 World Journal of AIDS, 2011, 1, 94-99 doi:10.4236/wja.2011.13014 Published Online September 2011 (http://www.SciRP.org/journal/wja) Copyright © 2011 SciRes. WJA Antiretroviral Therapy among HIV-1 Infected Female Sex Workers in Benin: A Comparative Study with Patients from the General Population Souleymane Diabaté1,2, Djimon Marcel Zannou3, Nassirou Geraldo4, Annie Chamberland1, Jocelyn Akakpo3, Carin Ahouada3, Marguerite Massinga Loembé5, Sévérin Anagonou3, Annie Claude Labbé6, Michel Alary2, Cecile Tremb la y 1 1Centre de Recherche du Centre Hospitalier de l’Université de Montréal, Montréal, Canada; 2URESP, Centre de recherche FRSQ du CHA universitaire de Québec, Québec, Canada; 3Centre National Hospitalier Universitaire de Cotonou, Cotonou, Bénin; 4Dispensaire IST, Centre de Santé de Cotonou I, Cotonou, Bénin; 5Institut de Médecine Tropicale d’Anvers, Anvers, Belgique; 6Université de Montréal, Hôpital Maisonneuve-Rosemont, Montréal, Canada. Email: dsouleym@hotmail.com Received June 7th, 2011; revised June 24th, 2011; accepted July 4th, 2011. ABSTRACT Objective: The aim of this study conducted in Benin was to compare HIV-1 infected female sex workers (FSW) and pa- tients from the general population (GP) to see whether there was a difference in adherence level, mortality rate and immuno-virologic response to antiretroviral therapy (ART). Methods: Fifty-tree FSW and 318 patients from the GP were recruited and followed for at least one year. We compared both cohorts according to poor-adherence (taking <95% of the p ills), CD4 count increa se, undetectable viral load (VL; ≤50 copies/mL) and crude mortality rate. We con- structed a multivariate regression model to assess factors associated with undetectable VL. Results: During the first year, the proportion of FSW with poor-adherence was significantly higher than that of the GP patients (19.3% versus 7.5%; p < 0.0001) and median gain in CD 4 count among FSW was slightly lower (103/mm3 versus 129/mm3; p = 0.085). In the multivariate model (including CD4 at ART initiation and the sub-coho rt i.e. FSW vs GP patients), duration under ART (p = 0.003) as well as CD4 count at enrolment in the study (p < 0.0001) and good-adherence (0.057) were inde- pendently associated with undetectable VL. When adherence was withdrawn from this model, there was a borderline significant asso ciation be tween detectable VL an d being a FSW (p = 0.074). The crude mortality rate was 1.11 per 100 persons-years among the GP patients and 4.65 per 100 persons-years among FSW. Conclusion: Response to ART was lower among FSW compared to GP patients, as a result of poorer adherence. Specific behavioural interventions are needed to improve adherence and response to ART among FSW. Keywords: ART, FSW, Adherence 1. Introduction In 2009, more than 22.5 million people (including ap- proximately 1.8 million new infections) were living with HIV in sub-Saharan Africa [1]. In this region, female sex workers (FSW) and their clients have played an impor- tant role in the extension of the epidemic to the general population (GP) [2,3]. Consequently, both groups have been targeted by various prevention interventions like promotion of condom use and diagnosis and treatment of sexually transmitted infections (STI). These interventions aiming at reducing HIV acquisition as well as transmis- sion have, indeed, contributed to an increase in condom use and a decline in STI prevalence [4-6]. Nonetheless, in sub-Saharan Africa, HIV prevalence among FSW is still 3 to 4 times higher than among the general popula- tion (19% versus 5.2%) [7]. It is therefore important to consider improving access to antiretroviral therapy (ART) for FSW and to scale-up other preventive interventions shown to be effective [8]. According to recent findings on the efficacy of ART for HIV prevention [9], providing early ART to HIV-infected female sex workers has the potential to have a considerable impact on HIV transmis- sion to their clients and therefore contribute to HIV pre- vention in the general population. Despite the increasing  Antiretroviral Therapy among HIV-1 Infected Female Sex Workers in Benin: A Comparative Study 95 with Patients from the General Population number of treated patients in sub-Saharan Africa, little is known about access to ART among FSW [1]. The objec- tive of this prospective study conducted in Cotonou (Be- nin) was to compare HIV-1 infected FSW and patients from the GP to see whether there was a difference in ad- herence level, mortality rate and immuno-virologic re- sponse to ART. 2. Methods From September 2008 to September 2010, we consecu- tively enrolled and followed 371 HIV-1 infected adults (aged ≥ 18 years) receiving a first-line ART composed of two nucleoside reverse transcriptase inhibitors (NRTI) plus a non-nucleoside reverse transcriptase inhibitor (NNRTI) or a protease inhibitor (PI)): Fifty-three (53) patients were FSW recruited at the Dispensaire IST (medical center dedicated since 1989 to routine check-up and STI treatment among FSW) and 318 patients were from the GP. The GP patients were recruited at the Cen- tre de traitement ambulatoire (CTA) du Centre national hospitalier universitaire de Cotonou (reference center for HIV treatment in Benin). After enrolment in the study, FSW were seen every 3 months and patients from the GP every 4 months (in accordance with existing follow-up visits’ chronograms in each study center). World Health Organisation (WHO) clinical stages III/IV or CD4 count ≤ 200 /mm3 were the main conditions for ART initiation in Benin. The study was approved by the research ethics committees of the Centre hospitalier de l’Université de Montréal and the CHA universitaire de Québec in Can- ada, and of the Ministry of Health in Benin. An informed consent form was signed by all the participants. Adherence counselling was routinely done in each study center and the interviewers were all trained on the study procedures. Adherence to ART was assessed through self-report of missed doses: taking < 95% of the pills prescribed during the four days preceding each visit was defined as poor adherence [10]. Adherence to ART and CD4 count (Flow cytometry) were measured at study entry and at each follow-up visit. For patients who initi- ated treatment before enrolment in the study, baseline CD4 count, age and WHO clinical classification were collected from the medical files. Viral load (VL, bDNA) was performed for all patients only at study entry. De- scriptive comparisons using chi-square or Fisher’s exact tests (proportions) and Student’s t test (means) were per- formed to compare both cohorts according to baseline characteristics. A univariate model was performed for each potential determinant of undetectable VL. All vari- ables with a P value ≤ 0.10 were included in a multivari- ate model to determine those independently associated with undetectable VL (≤50 copies/mL). Data were ana- lysed with SAS version 9.2 (SAS Institute, Cary, NC, USA). 3. Results At ART initiation, the median age (IQR) in both groups was comparable: thirty nine (39; (33 - 43)) years for the FSW and 37 (31 - 43) years for the patients from the GP (p = 0.179). The majority of the GP patients were fe- males (62.3%) and Beninese (91%). The FSW were from Benin (30%) and neighbouring countries like Togo (30%), Ghana (24%) and Nigeria (15%). The proportion of non educated FSW was higher (35.9% versus 22.6%; p = 0.034). At enrolment in the study, median duration (IQR) under ART was 21 (4 - 38) months for the GP patients and 4.3 (0 - 24.3) months for the FSW (p < 0.003). At ART initiation, most of the GP patients (73.9%) were classified as WHO clinical stages III/IV as compared with FSW (22.6%, p < 0.0001). The NRTI mostly prescribed was stavudine (71.7% for the GP patients and 77.4% for the FSW) whereas the NNRTI mostly prescribed was efavirenz for patients from the GP (72.4%) and nevirapine for FSW (73.6%; Table 1). During follow-up, 19 ART modifications due to side effects were observed among the GP patients (none among the FSW). These treatment modifications occurred after a median (IQR) duration under ART of 32 (7.8 - 53.8) months. In 18/19 cases (94.7%), stavudine was replaced by zidovudine because of lipodystrophy (lipoatrophy: 11/18, lipoatrophy plus lipohypertrophy: 3/18) and peripheral neuropathy (4/18). There were 13 (8.6%) switches to second-line ART in the GP patients. The Median time (IQR) from initiation to switch to sec- ond-line was 37.6 (27.9 - 49.0) months. Among the FSW, we observed 4 (7.5%) switches. The 17 switches oc- curred after availability of the VL measured at study en- try and were mostly related to insufficient viro-immu- nologic response (88.2%). During the first year, poor adherence, as assessed at follow-up visits, varied between 11.8% and 27.3% among the FSW and between 5.9% and 10.6% among the GP patients. Overall, the proportion of FSW with poor ad- herence was significantly higher than that of the GP pa- tients (19.3% versus 7.5%; p < 0.0001). There was no difference in adherence levels according to gender in the GP patients. The main reasons for missing doses reported by the FSW were being out of home (58.6%) and running out of pills (20.7%). At ART initiation, median CD4 count/mm3 (IQR) among the GP patients and the FSW was similar 115 (53 - 184) versus 134 (67 - 175, p=0.141), respectively. Dur- ing the first year of treatment, we observed an increase in CD4 count/mm3 among the FSW, but, median (IQR) Copyright © 2011 SciRes. WJA  Antiretroviral Therapy among HIV-1 Infected Female Sex Workers in Benin: A Comparative Study with Patients from the General Population Copyright © 2011 SciRes. WJA 96 Table 1. Antiretroviral combinations in 53 HIV-1 infected female sex workers and 318 patients from the general population, Cotonou, Benin. Antiretroviral combinations General population (number, %) Female sex workers (number, %) Stavudine + Lamivudine + Efavirenz 164 (51.6) 9 (17) Zidovudine + Lamivudine + Efavirenz 65 (20.4) 5 (9.4) Lamivudine + Didanosine + Efavirenz 2 (0.6) - Stavudine + Lamivudine + Nevirapine 63 (19.8) 32 (60.4) Zidovudine + Lamivudine + Nevirapine 23 (7.2) 7 (13.2) Stavudine + Lamivudine + Indinavir 1 (0.3) - Total 318 53 gain 103 (25 - 178) was to some extent lower as com- pared with the GP patients 129 (75 - 232; p = 0.085; Figure 1). At study entry, 57% of the GP patients and 34% of the FSW had undetectable VL. After adjustment for CD4 count at ART initiation, treatment duration, as well as CD4 count and good ad- herence at enrolment in the study were independently associated with undetectable VL (Table 2; model 1). When we withdrew adherence from this model, being recruited from the GP was found to be related to unde- tectable VL (Tabl e 2, model 2). The crude mortality rate was 1.11 per 100 person-years (3 deaths/270.33 per- son-years) among the GP patients and 4.65 per 100 per- son-years (2 deaths/43 person-years) among FSW. In a sub-group analysis, we observed that from ART initiation to year-3, CD4 count has increased progres- sively among the GP patients (t test for trend = 18.92; p < 0.0001; Figure 1). Median (IQR) gain in CD4 count during the same period was 263 (196 - 345)/mm3. There was no difference in CD4 progression according to the NNRTI used (nevirapine or efavirenz, p = 0.585). In ad- dition, the proportion of the GP patients recruited with undetectable VL was similar irrespective of the NNRTI prescribed (nevirapine, 60.5% and efavirenz, 55.7%; p = 0.366). Due to their small number, FSW were excluded from these sub-group analyses concerning follow-up > 1 year and the relation between the NNRTI regimen (nevi- rapine versus efavirenz) and immuno-virologic response. 4. Discussion Irrespective of their HIV status, FSW from Cotonou and its suburbs are requested to undergo each month a routine medical visit. This preventive approach is an opportunity for healthcare workers for early detection and treatment of STI and other morbidities. It also allows testing for HIV and prescription of cotrimoxazole prophylaxis if better clinical conditions at ART initiation among FSW needed. As a consequence, there was a trend towards as compared with the GP patients: the proportion of sub- jects with advanced WHO clinical stages (III/IV) was lower among FSW. This observation, in agreement with previous results showing a decrease in STI prevalence among FSW in Benin, supports the importance of main- taining and reinforcing preventive efforts towards high-risk groups [5,11]. As previously described, stavudine (the most pre- scribed NRTI in our study populations) was the main cause for most of the first-line ART modifications due to peripheral neuropathy and lipodystrophy [12,13]. This known stavudine toxicity profile has led the WHO to recommend less toxic agents such as tenofovir or zido- vudine [14]. Lipodystrophy did not lead to regimen mo- dification in FSW probably because of a lower median duration under ART [13,15]. Most of the switches to second-line ART were based on VL measured at enrol- ment in the study. This is a paradox since all (except one) patients with elevated viral load also had immunologic failure and could have been switched to second-line therapy earlier, based on this sole parameter. It appeared that clinicians were less confident if they had to base their decision of switching to second-line on CD4 counts only [16]. This conservative attitude needs to be ad- dressed by training activities aiming at strengthening physicians’ prescription abilities but also by facilitating access to VL, in particular for patients with insufficient immune response [17]. In general, FSW have higher challenges for treatment adherence because of their mobility motivated by many reasons such as looking for new clients and avoiding harassment from the police, from the brothel owners or sometimes from their boyfriends. To prevent the negative impact of this social instability on adherence level, care 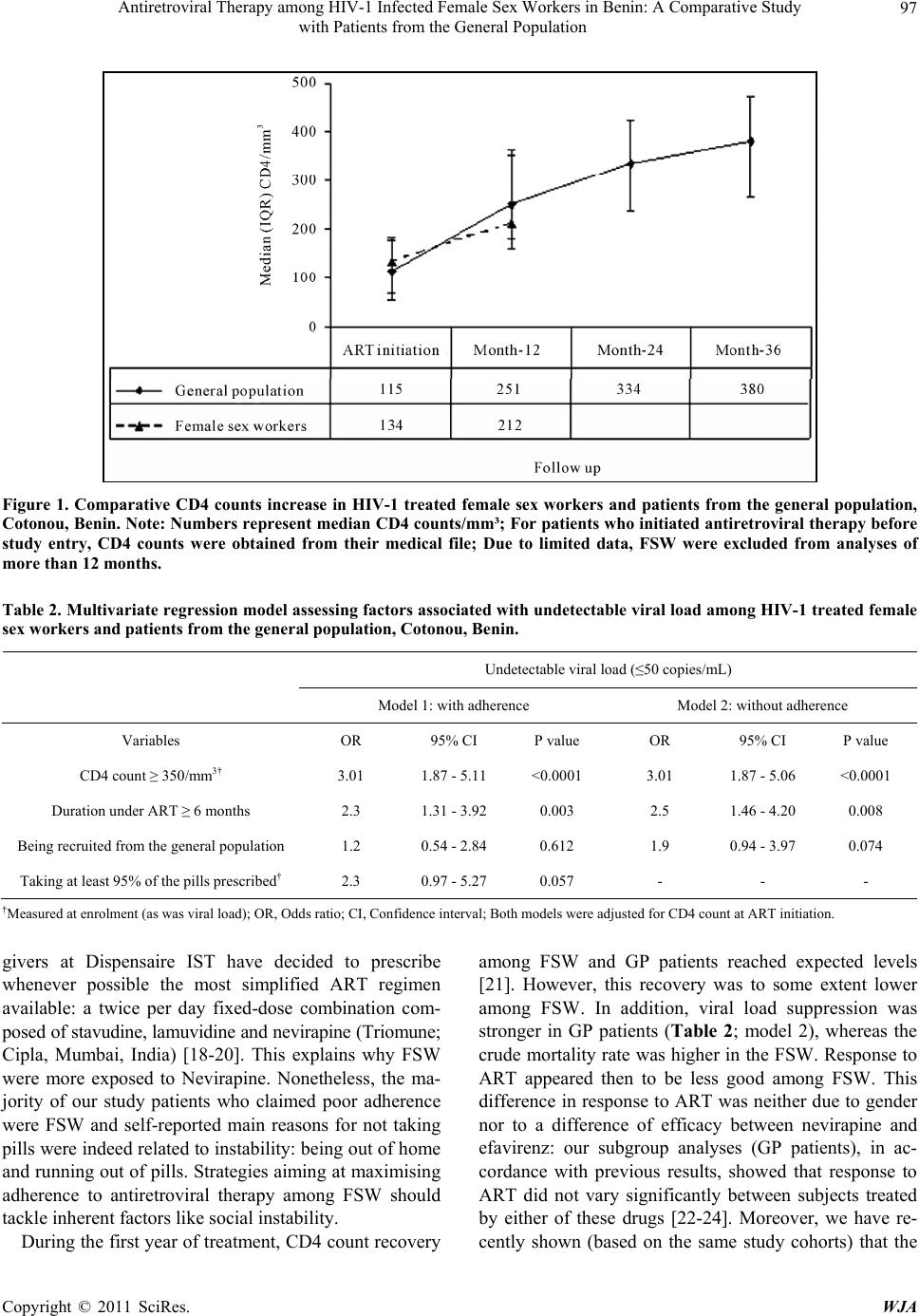 Antiretroviral Therapy among HIV-1 Infected Female Sex Workers in Benin: A Comparative Study 97 with Patients from the General Population Figure 1. Comparative CD4 counts increase in HIV-1 treated female sex workers and patients from the general population, Cotonou, Benin. Note: Number s represent median CD4 counts/mm³; For patients who initiated antiretroviral therapy before study entry, CD4 counts were obtained from their medical file; Due to limited data, FSW were excluded from analyses of more than 12 months. Table 2. Multivariate regression model asse ssing factors associated w ith undetectable viral load among HIV-1 tr eated female sex workers and patients from the general population, Cotonou, Benin. Undetectable viral load (≤50 copies/mL) Model 1: with adherence Model 2: without adherence Variables OR 95% CI P value OR 95% CI P value CD4 count ≥ 350/mm3† 3.01 1.87 - 5.11 <0.0001 3.01 1.87 - 5.06 <0.0001 Duration under ART ≥ 6 months 2.3 1.31 - 3.92 0.003 2.5 1.46 - 4.20 0.008 Being recruited from the general population 1.2 0.54 - 2.84 0.612 1.9 0.94 - 3.97 0.074 Taking at least 95% of the pills prescribed† 2.3 0.97 - 5.27 0.057 - - - †Measured at enrolment (as was viral load); OR, Odds ratio; CI, Confidence interval; Both models were adjusted for CD4 count at ART initiation. givers at Dispensaire IST have decided to prescribe whenever possible the most simplified ART regimen available: a twice per day fixed-dose combination com- posed of stavudine, lamuvidine and nevirapine (Triomune; Cipla, Mumbai, India) [18-20]. This explains why FSW were more exposed to Nevirapine. Nonetheless, the ma- jority of our study patients who claimed poor adherence were FSW and self-reported main reasons for not taking pills were indeed related to instability: being out of home and running out of pills. Strategies aiming at maximising adherence to antiretroviral therapy among FSW should tackle inherent factors like social instability. During the first year of treatment, CD4 count recovery among FSW and GP patients reached expected levels [21]. However, this recovery was to some extent lower among FSW. In addition, viral load suppression was stronger in GP patients (Table 2; model 2), whereas the crude mortality rate was higher in the FSW. Response to ART appeared then to be less good among FSW. This difference in response to ART was neither due to gender nor to a difference of efficacy between nevirapine and efavirenz: our subgroup analyses (GP patients), in ac- cordance with previous results, showed that response to ART did not vary significantly between subjects treated by either of these drugs [22-24]. Moreover, we have re- cently shown (based on the same study cohorts) that the Copyright © 2011 SciRes. WJA  Antiretroviral Therapy among HIV-1 Infected Female Sex Workers in Benin: A Comparative Study 98 with Patients from the General Population prevalence of primary resistance to ART was similar in both the FSW and the GP patients [25]. Then, the re- maining factor that could explain the difference in re- sponse to ART between both groups is cumulative ad- herence levels. Indeed, in our multivariate regression model 2, being a GP patient was associated with unde- tectable VL. In model 1, this association was dissipated by good adherence i.e. taking at least 95% of the pills prescribed by the doctors. Specific behavioural interven- tions targeting FSW are therefore needed to improve adherence and response to ART: as primary drug resis- tance has been found to be elevated in this population, poor adherence resulting in partial viral suppression can rapidly jeopardize ART benefits [10,25-26]. Our study has some limitations including the small number of HIV-1-treated FSW and the short-term follow-up. How- ever, our data provides important insights on the feasibil- ity and also on the challenges of ART delivery in the FSW population in sub-Saharan Africa. In conclusion, response to ART was not as good among female sex workers, compared with patients from the ge- neral population, as a result of poorer adherence. Efforts to improve response to ART among female sex workers should target inherent factors impacting adherence such as social instability. In addition, large size and long term follow-up studies are needed to better analyse the deter- minants and the extent to which response to ART among female sex workers is different from response among the general population patients. 5. Acknowledgements We would like to thank the patients and the staff at the Dispensaire IST and the Centre de traitement ambulatoire du Centre national hospitalier universitaire de Cotonou. We also thank the staff at the Centre de recherche du Centre hospitalier universitaire de Montréal and the Unité de recherche en santé des populations du Centre de recherche FRSQ du CHA universitaire de Québec for their administrative and technical assistance. REFERENCES [1] UNAIDS, “UNAIDS Report on the Global AIDS Epi- demic”, UNAIDS, Geneva, 2010. http://www.unaids.org/globalreport/documents/20101123 _GlobalReport_full_en.pdf [2] Z. Mukandavire and W. Garira, “Effects of Public Health Educational Campaigns and the Role of Sex Workers on the Spread of HIV/AIDS among Heterosexuals,” Theo- retical Population Biology, Vol. 72, No. 3, 2007, pp. 346- 365. doi:10.1016/j.tpb.2007.07.002 [3] C. M. Lowndes, M. Alary, H. Meda, C. A. Gnintoungbe, L. Mukenge-Tshibaka, C. Adjovi, et al., “Role of Core and Bridging Groups in the Transmission Dynamics of HIV and STIs in Cotonou, Benin, West Africa,” Sexually Transmitted Infections, Vol. 78, Supplement 1, 2002, pp. S69- S77. doi:10.1136/sti.78.suppl_1.i69 [4] P. D. Ghys, M.O. Diallo, V. Ettiegne-Traore, K. Kale, O. Tawil, M. Carael, et al., “Increase in Condom Use and Decline in HIV and Sexually Transmitted Diseases among Female Sex Workers in Abidjan, Côte d’Ivoire, 1991-1998,” AIDS, Vol. 16, No. 2, 2002, pp. 251-258. doi:10.1097/00002030-200201250-00015 [5] C. M. Lowndes, M. Alary, A. C. Labbe, C. Gnintoungbe, M. Belleau, L. Mukenge, et al., “Interventions among Male Clients of Female Sex Workers in Benin, West Af- rica: An Essential Component of Targeted HIV Preven- tive Interventions,” Sexually Transmitted Infections, Vol. 83, No. 7, 2007, pp. 577-581. doi:10.1136/sti.2007.027441 [6] R. Hayes, D. Watson-Jones, C. Celum, J. van de Wijgert and J. Wasserheit, “Treatment of Sexually Transmitted Infections for HIV Prevention: End of the Road or New Beginning?” AIDS, Vol. 24, Suppl. 4, 2010, pp. S15-S26. doi:10.1097/01.aids.0000390704.35642.47 [7] UNAIDS, “AIDS Epidemic Update,” UNAIDS, Geneva, 2009. http://data.unaids.org/pub/Report/2009/jc1700_epi_updat e_2009_en.pdf [8] N. Malunguza, S. Mushayabasa, C. Chiyaka and Z. Mu- kandavire, “Modelling the Effects of Condom Use and Antiretroviral Therapy in Controlling HIV/AIDS among Heterosexuals, Homosexuals and Bisexuals,” Computa- tional and mathematical methods in medicine, Vol. 11, No. 3, 2010, pp. 201-22. doi:10.1080/17486700903325167 [9] Editorial, “HIV Treatment as Prevention—It Works,” Lancet, Vol. 377, No. 9779, 2011, p.1719. doi:10.1016/S0140-6736(11)60713-7 [10] N. Ford, M. Darder, T. Spelman, E. Maclean, E. Mills and A. Boulle, “Early Adherence to Antiretroviral Medi- cation as a Predictor of Long-Term HIV Virological Sup- pression: Five-Year Follow Up of an Observational Co- hort,” PLoS One, Vol. 5, No. 5, 2010, p. e10460. doi:10.1371/journal.pone.0010460 [11] P. Vickerman, F. Ndowa, N. O'Farrell, R. Steen, M. Alary and S. Delany-Moretlwe, “Using Mathematical Model- ling to Estimate the Impact of Periodic Presumptive Trea- tment on the Transmission of Sexually Transmitted Infec- tions and HIV among Female Sex Workers,” Sexually Transmitted Infections, Vol. 86, No. 3, 2010, pp. 163-168. doi:10.1136/sti.2008.034678 [12] J. van Griensven, L. de Naeyer, T. Mushi, S. Ubarijoro, D. Gashumba, C. Gazille and R. Zachariah, “High Preva- lence of Lipoatrophy among Patients on Stavudine-Con- taining First-Line Antiretroviral Therapy Regimens in Rwanda,” Transactions of the Royal Society of Tropical Medicine and Hygiene, Vol. 101, No. 8, 2007, pp. 793- 798. doi:10.1016/j.trstmh.2007.02.020 [13] S. Mercier, N. F. Gueye, A. Cournil, A. Fontbonne, N. Copyright © 2011 SciRes. WJA  Antiretroviral Therapy among HIV-1 Infected Female Sex Workers in Benin: A Comparative Study with Patients from the General Population Copyright © 2011 SciRes. WJA 99 Copin, I. Ndiaye, et al., “Lipodystrophy and metabolic Disorders in HIV-1-Infected Adults on 4- to 9-Year Anti- retroviral Therapy in Senegal: A Case-Control Study,” Journal of Acquired Immune Deficiency Syndrom, Vol. 51, No. 2, 2009, pp. 224-230. [14] World Health Organisation, “Antiretroviral Therapy for HIV Infection in Adults and Adolescents. Recommada- tion for a Public Health approach: 2010 Revision,” WHO, Geneva, 2010. http://whqlibdoc.who.int/publications/2010/97892415997 64_eng.pdf [15] D. M. Zannou, L. Denoeud, K. Lacombe, D. Amoussou- Guenou, J. Bashi, J. Akakpo, et al., “Incidence of Li- podystrophy and Metabolic Disorders in Patients Starting Non-Nucleoside Reverse Transcriptase Inhibitors in Be- nin,” Antiviral Therapy, Vol. 14, No. 3, 2009, pp. 371- 380. [16] O. Keiser, H. Tweya, P. Braitstein, F. Dabis, P. MacPhail and A. Boulle, “Mortality after Failure of Antiretroviral Therapy in Sub-Saharan Africa,” Tropical medicine & international health, Vol. 15, No. 2, 2010, pp. 251-258. doi:10.1111/j.1365-3156.2009.02445.x [17] O. Keiser, H. Tweya, A. Boulle, P. Braitstein, M. Schec- ter and M. W. Brinkhof, “Switching to Second-Line Antiretroviral Therapy in Resource-Limited Settings: Comparison of Programmes with and without Viral Load Monitoring,” AIDS, Vol. 23, No. 14, 2009, pp. 1867-1874. doi:10.1097/QAD.0b013e32832e05b2 [18] E. J. Mills, J. B. Nachega, D. R. Bangsberg, S. Singh, B. Rachlis, P. Wu, et al., “Adherence to HAART: A Sys- tematic Review of Developed and Developing Nation Pa- tient-Reported Barriers and Facilitators,” PLoS Medicine, Vol. 3, No. 11, 2006, p. e438. [19] C. Protopopescu, F. Raffi, P. Roux, J. Reynes, P. Della- monica, B. Spire, et al., “Factors Associated with Non- Adherence to Long-Term Highly Active Antiretroviral Therapy: A 10 Year Follow-Up Analysis with Correction for the Bias Induced by Missing Data,” The Journal of Antimicrobial Chemotherapy, Vol. 64, No. 3, 2009, pp. 599-606. doi:10.1093/jac/dkp232 [20] A. D. Bouhnik, M. Chesney, P. Carrieri, H. Gallais, J. Moreaum J. P. Moatti, et al., “Nonadherence among HIV-Infected Injecting Drug Users: The Impact of Social Instability,” Journal of acquired immune deficiency syn- dromes, Vol. 31, Supplement 3, 2002, pp. S149-S153. doi:10.1097/00126334-200212153-00013 [21] D. Nash, M. Katyal, M. W. Brinkhof, O. Keiser, M. May R. Hughes, et al., “Long-Term Immunologic Response to Antiretroviral Therapy in Low-Income Countries: A Col- laborative Analysis of Prospective Studies,” AIDS, Vol. 22, No. 17. 2008, pp. 2291-2302. [22] P. de Beaudrap, J. F. Etard, F. N. Gueye, M. Gueye, R. Lan- dman, P. Girard, et al., “Long-Term Efficacy and Toler- ance of Efavirenz- and Nevirapine-Containing Regimens in Adult HIV Type 1 Senegalese Patients,” AIDS re- search and human retroviruses, Vol. 24, No. 6, 2008, pp. 753-760. doi:10.1089/aid.2007.0295 [23] S. Toure, B. Kouadio, C. Seyler, M. Traore, N. Da- koury-Dogbo, J. Duvignac, et al., “Rapid Scaling-Up of Antiretroviral Therapy in 10,000 Adults in Côte d'Ivoire: 2-Year Outcomes and Determinants,” AIDS, Vol. 22, No. 7, 2008, pp. 873-882. doi:10.1097/QAD.0b013e3282f768f8 [24] E. Nicastri, S. Leone, C. Angeletti, L. Palmisano, L. Sar- mati, A. Chiesi, et al., “Sex Issues in HIV-1-Infected Persons during Highly Active Antiretroviral Therapy: A Systematic Review,” The Journal of antimicrobial che- motherapy, Vol. 60, No. 4, 2007, pp. 724-732. doi:10.1093/jac/dkm302 [25] A. Chamberland, S. Diabaté, M. Sylla, Y. S. Anagounou, N. Geraldo, D. M. Zannou, et al., “HIV-1 Primary Drug Resistance Prevalence and Transmission in Cotonou, Be- nin,” 5th Conference of French Speeking Countries on HIV/ AIDS, Casablanca, March 2010, p. 110. [26] D. L. Hanson, C. Adje-Toure, N. Talla-Nzussouo, P. Eby, M. Y. Borget, L. Y. Kouadio, et al., “HIV Type 1 Drug Resistance in Adults Receiving Highly Active Antiretro- viral Therapy in Abidjan, Côte d'Ivoire,” AIDS Research and Human Retroviruses, Vol. 25, No. 5, 2009, pp. 489- 495. doi:10.1089/aid.2008.0273
|