 225-239. http://dx.doi.org/10.1007/978-1-62703-580-4_12
- Serrat, X., Esteban, R., Guibourt, N., Moysset, L., Nogués, S. and Lalanne, E. (2014) EMS Mutagenesis in Mature Seed-Derived Rice Calli as a New Method for Rapidly Obtaining TILLING Mutant Populations. Plant Methods, 10, 5. http://dx.doi.org/10.1186/1746-4811-10-5
- Uchida, N., Sakamoto, T., Tasaka, M. and Kurata, T. (2014) Identification of EMS-induced Causal Mutations in Arabidopsis Thaliana by Next-Generation Sequencing. Methods in Molecular Biology, 1062, 259-270. http://dx.doi.org/10.1007/978-1-62703-580-4_14
- Kim, Y., Schumaker, K.S. and Zhu, J.K. (2006) EMS Mutagenesis of Arabidopsis. Methods in Molecular Biology, 323, 101-103. http://dx.doi.org/10.1385/1-59745-003-0:101
- Kharkwal, M.C. (1999) Induced Mutations in Chickpea (Cicer arietinum L.) III. Frequency and Spectrum of Viable Mutations. Indian Journal of Genetics and Plant Breeding, 59, 451-464. http://www.indianjournals.com/ijor.aspx?target=ijor:ijgpb&volume=59&issue=4&article=010
- Waghmare, V.N. and Mehra, R.B. (2001) Induced Chlorophyll Mutations, Mutagenic Effectiveness and Efficiency in Lathyrus sativus L. Indian Journal of Genetics and Plant Breeding, 61, 53-56. http://www.indianjournals.com/ijor.aspx?target=ijor:ijgpb&volume=61&issue=1&article=014
NOTES
![]()
*Corresponding author.


