International Journal of Clinical Medicine
Vol.3 No.2(2012), Article ID:17880,10 pages DOI:10.4236/ijcm.2012.32024
Intralesion and CuradermBEC5 Topical Combination Therapies of Solasodine Rhamnosyl Glycosides Derived from the Eggplant or Devil’s Apple Result in Rapid Removal of Large Skin Cancers. Methods of Treatment Compared
![]()
“Villa Del Aripo”, Sheldon, Australia.
Email: bill.cham@gmail.com
Received December 12th, 2011; revised January 17th, 2012; accepted February 9th, 2012
Keywords: Cancer; Devil’s Apple; Eggplant; Curaderm; Solamargine; Apoptosis
ABSTRACT
Solasodine rhamnosyl glycosides (SRGs) are chemotherapeutic agents for the treatment of cancer. SRGs in a cream formulation, CuradermBEC5, is very effective for the treatment of nonmelanoma skin cancers with excellent cosmetic end results. Intralesion injection of SRGs successfully dispose of very large tumours in animals without any clinical adverse effects. The mode of action of SRGs is by apoptosis. In this study, it is shown that small to large basal cell carcinomas are effectively treated only with topical application of CuradermBEC5. Here it is reported for the first time that combination of intralesion SRG injection and topical application with CuradermBEC5 in humans reduces the treatment time period by more than half when compared with topical application as the sole treatment regime. Two intralesion injections of very low doses of SRGs rapidly and effectively remove a large melanoma on a horse. If rapid removal of large troublesome skin cancers is required then this can be achieved by intralesion and topical treatments. Intralesion or combination therapy with SRGs may have some applications for melanomas in situ such as lentigo maligna.
1. Introduction
Existing antimitotic chemotherapeutic agents appear to affect fast growing cells by killing such cells when they are dividing (proliferating cells). When such cells are not dividing (non-proliferating cells) the existing antimitotic chemotherapeutic agents have very limited effects upon these cells. Consequently, the time course for treating cancer cells with existing antimitotic chemotherapeutic agents is long and repetitive treatments are required. In addition, certain cancers have developed drug-resistance which seriously complicates antimitotic therapy. The side effects of antimitotic chemotherapy are well know.
Solasodine rhamnosyl glycosides (SGRs) are very promising antineoplastics [1-24]. Oral, intravenous, intraperitoneal and intralesion studies with these glycoalkaloids have been described [13,19-21,25]. Many studies have reported the high antineoplastic efficacy, low toxicity of solamargine, solasonine and a constant mixture of these glycoalkaloids (BEC) [4-25].
Unlike established antimitotic drugs, BEC and its individual glycoalkaloids, solamargine and solasonine, are not antimitotic in their actions [21,24,26-30]. BEC induces apoptosis in cancer cells by up-regulating the expressions of external death receptors, such as tumour necrosis factor receptor 1 (TNFR-1), Fas receptor, TNFR-1 associated death domain and Fas-associated death domain. BEC also enhances the intrinsic ratio of Bax to Bcl-2 by up-regulating Bax and down-regulating Bcl-2 and Bcl-xL expressions. These effects result in activation of Caspase –8, –9 and –3 in cancer cells, indicating that BEC triggers extrinsic and intrinsic apoptotic pathways in cancer cells [7,21,22,26-32].
Solamargine, the main component of BEC, also kills cancer cells by oncosis. After interaction of solamargine with cancer cells, marked changes in cell shape and volume occur. The cells get blebs on the membrane, the mitochondria swell, the contents of the nuclei clump and the cells die. It has been proposed that apoptosis and oncosis share certain mechanisms and alterations within the cell before they die by bursting. At low concentrations solamargine kills cancer cells by apoptosis and at higher doses, solamargine kills cancer cells by oncosis, and both types of cell death are induced by intermediate concentrations of solamargine [32].
It has recently been shown that, in addition to causing apoptosis in cancer cells, intralesion administration of BEC also stimulates lasting immunity against cancer [24].
Intravenous (i.v.) administrations of drugs pose a number of serious limitations including toxicity to body organs. Because of the dilution effect of the drug within the vascular system and the pharmacokinetics of the drug by various body organs, a relatively high dose of the drug has to be administered frequently for it to have a therapeutic effect on the targeted cancer cells and this high dose may result in serious toxicological effects. Intravenous doses are dependent on body weight or volume. It was shown that intralesion injection of BEC is far superior to i.v. injection if the targeted lesion being treated is rapidly and specifically degraded by the chemotherapeutic agent. Intralesion injections of very low doses of BEC into large chondrosarcomas and squamous cell carcinomas in animals completely eliminate these tumours. The required dose of intralesion injections are tumour weight dependent and not body weight dependent and are 100 times lower than for i.v. infusions [21].
CuradermBEC5 is a topical cream which contains a mixture of solasodine glycosides (BEC) and is very effective for treating nonmelanoma skin cancers. Advantages of treatment of these lesions with CuradermBEC5 compared with well established surgical interventions and other therapies have previously been reported [1-3,19,20,24,28,29,33-37].
The treatment time period with topical CuradermBEC5 to completely eliminate skin cancers varies from days to months depending on size and type of skin tumours. Large nonmelanoma skin cancers can successfully be treated with CuradermBEC5 and it may take several months of treatment to eliminate these large tumours [1-3,19,20,24, 28,29,33,33-37].
The incidences of nonmelanoma skin cancers such as basal cell carcinoma (BCC) and squamous cell carcinoma (SCC) have increased more than 300 percent since 1992 [1,2,38]. For malignant melanoma, increased rates of over 500 percent have been reported from 1975 to 2008 [1,39].
Limitations and costs of current treatments for skin cancers pose many shortfalls and many patients afflicted with these diseases do not seek proper treatment, resulting in increased morbidity and mortality [1,3,40,41].
This report describes the successful treatments of various large BCCs by topical application of a cream formulation. Combination therapy of intralesion and topical application of the same antineoplastic agents, BEC, result in rapid removal of a large BCC. Combination therapy of intralesion and topical treatment of BEC reduces skin cancer treatment period by about half when compared with topical treatment alone. The cosmetic end results of the treatments are excellent.
This report also shows that intralesion injection of BEC alone, without CuradermBEC5 cream combination therapy, into a large melanoma of a horse results in the elimination of this potentially deadly skin cancer.
2. Materials and Methods
2.1. Patient Selection
A 68 year old farmer was referred for consultation because he had multiple basal cell carcinomas of varying sizes. In particular, he also had a very large BCC on his back that often bled and was painful. This large BCC was a recurrence of a smaller BCC that was previously removed surgically with the involvement of a skin flap. The patient who had these lesions for several years elected to treat various lesions with the cream formulation CuradermBEC5. However, because of the size and pain associated with the large BCC, he requested to have this lesion treated by intralesion injection using the active ingredients solasodine glycosides (BEC) which are also present in the CuradermBEC5 cream formulation. Patient’s informed consent for the treatment was obtained.
The sizes of the BCCs ranged from less than 2 cm in diameter (6 lesions) to approximately 3 cm × 3 cm × 0.5 cm (4 lesions) to 5 cm × 5 cm × 1.5 cm (1 lesion). Histological analyses of biopsies revealed that these lesions were BCCs.
A horse had an histologically diagnosed melanoma on the chest. This lesion was present for at least six months. The surface area of the lesion measured 5 cm × 4 cm and protruded approximately 1 cm. The owner of the horse was referred to our centre by his veterinarian and requested treatment with BEC for his horse’s melanoma.
2.2. Treatments Administered
2.2.1. BEC—Solasodine Rhamnosyl Glycosides
Glycoalkaloids (BEC) were extracted from the fruit of S. sodomaeum also known as S. linnaeanum (devil’s apple) and S. melongena (eggplant) essentially as described earlier [42]. BEC is a mixture of solasodine glycosides consisting of the triglycosides solasonine (β-solatriose) (33%), solamargine (β-chacotriose) (33%), and di-and monoglycosides (34%). All the glycosides contain the same aglycone, solasodine [2,4,5,24,28,42]. Figure 1 shows the chemical structures of solasonine and solamargine [2,4,5,24, 28,42] and Figure 2 shows their mass spectra [2,24]. In the present studies the extract containing the mixture of these glycoalkaloids was investigated. Just prior to the current set of experiments BEC was analyzed by HPLC [24,42] and it was shown to consist of 33% solasonine, 33% solamargine and 34% of diand monoglycosides of solasodine.
2.2.2. CuradermBEC5 Topical Cream Formulation
The cream formulation CuradermBEC5 is available to patients in several countries. CuradermBEC5 contains the glycoalkaloids BEC at 0.005% as a topical cream formulation. The cream was applied twice daily (when possible every 12 hours) under occlusive dressing (micropore paper tape) until the lesion had clinically regressed [1,41].
2.2.3. BEC in DMSO Solution for Intralesion Injection
A sterile solution of 10% BEC in dimethylsulfoxide, w/w, was used in this study [21].
The weight of the large BCC on the back of the human was approximately 50 g. The doses injected intralesionally were approximately 100 mg BEC per 1 kg tumour weight [21]. In the human subject, with the BCC tumour weight of approximately 50 g, the dose was 50 microlitres of the 10% BEC solution and for the horse, with the melanoma tumour weight of 25 g, the dose was 25 microlitres of the 10% BEC solution.
The human and horse were injected intralesionally on 2 separate occasions. The second injection occurred approximately 48 hours after the first injection. Each injection was done intralesionally on multiple sites of the tumour including at the juncture of the lesions with the surrounding normal skin. No anaesthetic was used during the treatment regimes.
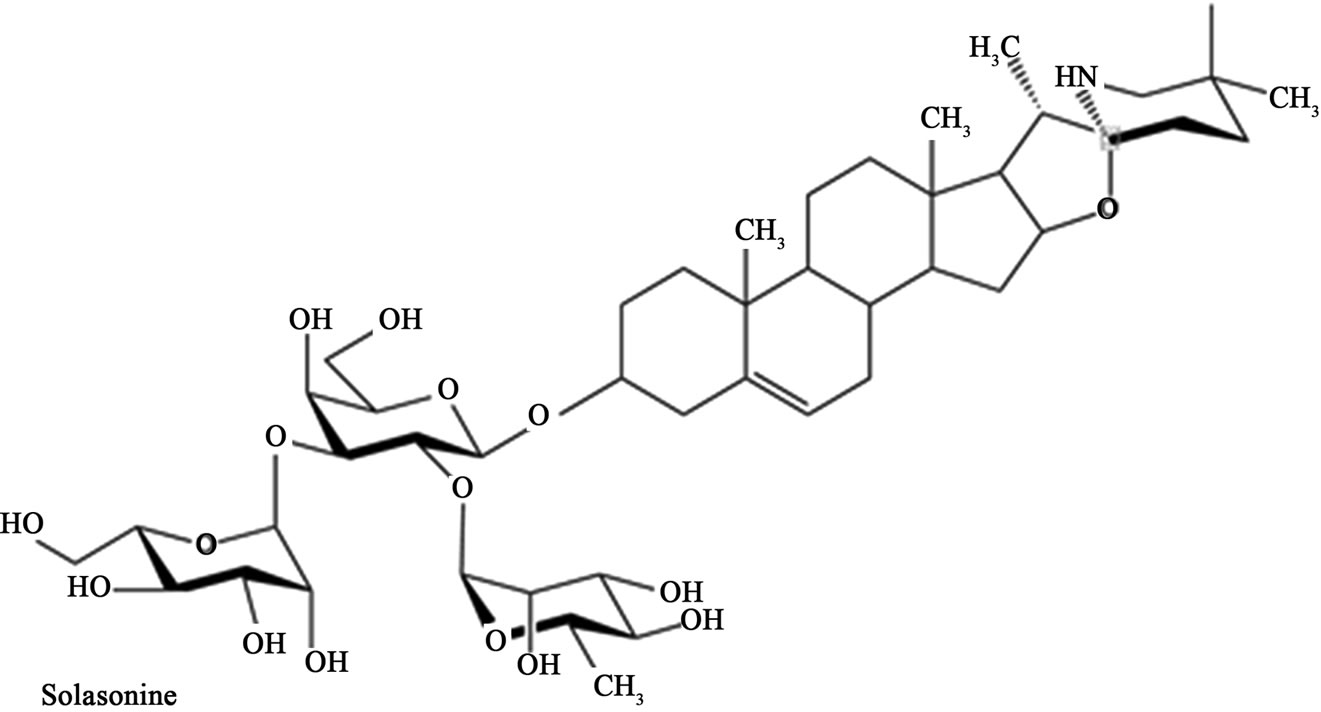

Figure 1. Chemical structures of solasonine and solamargine.

Figure 2. Mass spectra of solasonine and solamargine.
3. Results
The patient had several BCCs that were treated only with the topical cream formulation CuradermBEC5. The six BCC lesions which were less than 2 cm in diameter all regressed after 6 weeks with CuradermBEC5 topical treatment (not shown).
Figure 3(a) shows a BCC before CuradermBEC5 topical treatment. The lesion’s size was approximately 3 cm × 4 cm and protruded 0.5 cm from the skin’s surface. Figure 3(b) shows that the lesion was reduced considerably after six weeks of treatment. Figure 3(c) shows that the lesion was completely ablated 16 weeks after commencement of CuradermBEC5 therapy.
Similarly, Figure 4 shows another BCC of approximately 3 cm × 3 cm × 0.5 cm before (Figure 4(a)), six weeks during treatment (Figure 4(b)). After 16 weeks the lesion had clinically regressed (Figure 4(c)).
Figure 5(a) illustrates what appears to be three indi vidual BCCs. Closer clinical examination revealed that the three BCC lesions were interconnected. Six weeks of topical CuradermBEC5 therapy resulted in the erosion of the entire area (4 cm × 4 cm) including the spaces between the previously apparent distinctive lesions (Figure 5(b)). After 16 weeks there was no evidence of residual tumour (Figure 5(c)).
Figures 6(a) and (b) show a raised BCC of approximately 3 cm × 3 cm × 1 cm before CuradermBEC5 treatment. Six weeks of treatment resulted in haemorrhagic necrosis of the tumour mass (Figure 6(c)) and 16 weeks after commencement of treatment, complete remission had occurred (Figure 6(d)).
Clinical evaluation indicated that the lesions of Figures 3-6 were eliminated 14 weeks after topical CuradermBEC5 treatments. The cosmetic end results, as shown at 16 weeks after commencement of treatments, were outstanding and there were no scar tissues at the sites of the treated lesions. During the first week of treatment, the patient experienced a stinging to mild burning sensation which lasted for approximately 20 minutes after topical application of CuradermBEC5.
Figure 7(a) shows a large 5 cm × 5 cm BCC before combination treatment with intralesion injection followed by CuradermBEC5 topical treatment. Figure 7(b) illustrates that this lesion protruded 1.5 cm from the skin’s surface. Two days after the first injection of BEC in DMSO, but before the second injection, the lesion had already started to breakdown (Figure 7(c)). The odour of DMSO and necrosing tissue were noticeable. Two days after the second and final injection of BEC in DMSO, the necrosing lesion had released itself from the skin Figure 7(d). The patient experienced some pain when BEC in DMSO was
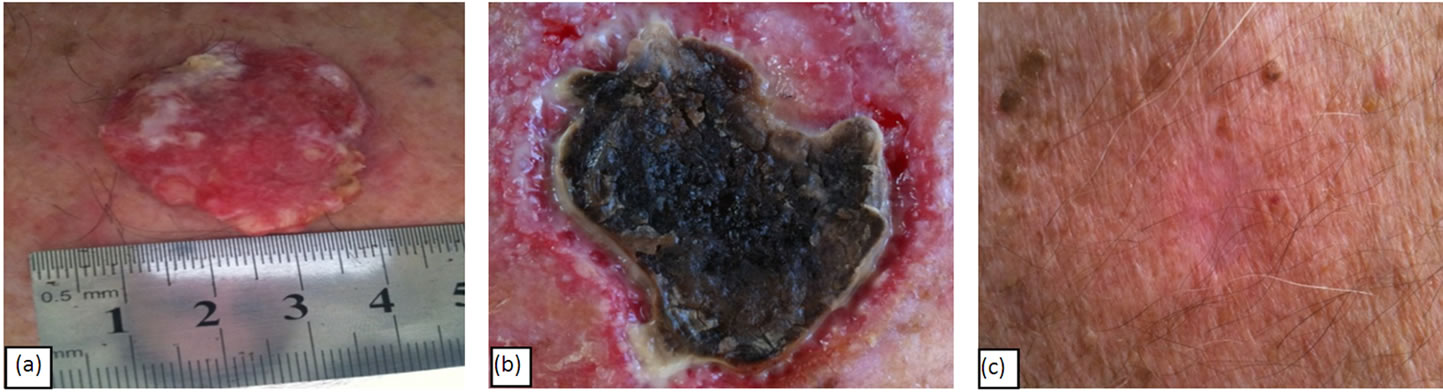
Figure 3. A BCC of approximately 3 cm × 4 cm × 0.5 cm before (a), during (b) and after (c) CuradermBEC5 topical treatment. Complete clinical remission was observed 14 weeks after topical CuradermBEC5 treatment.
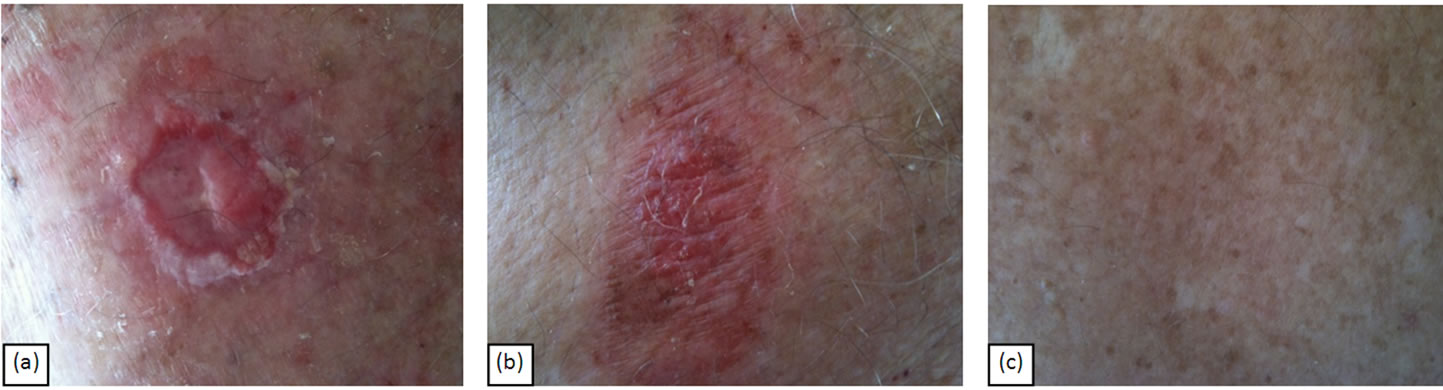
Figure 4. BCC of approximately 3 cm × 3 cm × 0.5 cm before treatment (a), during treatment (b) and after (c) CuradermBEC5 topical treatment. After 14 weeks of topical application with CuradermBEC5 the entire lesion had completely regressed.
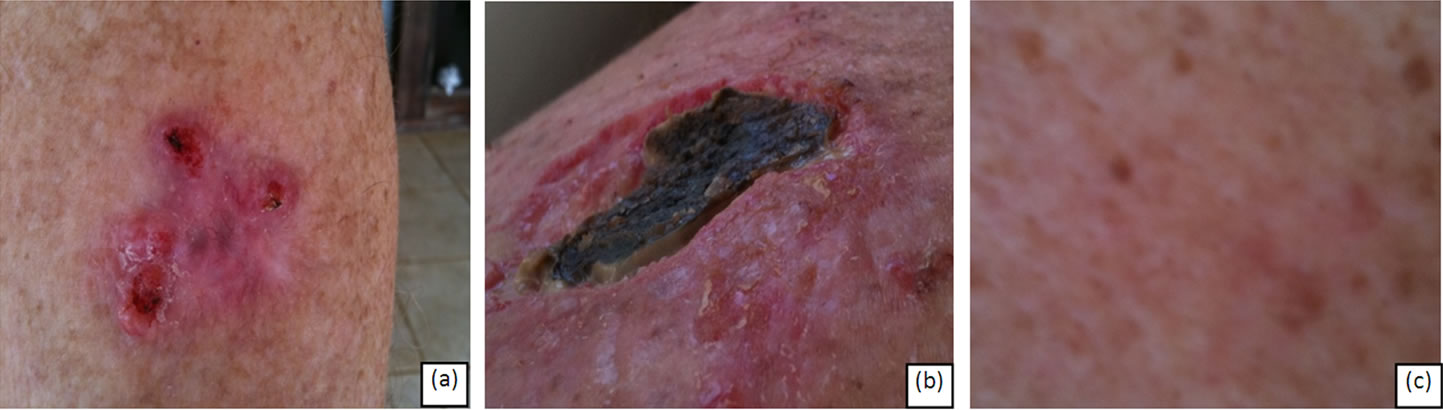
Figure 5. A large BCC with three distinct focal areas measuring approximately 4 cm × 4 cm (a). During CuradermBEC5 topical application erosion of the entire area occurred and it can be seen that the original distinct areas constituted a large interconnected BCC (b). The entire lesion was eliminated after treatment (c). Treatment with CuradermBEC5 topical application was for 14 weeks.
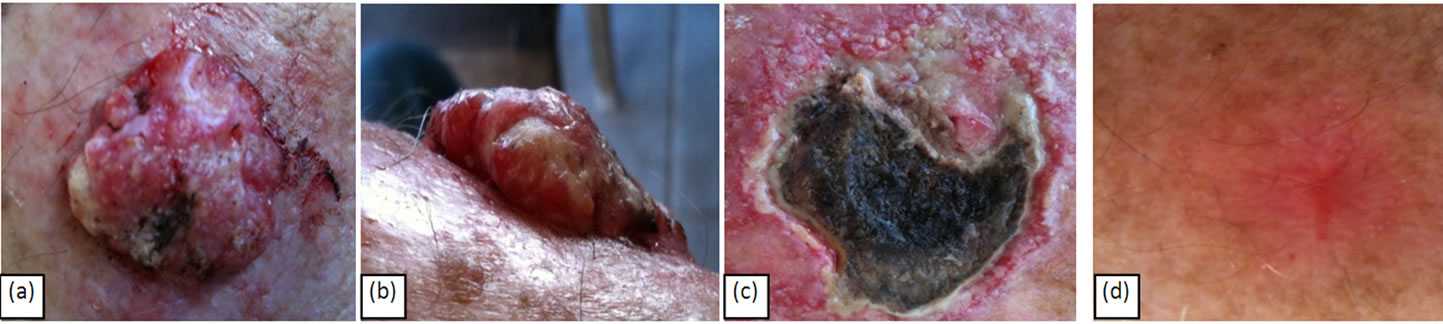
Figure 6. A large (a); protruding (b); BCC of approximately 1 cm × 3 cm × 3 cm before treatment. Haemorrhagic necrosis of the tumour mass occurred during treatment with CuradermBEC5 (c). After 14 weeks of CuradermBEC5 topical application the lesion had completely regressed. The impressive cosmetic end result (d) is seen 16 weeks after commencement of
CuradermBEC5 therapy.


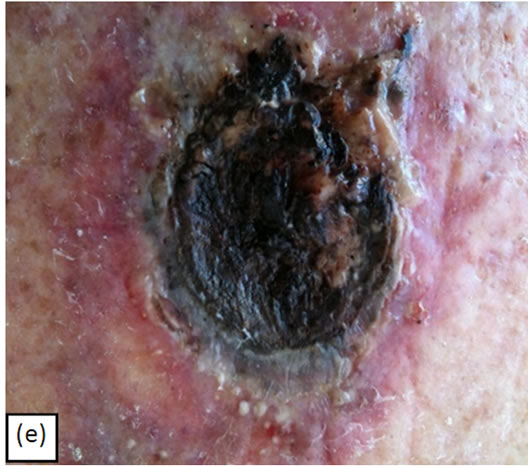
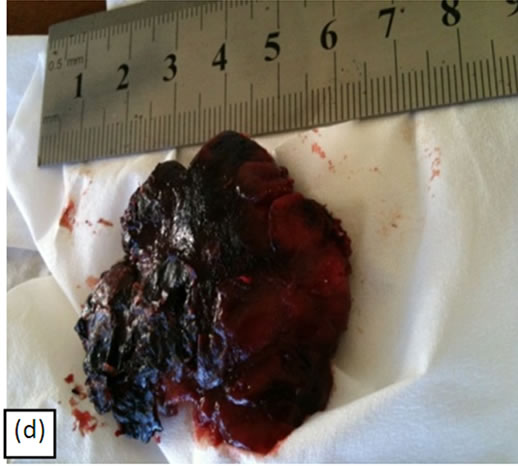
Figure 7. A large BCC, 5 cm × 5 cm (a). protruding 1.5 cm from the skin (b) before combination intralesion—topical applications. Two days after the first intralesion injection the lesion started to breakdown (c). Two days after the second and final intralesion injection the lesion released itself from the skin (d). There was an indentation in the skin where the lesion had separated itself (e). From here-on the lesion was then treated for six weeks with topical application of CuradermBEC5 treatment only. Sixteen weeks after commencement of the combination treatment, there was no evidence of residual tumour (f). The observed scar tissue was probably from a previous surgical procedure. The cosmetic result was astonishing.
injected, especially at the site of the juncture of the lesion with the surrounding normal skin. The pain lasted for several minutes. After the bulk of the lesion was separated from the skin Figure 7(e), the patient followed up with CuradermBEC5 topical application. Clinically this lesion was eliminated after 6 weeks of CuradermBEC5 topical treatment. Sixteen weeks after commencement of treatment, there was no evidence of residual tumour Figure 7(f). However, some scar tissue could be seen. The patient recollected that the scar tissue was due to a previous surgical procedure where a skin-flap was performed. However, after the previous surgical removal, the lesion reoccurred with rapid growth.
Figure 8 illustrates that intralesion injection of BEC solution alone, without additional topical CuradermBEC5 therapy, eliminates a large melanoma on a horse. In this case it can be seen clearly that there is a serumal exudate caused by the injection. This indicates that acute wound healing is in process despite that the cancer is being eliminated during treatment. Thus, while the cancer cells are being killed in the presence of BEC, normal cells are replacing the killed cancer cells and normal cells are not killed by BEC therapy.
4. Discussion
SRGs and thus BEC are a new class of antineoplastic chemotherapeutic agents. BEC is extracted from various Solanum plant materials and expresses its antineoplastic activities by highly ordered specificity and cancer cell destroying ability due to apoptosis and oncosis triggered by extrinsic and intrinsic apoptotic pathways. It has also been reported that BEC or its individual component solamargine is far more effective than other established antineoplastics—taxol, cisplatin, gemcitabine, camphothecin, vinblastine, methotrexate, 5-fluorouracil, epirubicin and cyclophosphamide [1,8,21].
The observations in this current report that CuradermBEC5 eliminates BCCs confirm many other published studies [1-3,19,20,24,28,29,33,35-37,41]. The cosmetic end results after CuradermBEC5 therapy are, as previously reported, excellent. This study confirms previous reports that topical CuradermBEC5 therapy removes smaller BCCs within 6 weeks of treatment and for larger BCCs it takes approximately 14 weeks [1-3,19,20,24,28,29,33,35-37,41].
Previous studies have shown that intralesion administration of BEC in DMSO into very large skin tumours in
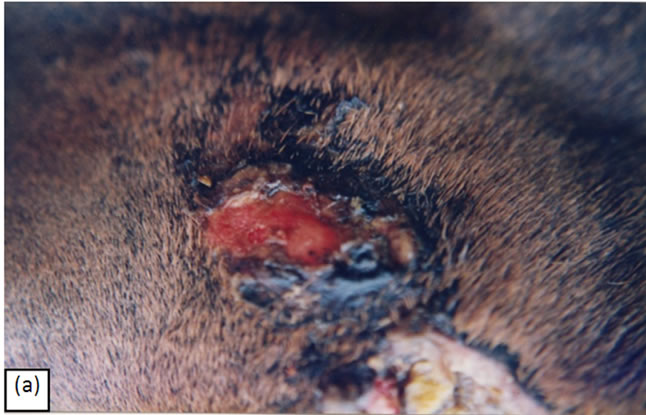
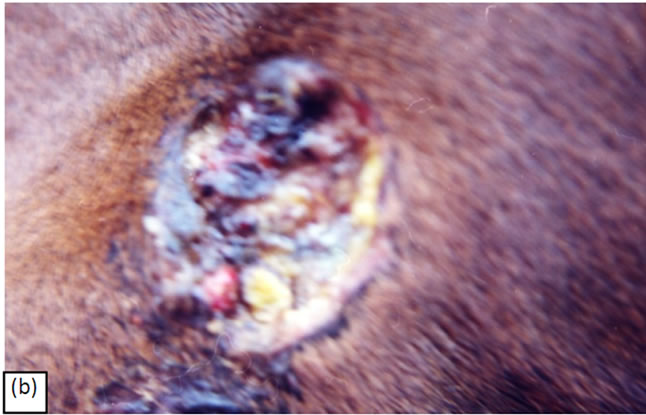


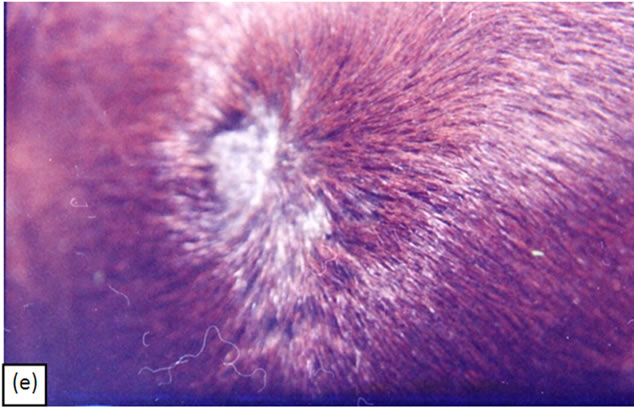
Figure 8. A melanoma on the chest of a horse before intralesion injection of BEC (a). After the first injection the lesion started to breakdown (b) and after the second and last intralesion administration of BEC, massive necrosis of the tumour was observed (c), (d). No further treatment was done after the second intralesion injection. Twelve weeks after intralesion injection of BEC, the melanoma was completely eliminated (e). There was no recurrence for at least 5 years after treatment.
animals resulted in removal of such lesions with excellent cosmetic outcomes [1-3,21]. A report on a human subject has shown that 2 injections of BEC into a large SCC on the head resulted in the tumour collapsing and reduction in size [1-3,21]. The large BCC injected with BEC in DMSO in this current study also exhibited rapid reduction in size. BEC intralesion administration resulted in the removal of the large extruding mass of the tumour two days after the second injection. To ensure complete removal of any possible remaining tumour cells after the intralesion injections the patient continued treatment with the CuradermBEC5 cream. After 6 weeks of CuradermBEC5 therapy the lesion was clinically ablated. Clinical observations indicate that the initial intralesion injections vastly reduced the subsequent time span treatment period with CuradermBEC5 therapy. Because of the similar sizes of lesions treated by CuradermBEC5 alone, and the lesion treated with intralesion and CuradermBEC5, it appears that the combination therapy reduced the treatment period from 14 weeks to 6 weeks. Again, it was shown that the cosmetic result was excellent, although it cannot be excluded that the observed scar tissue was the same scar that was the result of the previous surgical procedure or if BEC may have contributed to its existence. Nevertheless, the cosmetic end result was very impressive.
Toxicity, lack of specificity and lack of efficacy with current antimitotic chemotherapeutic agents have led to more and more use of combination therapies when treating cancer. Indeed, it has been shown that a combination of BEC with the widely used anticancer drug cisplatin has resulted in the effective treatment of cisplatin-resistant cancer cells, particularly lung cancer cells [10,11] and breast cancer cells [22,30].
Topical CuradermBEC5 therapy successfully treats squamous cell carcinoma on the penis of humans [2,3,19,20]. Intralesion administration of BEC into large multiple squamous cell carcinomas on the penis of a horse completely removes these tumours with excellent outcomes [2,3,21].
Successful treatment of Bowen’s disease (SCC in situ) of the penis with CuradermBEC5 was recently reported [43]. This tissue-sparing technique preserves functionality of the penis, is superior to other more-destructive treatment options for Bowen’s disease of the penis and confirms the efficacy of BEC and CuradermBEC5 when treating SCCs on the penis [2,3,20,21].
The main purpose of using combination therapy of BEC with other antimitotic drugs is because of the different modes of actions of the individual antineoplastic agents.
The purpose for using BEC combination therapy as a liquid intralesion and topical CuradermBEC5 cream, in this current communication, is not because there is a difference in the mode of action of the combination therapy. In the current study it is the bioavailability of BEC to the cancer cells that is much enhanced when using the intralesion injections.
Massive haemorrhagic necrosis of the BCC tumour mass in the human patient occurred after intralesion injection. The large tumour separated entirely and dislocated itself from the skin. These observations are similar to a previous report where it was shown that a horse with multiple large SCCs on its penis was injected intralesionally with BEC [1-3,21].
Intralesion injection of BEC in the large melanoma in a horse resulted in complete removal of the lesion. There were no apparent adverse effects. There was no recurrence after more than 5 years follow-up. The results obtained in this study confirms previously reported studies where it was shown that intralesion therapy with BEC of large tumours of up to 0.5 kg were successfully removed. The specificity of BEC towards cancerous tissues was also shown in those studies. When normal tissue was injected with BEC no adverse effects occurred [1-3,21]. Again, as with humans, the final cosmetic outcome after intralesion administration into very large tumours, was excellent [1-3,21].
The current observations are important, because it was previously shown that human melanoma cells in cell culture studies are very susceptible to treatment with BEC [1-4]. Most recent studies have shown that intralesion administration of BEC may also have a very positive effect on immunological responses to tumours [24], how such observations relate to this current report is unknown. Further studies are warranted to establish whether intralesion injection of BEC into large BCCs, large nonmetastasized SCCs and non-metastasized melanoma (lentigo maligna or melanoma in situ) in man will result in the effective and rapid removal of these serious tumours.
Finally, the report confirms that smaller skin tumours can successfully be treated topically with CuradermBEC5 alone, resulting in excellent cosmetic outcomes. Studies of topical application of CuradermBEC5 have now been reported for malignant lesions on the face, trunk, limbs and penis [1-3,19-21,24,29,33-37,41,43]. During treatment, patients develop areas of inflammation in and around the tumour, which then usually necrose, leading to ulceration and healing by re-epithelialization. The endpoint of therapy is achieved with regrowth of new skin at the treatment site. The cosmetic end results are most impressive. As little as two intralesion injections of low doses of BEC are required to eliminate a large melanoma, and, also reduces treatment time by more than half when very large skin cancers are treated with CuradermBEC5. The cosmetic outcomes after CuradermBEC5 are almost unthinkable.
REFERENCES
- T. R. Chase, “Curaderm BEC5 for Skin Cancers, Is It? An Overview,” Journal Cancer Therapy, Vol. 2, No. 5, 2011, pp. 728-745. doi:10.4236/jct.2011.25099
- B. E. Cham, “Eggplant Wins War against Skin Cancer. Conquest on the Horizon for Internal Cancers?” in Preparation.
- B. E. Cham, “The Eggplant Cancer Cure. A Treatment for Skin Cancer and New Hope for Other Cancers from Nature’s Pharmacy,” Smart Publications, Petaluma, 2007, p. 122.
- B. E. Cham, M. Gilliver and L. Wilson, “Antitumour Effects of Glycoalkaloids Isolated from Solanum sodomaeum L.,” Planta Medica, Vol. 53, No. 1, 1987, pp. 34- 36. doi:10.1055/s-2006-962612
- B. E. Cham and B. Daunter, “Solasodine Glycosides. Selective Cytoxicity for Cancer Cells and Inhibition of Cytotoxicity by Rhamnose in Mice with Sarcoma 180,” Cancer Letters, Vol. 55, No. 3, 1990, pp. 221-225. doi:10.1016/0304-3835(90)90122-E
- M. Freedman, K. R. Lee, H. J. Kim, I. S. Lee and N. Kozukue, “Anticarcinogenic Effects of Glycoalkaloids from Potatoes against Human Cervical, Liver, Lymphoma and Stomach Cancer Cells,” Journal Agriculture Food Chemistry, Vol. 53, No. 15, 2005, pp. 6162-6169. doi:10.1021/jf050620p
- K. W. Kuo, S. H. Hsu, Y. P. Li, W. L. Lin, L. F. Liu, L. C. Chang, C. C. Lin, C. N. Lin and H. M. Sheu, “Anticancer Activity Evaluation of the Solanum Glycoalkaloid Solamargine. Triggering Apoptosis in Human Hepatoma Cells,” Biochemistry Pharmacology, Vol. 60, No. 2, 2000, pp. 1865-1873. doi:10.1016/S0006-2952(00)00506-2
- K. W. Kuo and C. N. Lin, “Pharmacological Composition for Treating Cancer Cells,” United States Patent 6, 214, 803, 1999.
- K. R. Lee, N. Kozukue, J. S. Han, J. H. Park, E. Y. Chang, E. J. Back, J. S. Chang and M. Friedman, “Glycoalkaloids and Metabolites Inhibit the Growth of Human Colon (HT29) and Liver (HepG2) Cancer Cells,” Journal Agriculture Food Chemistry, Vol. 52, No. 10, 2004, pp. 2832- 2839. doi:10.1021/jf030526d
- C. H. Liang, L. F. Liu, L. Y. Shiu, Y. S. Huang, L. C. Chang and K. W. Kuo, “Action of Solamargine on TNFs and Cisplatin-Resistant Human Lung Cancer Cells,” Biochemistry Biophysics Research Communication, Vol. 322, No. 3, 2004, pp. 751-758. doi:10.1016/j.bbrc.2004.07.183
- C. H. Liang, L. Y. Shiu, L. C. Chang, H. M. Sheu and K. W. Kuo, “Solamargine Upregulation of Fas, Downregulation of HER 2, and Enhancement of Cytotoxicity Using Epirubicin in NSCLC Cells,” Molecular Nutrition Food Research, Vol. 51, No. 8, 2007, pp. 999-1005. doi:10.1002/mnfr.200700044
- L. F. Liu, C. H. Liang, L. Y. Shiu, W. L. Lin, C. C. Lin and K. W. Kuo, “Action of Solamargine on Human Lung Cancer Cells-Enhancement of the Susceptibility of Cancer Cells to TNFs,” FEBS Letters, Vol. 577, No. 1-2, 2004, pp. 67-74. doi:10.1016/j.febslet.2004.09.064
- M. Millward, A. Powell, P. Daly, S. Tyson, R. Ferguson and S. Carter, “Results of Phase I Clinical Trials of Coramsine in Patients with Advanced Solid Tumors,” Journal Clinical Oncology, Vol. 2, No. 18, 2006, p. 2070.
- T. Nakamura, C. Komori, Y. Lee, F. Hashinoto, S. Yahara, T. Nohara and A. Ejina, “Cytotoxic Activities of Solanum Steroidal Glycosides,” Biological Biopharmacology Bulletin, Vol. 19, No. 4, 1996, pp. 564-566. doi:10.1248/bpb.19.564
- K. Ono, S. O. Kin and J. Han, “Susceptibility of Lysosomes to Rupture Is a Determinant for Plasma Membrane Disruption in Tumor Necrosis Factor Alpha-Induced Cell Death,” Molecular Cell Biology, Vol. 23, No. 2, 2003, pp. 665-676. doi:10.1128/MCB.23.2.665-676.2003
- M. Ono, K. Nishimura, K. Suzuki, T. Fukushima, K. Igoshi, H. Yoshimitsu, T. Ikeda and T. Nohara, “Steroidal Glycosides from the Underground Parts of Solanum Sodomaeum,” Chemistry Pharmacology Bulletin, Vol. 54, No. 2, 2006, pp. 230-233. doi:10.1248/cpb.54.230
- J. G. Roddick, A. L. Rÿrenber and M. Weissenber, “Membrane-Disrupting Properties of the Steroidal Glycoalkaloids Solasonine and Solamargine,” Phyto Chemistry, Vol. 29, No. 5, 1990, pp. 1513-1518. doi:10.1016/0031-9422(90)80111-S
- P. Vijayan, K. S. Vinod, S. A. Dhanaraj, S. Badami and B. Suresh, “In Vitro Cytotoxicity and Anti-Tumour Properties of the Total Alkaloid Fraction of Unripe Fruits of Solanum pseudocapsicum,” Pharmaceutical Biology, Vol. 40, No. 6, 2002, pp. 456-460. doi:10.1076/phbi.40.6.456.8444
- B. E. Cham, “Solasodine Rhamnosyl Glycosides Specifically Bind Cancer Cell Receptors and Induce Apoptosis and Necrosis. Treatment for Skin Cancer and Hope for Internal Cancers,” Research Journal Biological Sciences, Vol. 2, No. 7, 2007, pp. 503-514.
- B. E. Cham, “Solasodine Rhamnosyl Glycosides in a Cream Formulation Is Effective for Treating Large and Troublesome Skin Cancers,” Research Journal Biological Sciences, Vol. 2, No. 7, 2007, pp. 749-761.
- B. E. Cham, “Cancer Intralesion Chemotherapy with Solasodine Rhamnosyl Glycosides,” Research Journal Biological Sciences, Vol. 3, 2008, pp. 1008-1017.
- L. Y. Shiu, L. C. Chang, C. H. Liang, Y. S. Huang, H. M. Sheu and K. W. Kuo, “Solamargine Induces Apoptosis and Sensitizes Breast Cancer Cells to Cisplatin,” Food Chemistry Toxicology, Vol. 45, No. 11, 2007, pp. 2155- 2164. doi:10.1016/j.fct.2007.05.009
- Solamargine anticancer. Google Search
- B. E. Cham and T. R. Chase, “Solasodine Rhamnosyl Glycosides Cause Apoptosis in Cancer Cells, Do They also Prime the Immune System Resulting in Long Term Protection against Cancer?” Planta Medica, Vol. 78, 2012, pp. 349-353. doi:10.1055/s-0031-1298149
- R. G. Van der Most, R. Himbeck, S. Aarens, S. J. Carter, I. Larma, C. Robinson, A. Currice and R. A. Lake, “Antitumor Efficacy of the Novel Chemotherapeutic Agent Coramsine Is Potentiated by Cotreatment with CpGContaining Oligodeoxynucleotides,” Journal Immunotherapy, Vol. 29, No. 2, 2006, pp. 134-142. doi:10.1097/01.cji.0000187958.38179.a9
- B. Daunter and B. E. Cham, “Solasodine Glycosides. In Vitro Preferential Cytotoxicity for Human Cancer Cells,” Cancer Letters, Vol. 55, No. 3, 1990, pp. 209-220. doi:10.1016/0304-3835(90)90121-D
- X. Li, Y. Zhao, W. K. K. Wu, S. Liu, M. Cui and H. Lou, “Solamargine Induces Apoptosis Associated with p53 Transcription-Dependent and Transcription-Independent Pathways in Human Osteosarcoma U20S Cells,” Life Science, Vol. 88, No. 7-8, 2011, pp. 314-321. doi:10.1016/j.lfs.2010.12.006
- B. E. Cham, “Monograph on the Compound BEC,” Drugs of the Future, Vol. 13, 1988, pp. 714-716.
- B. E. Cham, “Solasodine Glycosides as Anti-Cancer Agents: Pre-Clinical and Clinical Studies,” Asia Pacific Journal Pharmacology, Vol. 9, No. 2, 1994, pp. 113-118.
- L. Y. Shiu, C. H. Liang, Y. S. Huang, H. M. Sheu and K. W. Kuo, “Downregulation of HER2/neu Receptor by Solamargine Enhances Anticancer Drug-Mediated Cytotoxicity in Breast Cancer Cells with High-Expressing HER2/ neu,” Cellular Biology Toxicology, Vol. 24, No. 1, 2008, pp. 1-10. doi:10.1007/s10565-007-9010-5
- C. Paquet, A. T. Sané, M. Beauchemin and R. Bertrand, “Caspaseand Mitochondrial Dysfunction Dependent Mechanisms of Lysosomal Leakage and Cathepsin B Activation in DNA Damage-Induced Apoptosis,” Leukemia, Vol. 19, No. 5, 2005, pp. 784-791. doi:10.1038/sj.leu.2403717
- L. Sun, Y. Zhao, H. Yuan, X. Li, A. Cheng and H. Lou, “Solamargine, a Steroidal Alkaloid Glycoside, Induces Oncosis in Human K562 Leukemia and Squamous Cell Carcinoma KB Cells,” Cancer Chemotherapy Pharmacol, Vol. 65, No. 4, 2010, pp. 1125-1130.
- B. E. Cham and B. Daunter, “Topical Treatment of Pre-Malignant and Malignant Skin Cancers with Curaderm,” Drugs of Today, Vol. 26, No. 3, 1990, pp. 55-58.
- B. E. Cham, B. Daunter and R. Evans, “Topical Treatment of Malignant and Premalignant Skin Cancers by Very Low Concentrations of a Standard Mixture of Solasodine Glycosides,” Cancer Letters, Vol. 59, No. 3, 1991, pp. 183-192. doi:10.1016/0304-3835(91)90140-D
- B. E. Cham and M. M. Meares, “Glycoalkaloids from Solanum sodomaeum L. Are Effective in the Treatment of Skin Cancers in Man,” Cancer Letters, Vol. 36, No. 2, 1987, pp. 111-118. doi:10.1016/0304-3835(87)90081-4
- S. Punjabi, I. Cook, P. Kersey, R. Marks, A. Finlay and G. Sharpe, “A Double Blind, Multi-Centre Parallel Group Study of BEC-5 Cream in Basal Cell Carcinoma,” European Academy Dermatology Venereology, Vol. 14, 2000, pp. 47-60.
- S. Punjabi, L. J. Cook, P. Kersey, R. Marks and R. Cerio, “Solasodine Glycoalkaloids: A Novel Topical Therapy for Basal Cell Carcinoma. A Double-Blind, Randomized, Placebo-Controlled, Parallel Group, Multicentre Study,” International Journal Dermatology, Vol. 47, No. 1, 2008, pp. 78-82. doi:10.1111/j.1365-4632.2007.03363.x
- H. W. Rogers, N. A. Weinstock, A. R. Harris, M. R. Hinckley, S. R. Feldman, A. B. Fleischer and B. M. Coldiron, “Incidence Estimate of Nonmelanoma Skin Cancer in the United States,” Archives Dermatology, Vol. 146, No. 3, 2010, pp. 283-287. doi:10.1001/archdermatol.2010.19
- M. B. Lens and M. Dawes, “Epidemiological Trends of Cutaneous Malignant Melanoma: Incidence of Cutaneous Malignant Melanoma,” British Journal of Dermatology, Vol. 150, No. 2, 2004, pp. 179-185. doi:10.1111/j.1365-2133.2004.05708.x
- L. A. E. Sussman and D. F. Liggins, “Incompletely Excised Basal Cell Carcinoma a Management Dilemma?” Australia New Zealand Journal Surgery, Vol. 66, No. 5, 1996, pp. 276-278. doi:10.1111/j.1445-2197.1996.tb01184.x
- B. E. Cham, “Topical Solasodine Rhamnosyl Glycosides Derived from the Eggplant Treats Large Skin Cancers: Two Case Reports,” International Journal Clinical Medicine, Vol. 2, No. 4, 2011, pp. 473-477. doi:10.4236/ijcm.2011.24080
- B. E. Cham and L. Wilson, “HPLC of Glycoalkaloids from Solanum sodomaeum,” Planta Medica, Vol. 53, No. 1, 1987, pp. 34-36. doi:10.1055/s-2006-962612
- L. H. Goldberg, J. M. Landau, M. N. Moody and I. J. Vergilis-Kalner, “Treatment of Bowen’s Disease on the Penis with Low Concentration of a Standard Mixture of Solasodine Glycosides and Liquid Nitrogen,” Dermatologic Surgery, Vol. 37, No. 6, 2011, pp. 858-861. doi:10.1111/j.1524-4725.2011.02014.x

