Natural Resources
Vol.5 No.8(2014), Article ID:47496,11 pages
DOI:10.4236/nr.2014.58038
Linking Climate Variables with Tuber borchii Sporocarps Production
Elena Salerni1*, Claudia Perini1, Lorenzo Gardin2
1BIOCONNET, Biodiversity and Conservation Network, Department of Life Science, University of Siena, Siena, Italy
2Via Guerrazzi, Firenze, Italy
Email: *elena.salerni@unisi.it
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 27 March 2014; revised 29 April 2014; accepted 21 May 2014
ABSTRACT
Tuber borchii is an ectomycorrhizal edible truffle, commonly called “bianchetto” (whitish truffle) to distinguish it from the more valuable white truffle found in Italy (T. magnatum). Although Tuber borchii also has a fairly high commercial value, information on its ecology, and especially its optimum rainfall and temperature values are lacking. In recent years the issue of climate change has steadily grown in importance, not only in the scientific world, but also politically and in civil society. Over the last century there has been a general increase in the temperature in Italy of about 1˚C. Several studies have underlined how climatic changes influence the optimum growth and distribution of various species of truffle. This contribution aims to illustrate the fluctuation of T. borchii sporocarps production in different timescales and show how these alterations are driven by rainfall and temperature factors. The research, carried out in five different natural T. borchii production areas, reveals that the production of truffles is significantly higher after autumn months characterized by abundant rainfall and cold temperatures.
Keywords:Tuber borchii, Rainfall and Temperature Variations, Fruiting Production, Multiple Linear Regression, General Linear Model

1. Introduction
Truffles are ectomycorrhizal subterranean fruiting bodies of ascomycete fungi belonging to the genus Tuber (Trappe, 1979). While the worldwide number of Tuber species presumably exceeds 180 [1] [2] , only a few have edible sporocarps (truffles) of considerable economic and culinary importance [3] -[5] . The most commercially important species are Tuber magnatum Pico and Tuber melanosporum Vittad., but other species such as Tuber aestivum Vittad. and Tuber borchii Vittad. provide excellent truffles and are becoming increasingly popular in the marketplace [4] [6] . In Italy the truffle Tuber borchii is commonly called “bianchetto” (whitish truffle) to distinguish it from the more expensive Tuber magnatum [4] . Tuber borchii growth mainly depends on mutual relationships with a wide range of broadleaf trees, including oak (Quercus spp.), hazel (Corylus avellana L.), poplar (Populus spp.), linden (Tilia spp.), chestnut (Castanea sativa Miller) and alder (Alnus spp.), and coniferous species such as pine (Pinus spp.) and cedar (Citrus spp.) [1] [4] [7] . It is reported to have a wide distribution in Europe, being found from southern Finland to Sicily, and from Ireland to Hungary and Poland [4] . Since its first cultivation in Italy in 2000 T. borchii has become an increasingly popular species: it has high ecological adaptability and low host specificity [6] . However, information on its ecology is lacking and limited to studies of soil, vegetation geology and ectomycorrhizal communities in northern and central Italy [6] [8] [9] , while the aspect of the rainfall and temperature factors on sporocarps production have been virtually ignored.
Sporocarps production varies from year to year, and that the amount, duration and frequency of fruiting are influenced by numerous environmental factors e both biotic and abiotic, as well as complex interactions among them. Several short and long-term field surveys have related fruit body yields recorded as weight to environmental factors, including climate variation, especially rainfall and ambient temperature [10] -[19] .
In recent years the issue of climate change has steadily gained importance—not only in the scientific world, but also politically and in civil society. This has led to a new awareness: it is now widely understood that climate change is not a remote phenomenon, but is already taking place, and that its effects could be very serious. According to the Stern Report [20] , the differences in water availability between regions will become more pronounced in the near future: areas that are already relatively dry, such as the Mediterranean, will see further reductions in the availability of water (up to 30% per year), accompanied by an increase in temperature of 2˚C. According to the National Conference on Climate Change [21] there has been a general increase in temperature in Italy of about 1˚C over the last hundred years. This value is higher than those of both other European regions and the global average (0.76˚C). The areas particularly affected by this increase are the southern regions and some areas of central Italy. Some recent estimates predict a further warming of about 2˚C in the Mediterranean area over the next 50 years.
Several studies have pointed out that climatic changes influence the optimum growth and the distribution of several species of truffle [14] [15] [22] [23] . In this context our investigation seeks to clarify how T. borchii sporocarps production fluctuates, and to what extent these alterations are driven by climatic factors. Climate data (rainfall and temperature), in tandem with split period approaches and multiple linear regression (GLM) models were used to detect temporal variability in the response patterns of T. borchii sporocarps productivity.
2. Materials and Methods
2.1. Study Area
The research was conducted in five different natural T. borchii production areas (A, B, C1, C2 and P), allocated in the Parco Regionaledella Maremma [Marina di Alberese, province of Grosseto, Tuscany: lat. 42˚39'N 11˚2'E, altitude 5 m above sea level (a.s.l.)]. The areas, covering 1121 mq (C1 and C2), 2148 mq (P) and 2238 mq (A and B), are characterized by coniferous woods, where Pinus pinea and P. pinaster are the dominant species, with Erica multiflora L., Pistacia lentiscus L., Myrtus communis L., Juniperus oxycedrus L. subsp. macrocarpa (S. and S.) Bull., Phillyrea angustifolia L., Juncus spp. and Inula spp. present in the natural shrub layer. The areas are characterized by a Mediterranean climate (mean annual precipitation 590 mm and mean annual temperature 14.7˚C), with August being the hottest month (mean temperature over 23.5˚C) and January usually the coldest one (7.3˚C). The most abundant rains are recorded in autumn (October and November), with an average of 90 mm per month, while June and July are the driest months, with only 20 mm of rain per month. The soil, observed in a small profile, is sandy, single grain and calcareous in all horizons (13% - 21% of total CaCO3), with a pH of 8.0 - 8.3. It is similar to that described as representative of the Marina Soil Typological Unit in the Tuscan Regional Soil Map at 1:250.000 scale [24] . According to Soil Taxonomy [25] it belongs to the group of “Typic Xeropsamments, mixed, calcareous, thermic”.
2.2. Climate Assessment and Truffle Production
To perform the climatic analysis, data for the period 1943-1999 were obtained from the meteorological monitoring station situated in Casello Idraulico (Alberese, province of Grosseto). This thermopluviometric data was used to calculate the seasonal precipitation concentration index (the ratio between the total rainfall for each season and a quarter of the mean annual total), the modified Fournier index as modified by [26] (also known as the FFAO index, which expresses the monthly concentration of rainfall, given the sum of the squared monthly rainfall divided by the annual total), as well as the series of other indices (Lang’s rain factor; De Martonne’s index and Emberger’s pluviometric quotient) traditionally used for climatic analysis [27] [28] . Truffle production was assessed weekly from January to April for four years (1996-1999) (190 surveys in total) using trained dogs. All truffles collected were identified morphologically, counted, weighed, measured and recorded.
2.3. Statistical Analysis
The Pearson correlation coefficient was calculated to determine correlations between the number and weight of T. borchii fruiting bodies and parameters (rainfall and temperature) derived from the climate data. The data collected were grouped by area, year and month of sampling, Table 1 shows the variables used in the analysis. The P-value was set at the 5% significance level. Statistical analyses were performed using the STATISTICA 5.0 (Stat Soft. Inc.) package of programmes.The weight of the different variables (rainfall: Pm, Pm-1, Pm-2, Pm-3, Pm-4, WPm; temperature: Tm, Tm-1, Tm-2, Tm-3, Tm-4, WTm) in relation to the production of fruiting bodies was assessed by estimating the significance of the parameters of multiple linear regression in relation to the data derived through the application of a general linear model (GLM). Statistical analysis was performed using the PROC REG procedure in SAS/STAT software (SAS Institute Inc., Cary, NC, USA).
3. Results
During the study period, 1033 (total weight: 5657 g) T. borchii fruiting bodies were collected. The largest number of truffles (527) was collected in area P (2877.25 g), while truffles of greater size were observed in area B (mean 20.47 mm). All truffles were found at an average depth of 3 cm (Table 2).
Table 3 compares the main meteorological information recorded in the area during the study period (1996- 1999) with that recorded 7 and 44 years before, respectively. During the study period (1996-1999) the mean annual temperature was 14.7˚C. The coldest year was 1998, while the warmestwas1996. The average number of days with a mean temperature higher than or equal to 10˚C-considered as an index of the potential duration of the growing season [29] —was relatively high, particularly in 1997 (Table 3). Main meteorological data for the study areas.
Table 2. Truffle production (number of fruiting bodies, total weight, mean diameter and mean depth of discovery) in the five truffières.
Table 4. Truffle production (number of fruiting bodies, total weight, mean diameter and mean depth of discovery) in the five truffières.
The mean annual precipitation during the study period was around 564 mm. In contrast to the temperature, which remained essentially the same compared to the previous 40 years, on average precipitation decreased (Table 3). The mean seasonal distribution of rainfall was not homogeneous, as the seasonal precipitation concentration indices ranged from 0.28 to 2.43 (Table 3). The lowest number of rainy days (1 - 4 days) was always in typically summer months (June and July), except in 1997, when the months with the lowest number of rainy days were February and September.
The modified Fournier index (FFAO) revealed a modest degree of concentration of the monthly rainfall, mostly below 100, with the exception of 1999 when it was 130 and marked a situation of climatic aggressiveness.
In comparison to the previous 40 years, in which Lang’s rain factor (R) was >40, this index was lower during the study period (1996-1999) (Table 3), identifying climatic conditions of great aridity. The same trend was also observed for the De Martonne aridity index (Am), even if the differences were less marked. Emberger’s pluviometric quotient (Q) indicated a sub-humid Mediterranean climate, with the exception of 1998 in which Q = 44.4, indicating a semi-arid Mediterranean climate.
Figure 1 represents the thermopluviometric trend recorded in the study area over the four years, in relation to total truffle production. The highest production of truffles was observed in the months of February and March, especially in February 1997 when 130 truffles (789.14 g) were collected. Of the 4 years of the study, 1998 was the worst year for fruiting: 137 truffles (802.84 g) were collected, compared to 336 (1833.33 g) in the previous year (Figure 1).
In 1996 the rainfall was abundant and well distributed throughout the seasons, while in the following years the rainfall decreased and in 1997 it was concentrated in the month of November, when 227 mm of the annual total of 556 mm of rainfall.
In general, the production of truffles seems to decrease during particularly rainy months (Figure 1), while it seems not to be particularly affected by the average monthly temperature. In fact, the production decreased linearly with increases in rainfall of only 30 - 40 mm per month (Figure 2), while higher monthly rainfalls did not seem to be associated with any regular pattern.
Overall, the precipitation have a negative effect on the production of sporocarps in fact statistically significant negative correlations (p < 0.01) were found (Table 4) between monthly rainfall (Pm) and the number of truffles (reported per hectare) for each month, and year of sampling (Freq-ha) and the sum of their weights (reported per hectare), as well as weight month-to-month (Prod-ha).
Using a multiple linear regression model with the stepwise method and the parameters Pm-3, WPm and WTm a significant amount of the variance was explained, which was not explained by total monthly rainfall (Table 5(a)). When we combined these parameters into a single regression model (Table 5(b)), Pm-3 and WPm had a significant effect on both the number and weights of T. borchii. WTm, which had a significant (negative) effect on Prod-ha (Table 5(a)), had no significant effect when the rate of variance due to precipitation was removed from the general linear model (Table 5(b)).
Table 6 reports the relationship between climatic parameters in the autumn months before truffle production (WPm and WTm) and the weight of the truffles in the five study areas, using a multiple regression model. With the
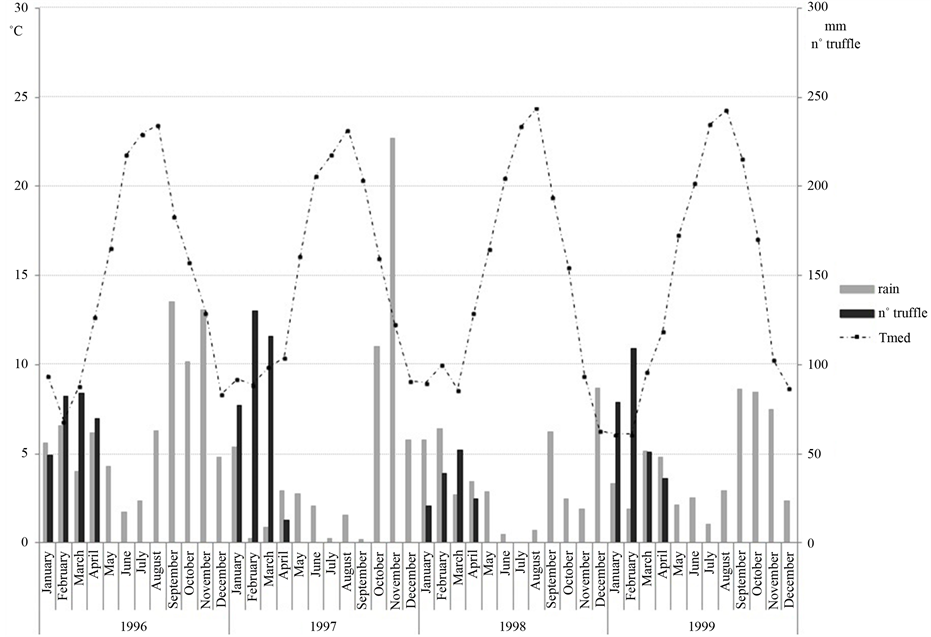
Figure 1. Graph of mean monthly temperature, total monthly rainfall (mm), and number of T. borchii sporocarps in the study period.
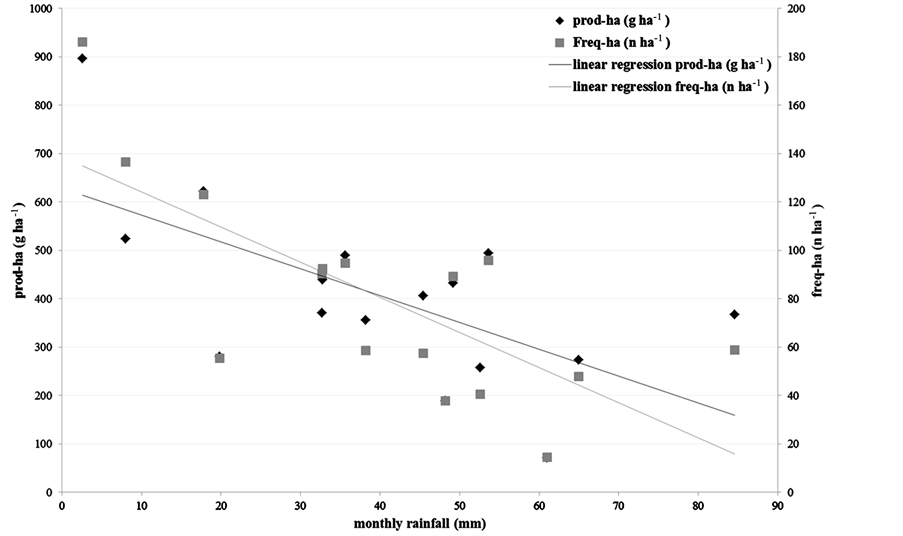
Figure 2. Linear regression analysis between monthly rainfall in the month of collection and truffle production.
Table 4. Pearson correlation coefficient between climatic parameters (rain in mm and average monthly temperature in ˚C) and truffle production. The data collected were grouped by area, year and month of sampling. For each area, we calculated the number of truffles (reported per hectare) for each month and year of sampling (Freq-ha) and the sum of their weights (reported per hectare), as well as their weights month-to-month (Prod-ha). For each cell the following are shown: the correlation coefficient (first row), the significance level (second row) and sample (third row).
exception of area B, WTm had a significant negative effect (p < 0.05) on the weight of truffles in all areas considered.
4. Discussion
This study evaluated the effect of rainfall and temperature conditions in natural T. borchii areas for the first time. Ecological responses to climate variations have been reported for a broad range of taxa, including fungi [14] [30] -[35] , across a variety of spatiotemporal scales [36] [37] . Previous authors have studied only epigeous mushroom and shown how climate change over the last 50 years has extended the fruiting period of several fungal species. Ranging from local surveys [14] [30] [38] to national and even international networks [31] [32] [39]
Table 5 . (a) Results of the multiple linear regression with the stepwise method. For each dependent variable (Freq-ha; Prod-ha) the estimates of the regression parameters were reported for the significant results; (b) Analysis of multiple linear regression using a general linear model and the significant parameters results in (a) as a predictor.
there is clear evidence of a longer mushroom fruiting season in temperate forest ecosystems, which parallels an extended vegetation period [40] . However, these climate changes have not had the same effect on hypogeous species, in particular of the genus Tuber. In fact, species such as Tuber magnatum and T. melanosporum have declined in their natural Mediterranean habitat despite efforts at their cultivation since the 1970s [3] [5] , while others (e.g. T. aestivum) have expanded their range of distribution [15] .
The results of our study indicate that variations in rainfall and temperature parameters have significant effects on Tuber borchii truffle production. The correlation analysis showed the negative effect of average monthly rainfall (Pm) on productivity (number and weight) of Tuber borchii sporocarps. This indicates that exceeding the certain degree of soil moisture essential for the fructification of most fungi [18] [41] [42] becomes a limitation to
Table 6 . Relationship between climatic parameters in the autumn months before truffle production (WPm and WTm) and the size of the truffles found in the 5 study areas.
the fruiting of T. borchii. Indeed ectomycorrhizal species, in contrast to saprotophs, receive water from the host tree through hydraulic lift (nocturnal water transfer from the tree to the associated mycorrhizal symbiont) [43] , and transfer this water to the sporocarps [44] : if the water content becomes too high aeration is reduced, which can be inhibitory [42] .
The production of T. borchii is positively correlated with the rainfall of the previous three months (Pm-3) and in general with those of the autumn months prior to collection (WPm). This confirms the results of several studies [10] [12] [17] which have shown that the production of sporocarps is delayed in relation to rainfall, to degrees that vary according to latitude [42] . This period is necessary for the acquisition of energy and nutrients by perennial mycelia [42] . Moreover, high yields in one year are often followed by low yields the following year [12] , presumably due to the insufficient time to build up resources to be allocated to fruiting again. Although our data are based on only four years of investigation a similar case has been observed, in fact a year of high productivity (1997) was followed by one (1998) in which the number and weight of truffles was drastically reduced.
Evapotranspiration is also an important factor for the production of sporocarps [13] , especially in the Mediterranean basin where soil water availability can be subject to significant fluctuations [22] . In line with the findings reported by the author cited, our results show that low temperatures in the autumn months prior to truffle production (WTm) correspond to larger truffles. This result is more difficult to interpret from an ecological point of view, and could be due to complex interactions with other soil microorganisms, which are restricted by colder temperatures in the months prior to collection and could therefore indirectly lead to an adaptive advantage for the mycelium, or to complex chemical-physical interactions at soil level, such as a greater availability of nutrients in relation to the coldest temperatures. These hypotheses are not currently supported by the data analyzed, as further and more detailed investigations are required to clarify the ecological mechanisms behind the results obtained. Considering the modest share of variance explained by individual factors (max. 20%) other factors not considered in this analysis are probably able to explain more than 80% of the variation from year to year and from month to month.
Although the data (climate and production) refer to almost 10 years ago, it was still possible to highlight how rainfall and temperature variations significantly affect the production of these valued crops. In comparison to the highly prized Tuber magnatum and T. melanosporum, T. borchii has a high ecological adaptability, low host specificity and is highly competitive with other mycorrhizal fungi, thus being easier to cultivate [7] . However, changes in this parameters could lead to changes in the geographical distribution of this species too, as observed by Büntgen et al. [14] [15] for Tuber aestivum, and to a drastic decrease in productivity, as already witnessed for T. magnatum and T. melanosporum [3] [5] . Further studies involving larger datasets and genetic techniques, combining ecological (including climate) and phenological details, may be useful to elucidate the distribution of Tuber borchii and identify potential drivers of sporocarps production.
Acknowledgements
We are grateful to Gabriele Bucci for his help in the statistical analysis of the data and to Emma Thorley, a language technology consultant, for the improvement of the English manuscript.
References
- Bonito, G., Gryganskyi, A.P., Trappe, J.M. and Vilgalys, R. (2010) A Global Meta-Analysis of Tuber ITS rDNA Sequences: Species Diversity, Host Associations and Long-Distance Dispersal. Molecular Ecology, 19, 4994-5008. http://dx.doi.org/10.1111/j.1365-294X.2010.04855.x
- Bonito, G., Smith, M.E., Nowak, M., Healy, R.A., Guevara, G., et al. (2013) Historical Biogeography and Diversification of Truffles in the Tuberaceae and Their Newly Identified Southern Hemisphere Sister Lineage. PLoS ONE, 8, Article ID: E52765. http://dx.doi.org/10.1371/journal.pone.0052765
- Hall, I., Wang, Y. and Amicucci, A. (2003) Cultivation of Edible Ectomycorrhizal Mushrooms. Trends in Biotechnology, 21, 433-438. http://dx.doi.org/10.1016/S0167-7799(03)00204-X
- Hall, I., Brown, G. and Zambonelli, A. (2007) Taming the Truffle. The History, Lore, and Science of the Ultimate Mushroom. Timber Press, Portland.
- Mello, A., Murat, C. and Bonfante, P. (2006) Truffles: Much More than a Prized and Local Fungal Delicacy. FEMS Microbiology Letters, 260, 1-8. http://dx.doi.org/10.1111/j.1574-6968.2006.00252.x
- Zambonelli, A., Iotti, M., Rossi, I. and Hall, I. (2000) Interaction between Tuber borchii and Other Ectomycorrhizal Fungi in a Field Plantation. Mycological Research, 104, 698-702. http://dx.doi.org/10.1017/S0953756299001811
- Zambonelli, A., Iotti, M., Giomaro, G., Hall, I. and Stocchi, V. (2002) T. borchii Cultivation: An Interesting Perspective. Proceedings of the Second International Conference on Edible Mycorrhizal Mushrooms, Christchurch, 1-7.
- Gardin, L. (2005) I tartufi minori in Toscana. Gli ambienti di crescita dei tartufi marzuolo e scorzone. Quaderno ARSIA, January 2005.
- Iotti, M., Lancellotti, E., Hall, I. and Zambonelli, A. (2010) The Ectomycorrhizal Community in Natural Tuber borchii Grounds. FEMS Microbiology Ecology, 72, 250-260. http://dx.doi.org/10.1111/j.1574-6941.2010.00844.x
- Yang, X., Luedeling, E., Chen, G., Hyde, K.D., Yang, Y., Zhou, D., Xu, J.C. and Yang, T. (2012) Climate Change Effects Fruiting of the Prize Matsutake Mushroom in China. Fungal Diversity, 56, 189-198. http://dx.doi.org/10.1007/s13225-012-0163-z
- Baptista, P., Martins, A., Tavares, R.M. and Lino-Neto, T. (2010) Diversity and Fruiting Pattern of Macrofungi Associated with Chestnut (Castanea sativa) in the Trás-os Montes Region (Northeast Portugal). Fungal Ecology, 3, 9-19. http://dx.doi.org/10.1016/j.funeco.2009.06.002
- Krebs, C.J.K.C., Carrier, P.C.P., Boutin, S.B.S., Boonstra, R.B.R. and Hofer, E.H.E. (2008) Mushroom Crops in Relation to Weather in the Southwestern Yukon. Botany, 86, 1497-1502. http://dx.doi.org/10.1016/j.funeco.2009.06.002
- Martínez de Aragón, J., Bonet, J.A., Fischer, C.R. and Colinas, C. (2007) Productivity of Ectomycorrhizal and Selected Edible Saprotrophic Fungi in Pine Forests of the Pre-Pyrenees Mountains, Spain: Predictive Equations for Forest Management of Mycological Resources. Forest Ecology and Management, 252, 239-256. http://dx.doi.org/10.1016/j.foreco.2007.06.040
- Büntgen, U., Egli, S., Tegel, W., Stobbe, S., Sproll, L., Elburg, R., Peter, M., Nievergelt, D., Cherubini, P. and Stenseth, N. C. (2012) Illuminating the Mysterious World of Truffles. Frontiers in Ecology and the Environment, 10, 462-463. http://dx.doi.org/10.1890/12.WB.021
- Büntgen, U., Tegel, W., Egli, S., Stobbe, U., Sproll, L. and Stenseth, N.C. (2011) Truffles and Climate Change. Frontiers in Ecology and the Environment, 9, 150-151. http://dx.doi.org/10.1890/11.WB.004
- Straatsma, G., Ayer, F. and Egli, S. (2001) Species Richness, Abundance, and Phenology of Fungal Fruit Bodies over 21 Years in a Swiss Forest Plot. Mycological Research, 105, 515-523. http://dx.doi.org/10.1017/S0953756201004154
- Egli, S., Ayer, F., Peter, M., Eilmann, B. and Rigling, A. (2010) In Forest Mushroom Productivity Driven by Tree Growth? Results from a Thinning Experiment. Annals of Forest Science, 67, 509.http://dx.doi.org/10.1051/forest/2010011
- Salerni, E., Laganà, A., Perini, C., Loppi, S. and De Dominicis, V. (2002) Effects of Temperature and Rainfall on Fruiting of Macrofungi in Oak Forests of the Mediterranean Area. Israel Journal of Plant Science, 50, 189-198.
- Eveling, D.W., Wilson, R.N., Gillespie, E.S. and Bataille, A. (1990) Environmental Effects on Sporocarp Counts over Fourteen Years in a Forest Area. Mycological Research, 94, 998-1002. http://dx.doi.org/10.1016/S0953-7562(09)81320-8
- Stern, N. (2006) Stern Review on the Economics of Climate Change. HM Treasury, Cabinet Office. www.sternreview.org.uk
- dei Deputati, C. (2007) Relazione della VII Commissione Ambiente sulle tematiche relative ai Cambiamenti Climatici. Doc. XVI-N.1, 2 luglio.
- Büntgen, U., Egli, S., Camarero, J.J., Fischer, E.M., Stobbe, U., Kauserud, H., Tegel, W., Sproll, L. and Stenseth, N.C. (2012) Drought-Induced Decline in Mediterranean Truffle Harvest. Nature Climate Change, 2, 827-829. http://dx.doi.org/10.1038/nclimate1733
- Splivallo, R., Rittersma, R., Valdez, N., Chevalier, G., Molinier,V., Wipf, D. and Karlovsky, P. (2012) Is Climate Change Altering the Geographic Distribution of Truffles? Frontiers in Ecology and the Environment, 10, 461-462.http://dx.doi.org/10.1890/12.WB.020
- Gardin, L. and Vinci, A. (2006) Carta dei suoli della Regione Toscana in scala 1:250.000. http://sit.lamma.rete.toscana.it/websuoli/
- Soil Survey Staff (2010) Keys to Soil Taxonomy. 11th Edition, USDA-NRCS, Washington DC.
- Arnoldus, H.M.J. (1977) Methodology Used to Determine the Maximum Potential Range Average Annual Soil Loss to Sheet and Rill Erosion in Morocco. Assessing Soil Degradation, FAO Soils Bulletin (FAO), 34, 39-48.
- Pinna, M. (1977) Climatologia. UTET, Torino.
- Blasi, C. (1998) Clima e fitoclima. In: Pignatti, S., Ed., I boschi d’Italia: Sinecologia e Biodiversità, UTET, Torino, 33-71.
- Gregori, E., Sani, L., Finora, M. and Meli, C. (2004) Caratterizzazione mesoclimatica di un’area a vocazionetartuficola nelle Marche. EM-Linea Ecologica, 36, 35-44.
- Gange, A.C., Gange, E.G., Sparks, T.H. and Boddy, L. (2007) Rapid and Recent Changes in Fungal Fruiting Patterns. Science, 316, 71. http://dx.doi.org/10.1126/science.1137489
- Kauserud, H., Stige, L.F., Vik, J.O., Okland, R.H., Hoiland, K. and Stenseth, N.C. (2008) Mushroom Fruiting and Climate Change. Proceedings of the National Academy of Science of the United States of the America, 105, 3811-3814.http://dx.doi.org/10.1073/pnas.0709037105
- Kauserud, H., Heegaard, E., Semenov, M.A., Boddy, L., Halvorsen, R., Stige, L.C., Sparks, T.H., Gange, A.C. and Stenseth, N.C. (2010) Climate Change and Spring-Fruiting Fungi. Proceedings of the Royal Society B, 277, 1169-1177. http://dx.doi.org/10.1098/rspb.2009.1537
- Büntgen, U., Kauserud, H. and Egli, S. (2011) Linking Climate Variability to Mushroom Productivity and Phenology. Frontiers in Ecology and the Environment, 14, 14-19. http://dx.doi.org/10.1890/110064
- Büntgen, U., Peter, M., Kauserud, H. and Egli, S. (2013) Unraveling Environmental Drivers of a Recent Increase in Swiss Fungi Fruiting. Global Change Biology, 19, 2785-2794. http://dx.doi.org/10.1111/gcb.12263
- Sato, H., Morimoto, S. and Hattori, T. (2012) A Thirty-Year Survey Reveals That Ecosystem Function of Fungi Predicts Phenology of Mushroom Fruiting. PLoS ONE, 7, Article ID: E49777. http://dx.doi.org/10.1371/journal.pone.0049777
- Menzel, A. and Fabian, P. (1999) Growing Season Extended in Europe. Nature, 397, 659.http://dx.doi.org/10.1038/17709
- Stenseth, N.C., Mysterud, A., Ottersen, G., Hurrell, J.W., Chan, K.S. and Lima, M. (2002) Ecological Effects of Climate Fluctuations. Science, 297, 1292-1296. http://dx.doi.org/10.1126/science.1071281
- Egli, S., Peter, M., Buser, C., Stahel, W. and Ayer, F. (2006) Mushroom Picking Does Not Impair Future Harvests e Results of a Long Term Study in Switzerland. Biological Conservation, 129, 271-276.
- Kauserud, H., Heegaard, E., Büntgen, U., Halvorsen, R., Egli, S., Senn-Irlet, B., Krisai-Greilhuber, I., Damon, W., Sparks, T., Nordén, J., Hoilanda, K., Kirk, P., Semenov, M., Boddy, L. and Stenseth, N.C. (2012) Warming-Induced Shift in European Mushroom Fruiting Phenology. Proceedings of the National Academy of Science of the United States of the America, 109, 14488-14493. http://dx.doi.org/10.1073/pnas.1200789109
- Menzel, A., Sparks, T.H., Estrella, N., Koch, E., Aasa, A., Ahas, R., Alm-Kübler, K., Bissolli, P., Braslavaská, O., Briede, A., et al. (2006) European Phenological Response to Climate Change Matches the Warming Pattern. Global Change Biology, 12, 1969-1976. http://dx.doi.org/10.1111/j.1365-2486.2006.01193.x
- Barroetavena, C., La Manna, L. and Alonso, M.V. (2008) Variables Affecting Suillus luteus Fructification in Ponderosa Pine Plantations in Patagonia (Argentina). Forest Ecology and Management, 256, 1868-1874.http://dx.doi.org/10.1016/j.foreco.2008.07.029
- Boddy, L., Büntgen, U., Egli, S., Gange, A.C., Heegaard, E., Kirk, P.M., Mohammad, A. and Kauserud, H. (2013) Climate Variation Effects on Fungal Fruiting. Fungal Ecology, 10, 20-33. http://dx.doi.org/10.1016/j.funeco.2013.10.006
- Querejeta, J.I., Egerton-Warburton, L.M. and Allen, M.F. (2003) Direct Nocturnal Water Transfer from Oaks to Their Mycorrhizal Symbionts during Severe Soil Drying. Oecologia, 134, 55-64.http://dx.doi.org/10.1007/s00442-002-1078-2
- Lilleskov, E.A., Bruns, T.D., Dawson, T.E. and Camacho, F.J. (2009) Water Sources and Controls on Water-Loss Rates of Epigeous Ectomycorrhizal Fungal Sporocarps during Summer Drought. New Phytologist, 182, 483-494.http://dx.doi.org/10.1111/j.1469-8137.2009.02775.x
NOTES

*Corresponding author.


