 International Journal of Organic Chemistry, 2011, 1, 87-96 doi:10.4236/ijoc.2011.13014 Published Online September 2011 (http://www.SciRP.org/journal/ijoc) Copyright © 2011 SciRes. IJOC Synthesis, Spectroscopy and Electrochemis try of New 3-(5-Aryl-4,5-Dihydro-1H-Pyrazol-3-yl)-4-Hydroxy-2H- Chromene-2-One 4, 5 as a Novel Class of Potential Anti- bacterial and Antioxidant Derivatives Abdullah Sulaiman Al-Ayed College of Science and Arts at Ar-Rass, Qassim University, Kingdom of Saudi Arabia E-mail: salayedabdualla1@yahoo.com Received June 24, 2011; revised August 1, 2011; accepted August 10, 2011 Abstract 3-((2E)-3(aryl)prop-2-enoyl)-2H-chromen-2-one 3 was synthesized from 4-hydroxy coumarin by refluxing 3-acetyl-4-hydroxy coumarin with aromatic aldehydes in chloroform in the presence of a catalytic amount of piperidine. 3 was converted to pyrazoles 4, 5 by treatment with hydrazine and phenylhydrazine in toluene, respectively. The structures of the new compounds were confirmed by elemental analysis, IR, and multinu- clear/multidimensional NMR spectroscopy (1H, 13C-NMR, NOESY, HMBC) which allowed us to assign the complete network of proton and carbon atoms. All the compounds exhibited one quasireversible redox proc- ess. All the newly synthesized compounds were screened for their antibacterial and antioxidant activities. Antimicrobial studies revealed that 3-(5-(2,5-dimethylphenyl)-1-phenyl-4,5-dihydro-1H-pyrazol-3-yl)-4- hydroxy-2H-chromene-2-one 5c showed significant antibacterial activity against Escherichia coli and Pseu- domonas Aeruginosa 27853. Furthermore, 3-(5-(aryl)-4,5-dihydro-1H-pyrazol-3-yl)-4-hydroxy-2H-chromene- 2-ones 4, 5 showed antioxidant activities of different extents with respect to individual compounds as well as to the antioxidant methods. The 3-(5-(phenyl)-4,5-dihydro-1H-pyrazol-3-yl)-4-hydroxy-2H-chromene-2-ones 4a was found to be the most active antioxidant in the series and more active than trolox which makes the in- vestigated complexes a new promising class of antibacterial compounds. Keywords: 4-Hydroxycoumarin, Pyrazole, Antibacterial Activity, Antioxydant Activity 1. Introduction Chalcones are important precursors of flavonoids and isoflavonoids [1]. A large number of chalcones have been prepared by Claisen-Schmidt condensation of al- dehydes with methyl ketones under basic conditions [2]. These compounds have shown in vitro antimalarial ac- tivity against chloroquine-sensitive and chloroquine-re- sistant strains of Plasmodium falciparum [3]. Recently authors have reported the synthesis of chalcones under acidic conditions using perchloric acid and acetic acid [4]. The activity of a variety of chalcones as potent tyrosinase inhibitors and antioxidants has been also reported which are, thus, used as new depigmentation agents [5]. Nitro- gen heterocycles containing chalcone moiety have been reported as active compounds against herpes simplex virus-1 (HSV-1) and human immunodeficiency virus 1 (HIV-1) [6,7]. This class of compounds also exhibits cytotoxic activity towards leukemia cell lines [8,9]. Various other chalcones exhibit insecticidal, antichino- viral, and antipicorniviral properties [10]. On the other hand, coumarins and structurally related compounds have been shown to inhibit replication of HIV and thus exhibit a therapeutic potential [11]. A large number of structurally novel coumarin derivatives have been reported to show substantial cytotoxic and anti-HIV activity both in vitro and in vivo [12,13]. A variety of synthetic coumarins have unique action mechanisms referring to the different stages of HIV replication [14]. Thus, coumarins are important lead compounds for the development of antiviral and/or virucidal drugs against HIV [15-17]. In view of the variety of pharmacological properties exhibited by chalcones, we were prompted to undertake 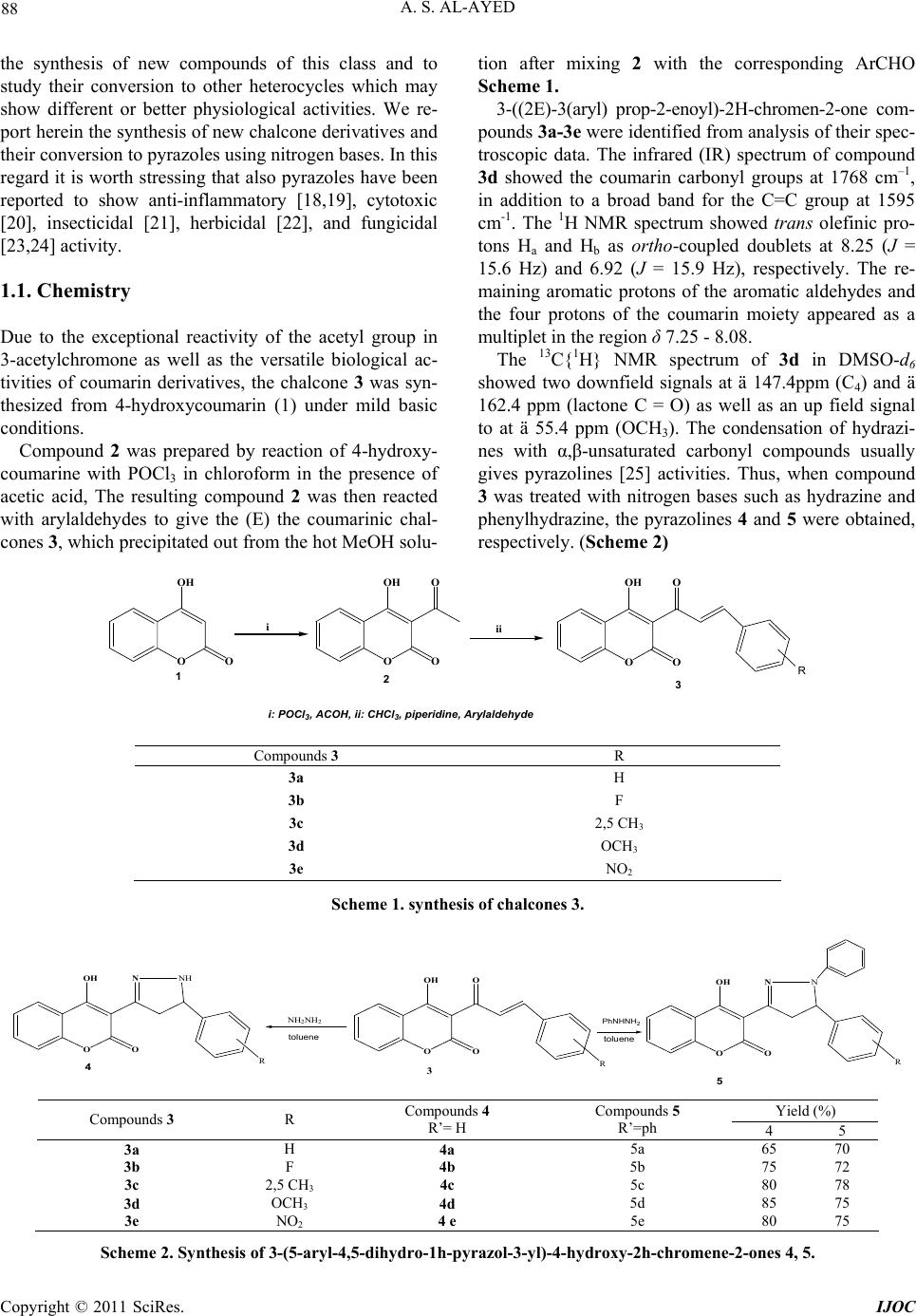 A. S. AL-AYED 88 the synthesis of new compounds of this class and to study their conversion to other heterocycles which may show different or better physiological activities. We re- port herein the synthesis of new chalcone derivatives and their conversion to pyrazoles using nitrogen bases. In this regard it is worth stressing that also pyrazoles have been reported to show anti-inflammatory [18,19], cytotoxic [20], insecticidal [21], herbicidal [22], and fungicidal [23,24] activity. 1.1. Chemistry Due to the exceptional reactivity of the acetyl group in 3-acetylchromone as well as the versatile biological ac- tivities of coumarin derivatives, the chalcone 3 was syn- thesized from 4-hydroxycoumarin (1) under mild basic conditions. Compound 2 was prepared by reaction of 4-hydroxy- coumarine with POCl3 in chloroform in the presence of acetic acid, The resulting compound 2 was then reacted with arylaldehydes to give the (E) the coumarinic chal- cones 3, which precipitated out from the hot MeOH solu- tion after mixing 2 with the corresponding ArCHO Scheme 1. 3-((2E)-3(aryl) prop-2-enoyl)-2H-chromen-2-one com- pounds 3a-3e were identified from analysis of their spec- troscopic data. The infrared (IR) spectrum of compound 3d showed the coumarin carbonyl groups at 1768 cm–1, in addition to a broad band for the C=C group at 1595 cm-1. The 1H NMR spectrum showed trans olefinic pro- tons Ha and Hb as ortho-coupled doublets at 8.25 (J = 15.6 Hz) and 6.92 (J = 15.9 Hz), respectively. The re- maining aromatic protons of the aromatic aldehydes and the four protons of the coumarin moiety appeared as a multiplet in the region δ 7.25 - 8.08. The 13C{1H} NMR spectrum of 3d in DMSO-d6 showed two downfield signals at ä 147.4ppm (C4) and ä 162.4 ppm (lactone C = O) as well as an up field signal to at ä 55.4 ppm (OCH3). The condensation of hydrazi- nes with α,β-unsaturated carbonyl compounds usually gives pyrazolines [25] activities. Thus, when compound 3 was treated with nitrogen bases such as hydrazine and phenylhydrazine, the pyrazolines 4 and 5 were obtained, respectively. (Scheme 2) O OH O O OH O O O OH O O iii R i: POCl 3 , ACOH, ii: CHCl 3 , piperidine, Arylaldehyde 123 Compounds 3 R 3a H 3b F 3c 2,5 CH3 3d OCH3 3e NO2 Scheme 1. synthesis of chalcones 3. O OH O O 3 NH 2 NH 2 O OH O NNH 4 O OH O NN 5 R RR PhNHNH 2 toluene t oluene Yield (%) Compounds 3 R Compounds 4 R’= H Compounds 5 R’=ph 4 5 3a H 4a 5a 65 70 3b F 4b 5b 75 72 3c 2,5 CH3 4c 5c 80 78 3d OCH3 4d 5d 85 75 3e NO2 4 e 5e 80 75 Scheme 2. Synthesis of 3-(5-aryl-4, 5-dihydro-1h-pyr azol-3-yl)-4-hydroxy-2h-chromene-2-ones 4, 5. Copyright © 2011 SciRes. IJOC  A. S. AL-AYED 89 All the new 3-(5-aryl-4,5-dihydro-1H-pyrazol-3-yl)-4- hydroxy-2H-chromene-2-ones 4, 5 were characterized by IR, 1H, 13C-NMR spectra as well as by NOESY and HMBC 2D-NMR experiments to elucidate their struc- tures and assign completely the structural network of both protons and carbons. The spectral data were in ac- cordance with the proposed structures (see experimental section). The IR spectrum of 4d showed broad band at 3207 cm–1 due to the presence of the NH group. A sharp and strong absorption band at 1668 cm–1 indicated a carbonyl group in the compound. Since chromone carbonyl groups usually appear as sharp absorption bands in the region 1620 - 1650 cm–1 [26], the band at 1684 cm–1 was assigned to coumarin rather than the chromone carbonyl group. In addition, the detection of a strong C=N stretching band at 1608 cm−1 evidenced the formation of the pyra- zole ring. The 1H NMR spectra of 4d displays a signal at δ 4.12 ppm ascribable to the CH2 protons of the pyrazole ring. A characteristic singlet proton signal at δ 4.81 ppm was assigned to CH proton from the pyrazole fragment. In addition, the aromatic protons (both coumarinic and aromatic) are observed between δ 6.82 and δ 8.1ppm (see experimental). Full assignment of the 1H NMR spectra of 4d was de- duced from the NOESY spectrum. An observed NOE cross peak between H-1’ and aromatic protons confirms that these two units are located on the same side of the pyrazole ring. The structure of 4d was finally elucidated through the analysis of the 1H, 13C HMBC spectrum, which corre- lates the protons at δ 4.78 ppm with C5' (δ 55.05 ppm) and C2’ (δ 153.3 ppm). The aromatic protons correlate with C5’ (δ 55, 05 ppm). (Table 1) Table 1. Correlations between HMBC and NOESY for com- pound 4d. Proton H-n HMBC H-n-C-j NOESY H-n-H-j H-1’ 2, 2’, 5’ Harom H-5’ 1’,7’ H-arom 5’ A mechanistic rationalization for this reaction is straightforward and is provided in Scheme 3. The first reaction step consists of a nucleophilic attack of the final hydrazine nitrogen atom to the carbonyl function followed by the elimination of water. The in- termediate which forms may easily rearrange to afford the corresponding pyrazoles 4, 5. 1.2. Results and Discussion 1.2.1. Electrochemistry Electrochemical studies of 3-(5-aryl-4,5-dihydro-1H- pyrazol-3-yl)-4-hydroxy-2H-chromene-2-one, 5, are of in- terest due to the electron-deficient nature of the pyrazole unit. Hence, the electrochemical properties of compounds 5a-e, were determined by cyclic voltammetry in CH3CN (1 × 10–3 M) solutions, using 0.1 M tetrabutylammonium bromide (C4H12BrN) as the supporting electrolyte. Both platinum and gold were used as working electrodes, Ag/AgCl (0.1 M) as the reference electrode, and plati- num as the counter electrode. Under these electrochemi- cal conditions, 5 shows a quasi reversible behavior for the first reduction process. This can be deduced from the fact that the cathodic–anodic peak separations (Epc - Epa) are ca. 100 mV. The ratio of the peak current intensity for the cathodic and anodic processes is about 0.5 - 0.7. O OH O O R 3 O OH O NNR' 4,5 NH 2 NH R' O OH O OH NH NHR' -H 2 O O OH O NNHR' (i) (ii) RR R R': H, Ph Scheme 3. Proposed mechanism for the synthesis of 4, 5. Copyright © 2011 SciRes. IJOC  A. S. AL-AYED 90 As expected, the reduction peak potential of the 3-(5-aryl- 4,5-dihydro-1H-pyrazol-3-yl)-4-hydroxy-2H-chromene- 2-one 5 are strongly influenced by the para substitutent on the phenylene ring. Compared to the unsubstituted compound 5, the presence of electron donor groups such as the methoxy group shifts the reduction peak potential of 5 to more negative values. The redox behavior of all the new pyrazoles are summarized in Figure 1 All the compounds exhibited one quasireversible redox processes. For example, compounds 5d showed one quasirever- sible reduction at –0.8 V and –1.15 V, respectively. We assume that the curve at lower reduction potential may be due to the more electron-deficient dications in the ring system, and the curve at higher reduction potential may be attributed to the redox behavior of the pyrazole unit. 1.2.2. Antiba cterial and Antioxi dant Studies 1.2.2.1. Free radical scavenging activity assay The free radical scavenging activity of the new 3-(5-aryl- 4,5-dihydro-1H-pyrazol-3-yl)-4-hydroxy-2H-chromene- 2-one 4, 5 was tested by utilizing DPPH scavenging [34]. DPPH is a free radical and accepts one electron or one hydrogen radical to become a stable diamagnetic mole- cule [27]. The reduction capability of DPPH radical was determined by the decrease in absorbance induced by 3-(5-aryl-4,5-dihydro-1H-pyrazol-3-yl)-4-hydroxy-2H- chromene-2-one 4, 5. Briefly, 1.5 ml ethanolic solution of the synthesized compounds (0.2 mM) was added to 1.5 ml (0.2 mM) solution of DPPH radical in ethanol (final concentration of DPPH and synthesized com- pounds was 0.1 mM). The mixture was shaken vigorously and allowed to stand for 30 min. After this, the absorb- ance at 534 nm was determined and the percentage of scavenging activity was calculated using the following formula: Figure 1 Cyclic voltammograms of compounds 5d, 5b and 5e (1 × 10–3 M) in CH3CN, scan rate 100 mV·s–1. Scavenging activity = {[(Ab+ As) – Am]/Ab}×100% Ab: absorbance of 0.1 mM ethanolic solution of DPPH at 534 nm, As: absorbance of 0.1 mM ethanolic solution of test compound at 534 nm, Am: absorbance of ethanolic mixture of the drug and DPPH at 534 nm. Trolox was used as reference compound. All tests and analyses were done on triplicate and averaged on three samples. The results are given in Scheme 4. Among the compounds from the 3-(5-aryl-4,5-dihydro- 1H-pyrazol-3-yl)-4-hydroxy-2H-chromene-2-one 4 series, 4b showed moderate antioxidant activity. The activity exhibited by the compound 4e was the highest. In addition the experimental data show that compound 4a scavenges free radical better than Trolox. According to the experimental results, we notice that increasing the concentration of pyrazole percentage of inhibition reaches 90% for a concentration about 1 µM for all the synthesized products. Thus, we can conclude that substituents on the aryl group do not influence sig- nificantly the anti-oxidant activity. One parameter that has been introduced recently for the interpretation of the results from the DPPH method is the efficient concentration or EC50 value (otherwise called the IC50 value), which is defined as the concentration of substrate that causes 50% loss of the DPPH activity (color) and corresponds to the endpoint of the titration. In all cases, any residual (yellow) color from the reduced form or any non specific absorbance from the sample should be considered in defining the ‘‘endpoint’’ of the titration, i.e., the 50% point. Additionally, this IC50 pa- rameter has also the drawback that the higher the anti- oxidant activity, the lower is the value of EC50. The EC50 values exhibited by 3-(5-aryl-4,5-dihydro- 1H-pyrazol-3-yl)-4-hydroxy-2H-chromene-2-one 4, 5 are summarized in the following Table 2. From inspection of Table 2, it is evident that pyra- zoles 4,5 are more active than trolox. Table 2. The EC50 values exhibited by 3-(5-aryl-4,5-dihydro- 1H-pyrazol-3-yl)-4-hydroxy-2H-chromene-2-one 4, 5 Compounds 4, 5 CI50 (µmol.L–1). 4a 4,8 4b 5 4c 3,8 4d 4,2 5a 3,8 5b 3,2 5c 2,7 5d 3 trolox 7,5 Copyright © 2011 SciRes. IJOC 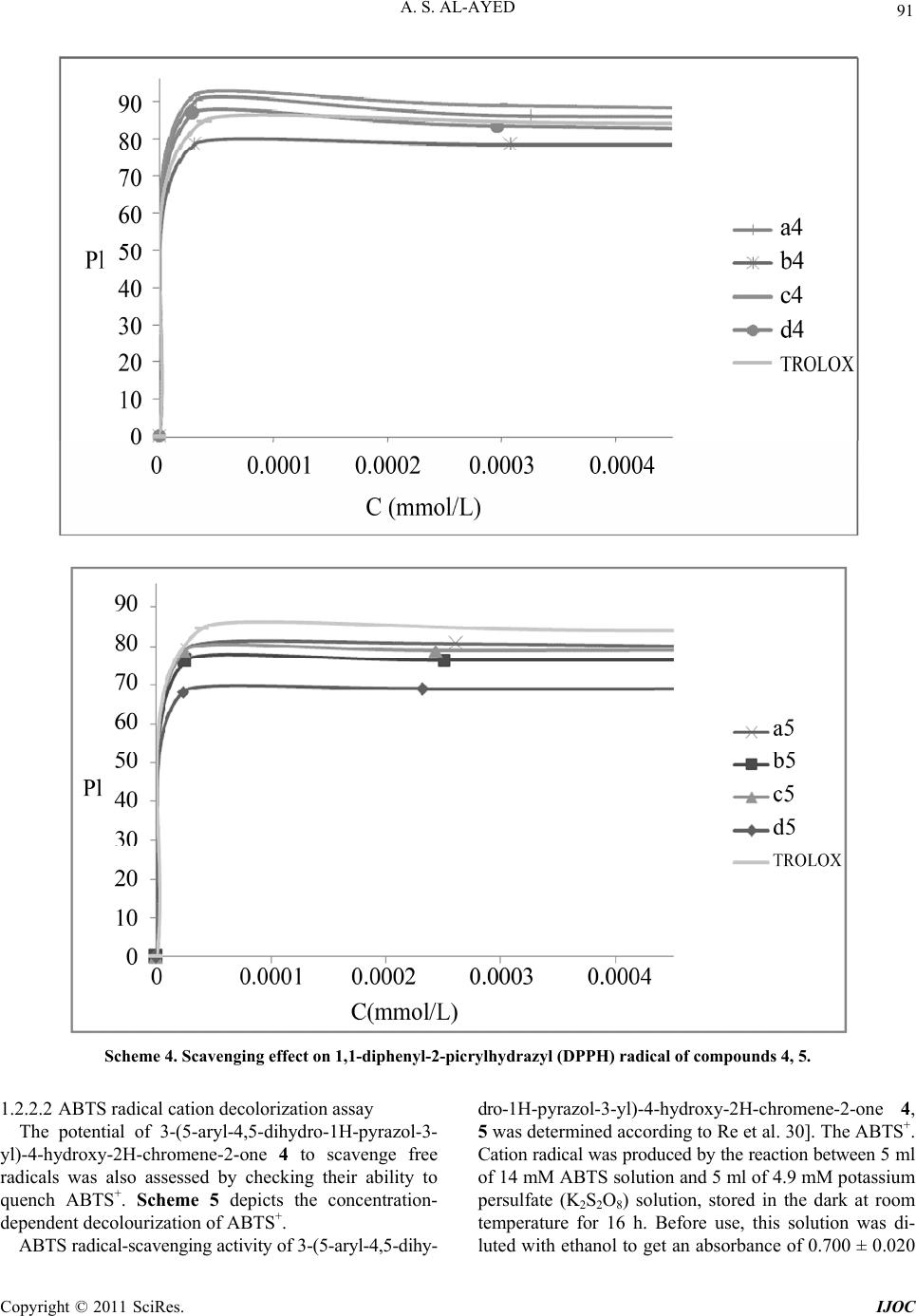 A. S. AL-AYED 91 Scheme 4. Scavenging effect on 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical of compounds 4, 5. 1.2.2.2 ABTS radical cation decolorization assay The potential of 3-(5-aryl-4,5-dihydro-1H-pyrazol-3- yl)-4-hydroxy-2H-chromene-2-one 4 to scavenge free radicals was also assessed by checking their ability to quench ABTS+. Scheme 5 depicts the concentration- dependent decolourization of ABTS+. ABTS radical-scavenging activity of 3-(5-aryl-4,5-dihy- dro-1H-pyrazol-3-yl) -4-hydrox y-2H-chro mene-2-one 4, 5 was determined according to Re et al. 30]. The ABTS+. Cation radical was produced by the reaction between 5 ml of 14 mM ABTS solution and 5 ml of 4.9 mM potassium persulfate (K2S2O8) solution, stored in the dark at room temperature for 16 h. Before use, this solution was di- luted with ethanol to get an absorbance of 0.700 ± 0.020 Copyright © 2011 SciRes. IJOC 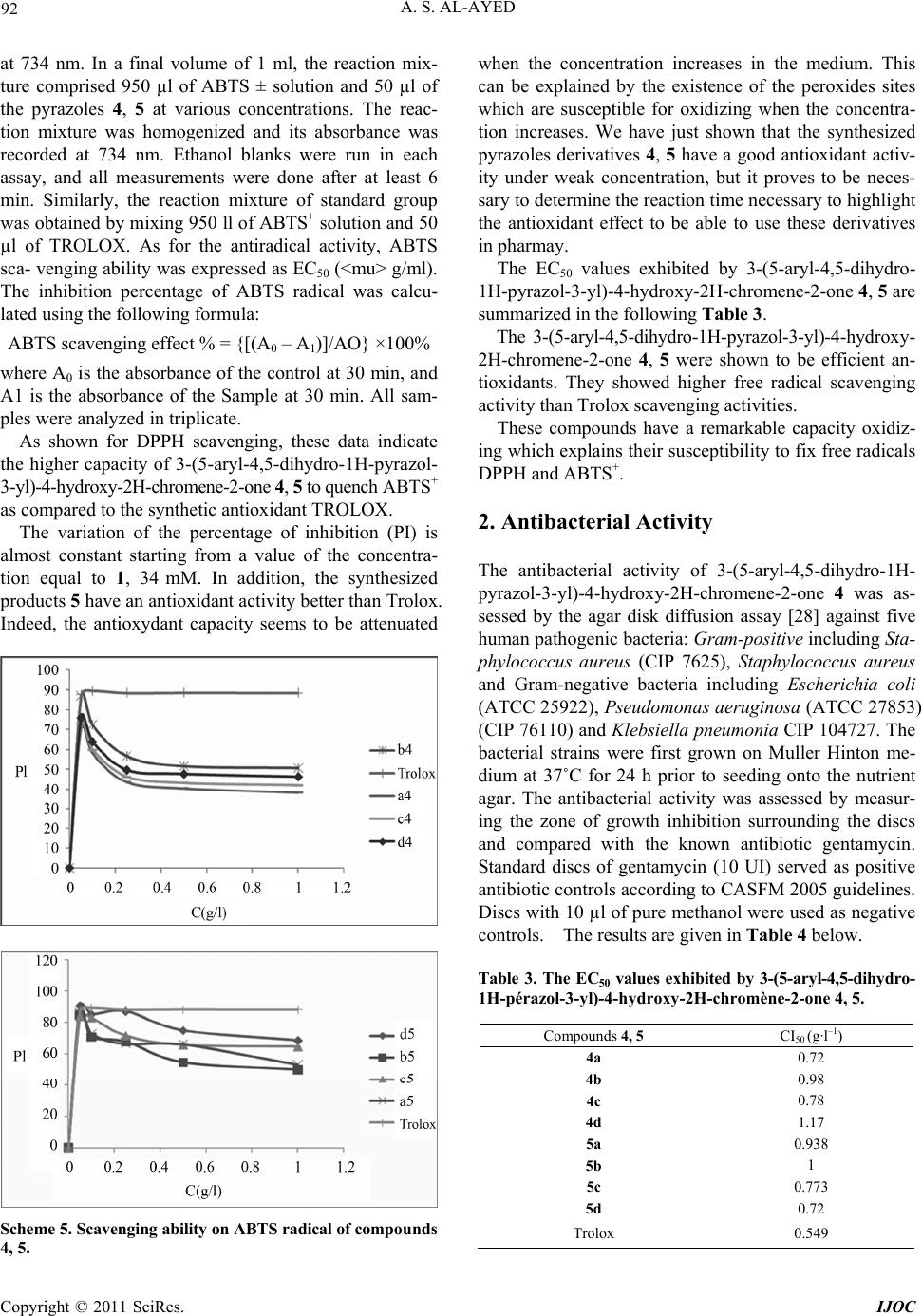 A. S. AL-AYED 92 at 734 nm. In a final volume of 1 ml, the reaction mix- ture comprised 950 µl of ABTS ± solution and 50 µl of the pyrazoles 4, 5 at various concentrations. The reac- tion mixture was homogenized and its absorbance was recorded at 734 nm. Ethanol blanks were run in each assay, and all measurements were done after at least 6 min. Similarly, the reaction mixture of standard group was obtained by mixing 950 ll of ABTS+ solution and 50 µl of TROLOX. As for the antiradical activity, ABTS sca- venging ability was expressed as EC50 (<mu> g/ml). The inhibition percentage of ABTS radical was calcu- lated using the following formula: ABTS scavenging effect % = {[(A0 – A1)]/AO} ×100% where A0 is the absorbance of the control at 30 min, and A1 is the absorbance of the Sample at 30 min. All sam- ples were analyzed in triplicate. As shown for DPPH scavenging, these data indicate the higher capacity of 3-(5-aryl-4,5-dihydro-1H-pyrazol- 3-yl)-4-hydroxy-2H-chromene-2-one 4, 5 to quench ABTS+ as compared to the synthetic antioxidant TROLOX. The variation of the percentage of inhibition (PI) is almost constant starting from a value of the concentra- tion equal to 1, 34 mM. In addition, the synthesized products 5 have an antioxidant activity better than Trolox. Indeed, the antioxydant capacity seems to be attenuated Scheme 5. Scav enging abili ty on ABTS radical of comp o u n ds 4, 5. when the concentration increases in the medium. This can be explained by the existence of the peroxides sites which are susceptible for oxidizing when the concentra- tion increases. We have just shown that the synthesized pyrazoles derivatives 4, 5 have a good antioxidant activ- ity under weak concentration, but it proves to be neces- sary to determine the reaction time necessary to highlight the antioxidant effect to be able to use these derivatives in pharmay. The EC50 values exhibited by 3-(5-aryl-4,5-dihydro- 1H-pyrazol-3-yl)-4-hydroxy-2H-chromene-2-one 4, 5 are summarized in the following Table 3. The 3-(5-aryl-4,5-dihydro-1H-pyrazol-3-yl)-4-hydroxy- 2H-chromene-2-one 4, 5 were shown to be efficient an- tioxidants. They showed higher free radical scavenging activity than Trolox scavenging activities. These compounds have a remarkable capacity oxidiz- ing which explains their susceptibility to fix free radicals DPPH and ABTS+. 2. Antibacterial Activity The antibacterial activity of 3-(5-aryl-4,5-dihydro-1H- pyrazol-3-yl)-4-hydroxy-2H-chromene-2-one 4 was as- sessed by the agar disk diffusion assay [28] against five human pathogenic bacteria: Gram-positive including Sta- phylococcus aureus (CIP 7625), Staphylococcus aureus and Gram-negative bacteria including Escherichia coli (ATCC 25922), Pseudomonas aeruginosa (ATCC 27853) (CIP 76110) and Klebsiella pneumonia CIP 104727. The bacterial strains were first grown on Muller Hinton me- dium at 37˚C for 24 h prior to seeding onto the nutrient agar. The antibacterial activity was assessed by measur- ing the zone of growth inhibition surrounding the discs and compared with the known antibiotic gentamycin. Standard discs of gentamycin (10 UI) served as positive antibiotic controls according to CASFM 2005 guidelines. Discs with 10 µl of pure methanol were used as negative controls. The results are given in Table 4 below. Table 3. The EC50 values exhibited by 3-(5-aryl-4,5-dihydro- 1H-pérazol-3-yl)-4-hydroxy-2H-chromène-2-one 4, 5. Compounds 4, 5 CI50 (g·l–1) 4a 0.72 4b 0.98 4c 0.78 4d 1.17 5a 0.938 5b 1 5c 0.773 5d 0.72 Trolox 0.549 Copyright © 2011 SciRes. IJOC  A. S. AL-AYED 93 Table 4. Antibacterial activity spectrum of compounds 5a-e. Indicator organism inhibition zone (mm) compounds 34 5a 27 5b 28 5c 33 5d 34 5e Staphylococcus aureus (CIP 7625) 24 - 28 Gentamycin 26 5a 28 5b 27 5c 33 5d 34 5e Staphylococcus aureus* 24 Gentamycin 30 5a 27 5b 35 5c 32 5d 31 5e Escherichia coli ATCC 25922 22 - 26 Gentamycin 25 5a 27 5b 31 5c 30 5d 28 5e Klebsiella pneumonia CIP 104727 21 Gentamycin 30 5a 26 5b 34 5c 33 5d 32 5e PseudomonasAeruginosa 27853 (CIP 76110) 15 - 22 Gentamycin The residual antibacterial activity of the compounds was tested by disc diffusion assay against the indicator strain in LB medium at 28˚C ATCC: American Type Culture Collection, USA; CIP: Collection de l’Institut Pasteur, Paris, France LM: Laboratoire de Microbiologie, Centre National de Greffe de Moelle Osseuse, Tunis, Tunisia Methicillin-resistant clinical isolates An examination of the data reveals that all the com- pounds showed antibacterial activity ranging from 25 to 100 μg ml–1. The compounds 5a and 5e were highly active against all the five organisms employed. Compound 5c was highly active against E. coli. From the screened results, it is observed that the presence of methoxy/NO2 group at the phenyl ring increases the antibacterial activity. The highest activity was found in compound 5b bearing a methoxy group at 4-position. 3. Conclusions A new versatile synthetic route to 3-(5-aryl-4,5-dihydro- 1H-pyrazol-3-yl)-4-hydroxy-2H-chromene-2-one 4, 5 by the treatment of 4-hydroxycoumarine with different re- agents is described. The method is easy, rapid and yielded the title compounds 4, 5 in good yields. The structures of the novel compounds were verified by, IR 1D/2D NMR spectroscopy. All the newly synthesized compounds were screened for their antibacterial and antioxidant activities. Among the screened samples, compounds 5a and 5b showed excellent antibacterial activity against E. coli. Compounds with 4-phenyl, 4-methoxyphenyl, 4-fluoro- phenyl and 4-nitrophenyl substituents in the pyrazole ring exhibited higher antioxidant activity than trolox while the pyrazole bearing a p-methoxy substituent in the phenyl ring exhibited enhanced antioxidant activity. 4. Experimental Section 4.1. General All reactions were magnetically stirred. Commercially available reagents were used without further purification. All chemicals were supplied from Aldrich, Merck and- Fluka Co. Melting points were determined by open cap- illary method and were uncorrected. All reactions were monitored by thin layer chroma- tography (TLC). Compounds were visualized with UV light at 254 and 365 nm. Melting points were measured on a WRX-1S instrument. Infrared (IR) spectra were re- corded with a Perkin-Elmer spectrum one B spectrometer. 1H and 13C{1H} NMR spectra were recorded on a Var- ian-Unity spectrometer at 300 MHz using tetramethylsi- lane (TMS) as an internal standard. Cyclic voltammetry (CV) was performed on a BAS 100 BW electrochemical workstation. All CV measurements were carried out us- ing tetrabutylammonium bromide (C4H12BrN) as a sup- porting electrolyte, purging with nitrogen prior to con- duct the experiment. Platinum wire (MF-2013) was used as a working electrode, Ag/AgCl as a reference electrode, and another platinum wire (MF-1032) as a counter elec- trode. 3-Acetyl-4-hydroxycoumarin To a solution of 4-hydroxy-2H-chromen-2-one (3.0 g, 1.86mmol) in acetic acid (16 ml) phosphorus oxych- loride (5.6 ml) was added. The mixture was heated at reflux for 30 min. After cooling to room temperature, the precipitate which separated out was collected by filtra- tion and recrystallized from ethanol to give 3-acetyl-4- hydroxy-2Hchromen-2-one as white needles. Yield 2.7 g (90%); Mp = 135˚C. IR spectrum, ν cm–1: 3185 (OH); 1705 (CO); 1700 (O-CO lactone). 1H NMR spectrum (CDCl3). δ: 2.72 (3H, s, CH3); 7.98 (1H, s, H-5); 7.95 (1H, dd, 3J 7.8, 8.35, 4J 6.8, 1.2 Hz, H-8); 7.1 - 7.4 (2H, m, H-6 + H-7); 17.7 (1H, s, OH).13C{1H} NMR spec- trum (CDCl3), : 29.9 (CH3); 178.5 (CO);159.8 (C-4); 154.6 (C-2); 101.3 (C-3); 115.0 - 136.0 (Carom). Copyright © 2011 SciRes. IJOC  A. S. AL-AYED 94 General procedure for the preparation of the cou- marinic chalcones 4a-i 3-acetyl-4-hydroxy-2Hchromen-2-one (0.031 mol) and the substituted aromatic aldehyde (0.03 mol) were dis- solved in 30 mL of chloroform. A catalytic amount of piperidine (0.02 mol) was added and the reaction mixture was refluxed for 1.5 h. The chloroform was removed under vacuum and the residue was washed with methanol Yield 65%; mp 234˚C. IR spectrum, ν cm–1: 1683 (C=O lactone), 1608 (C=N), 3230 (NH). 1H NMR spectrum (DMSO-d6, 300 MHz, tamb) 3-((2E)-3(phenyl)prop-2-enoyl)-2H-chromen-2-one: 3a Yield 85%; mp 265˚C. IR spectrum, ν cm–1: 1490 (C=O), 1728 (O-C-O), 1529 (C=C). 1H NMR spectrum (CDCl3). δ ppm: 6.5 - 8.3 (m, 11H, Harom, Heth); 18.56 (s, 1H, OH). 13CNMR spectrum (CDCl3). δ ppm: 116.9 (C3); 135.8 (C2); 147.1 (C4); 134.5 (Cethyl) 3-((2E)-3(4-fluorophenyl)prop-2-enoyl)-2H-chromen- 2-one: 3b Yield 80%; mp 215˚C. IR spectrum, ν cm–1: 1494 (C=O), 1716 (O-C-O), 1531 (C=C). 1H NMR spectrum (CDCl3, 400MHz). δ ppm: 6.51 - 8.21 (m, 10H, Harom+éthyl ); 18,4(s, 1H, OH). 13CNMR spectrum (CDCl3). δ ppm: 100.9 (C3); 154.8 (C2); 166 (C4); 181.5 (CO); 136.2 (Céthyl1); 131.5 (Céthyl2); 116.5 - 131.51 (Carom). NMR 19F (CDCl3, 400 MHz, tamb) ppm: –107.8 *3-((2E)-3(2, 5-dimethylphenyl)prop-2-enoyl)-2H-chro- men-2-one: 3c Yield 75%; mp 224˚C. IR spectrum, ν cm–1: 1490 (C=O), 1720 (O-C-O), 1527 (C=C). 1H NMR spectrum (CDCl3). δ ppm: 2,41 (s, 1H, CH3), 3,21 (s, 3H, CH3), 6.82 – 8.21 (m, 9H, Harom+éthy); 13,81 (s, 1H, OH). 13CNMR spectrum (CDCl3). δ ppm: 91.4 (C3); 135.8 (C2); 161.4 (C4); 139.1 (Céthyl), 134.1 (Céthy2); 116.4 – 133.2 (Crom), 176.2 (CO) *3-((2E)-3(4-methoxyphenyl) prop-2-enoyl)-2H-chromen- 2-one: 3d Yield 75%; mp 194˚C. IR spectrum, ν cm–1: 1494 (C=O), 1708 (O-C-O), 1595 (C=C). 1H NMR spectrum (CDCl3). δ ppm: 3.84 (s, 3H, OCH3); 6.92 (d, 1H, CH); 8.25 (d, 1H, CH); 7.25 - 8.08 (m, 8H, Harom); 18.05 (s, 1H, OH). 13CNMR spectrum (CDCl3). δ ppm: 55.4 (OCH3); 114.4 (C3); 116.8 – 131.3 (Carom); 135.6 (C2); 147.48(C4); 162.4 (CO). *3-((2E)-3(4-nitrophenyl)prop-2-enoyl)-2H-chromen- 2-one: 3e Yield 72%; mp 194˚C. IR spectrum, ν cm–1: 1489 (C=O), 1712 (O-C-O), 1535 (C = C). 1H NMR spectrum (CDCl3). δ ppm: 7.1 - 8.6 (m,1OH, Harom+ethyl); 18.4 (s1, H, OH). 13CNMR spectrum (CDCl3). δ ppm: 98.7 (C3); 151.8 (C2); 178.7 (C4); 182.5 (CO); 151.86 (Cethyl1); 122.5 (Cethyl2). 3-(5-(phenyl)-4,5-dihydro-1H-pyrazol-3-yl)-4-hydroxy -2H-chromene-2-ones: 4a ppm: 1.75(d, 2H, CH2), 3.74 (t, 1H, CH), 6,82-7,27 (m, 9H, Harom). 13CNMR spectrum (DMSO-d6, 75 MHz, tamb) ppm: 40, 6 (CH2) 49,5 (CH), 91,1 (C3); 158,6 (C2); 166,4 (C4); 155,7 (C = N), 121,5-127,1 (Carom). 3-(5-(4-fluorophenyl)-4,5-dihydro-1H-pyrazol-3-yl)-4- hydroxy-2H-c hromene-2-ones: 4b Yield 70%; mp 220˚C. IR spectrum, ν cm–1: 1668 (C=O lactone), 1612 (C=N), 3192 (NH). 1H NMR spectrum (DMSO-d6, 300 MHz, tamb) ppm: 1,76(d, 2H, CH2), 3.64 (t, 1H, CH), 6.79 – 7.35(m,8H, Harom). 13CNMR spectrum (DMSO-d6, 75 MHz, tamb) ppm: 40.2 (CH2) 48.7 (CH), 91.1 (C3); 158.5 (C2); 166.7 (C4); 155.2 (C=N), 120.8 - 128.3 (Carom). NMR 19F (DMSO-d6, 300 MHz, tamb) ppm: –139.26 3-(5-(2,5-dimethylphenyl)-4,5-dihydro-1H-pyrazol-3- yl)-4-hydroxy-2H-chromene-2-ones: 4c Yield 80%; mp 252˚C. IR spectrum, ν cm–1: 1683 (C=O lactone), 1606 (C=N), 3230 (NH). 1H NMR spectrum (DMSO-d6, 300 MHz, tamb) ppm: 1.68 (d, 2H, CH2), 3,68 (t, 1H, CH), 2,34 (s, 6H, 2CH3), 6.85 - 7.32(m, Harom). 13CNMR spectrum (DMSO-d6,75 MHz, tam ) b ppm: 18.6 (CH3), 24.5 (CH3), 40.2 (CH2), 42.6 (CH), 91.7 (C3); 158.4 (C2); 167.6 (C4); 155.7 (C=N), 121.7 - 132.8 (Carom). 3-(5-(4-methoxyphényl)-4,5-dihydro-1H-pyrazol-3-yl) -4-hydroxy -2 H -chr omene-2-ones: 4d Yield 85%; mp 210˚C. IR spectrum, ν cm–1: 1668 (C=O lactone), 1608 (C=N), 3207 (NH). 1H NMR spectrum (DMSO-d6,300 MHz,tamb) ppm: 3.84 (s, 3H, OCH3), 6.82 - 8.1 (m, 8H, Harom), 4.12 (d, 2H, CH2), 4,81 (t, 1H, CH). 13CNMR spectrum (DMSO-d6, 75 MHz, tamb) ppm: 58.2 (OCH3), 55.0 (CH), 43.7 (CH2), 91.4 (C3); 158.7 (C2); 161.6 (C4); 153.3 (C=N), 113.8 – 133.4 (Carom). 3-(5-(phenyl)-1-phenyl-4,5-dihydro-1H-pyrazol-3-yl)- 4-hydroxy-2H-chromene- 2-one:5a Yield 70%; mp 230˚C. IR spectrum, ν cm–1: 1668 (C=O lactone), 1608 (C=N), 3110 (NH). 1H NMR spectrum (DMSO-d6, 300 MHz, tamb) ppm: 1.65 (d, 2H, CH2), 3.75 (t, 1H, CH), 6.42 - 7.35 (m Harom). 13CNMR spectrum (DMSO-d6, 75 MHz, tamb) ppm: 36.4 (CH2) 54.8 (CH), 92.3 (C3); 159.7 (C2); 167.8 (C4); 158.1 (C=N), 113.5 - 133.1 (Carom). Copyright © 2011 SciRes. IJOC  A. S. AL-AYED 95 3-(5-(4-fluorophenyl)-1-phenyl-4,5-dihydro-1H-pyraz ol-3-yl)-4-h ydroxy-2H-chromene- 2-one: 5b Yield 72%; mp 226˚C. IR spectrum, ν cm–1: 1668 1683 (C=O lactone), 1606 (C=N), 3230 (NH). 1H NMR spe- ctrum (DMSO-d6, 300 MHz, tamb) ppm: 1.62 (d, 2H, CH2), 3.65 (t, 1H, CH), 6.35 – 7.45 (m Harom). 13CNMR spectrum (DMSO-d6,75 MHz, tamb) ppm: 35.3 (CH2) 52.7 (CH), 92.1 (C3); 159.2 (C2); 167.8 (C4); 152.6 (C=N), 120.5 - 134.1 (Carom). NMR 19F (DMSO-d6, 300 MHz, tamb) ppm: –129,42 3-(5-(2,5-dimethylphenyl)-1-phenyl-4,5-dihydro-1H-p yrazol-3-yl)-4-hydroxy-2H-chromene-2-one: 5c Yield 78%; mp 265˚C. IR spectrum, ν cm–1: 1681 (C=O lactone), 1608 (C=N), 3220 (NH). 1H NMR spectrum (DMSO-d6, 300 MHz, tamb) ppm: 1,63 (d, 2H, CH2), 3.68 (t, 1H, CH), 6.35 – 7.68 (m Harom). 13CNMR spec- trum (DMSO-d6, 75 MHz, tamb) ppm: 35.3 (CH2) 52.8 (CH), 92.1 (C3); 158.8 (C2); 168.4 (C4); 159.6 (C=N), 126.5 - 142.1 (Carom). 3-(5-(4-methoxyphenyl)-1-phenyl-4,5-dihydro-1H-pyr azol-3-yl)-4-hydr ox y -2 H -chromene-2-one: 5d Yield 75%; mp 216˚C. IR spectrum, ν cm–1: 1681 (C=O lactone), 1608 (C=N), 3234 (NH). 1H NMR spectrum (DMSO-d6, 300 MHz, tamb) ppm: 1.62 (d, 2H, CH2), 3.68 (t, 1H, CH), 3.78 (s, 3H, CH3), 6.47 – 7.42 (m Harom). 13CNMR spectrum (DMSO-d6, 75 MHz, tamb) ppm: 32.4 (CH2) 51.4 (CH), 55.8 (OCH3), 93.3 (C3); 156.4 (C2); 167.3 (C4); 152.5 (C=N), 113.4 – 132.6 (Carom). 5. References [1] F. Aqil and I. Ahmad, “Rapid and Accurate Spe- cies―Specific Detection of Phytophthora Infestans throu- gh Analysis of Its Regions in Its rDNA,” Journal of Mi- crobiology and Biotechnology, Vol. 19, No. 6, 2000, pp. 653-657. [2] A. W. Bauer, W. M. M. Kirby, J. C. Sherris and M. Turch, “Antibiotic Susceptibility Testing by a Standardized Sin- gle Disk Method,” American Journal of Clinical Pathol- ogy, Vol. 45, No. 5, 1998, pp. 493-496. [3] O. Bruno, A. Ranise, F. Bonadavalli, P. Schenone, M. D’Amico, W. Felipplli, A. Filippelli and F. Rossi, “3,5- Diphenyl-1H-Pyrazole Derivatives. XI. N-Aryl-5(3)- Phenyl-4-(3,5-Diphenyl-1-Pyrazolyl)-3(5)-Pyrazole Ami- nes, 5-Substituted 4,5-Dihydro-3-Phenyl-4-(3,5-Diphenyl -1-Pyrazolyl)-1H-Pyrazoles and 2,6-Disubstituted 1,6-Di- hydro-4-Phenyl-5-(3,5-Diphenyl-1-Pyrazolyl)Pyrimidines with Antipyretic, Anti-Inflammatory and Other Activi- ties,” Farmaco, Vol. 48, No. 4, 1998, pp. 949-966 [4] J. R. Dimmock, S. K. Raghavan, et al., “Antileukemic Evaluation of Some Mannich Bases Derived from 2- Arylidene-1,3-Diketones,” European Journal of Medicinal Chemistry, Vol. 18, No. 4, 1998, pp. 248-254. [5] H. R. Eisenhauer and K. P. Link,“ Studies on 4-Hydroxy- Coumarins. XIII. The Mechanism for the Reaction of 4- Hydroxycoumarin with Aliphatic Acid Chlorides,” Jou- rnal of the American Chemical Society, Vol. 75, No. 9, 1953, pp. 2044-2050. doi:10.1021/ja01105a006 [6] A. A. El-Barbary, A. I. Khodair, E. B. Pederson and C. Nielsen, “S-Glucosylated Hydantoins as New Antiviral agents,” Journal of Medicinal Chemistry, Vol. 37, No. 1, 1994, pp. 73-77. doi:10.1021/jm00027a009 [7] E. S. M. El-khawass and A. E. Bistawroos, “Synthesis and Biological Activity of Heterocycles from Chalcone,” Medicinal Chemistry Research, Vol. 17, No. 2-7, 2008, pp. 318-325 [8] H. I. El-Subbagh, S. M. Abu-Zaid, M. A. Mahran, F. A. Badaria, Al-Obaid,” Synthesis and Biological Evaluation of Certain α,β-Unsaturated Ketones and Their Corre- sponding Fused Pyridines as Antiviral and Cytotoxic Agents,” Journal of Medicinal Chemistry, Vol. 43, No. 15, 2000, pp. 2915-2921. doi:10.1021/jm000038m [9] E. O. Nuzum, P. J. Rosenthal and J. H. Mckerrow, “In Vitro Antimalarial Activity of Chalcones and Their De- rivatives,” Journal of Medicinal Chemistry, Vol. 38, No. 26, 1995, pp. 5031-5037. doi:10.1021/jm00026a010 [10] A. Nayak and A. S. Mittra, “4,4-Bis-5-Pyrazolones and Their 4,4-Unsaturated Products for Possible Use as Fun- gicides,” Journal of the Indian Chemical Society, Vol. 57, 1980, pp. 643-649 [11] N. Marquez, R. Sancho, L. M. Bedoya, J. Alcamy, J. L. Lopez-Perez, A. S. Feliciano, B. L. Fiebich and E. Munoz, “Mesuol, a Natural Occurring 4-Phenylcoumarin, Inhibits HIV-1 Replication by Targeting the NF-κB Pathway,” Antiviral Research, Vol. 66, No. 2-3, 2005, pp. 137-143. doi:10.1016/j.antiviral.2005.02.006 [12] C. Spino, M. Dodier and S. Sotheeswaran, “Anti-HIV Coumarins from Calophyllum Seed Oil,” Bioorganic & Medicinal Chemistry Letters, Vol. 8, No. 24, 1998, pp. 3475-3481. doi:10.1016/S0960-894X(98)00628-3 [13] S. Thaisrivongs, K. D. Watenpaugh, W. J. Howe, et al., “Structure-Based Design of Novel HIV Protease In- hibitors: Carboxamide-Containing 4-Hydroxycoumarins and 4-Hydroxy-2-Pyrones as Potent Nonpeptidic Inhibi- tors,” Journal of Medicinal Chemistry, Vol. 38, No. 18, 1995, pp. 3624-3630. doi:10.1021/jm00018a023 [14] D. Yu, M. Suzuki, L. Xie, S. L. Morris-Natschke and K. H. Lee, “Recent Progress in the Development of Cou- marin Derivatives as Potent Anti-HIV Agents,” Medici- nal Research Reviews, Vol. 23, No. 3, 2003, pp. 322-328. doi:10.1002/med.10034 [15] J. W. Erickson and S. K. Burt, “Structural Mechanisms of HIV Drug Resistance,” Annual Review of Pharmacology and Toxicology, Vol. 36, 1996, pp. 545-571. doi:10.1146/annurev.pa.36.040196.002553 [16] I. Kostova, S. Raleva, P. Genova and R. Argirova, “Structural Features of Antitumor Titanium Agents and Related Compounds,” Bioinorganic Chemistry and Appli- Copyright © 2011 SciRes. IJOC  A. S. AL-AYED Copyright © 2011 SciRes. IJOC 96 cations, Vol. 3, No. 3-4, 2005, pp. 317-329. doi:10.1155/BCA.2005.317 [17] A. J. Vlietinck, T. de Bruyne, S. Apers and L. A. Pieters, “Plant-Derived Leading Compounds for Chemotherapy of Human Immunodeficiency Virus (HIV) Infection”, Medicinal Chemistry Research, Vol. 64, 1998, pp. 97- 102. [18] J. B. Harborne and T. J. Mabry, “Flavonoid Chemistry of chenopodium fremontii. Infraspecific Variation and Sys- tematic Implications at the Interspecific Level,” Chemical Abstracts, Vol. 119, pp. 180774-180776. [19] K. Kostka, “Chromone Derivatives. VI. Reaction of Phenylhydrazine with Chromone and x-Formyl-o-Hydro- Xyacetophenone,” Polish Journal of Chemistry, Vol. 47, 1973, pp. 305-313 [20] E. O. Nuzum, P. J. Rosenthal and J. H. Mckerrow, “In Vitro Antimalarial Activity of Chalcones and Their De- rivatives,” Journal of Medicinal Chemistry, Vol. 38, No. 26, 1995, pp. 5031-5037. doi:10.1021/jm00026a010 [21] A. Nayak and A. S. Mittra “4,4-Bis-5-Pyrazolones and Their 4,4-Unsaturated Products for Possible Use as Fun- gicides,” Journal of the Indian Chemical Society, Vol. 57, 1980, pp. 643-649 [22] O. Nerya, R. Musa, S. Khatib, S. Tamir, Vaya, “Chalco- nes as Potent Tyrosinase Inhibitors: The Effect of Hydro- xyl Positions and Numbers,” Phytochemistry, Vol. 65, No. 10, 2004, pp. 1389-1395. doi:10.1016/j.phytochem.2004.04.016 [23] Z. N. Siddiqui, G. Khuwaja and M. Asad, “Synthesis of Novel Heterocycles from 3-(3-Alkyl-5-Mercapto-1,2,4- Triazolyliminomethyl) Chromones,” Heterocyclic Com- mun, Vol. 12, No. 6, 2006, pp. 443-448 [24] L. Santana, E. Uriarte, L. via Dalla and O. Gia, “A New Benzoangelicin with Strong Photobiological Activity,” Bioorganic & Medicinal Chemistry Letters, Vol. 10, No. 6, 2000, pp. 135-137. doi:10.1016/S0960-894X(99)00640-X [25] R. H. Wiley and C. H. Jarboe, “Synthesis of Pyrazolines by the Reactions of α,β-enones with Diazomethane and Hydrazines,” Chemistry of Heterocyclic Compounds, Vol. 33, No. 6, 1997, pp. 647-659, doi:10.1007/BF02291794 [26] K. C. Fylaktakidou, D. J. Hadjipavlou-Litina, K. E. Litinas and D. N. Nicolaides, “Natural and Synthetic- coumarin Derivatives with Anti-Inflammatory and Anti- oxidant Activity,” Current Pharmaceutical Design, Vol. 10, No. 6, 2004, pp. 3813-3833 [27] S. Kirkiachiarian, R. Bakhchinian, H. Chidiak, M. Mazmanian and C. Planche, “Two New Flavonoids and Other Constituents in Licorice Root; Their Relative As- tringency and Radical Scavenging Effects,” Annales Pha- rmaceutiques Françaises, Vol. 57, No. 5, 1999, pp. 251- 259. [28] J. R. Soares, T. C. P. Dins, A. P. Cunha and L. M. Ameida, “Antioxidant Activity of Some Extracts of Thymus Zy- gis,” Free Radical Research, Vol. 26, No. 5, 1997, pp. 469-476. doi:10.3109/10715769709084484 NOESY: Nuclear Overhauser effect spectroscopy HMBC: Heteronuclear Multiple Bond Correlation experiment
|