Open Journal of Thoracic Surgery
Vol.3 No.2(2013), Article ID:32640,5 pages DOI:10.4236/ojts.2013.32013
A Primary Epithelioid Hemangioendothelioma of the Right Atrium: Report of a Case and Literature Review
![]()
Department of Cardiovascular Surgery, Tachikawa Medical Center, Nagaoka, Japan.
Email: tsugimoto-cvs@umin.ac.jp
Copyright © 2013 Tsutomu Sugimoto et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received February 15th, 2013; revised March 17th, 2013; accepted March 25th, 2013
Keywords: Epithelioid Hemangioendothelioma; Heart; Right Atrium
ABSTRACT
Primary cardiac epithelioid hemangioendothelioma (EHE) is extremely uncommon. To our knowledge, only fourteen cases have been reported. We report a case of a primary EHE of the right atrium which incidentally detected in 77-year-old woman. Echocardiograhpy revealed a 26 × 16 mm right atrial mass attached to the base of the interatrial septum and moderate regurgitation of the tricuspid valve. The tumor and stalk were completely resected from the interatrial septum with a partial resection of the septal leaflet of the tricuspid valve. The patient had an uneventful postoperative course and no signs of recurrence nor metastasis of EHE 4 years after operation. Because of malignant potential of EHE, a longer period of close follow up is necessary.
1. Case Report
Epithelioid hemangioendothelioma (EHE) most often presents as a painful mass in adult superficial and deep soft tissue. Primary lesions are known to present elsewhere, namely lung, liver and bone; however, cardiac occurrences are extremely rare. EHE belongs to a family of vascular lesions characterized by epithelioid endothelial cells, which also includes epithelioid haemangioma and epithlioid angiosarcoma. Although previously be considered as a borderline or low grade malignant vascular lesion, EHE is regarded in the World Health Organization classification as a malignant tumor along with angiosarcomas, because of its local aggressiveness and its metastasizing potential.
In this report, we describe a patient with an EHE of the right atrium that was successfully resected.
2. Clinical Summary
A 77-year-old woman undergoing treatment for hypertension and hyperlipidemia was referred to our hospital because her routine UCG showed an intracardiac mass. Physical examination did not reveal any abnormalities. ECG showed a QS pattern in V1 ~ V3 and slight ST elevation in V2 ~ V4. Chest X-ray showed slight cardiomegaly with a cardiothoracic ratio of 57.9%. Echocardiograhpy revealed a 26 × 16 mm right atrial mass attached to the base of the interatrial septum and moderate regurgitation of the tricuspid valve. CT showed a 26 × 16 mm noncalcified tumor located on the right interatrial septum with no detect signs of metastasis.
The operation was performed through a median sternotomy, and cardiopulmonary bypass was established with aortic and bicaval cannulation. Oblique right atriotomy revealed a tumor mass extending to near the traingle of Koch with a single stalk and adhesion to the septal leaflet of the tricuspid valve (Figure 1). The tumor and stalk were completely resected from the inter atrial septum with a partial resection of the septal leaflet. The partial defect of septal leaflet was repaired by direct closure.
On gross examination, the tumor was 25 mm. The cross-section of the tumor showed solid and monolocular cystic mass with mucinous contents. Histological examination showed immature pattern of vascular proliferation with many vessels poorly canalized in a myxoid matrix (Figure 2). These vessels were lined by epithelioid endothelial cells which were typified by rich eosinophilic cytoplasm and large hyperchromatic nuclei. Pleomorphism or mitotic figures were not seen (Figure 3). Immunohistochemical examination showed cytoplasmic expression of CD31, CD34, Factor VIII, and Vimentin.
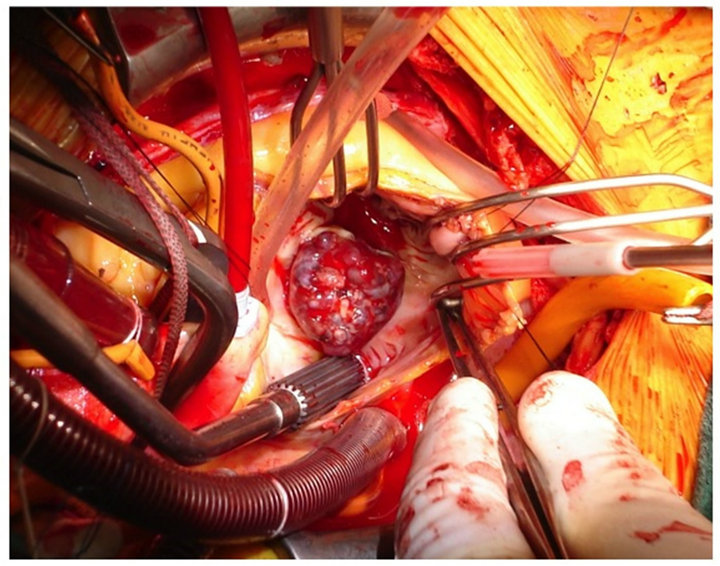
Figure 1. Operative findings.

Figure 2. Histological examination. The tumor revealed the tumor composed of plump, rounded endothelial cells arranged in nests or cords (Hematoxylin-eosin, original magnification ×100).

Figure 3. Histological examination (Hematoxylin-eosin, original magnification ×200).
The patient had an uneventful postoperative course and was discharged on the 21st postoperative day. She has remained well with no signs of recurrence nor metastasis of EHE 4 years after operation.
3. Discussion
Although EHE has been reported to generate from the liver, lung, bone, superficial soft tissue, mediastinum, lymph node, spleen, the thyroid gland and brain, cardiac occurrences are extremely rare. In the reports of liver and lung, the tumors were most apt to occur in middle age females. Although participation of estrogen receptor in the tumor was considered as the reason of predominance in woman, it has not been proved yet [1].
According to literature, primary cardiac EHE has only been reported in 14 patients including this case [2-12] (Table 1). The average age was 49.3 years (2 months - 77 years old), and the ratio of man to woman was 5 to 9. EHE occurs with similar frequencies in both atriums, 3 patients in the right atrium and 4 patients in the left atrium. Tumor located on the cardiac valves was seen in 5 patients, of which 2 were on the mitral valve, and 1 each on the aortic valve, pulmonary valve and tricuspid valve. Only 1 patient showed occurrence in the right ventricle.
Vascular tumors are a heterogenous group of neoplasms consisting of a spectrum of tumors from the extremely benign hemagiomas, to intermediate lesions that are locally aggressive and metastasize, to relatively rare, highly malignant angiasarcomas. Vascular neoplasms are categorized as benign or malignant on the basis of two major anatomic characteristics: 1) the degree of composed vascular cannel differentiation; and 2) the abundance and regularity of the endothelial cell proliferation. Ordinarily, benign neoplasms are composed largely of well-formed vessels with a significant amount of regular endothelial cell proliferation, whereas the malignant tumors are solidly cellular and anaplastic and reproduce slight numbers of only abortive vascular channels [13]. Although it is thought that EHE is positioned as borderline or low grade malignant vascular lesion, it is classified into malignant tumor along with angiosarcomas, according to the recent WHO (World Health Organization) classification, because of its local aggressiveness and its metastasizing potential. EHE is a member of a family of vascular leasions which include EH and epithelioid angiosarcoma. These are linked by the presence of eplithelioid endothelial cells. However, these neoplasms differ also in a pathological features and clinical behavior, and which are classified from begin to borderline to frankly malignant [14].
The major consequences of EHE are the development of various symptoms. The most common presentation is heart failure caused by tumor obstruction of the cardiac chamber, or valve orifice and tumor embolization. Of the seven patients whose tumor occurred in the right heart, 2 patients showed respiratory symptoms such as respiratory pain and pulmonary embolism, 1 patent presented with cough and edema of lower limbs, 1 patient was incidentally diagnosed while he was under observation for another disease, and 1 was diagnosed at autopsy. A variety of symptoms were also seen in the seven patients with tumors occurring in the left heart, such as palpitation, dizziness and fever in 1 patient, shortness of breath in 1 patient, chest pain in 1 patient, stroke in 1 patient,

Table 1. Primary cardiac EHE: Literature review.
and 2 patients were found out a routine echocardiography and 1 patient at a cardiac surgery.
EHE is a unique vascular tumor occurring around medium-sized and large vessels, usually a vein, in the soft tissue of adults. Primary cardiac EHE is extremely uncommon. In EHE, vascular differentiation proceeds through the formation of multicellular, canalized vascular channels. Vascular differentiation in these tumors is more primitive and is expressed primarily at the cellular level. The tumors are composed of short stands or solid nests of rounded to slightly spindled endothelial cells, characterized by an abundant eoshinophilic cytoplasm and large hyperchromatic nuclei [15]. In the literature, the term “hemangioendothelioma” is used to describe various entities including neoplastic angioendotheliomas, systemic angioendotheliomatosis, and malignant hemangioendotheliomas (angiosarcoma). EHE is distinctive and is characterized by an epithelioid appearance and frequent angiocantric location. Immunohistochemically, EHE usually reacts with at least one endothelial marker such as CD31, CD34, factor VIII, and is mostly negative for cytokeratin. CD31 is especially highly sensitive and thought to be the most specific marker of endothelial differentiation [16]. Sarcoma and carcinomas show the opposite immuohistochemical observation. The expression of vascular endothelial markers rules out metastatic adenocarcinaoma or melanoma.
EHE was initially considered as a low grade malignant tumor, nonetheless considering all site of EHE, local recurrence and metastases occur in approximately 15% to 30% of cases, respectively, and mortality varies from 10% ~ 20% [14,17]. Histological factors predictive of malignancy include at least 1 - 2 mitotic figures per ten high power fields, cellular pleomorphism, and tumor necrosis [18]. Weiss and Enzinger reported that tumors classified malignant by these criteria had a 53% rate of metastasis, while benign lesions had a 17%. Similarly, while 31% of malignant tumors were fatal, only 3% of benign lesions result in the death [14].
Benign lesions are best treated by wide local resection. As malignant lesions often metastasize to lymph nodes, and lung, they are best treated by radical exicision [4]. In cardiac EHE, the report of excluding the autopsy case, 12 patients were underwent local or radical surgical excision of the heart and one patient underwent a cardiac transplantation after chemotherapy combining doxorubicin and ifoframide [10]. Follow up information in these reports revealed that 3 patients developed metastasis. Two patients were 4 months after surgery [4] and 1 patient was 2 years after operation [10]. There is no standard therapy for EHE, and recommended treatment varies with it’s malignant potential which should be assessed by the presence of metastasis at time of diagnosis or pathological features. Adjuvant therapy is not suggested. Because of its malignant potential, a longer period of close follow up is necessary.
REFERENCES
- N. P. Ohri, S. A. Yousem, E. Sonmez-Alpan and T. V. Colby, “Estrogen and Progesterone Receptors in Lymphangioleiomyomatosis, Epithelioid Hemangioendothelioma, and Sclerosing Hemangioma of Lung,” American Journal of Clinical Pathology, Vol. 96, No. 4, 1991, pp. 529-535.
- R. H. Hayward and F. L. Korompai, “Endothelioma of the Mitral Valve,” The Annals of Thoracic Surgery, Vol. 28, No. 1, 1979, pp. 87-89. doi:10.1016/S0003-4975(10)63398-X
- K. Singal, “Intracardiac Mass in a Young Woman with a History of Brain Tumor,” Chest, Vol. 92, No. 2, 1987, pp. 337-341. doi:10.1378/chest.92.2.337
- D. Marchiano, F. Fisher and S. Hofstetter, “Epihtlioid Hemangioendothelioma of the Heart with Distant Metastases. A Case Report and Literature Review,” The Journal of Cardiovascular Surgery, Vol. 34, No. 6, 1993, pp. 529-533.
- F. Bille’-turk, R. Padovani, R. Rosario, D. Heckenroth, M. Gracia, G. Duport, P. Bergeron and J. Bille, “Hemangioendothelioma of the Aortic Valve Revealed by Transient Ischemic Episodes,” La Presse Médicale, Vol. 22, No. 38, 1993, p. 1928.
- T. Tansel, U. Aydogan, D. Yilmazbayhan, B. Bilgic, M. Demiryount and E. Onursal, “Epitheloid Hemangioendothelioma of the Heart in Infancy,” The Annals of Thoracic Surgery, Vol. 79, No. 4, 2005, pp. 1402-1405. doi:10.1016/j.athoracsur.2003.10.053
- J. F. Val-Bernal, E. Gracia-Alberdi, J. A. Gutierrez and M. F. Garijo, “Incidental in Vivo of an Epithelioid Hemangioendothelioma of the Mitral Valve,” Pathology International, Vol. 55, 2005, pp. 644-648. doi:10.1111/j.1440-1827.2005.01883.x
- H. Kitamura, H. Okabayashi, M. Hanyu, Y. Soga, T. Nomoto and H. Johno, “Successful Enucleation of a Giant Cardiac Hemangioendothelioma Showing an Unusual Proliferation Pattern,” The Journal of Thoracic and Cardiovascular Surgery, Vol. 130, No. 4, 2005, pp. 1199- 1201. doi:10.1016/j.jtcvs.2005.05.018
- M. Lisy, W. Beierlein, H. Muller, B. Bultmann and G. Ziemer, “Left Atrial Epithelioid Hemangioendothelioma,” The Journal of Thoracic and Cardiovascular Surgery, Vol. 133, No. 3, 2007, pp. 803-804. doi:10.1016/j.jtcvs.2006.09.076
- N. Moulai, O. Chavanon, L. Guillou, M. Noirclerc, D. Blin and E. Brambilla, “Atypical Primary Epithelioid Hemangioendothelioma of the Heart,” Journal of Thoracic Oncology, Vol. 1, No. 2, 2006, pp. 188-189. doi:10.1097/01243894-200602000-00017
- J. Safirstein, S. Aksenov and F. Smith, “Cardiac Epithelioid Hemangioendothelioma with 8-Year Follow up,” Cardiovascular Pathology, Vol. 16, No. 3, 2007, pp. 183- 186. doi:10.1016/j.carpath.2006.10.003
- P. Messias, J. Bernardo and M. J. Antunes, “Primary Left Atrial Hemangioendothelioma,” Interactive CardioVasc Thoracic Surgery, Vol. 7, No. 5, 2008, pp. 945-946. doi:10.1510/icvts.2008.184085
- R. S. Cotran, V. Kumar and S. L Robbins, “Tumors of Blood Vessels,” In: W B. Saunders, Ed., Robbins Pathologic Basis of Disease, WB Saunders Company, Philadelphia, 1989, pp. 587-592.
- S. W. Weiss, K. G. Ishak, D. H. Dail, D. E. Sweet and F. M. Enzinger, “Epithelioid Hemangioendothelioma and Related Lesions,” Seminars in Diagnostic Pathology, Vol. 3, No. 4, 1986, pp. 259-287.
- F. M. Enziger and S. W. Weiss, “Benign Tumors and tumorlike Lesions of Blood Vessels,” In: F. M. Enziger and S. W. Weiss, Eds., Soft Tissue Tumors, 2nd Edition, Mosby, St Louis, 1988, pp. 489-580.
- B. R. De Young, H. F. Frierson Jr., M. N. Ly, D. Smith and P. E. Swanson, “CD31 Immunoreactivity in Carcinomas and Mesotheliomas,” American Journal of Clinical Pathology, Vol. 110, No. 3, 1998, pp. 374-377.
- T. Mentzel, A. Beham, E. Calonje, D. Katenkamp and C. D. Fletcher, “Epihtelioid Hemangioendothelioma of Skin and Soft Tissues: Clinicopathologic and ImmunohistoChemical Study of 30 Cases,” The American Journal of Surgical Pathology, Vol. 21, No. 4, 1997, pp. 363-374. doi:10.1097/00000478-199704000-00001
- S. W. Weiss and F. M. Enzinger, “Epithelioid Hemangioendothelioma: A Vascular Tumor Often Mistaken for Carcinoma,” Cancer, Vol. 50, No. 5, 1982, pp. 970-981. doi:10.1002/1097-0142(19820901)50:5<970::AID-CNCR2820500527>3.0.CO;2-Z

