American Journal of Plant Sciences
Vol.5 No.15(2014), Article
ID:47637,13
pages
DOI:10.4236/ajps.2014.515240
Diagnosis of Sugarcane White Leaf Disease Using the Highly Sensitive DNA Based Voltammetric Electrochemical Determination
Porntip Wongkaew1*, Suta Poosittisak2
1Plant Pathology Division, Faculty of Agriculture, Khon Kaen University, Khon Kaen, Thailand
2Department of Chemistry, Faculty of Science, Khon Kaen University, Khon Kaen, Thailand
Email: *porwon@kku.ac.th
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 17 April 2014; revised 10 June 2014; accepted 1 July 2014
ABSTRACT
A simple and highly sensitive analysis by electrochemical voltammetry has been developed for diagnosis of the most destructive crop disease in Thailand known as sugarcane white leaf (SCWL). Determination of the corresponding DNA interaction has been obtained from the voltammetric signals of electroactive redox methylene blue (MB) by means of cyclic and differential pulse voltammetry. In this study, a chitosan-modified glassy carbon electrode (GCE) was created by self-assembly to produce electrostatic platform for effective immobilization of the DNA. Fabrication of SCWL-DNA hybridization detection system was performed by immobilizing the ssDNA probe as a specific sensor onto chitosan-modified GCE. Hybridization of complementary DNA from the real samples could then be detected by its respective MB signal. This fabricated DNA probe sensor was shown to be capable for discriminative identification among the DNAs from SCWL plants, mosaic virus infected sugarcane and healthy sugarcane plants. Relationship between the specific hybridization signal and DNA target concentration was also observed under optimal condition. The detection limit of 4.709 ng/µl with the regression coefficient (R2) of 0.998 and overall RSD of 2.44% were obtained by response curve fit analysis. The actual SCWL-ssDNA immobilization and hybridizing event were subsequently confirmed by an observation under atomic force microscope. Thus these experiments demonstrate the first successful and effective DNA based voltammetric electrochemical determination for a verification of the specific pathogenic infection within plants from the real epidemic field.
Keywords:Sugarcane White Leaf Disease, Chitosan-Modified Glassy Carbon Electrode, DNA Hybridization Sensor, Methylene Blue, Voltammetric Signal, Atomic Force Microscope

1. Introduction
Sugarcane white leaf (SCWL) has long been considered as the most important disease of economic crops in Thailand due to its destructive capability to the country’s profit from agricultural products. The disease is one of the most serious factors of a low productivity in cane and sugar production of Thailand where the fifth biggest sugarcane plantation of the world is standing. It has become extensively noticed since the year 1962 and still holds on its primary cause of sugarcane production handicap [1] [2] . Over 30 million US dollars has been correspondent for the losses in Thai sugarcane industry each year due to this disease [2] [4] . An economic threshold of SCWL has also been reported in Laos, Myanmar, Cambodia, Taiwan, Bangladesh, Sri Lanka [2] [4] -[7] . The typical characteristics symptoms of the disease are bunchy white leaf proliferation on the dwarfed shoots. The disease symptoms can be pronounced in every growth stages of sugarcane plants from the earliest seed cane or cane sett germination until the latest maturity. The affected plants are soon dried as a result of heavy chlorosis and die before they can produce desirable sugar juicy stems. This disease is caused by a fastidious and unculturable phytoplasma, a wall-less prokaryote that localizes in phloem sieve elements and is transmitted by a leafhopper Matsumuratettix hiroglyphicus. Spreading of the disease throughout the country is accelerated by symptomless endemic cane left for the next season due to the lack of certified healthy cane sett or seedlings [8] .
Early diagnosis of SCWL disease is a crucial need for the affirmation of infection sources and epidemic spread that are essential for the right and effective disease control. Although there are several methods that have been used to detect the causal phytoplasma such as fluorescence and electron microscopy [9] , immunology [10] [11] and the DNA based methods using specific cloned-DNA and 16S rDNA probes for dot blot hybridization and several designed primers for PCR techniques [2] -[4] [11] [12] . However, these methods are laborious and very expensive, hence unsuitable for routine detection and real time diagnosis. Currently, the advances in nucleic acid recognition and a discovery of its electrochemical activity have contributed to an exciting progress in DNA behavior investigation and construction of its hybridization probe sensor as a new strategy for greater detection capability than conventional methods. Particularly the determination using electrochemical methods has attracted much interest because of their simple instrumentation, high specificity, sensitivity, speedy, and is inexpensive with potential for applications in molecular sensing devices [13] -[15] .
The DNA based electrochemical strategies in particular the voltammetric analyses have been used as a critical tool for a discriminative study of nucleic acid conformation and modification altogether with the simultaneous detection of all bases of the DNA without the necessity of a previous hydrolysis step [16] -[17] . A number of DNA based voltammetric detections have been reported for detection of pathogens responsible for diseased states such as the detection of hepatitis B virus [18] -[21] , Microcystis spp. [22] , HIV [23] , Yersinia enterocolitica [24] , Legionella pneumophilla [25] dengue virus [26] and Escherichia coli [27] . The detection generally includes the redox labels physically or covalently attached to specific DNA probe or target allocated on the active electrode surface. Recognition of DNA targets is immediately approved by measuring redox signal delivered from the manipulated events using methylene blue, an organic dye that belongs to the phenothiazine family as a preferential redox-active indication [22] [28] -[31] . Various modifications to activate the basal electrode have been executed for efficient immobilization of the DNA probe sensor. Chitosan has been introduced for this purpose because of its well known diverse properties of natural cationic biopolymer. The strong inter and intra-molecular hydrogen bonding interactions of chitosan due to the presence of the hydroxyl and amino groups thus enable an effective DNA entrapment on its matrix [32] -[36] .
The feasibility of rapid and cheap diagnosis of SCWL disease based electrochemical determination has been noticed from our previous preliminary studies on the establishment of high conductivity and bioaffinity modified electrode using the versatile chitosan biomaterial and the use of a short sequence of DNA probes specific to the causal SCWL-16S rDNA phytoplasma [2] [35] -[37] . In this present report, we have performed the investigation on the behavior of both double stranded DNA (dsDNA) and single stranded DNA (ssDNA) from healthy and SCWL diseased plants. Authenticate experiments were subsequently initiated for a specific DNA probe based on ssDNA of diseased plants that have been proved for the presence of the causal SCWL phytoplasma DNA to extend a simplicity of sensing probe preparation and cost reduction. As it has been stated that the whole genomic DNA probes can be used for the identification of several pathogenic bacteria in clinical laboratories, these probes were mostly employed in the detection of direct samples from patients [38] . The determinative analysis of the DNA is based on an immobilization of the DNA onto chitosan-modified glassy carbon electrode using methylene blue (MB) as a redox indicator. A comparative response was also carried out with another disease of sugarcane, the mosaic viral disease, to ensure a specificity of the constructed SCWL-DNA probe sensor. The evidence of specific DNA hybridization observed by atomic force microscopy (AFM) is concurrently illustrated.
2. Materials and Methods
2.1. Plant Materials
The sugarcane white leaf (SCWL) diseased plants collection maintained in the insect proof disease nursery of Plant Pathology Division of the Faculty of Agriculture, Khon Kaen University, was used as a pure source of SCWL-DNA extraction for the generation of specific DNA probe. Samples of field-grown sugarcane for the challenge examination such as healthy plants, SCWL diseased plants and sugarcane mosaic diseased plants were brought from plantation areas in Udornthani Provinces, northeastern Thailand. A preliminary screening for the absence or existence of the causal SCWL-phytoplasma was done by PCR with a set of specific primer pair of both the 16S rDNA and 16-23S rDNA reported earlier [3] .
2.2. Nucleic Acid Extraction and Reagents
The SCWL-DNA was extracted from midrib of the leaves according to the CTAB-phenol-chloroform method [39] . As it has been proved that the phytoplasma cells are colonized in high numbers within plant sieve elements [8] [9] [11] [12] . The extracted DNA was purified by repeating this procedure again to get at least the purity index of 1.8 at A260/A280 ratio for a suitable direct use as specific SCWL-DNA probe. Its concentration was then quantified spectrophotometrically from the value measured at A260. The Challenging target nucleic acid from healthy and SCWL diseased sugarcane and from mosaic virus diseased plants in the field were also extracted from midrib of the leaves by similar method.
Stock solution of each nucleic acid sample was maintained in 10 mM Tris HCL pH 8.0 containing 1 mM Sodium EDTA (TE buffer) and kept frozen. The ssDNA was achieved by denaturing its native dsDNA in 100˚C water bath for 10 min and followed by rapid cooling in ice bath. Chitosan at 85% degree of deacetylation with Mv of 1.11 × 105 Da was obtained from Bioline Lab, Co., Thailand. An analytical grade of methylene blue (MB) and other chemicals was used and all chemicals were purchased from available trades such as Fluka Chemica, Switzerland and Ajax Finechem Pty, Ltd., Australia. High purity deionized water of 18.2 MΩ produced by a Milli-Q RG system (Millipore Corporation, MA, USA.) was used in all solutions.
2.3. Preparation of DNA Probe Sensor
The glassy carbon electrode was polished serially with Al2O3 particles of 0.3 and 0.05 µm. Then it was dip into 70% ethanol for 3 minutes and ultrasonically washed twice in deionized water. The cleaned electrode was finally blown dry with high purity nitrogen prior for further operation. Chitosan-modified electrode was subsequently prepared by applying a 2.0 µl of 1% chitosan in 1% acetic acid solution onto the sensing area of freshly smoothed glassy carbon electrode (GCE). After drying naturally, it was immersed in 0.1 M NaOH for 30 min to fix the film and let it dried again. The SCWL-ssDNA probe was immobilized by dropping 2 µl of its 10 µM in TE buffer onto the chitosan-modified electrode surface and allowed to incubate overnight at room temperature. This probe-functionalized electrode was rinsed with 0.1% SDS phosphate buffer (pH 7.0) three times to remove the unbound ssDNA probe and dried at room temperature.
2.4. Hybridization
Hybridization was performed by immersing the SCWL-ssDNA probe-functionalized chitosan-modified glassy carbon electrode that acted as a specific DNA sensor in 1 ml of 2 × SSC solution containing 10 ng/µl target ssDNA or other testing concentration ranged from 0 - 100 ng/µl in this solution and incubating the reaction at 42˚C for 1 h. Afterward it was rinsed three times with 0.1% SDS phosphate buffer (pH 7.0). The intercalating methylene blue (MB) was accumulated onto the hybridized surface by immersing the electrode into stirred 20 mM Tris buffer saline (TBS) pH 7.0 containing 20 µM MB for 1 h. After that the reaction electrode was rinsed again with blank 20 mM TBS (pH 7.0) for 10 s and was ready for electrochemical measurement.
2.5. Electrochemical Detection
Electrochemical measurements were carried out with an ECoChemie Autolab PSTAT 30 controlled with the General Purpose Electrochemical System (GPES) 4.9 software (now Metrohm Autolab B.V., Utrecht, The Netherlands) coupled to a custom made 10 ml cell and electrode system. The three-electrode system consisted of a glassy carbon working electrode with diameter of 2.0 mm, an Ag/AgCl/3 M KCl reference electrode and a platinum wire counter electrode. All electrolyte buffer solutions were purge with nitrogen for 10 min to remove dissolved oxygen prior to the measurement. Configuration of the self assembled layer and immobilization was declared by cyclic voltammetry (CV) in a 0.1 M phosphate buffer saline (pH 7.0) containing 1.0 mM K3Fe(CN)6 at a scan rate of 25, 50, 100, 150 and 200 m∙Vs−1 and a scan from −0.0 to + 0.6 V. The accumulation of MB on the operated electrode surface was measured by CV and Differential pulse voltammetry (DPV) in 20 mM TBS (pH 7.0) solution at the potential interval 0.0 to −0.5 V with 0.025 V modulation amplitude, 0.05 s modulation time, 0.025 V step potential and 100 m∙Vs−1 scan rate, following a 5 s equilibration period. The reduction peak current in accordance to MB response around −0.22 to −0.27 V was implied as the measurement signal.
2.6. Atomic Force Microscopy (AFM) Investigation
An experiment on hybridization of the SCWL-ssDNA probe with ssDNA target either from diseased plant or healthy plant was performed on chitosan thin film located onto a 9 × 9 mm mica disc (Ted Pella, Inc., CA, USA). The immobilization of ssDNA probe and the hybridization assay were precisely initiated as previously described. Evidence of the hybridization was observed by a XE-70 AFM (Park Systems Corp., Suwon, Korea) controlled with XEP software for data acquisition and XEI software for image processing and analysis with an x-y accessible area of 50 × 50 µm, a position accuracy at 0.6 nm and a Z scanner accuracy of 0.2 nm at 12 µm. The image processing was based on true non contact mode with a PPP-NCHR silicon cantilever consisting of a < 10 nm tip radius and 42 N/m force constant (Nanosensors TM, Neuchâtel, Switzerland).
3. Results and Discussion
3.1. Immobilization of SCWL-ssDNA on Chitosan-Modified GCE
The stability of SCWL-ssDNA immobilization on chitosan-modified GCE was investigated by cyclic voltammetry analysis in 1.0 mM K3Fe(CN)6 solution. As the system comprised both self-assembled chitosan polymer modification and the immobilized ssDNA, so the electrochemical behavior was reflect their interaction between polycationic nature of chitosan and polyanionic nature of DNA. Overall counteraction of the system has previously been identified through comparative redox peak currents in the ferricyanide solution among the bare GCE, chitosan-modified GCE and SCWL-ssDNA immobilized chitosan-modified GCE [35] -[37] . In this experiment, well defined reversible electro-redox responses with a high level of both anodic peak (Ipa) and cathodic peak (Ipc) were obtained at various scan rates from 25 to 200 mv in the system as shown in Figure 1. Linear relationships between the peak currents and the square root of scan rate in both Ipc and Ipa with the corresponding correlation coefficient of 0.9992 and 0.9979 were subsequently gained (Figure 1) for the indication of excellent ion diffusion and the actuality of a fast electron transfer. Their linear curve fitting gives the following equations: Ipa = (−0.8816) µA + 0.444273 µA [mV/s]−1/2 and Ipc = (−0.4975) µA + (−0.30843) µA [mV/s]−1/2, respectively. Both evidences have signified a uniform dispersion of SCWL-ssDNA on chitosan-modified GCE and an integrity of this functionalized electrode surface as suggested by other similar cases [29] [3] [34] . Thus successful DNA immobilization was achieved to fulfill a prerequisite aspect of the development of effective SCWL specific diagnostic sensor.
3.2. Electrochemical Responses of MB-Bound SCWL-ssDNA Probe
Methylene blue (MB) is an organic phenothiazine dye and is known as a redox-active molecule interacting with DNA. The affinity of MB towards guanine bases also affects the electrochemical signals at DNA immobilized electrode surfaces and is commonly used as indicator for the detection of DNA hybridization [19] [22] . Figure 2 shows the CV responses of 20 µM MB bound SCWL-ssDNA functionalized electrode at increasing scan rate
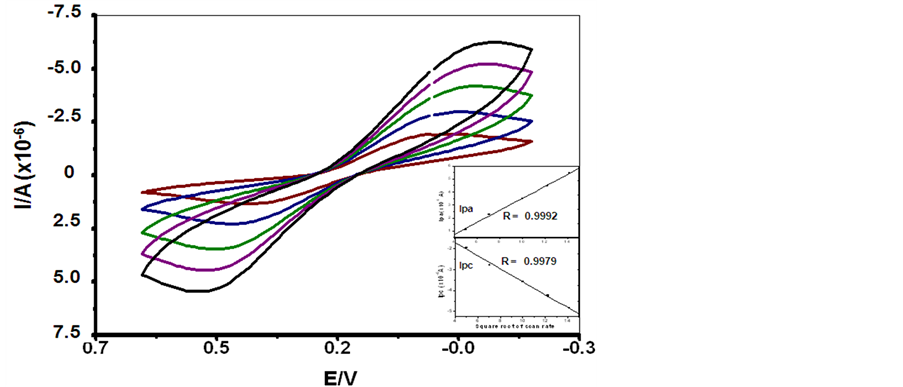
Figure 1. Cyclic voltammograms of the chitosan-modified glassy carbon electrode immobilizing SCWL-ssDNA probe measured in 0.1 M PBS (pH 7.0) containing 1.0 mM K3Fe(CN)63−/K3Fe(CN)64− and 0.1 M KCl with increasing scan rate from inner to outer: 25, 50, 100, 150 and 200 m∙Vs−1. The inset shows plots of redox linear relationship of peak current vs. square root of the scan rate.
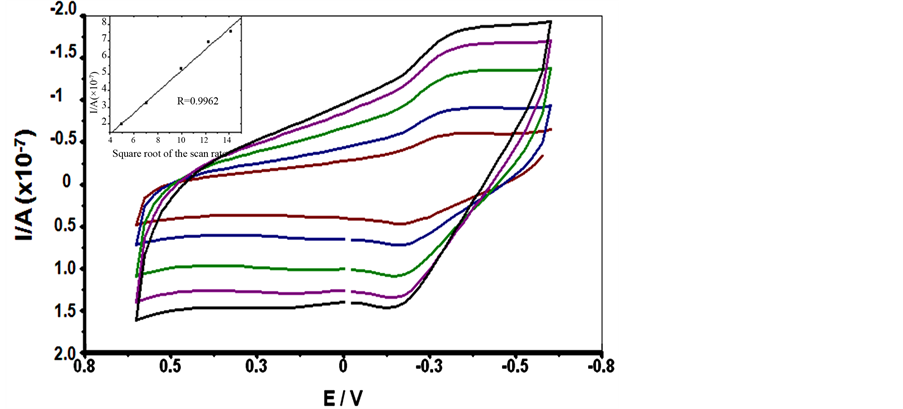
Figure 2. Cyclic voltammograms of current signal after incubation of 20 µM MB in 20 mM TBS (pH 7.0) at the chitosan-modified glassy carbon electrode immobilizing SCWL-ssDNA probe with increasing scan rate from inner to outer: 25, 50, 100, 150 and 200 m∙Vs−1. The inset shows a plot of reduction peak current vs. Square root of the scan rate.
from 25 to 200 mv in 20 mM Tris buffer saline. This obvious signals could indicated a rigid binding and electronically well coupled of an MB to this functionalized electrode surface. A plot of apparent peak current (Ip) versus the square root of scan rate as shown in the inset of Figure 2 is linear with a correlation coefficient of 0.9962 and the linear curve fitting equation of Ip = (−1.127) µA + 0.633 µA [mV/s]−1/2 indicating a required uniform diffusion phenomenon.
3.3. Hybridization Specificity of the SCWL-ssDNA Probe
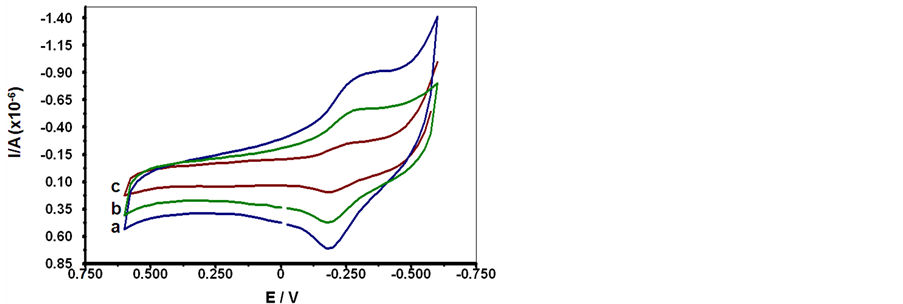
A specificity performance of SCWL-ssDNA probe was examined through hybridization assay with the DNAs from healthy sugarcane plants, SCWL-infected plants, and sugarcane mosaic virus infected plants. Their electrochemical responses regarding the MB signal current monitoring from both CV and DPV are shown in Figure 3(a) and Figure 3(b). As the MB could directly interact with guanine based on the DNA grooves and therefore an extent of electrochemical responses variation could be observed [40] . An obvious comparative bar chart could be illustrated according to their peak current signals in terms of the Ipa, Ipc from CV of the redox activity and the reduction peak Ip from DPV in Figure 3(c). Bar chart set of the originative SCWL-ssDNA probe responses appears with very high peak current signals which indicate a large amount of MB accumulation due to the strong affinity of MB to the free G bases in this ssDNA. On the other hand, hybridization of the SCWLssDNA probe with complementary DNA from SCWL-infected plants yields the lowest MB signal peak currents which indicate a successful and complete hybridization due to the inaccessibility of the guanine bases in a complete dsDNA duplex formation. A minor decrease of peak current values was seen after the SCWL-ssDNA probe was reacted with non-complementary DNA from sugarcane mosaic virus infected plants, indicating distant lower hybridization efficiency. While the control experiment with the DNA from healthy sugarcane plants yielded the highest peak current signals which implied much excess guanine residues due to the absence of pathogenic SCWL-DNA. Therefore, different orders of the MB signal currents were generated with corresponding to their different in matching capability. Since guanine was that mismatched base responsible for the increase in MB signal from a close interaction with the unbound guanine. The hybridization selectivity could then be recognized by comparing the MB peak current signal values from both CV and DPV. The results demonstrated that this SCWL-ssDNA probe displayed adequate specificity for the detection of its complementary DNA from SCWL diseased plants and the discrimination of the DNAs from healthy and sugarcane plants infected with other pathogen such as the sugarcane mosaic virus.
3.4. Electrochemical Responses of the Complementary SCWL-DNA Target with Different Concentration
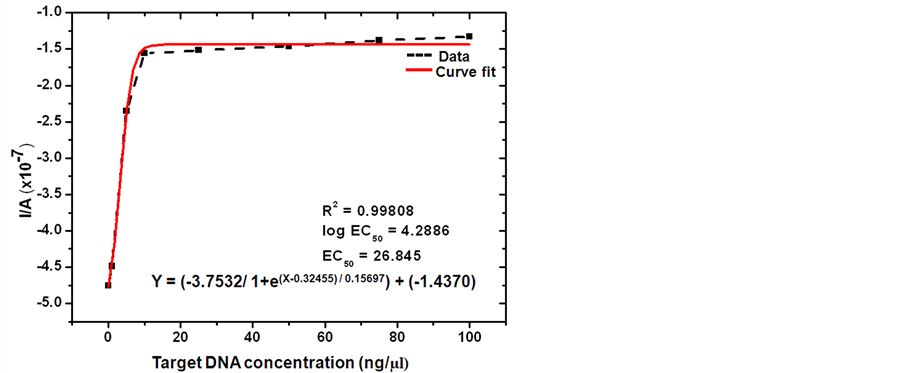
Sensitivity of the specific hybridization of SCWL-ssDNA probe with its complementary DNA from white leaf diseased sugarcane could be quantitatively analyzed by varying the target concentration (0 to 100 ng/µl). Electrochemical responses were detected by differential pulse voltammetry under the constant MB at 20 µM in 20 mM Tris buffer saline as it has been suggested that the DPV can remove capacitive currents and known to increase detection limits compared to non-pulsed voltammetry by two fold differences [25] [26] . The plots of overall obtaining reduction peak current of MB are displayed in Figure 4.
The MB reduction peak current was decreased steeply with the increase of complementary SCWL-DNA target concentration from 0 to10 ng/µl and slightly decreased with the target concentration from 10 up to100 ng/µl and tended to keep constant after that (Figure 4(a)). A plateau state at the final range of ssDNA target concentration hence indicated a saturate hybridization due to the limitation of capturing functional area at the electrode surface. By this concentration response curve, a 50% effective concentration (EC50) could be estimated to be 26.845 ng/µl of ssDNA target with the regression coefficient (R2) of 0.998 and the curve fit equation of Y = (−1.4370) + [(−5.1902) − (−1.4370)/1 + e(X−0.32455)/0.15697], where Y is the reduction peak value (Ip) and X is the DNA concentration. The detection limit following this concentration response curve fit based on 3 × SD/slope value was 4.709 ng/µl and the overall relative standard deviation (percentage of R.S.D.) was 2.44%. The apparent linear relationship could be raised within the target concentration ranging from 0 to 10 ng/µl with a correlation coefficient of 0.9962 and a regression fitting equation of Y = (−4.62677) + (0.33513)X where Y is the reduction peak value (Ip) and X is the DNA concentration (Figure 4(b)). The limit of the detection in this range was then assumed to be 4.458 ng/µl and the relative standard deviation was 3.63%. These linear equations might be useful in an analysis for narrowing the actual concentration within each suspected range. The detection capability is comparable to several published electrochemical voltammetric DNA detection such as the detection limit of 5.0 nM via ferrocene labeling and chitosan-modified electrode [33] [34] , 0.5 nM for Microcystis related 17 bases DNA sequence immobilized on CPE [22] , 0.252 nM of fish sperm DNA via chitosan/CNT/graphite electrode [28] , 0.2 nM via a hairpin DNA on gold electrode [29] , the detection range of 6.25 - 100 nM for 104 nt DNA sequence using PEGylated 20 nt ssDNA and mercaptoethanol on gold electrode [41] , 0.1 nM of the synthetic oligonucleotides according to HIV sequence using gold electrode with 6 sensor array [23] , the detection range of 10 - 60 ng/µl for genomic DNA and PCR product of Neisseria meningitides [42] and the detection limit of 36.72 ng/μL for the concentration range 100 - 500 ng/μl of genomic DNA from Vibrio cholera [43] .
 (a)
(a)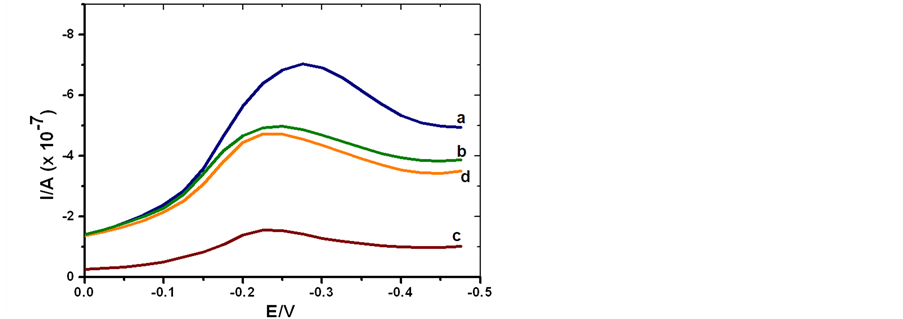 (b)
(b) (c)
(c)
Figure 3. (a) Cyclic voltammograms of MB current signal determined in 20 mM TBS (pH 7.0) at 100 m∙Vs−1 scan rate: a) hybridization of SCWL-ssDNA probe with healthy sugarcane plant ssDNA; b) hybridization of SCWL-ssDNA probe with sugarcane mosaic diseased plant ssDNA; c) hybridization of SCWL-ssDNA probe with its complementary SCWL diseased plant ssDNA. (b) Differential pulse voltammograms of MB current signal at similar condition: a) hybridization of SCWLssDNA probe with healthy sugarcane plant ssDNA; b) hybridization of SCWL-ssDNA probe with sugarcane mosaic diseased plant ssDNA; c) hybridization of SCWL-ssDNA probe with its complementary SCWL diseased plant ssDNA and d) SCWL-ssDNA probe only. (c) Bar chart of CV and DPV redox peak current responses in hybridization experiments. Histograms and error bars represent mean values and standard deviations, respectively. The relative standard deviations (percentage of R.S.D.) are less than 5%.
 (a)
(a)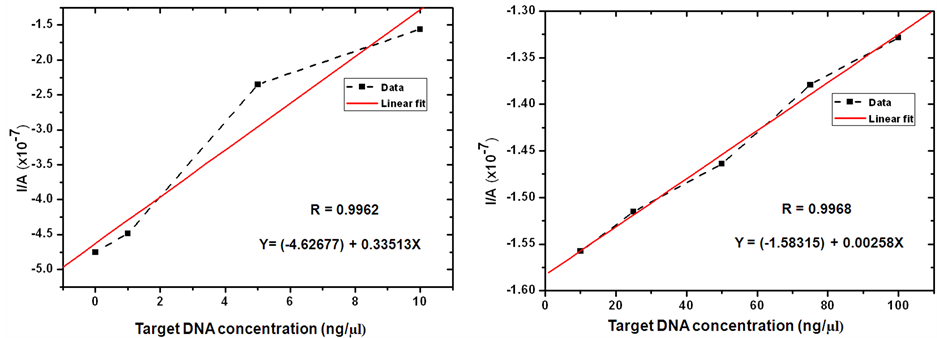 (b)
(b)
Figure 4. Response studies of SCWL-ssDNA probe-functionalized chitosan-modified glassy carbon electrode after hybridization with different concentration of complementary target DNA from SCWL diseased plant. (a) Dose response curve of MB peak current from DPV performance vs. the target concentration from 0 to100 ng/µl at 100 m∙Vs−1 scan rate in 20 mM TBS (pH 7.0); (b) Linear graph of peak current vs. the concentration ranged from 0 to 10 ng/µl; (c) Linear graph of peak current vs. the concentration ranged from 10 to 100 ng/µl. The relative standard deviations (percentage of R.S.D.) are all less than 5%.
The results thus demonstrate the great potential for practical application of this electrochemical response detection for the qualitative and quantitative analysis of SCWL with a very simple preparation using domestically available chitosan and self producible DNA probe.
3.5. AFM Investigation
Immobilization of SCWL-ssDNA probe and hybridization forming event was investigated by means of AFM that allowed a visual ultra-configuration of the DNA strands. The SCWL-ssDNA probe strand is clearly seen immobilizing on the chitosan matrix surface (Figure 5(a)). Consequently, an image of hybridization forming event in Figure 5(b) has revealed the uniformly coupling strands of the SCWL-ssDNA probe with its complementary ssDNA from diseased plant. The surface topographic characteristics reported by the AFM embedded XEI software for image processing and analysis with an x-y accessible area has provide an average roughness value for the plain chitosan matrix and after the SCWL-ssDNA immobilization as 0.657 nm and 3.3295 nm, respectively. The value seemed to increase following DNA immobilization which indicated a well dispersing layer of SCWL-ssDNA formed on the basal chitosan matrix. The average roughness of the surface was also raised up to a higher value of 8.083 nm when the immobilized SCWL-ssDNA was subjected to hybridization with its
 (a)
(a) (b)
(b)
Figure 5. (a) AFM images of SCWL-ssDNA immobilization on chitosan matrix surface within 5 × 5 µm2 scanning area; (b) Hybridization forming by joining the SCWL-ssDNA probe with its complementary ssDNA from SCWL diseased plant.
complementary DNA. An average size of the ssDNA strand on the surface measured within 5 × 5 µm2 scanning area as shown in Figure 5 was around 15.33 nm in term of ∆x value and 77.67 nm in term of ∆y value while the average size of coupling DNA including their linker distance in term of ∆y value was 299.33 nm with a nearly similar ∆x value of 15.71 nm. Our overall AFM results demonstrated successful immobilization and hybridization feature on the surface of chitosan-modified GCE together with measurable surface characteristic. Although our results from both electrochemical and AFM characterization in this investigation were contradictory to some other statements that claimed a strong stearic hindrance and signal suppression on hybridization using long DNA sequence and high surface density [44] [47] . However, another study on DNA coverage dependent using AFM and nanografting technique has also reported a new insight of little trouble in hybridizing efficacy even at high surface densities. Instead, the denser DNA the higher hybridization efficiency was performed [48] . Our results then could be more likely explained by the latter resembling case with a certain suitable circumstance for the optimum hybridization. Thus, this formulated ssDNA probe is effectively differentiated the SCWL-diseased plants from suspected population and can be used as an efficient tool for SCWL disease diagnosis.
4. Conclusion
A practicable DNA detection using electrochemical voltammetric techniques was first reported for reliable diagnosis of sugarcane white leaf disease in real sample from commercial planting fields through SCWL-ssDNA probe. Satisfactory detection capability could be achieved with the detection limit of 4.709 ng/µl DNA target. Discrimination of SCWL disease from the healthy one and other resembled disease features such as mosaic viral infected sugarcane plants was well demonstrated by the hybridization selectivity. The sensitivity of this DNA probe sensor can be comparable to several other electrochemical DNA probe sensor systems for the detection of DNA matching that have been reported. Fabrication of the SCWL-ssDNA probe sensor is incredibly simple and low cost via self producible DNA probe from previously identified SCWL-mother plants by our specific PCR primers [3] and the use of domestically available chitosan for a rapid and cheap MB redox determination system. Successfully specific hybridization was easily obtained despite a lengthy sequence chain of this DNA probe formulation. The actual immobilization of SCWL-ssDNA probe on chitosan matrix and corresponding hybridization coupling could be revealed by AFM images. These results thus have provided a very convenient tool for diagnosis of SCWL disease and also encourage further studies on the feasibility, the improvement for ultrasensitive detection and the development of a better defined condition for various specific choices and the requirement for production of portable device for real time assessment in the field.
Acknowledgements
This work run under the Biosensor Technology for Sustainable Development Research Group of Khon Kaen University and financial fund from the Division of Research and Technology Transfer Affair (No. 53-55-1101) is greatly acknowledged. We also thank Mr. Supatr Chuaymee and Mr. Vorrayuth Mekarkat for their AFM technical assistances.
References
- Kusalwong, A. (1980) Sugarcane Disease. In: Suriyapan, P., Ed., Sugarcane, Monograph Number 1, Department of Agriculture, Ministry of Agriculture and Cooperatives, Bangkok, 131-144.
- Wongkaew, P. (2012) Sugarcane White Leaf Disease Characterization, Diagnosis Development, and Control Strategies. Functional Plant Science Biotechnology, 6, 73-84.
- Wongkaew, P., Hanboonsong, Y., Sirithorn, P., Choosai, C., Boonkrong, S., Tinnangwattana, T., Kitchareonpanya, R., and Damak, S. (1997) Differentiation of Phytoplasmas Associated with Sugarcane and Gramineous Weed White Leaf Disease and Sugarcane Grassy Shoot Disease by RFLP and Sequencing. Theoretical Applied Genetics, 95, 660-663. http://dx.doi.org/10.1007/s001220050609
- Wongkaew, P. (2013) Plant Diseases Caused by Phytoplasma. Biosensor Technology for Sustainable Development Research Group, Khon Kaen University Publishing, Khon Kaen.
- Leu, L.S. (1983) White Leaf Disease. In: Agnihotri, V.P., Ed., Disease of Sugarcane, Oxford and IBH Publishing Co., Delhi, 250-267.
- Kumarrasinghe, N.C. and Jones, P. (2001) Identification of white Leaf Disease of Sugarcane in Sri Lanka. Sugar Technology, 3, 55-58. http://dx.doi.org/10.1007/BF02945532
- Win, N.K.K and Jung, H.Y. (2012) The Distribution of Phytoplasmas in Myanmar. Journal of Phytopathology, 160, 139-145. http://dx.doi.org/10.1111/j.1439-0434.2011.01875.x
- Wongkaew, P. and Fletcher, J. (2004) Sugarcane White Leaf Phytoplasma in Tissue Culture: Long-Term Maintenance, Transmission, and Oxytetracycline Remission. Plant Cell Reports, 23, 426-434. http://dx.doi.org/10.1007/s00299-004-0847-2
- Wongkaew, P., Sirithorn, P., Chaleeprom, W., Nakashima, K., Hayashi, T. and Koizumi, M. (1995) Detection of Sugarcane White Leaf Mycoplasma-Like Organism in Field Plants and Tissue Cultures by DNA Probes. In: Matangkasombat, P. and Yoshida, T., Eds., Microbial Utilization of Renewable Resources, ICCR Biotechnology, 9, 406-419.
- Sarindu, N. and Clark, M.F. (1993) Antibody Production and Identify of MLOs Associated with Sugarcane White Leaf Disease and Bermuda Grass White Leaf Disease from Thailand. Plant Pathology, 42, 396-402. http://dx.doi.org/10.1111/j.1365-3059.1993.tb01517.x
- Wongkaew, P. (1999) Sugarcane White Leaf Disease Management. Pimpatana Press, Khon Kaen.
- Nakashima, K., Chaleeprom, W., Wongkaew, P. and Sirithorn, P. (1994) Detection of Mycoplasmalike Organism Associated with White Leaf Disease of Sugarcane in Thailand Using DNA Probes. JIRCAS Journal, 1, 57-67.
- Palecek, E., Fojta, M. and Jelen, F. (2002) New Approaches in the Development of DNA Sensors: Hybridization and Electrochemical Detection of DNA and RNA at Two Different Surfaces. Bioelectrochemistry, 55, 85-90.
- Yin, B-C., Guan, Y-M. and Ye, B-C. (2012) An Ultrasensitive Electrochemical DNA Sensor Based on the ssDNA-Assisted Cascade of Hybridization Reaction. Chemical Communications, 48, 4208-4210. http://dx.doi.org/10.1039/c2cc30997a
- Wei, F., Lillehoj, P.B. and Ho, C-M. (2010) DNA Diagnostics: Nanotechnology-Enhanced Electrochemical Detection of Nucleic Acids. Pediatric Research, 67, 458-468. http://dx.doi.org/10.1203/PDR.0b013e3181d361c3
- Oliveira-Brett, A.M., Piedade, J.A.P., Silva, L.A. and Diculescu, V.C. (2004) Voltammetric Determination of All DNA Nucleotides. Analytical Biochemistry, 332, 321-329. http://dx.doi.org/10.1016/j.ab.2004.06.021
- Pedano, M.L. and Rivas, G.A. (2005) Immobilization of DNA at Glassy Carbon Electrodes: A Critical Study of Adsorbed Layer. Sensors, 5, 424-447. http://dx.doi.org/10.3390/s5060424
- Erdem, A., Kerman, K., Meric, B., Akarca, U.S. and Ozsoz, M. (1999) DNA Electrochemical Biosensor for the Detection of Short DNA Sequences Related to the Hepatitis B Virus. Electroanalysis, 11, 586-587. http://dx.doi.org/10.1002/(SICI)1521-4109(199906)11:8<586::AID-ELAN586>3.0.CO;2-J
- Erdem, A., Kerman, K., Meric, B., Akarca, U.S. and Ozsoz, M. (2000) Novel Hybridization Indicator Methylene Blue for the Electrochemical Detection of Short DNA Sequences Related to the Hepatitis B Virus. Analytica Chimica Acta, 422, 139-149. http://dx.doi.org/10.1016/S0003-2670(00)01058-8
- Ye, Y.K. and Ju, H.X. (2003) DNA Electrochemical Behaviors, Recognition and Sensing by Combining with PCR Technique. Sensors, 3, 128-145. http://dx.doi.org/10.3390/s30600128
- Guo, M.D., Li, Y.Q., Guo, H.X., Wu, X.Q. and Fan, L.F. (2007) Electrochemical Detection of Short Sequences Related to the Hepatitis B Virus Using MB on Chitosan-Modified CPE. Bioelectrochemistry, 70, 245-249. http://dx.doi.org/10.1016/j.bioelechem.2006.09.002
- Erdem, A., Kerman, K., Meric, B., Ozkan, D., Kara, P. and Ozsoz, M. (2002) DNA Biosensor for Microcystis spp. Sequence Detection by Using Methylene Blue and Ruthenium Complex as Electrochemical Hybridization Label. Turkish Journal of Chemistry, 26, 851-862.
- Zhang, D., Peng, Y., Qi, H., Gao, Q. and Zhang, C.X. (2010) Label-Free Electrochemical DNA Biosensor Array for Simultaneous Detection of the HIV-1 and HIV-2 Oligonucleotides Incorporating Different Hairpin-DNA Probes and Redox Indicator. Biosensors and Bioelectronics, 25, 1088-1094. http://dx.doi.org/10.1016/j.bios.2009.09.032
- Sun, W., Qin, P., Gao, H., Li, G. and Jiao, K. (2010) Electrochemical DNA Biosensor Based on Chitosan/Nano-V2O5/MWCNTs Composite Film Modified Carbon Ionic Liquid Electrode and Its Application to the LAMP Product of Yersinia enterocolitica Gene Sequence. Biosensors and Bioelectronics, 25, 1264-1270. http://dx.doi.org/10.1016/j.bios.2009.10.011
- Rai, V., Deng, J. and Toh, C.S. (2012) Electrochemical Nanoporous Alumina Membrane-Based Label-Free DNA Biosensor for the Detection of Legionella sp. Talanta, 98, 112-117. http://dx.doi.org/10.1016/j.talanta.2012.06.055
- Rai, V., Hapuarachchi, H.C., Ng, L.C., Soh, S.H., Leo, Y.S. and Toh, C.S. (2012) Ultrasensitive cDNA Detection of Dengue Virus RNA Using Electrochemical Nanoporous Membrane-Based Biosensor. PLoS ONE, 8, Article ID: e42346. http://dx.doi.org/10.1371/journal.pone.0042346
- Jiang, D., Liu, F., Liu, C., Liu, L. and Pu, X. (2013) An Electrochemical Sensor Based on Allosteric Molecular Beacons for DNA Detection of Escherichia coli O157:H7. International Journal of Electrochemical Science, 8, 9390-9398.
- Li, J., Liu, Q, Liu, Y., Liu, S. and Yao, S.Z. (2005) DNA Biosensor Based on Chitosan Film Doped with Carbon Nanotubes. Analytical Biochemistry, 346, 107-114. http://dx.doi.org/10.1016/j.ab.2005.07.037
- Jin, Y., Yao, X., Liu, Q. and Li, J.H. (2007) Hairpin DNA Probe Based Electrochemical Biosensor Using Methylene Blue as Hybridization Indicator. Biosensors and Bioelectronics, 22, 1126-1130. http://dx.doi.org/10.1016/j.bios.2006.04.011
- Pänke, O., Kirbs, A. and Lisdat, F. (2007) Voltammetric Detection of Single Base-Pair Mismatches and Quantification of Label-Free Target ssDNA Using a Competitive Binding Assay. Biosensors and Bioelectronics, 22, 2656-2662. http://dx.doi.org/10.1016/j.bios.2006.10.033
- Sun, W., Qin, P., Gao, H., Li, G. and Jiao, K. (2010) Electrochemical DNA Biosensor Based on Chitosan/Nano-V2O5/MWCNTs Composite Film Modified Carbon Ionic Liquid Electrode and Its Application to the LAMP Product of Yersinia enterocolitica Gene Sequence. Biosensors and Bioelectronics, 25, 1264-1270. http://dx.doi.org/10.1016/j.bios.2009.10.011
- Motomura, T. and Aoyama, Y. (1992) Multiple Electrostatic Interaction of Polycationic Chitosan Oligomers with Nucleic Acids: Polyamine Promotes Stabilization of the A-Form of RNA and DNA in Aqueous Solutions. Bulletin of the Chemical Society of Japan, 65, 1755-1760. http://dx.doi.org/10.1246/bcsj.65.1755
- Xu, C., Cai, H., He, P. and Fang, Y. (2001) Electrochemical Detection of Sequence-Specific DNA Using a DNA Probe Labeled with Aminoferrocene and Chitosan Modified Electrode Immobilized with ssDNA. Analyst, 126, 62-65. http://dx.doi.org/10.1039/b005847p
- Xu, C., Cai, H., Xu, Q., He, P. and Fang, Y. (2001) Characterization of Single Stranded DNA on Chitosan-Modified Electrode and Its Application to the Sequence-Specific DNA Detection. Fresenius’ Journal of Analytical Chemistry, 369, 428-432. http://dx.doi.org/10.1007/s002160000673
- Wongkaew, P. and Poosttisak, S. (2008) Direct Electrochemical DNA Sensor for Sugarcane White Leaf Disease Diagnosis Using Label Free DNA Probes and Oligochitosan Self Assembled Monolayer-Modified Glassy Carbon Electrodes. Proceeding of the 2nd International Conference on Technology and Innovation for Sustainable Development, Khon Kaen, 28-29 January 2008, 504-507.
- Wongkaew, P. and Poosittisak, S. (2012) Electro-Affinity of SCWL-dsDNA on Different High Deacetylation Degree Chitosans Deposited Glassy Carbon Electrode. Proceeding of the International Conference on Technological Challenges in Developing Affordable in Vitro Molecular Diagnostics, Mumbai, 11-14 March 2012, 249-258.
- Poosittisak, S. and Wongkaew, P. (2012) Electrochemical Characterization of the Hexacyanides of a Bare Screen Printed Carbon Electrode and Chitosan Thin Film Modified-Screen Printed Carbon Electrode. Proceeding of the International Conference on Technological Challenges in Developing Affordable in Vitro Molecular Diagnostics, Mumbai, 11-14 March 2012, 259-266.
- Venkateswaran, N. and Venkateswaran, K.S. (1991) Nucleic Acid Probes in Microbiology. Defence Science Journal, 41, 335-356.
- Kollar, A., Seemuller, E., Bonnet, F., Saillard, C. and Bove, J.M. (1990) Isolation of the DNA of Various Plant Pathogenic Mycoplasmalike Organisms from Infected Plants. Phytopathology, 80, 233-237. http://dx.doi.org/10.1094/Phyto-80-233
- Kara, P., Kerman, K., Ozkan, D., Meric, B., Erdem, A. and Ozsoz, M. (2002) Electrochemical Genosensor for the Detection of Interaction between Methylene Blue and DNA. Electrochemistry Communications, 4, 705-709. http://dx.doi.org/10.1016/S1388-2481(02)00428-9
- Henry, O.Y.F., Gutierrez Perez, J., Acero Sanchez, J.L. and O’Sullivan, C.K. (2010) Electrochemical Characterization and Hybridization Efficiency of Co-Assembled Monolayers of PEGylated ssDNA and Mercaptohexanol on Planar Gold Electrodes. Biosensor and Bioelectronics, 25, 978-983. http://dx.doi.org/10.1016/j.bios.2009.09.007
- Patel, M.K., Solanki, P.R., Kumara, A., Khare, S., Gupta, S. and Malhotra, B.D. (2010) Electrochemical DNA Sensor for Neisseria meningitidis Detection. Biosensors and Bioelectronics, 25, 2586-2591. http://dx.doi.org/10.1016/j.bios.2010.04.025
- Patel, M.K., Ali, A., Zafaryab, M., Agrawal, V.V., Rizvi, M.M.A., Ansari, Z.A., Ansari, S.G. and Malhotra, B.D. (2013) Biocompatible Nanostructured Magnesium Oxide-Chitosan Platform for Genosensing Application. Biosensors and Bioelectronics, 45, 181-188. http://dx.doi.org/10.1016/j.bios.2012.12.055
- Peterson, A.W., Heaton, R.J. and Georgiadis, R.M. (2001) The Effect of Surface Probe Density on DNA Hybridization. Nucleic Acids Research, 29, 5163-5168. http://dx.doi.org/10.1093/nar/29.24.5163
- Petrovynkn, D.Y., Kimura-Suda, H., Whitman, L.J. and Tarlov, M.J. (2003) Quantitative Analysis and Characterization of DNA Immobilized on Gold. Journal of American Chemistry Society, 125, 5219-5226. http://dx.doi.org/10.1021/ja029450c
- Steel, A.B., Herne, T.M. and Tarlov, M.J. (1998) Electrochemical Quantitation of DNA Immobilized on Gold. Analytical Chemistry, 70, 4670-4677. http://dx.doi.org/10.1021/ac980037q
- Ricci, F., Lai, R.Y., Heeger, A.J., Plaxco, K.W. and Sumner, J.J. (2007) Effect of Molecular Crowding on the Response of an Electrochemical DNA Sensor. Langmuir, 23, 6827-6834. http://dx.doi.org/10.1021/la700328r
- Mirmomtaz, E., Castronovo, M., Grunwald, C., Bano, F., Scaini, D., Ensafi, A.A., Scoles, G. and Casalis, L. (2008) Quantitative Study of the Effect of Coverage on the Hybridization Efficiency of Surface-Bound DNA Nanostructures. Nano Letters, 12, 4134-4139. http://dx.doi.org/10.1021/nl802722k
NOTES

*Corresponding author.

