Functionalization of Cobalt Ferrite Nanoparticles with Alginate Coating for Biocompatible Applications
1320
the intended application point of view.
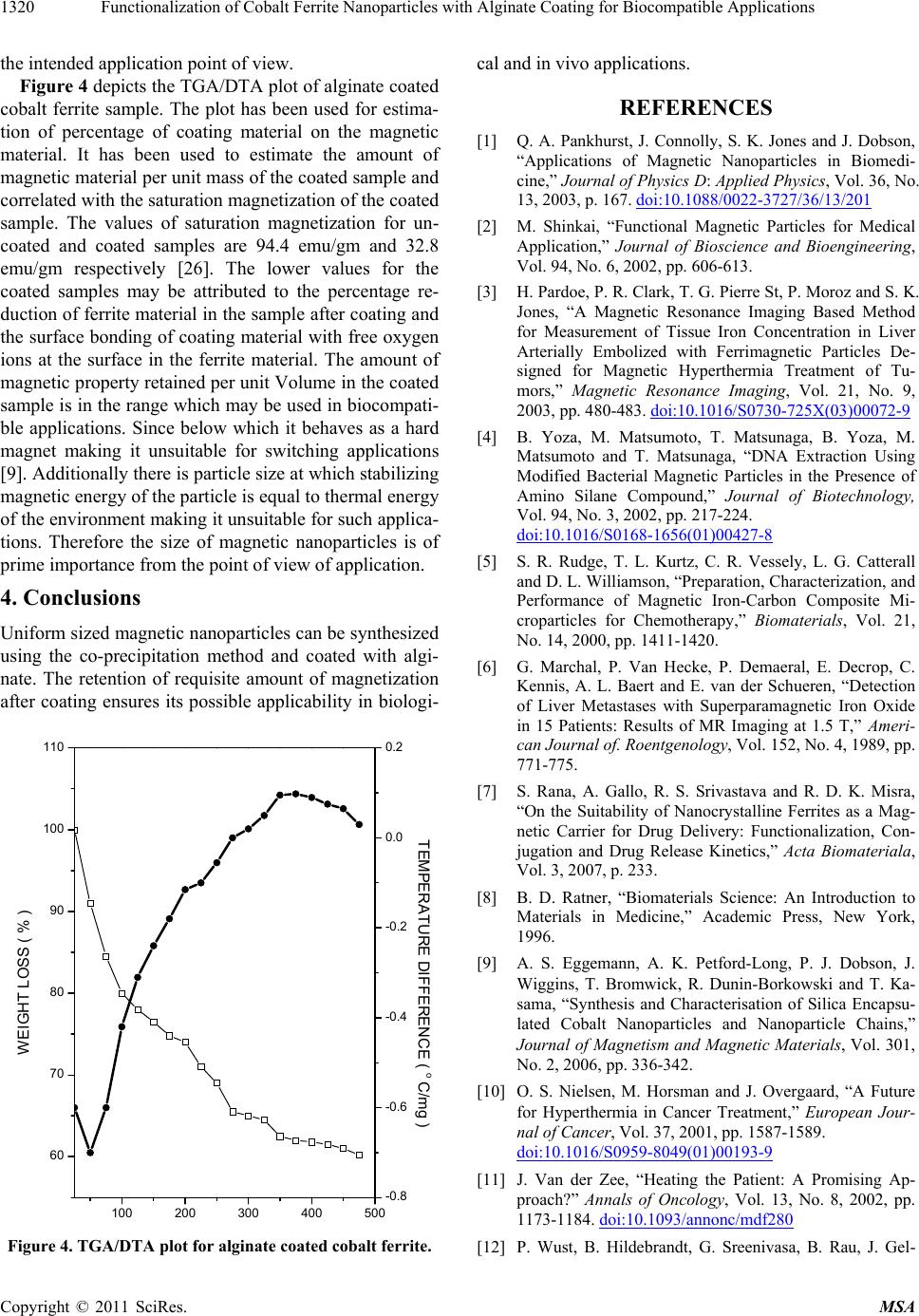
Figure 4 depicts the TGA/DTA plot of alginate coated
cobalt ferrite sample. The plot has been used for estima-
tion of percentage of coating material on the magnetic
material. It has been used to estimate the amount of
magnetic material per unit mass of the coated sample and
correlated with the saturation magnetization of the coated
sample. The values of saturation magnetization for un-
coated and coated samples are 94.4 emu/gm and 32.8
emu/gm respectively [26]. The lower values for the
coated samples may be attributed to the percentage re-
duction of ferrite material in the sample after coating and
the surface bonding of coating material with free oxygen
ions at the surface in the ferrite material. The amount of
magnetic property retained per unit Volume in the coated
sample is in the range which may be used in biocompati-
ble applications. Since below which it behaves as a hard
magnet making it unsuitable for switching applications
[9]. Additionally there is particle size at which stabilizing
magnetic energy of the particle is equal to thermal energy
of the environment making it unsuitable for such applica-
tions. Therefore the size of magnetic nanoparticles is of
prime importance from the point of view of application.
4. Conclusions
Uniform sized magnetic nanoparticles can be synthesized
using the co-precipitation method and coated with algi-
nate. The retention of requisite amount of magnetization
after coating ensures its possible applicability in biologi-
100 200 300400 500
60
70
80
90
100
110
WEIGHT LOSS ( % )
-0.8
-0.6
-0.4
-0.2
0.0
0.2
TEMPERATURE DIFFERENCE ( o C/mg )
Figure 4. TGA/DTA plot for alginate coated cobalt ferrite.
cal and in vivo applications.
REFERENCES
[1] Q. A. Pankhurst, J. Connolly, S. K. Jones and J. Dobson,
“Applications of Magnetic Nanoparticles in Biomedi-
cine,” Journal of Physics D: Applied Physics, Vol. 36, No.
13, 2003, p. 167. doi:10.1088/0022-3727/36/13/201
[2] M. Shinkai, “Functional Magnetic Particles for Medical
Application,” Journal of Bioscience and Bioengineering,
Vol. 94, No. 6, 2002, pp. 606-613.
[3] H. Pardoe, P. R. Clark, T. G. Pierre St, P. Moroz and S. K.
Jones, “A Magnetic Resonance Imaging Based Method
for Measurement of Tissue Iron Concentration in Liver
Arterially Embolized with Ferrimagnetic Particles De-
signed for Magnetic Hyperthermia Treatment of Tu-
mors,” Magnetic Resonance Imaging, Vol. 21, No. 9,
2003, pp. 480-483. doi:10.1016/S0730-725X(03)00072-9
[4] B. Yoza, M. Matsumoto, T. Matsunaga, B. Yoza, M.
Matsumoto and T. Matsunaga, “DNA Extraction Using
Modified Bacterial Magnetic Particles in the Presence of
Amino Silane Compound,” Journal of Biotechnology,
Vol. 94, No. 3, 2002, pp. 217-224.
doi:10.1016/S0168-1656(01)00427-8
[5] S. R. Rudge, T. L. Kurtz, C. R. Vessely, L. G. Catterall
and D. L. Williamson, “Preparation, Characterization, and
Performance of Magnetic Iron-Carbon Composite Mi-
croparticles for Chemotherapy,” Biomaterials, Vol. 21,
No. 14, 2000, pp. 1411-1420.
[6] G. Marchal, P. Van Hecke, P. Demaeral, E. Decrop, C.
Kennis, A. L. Baert and E. van der Schueren, “Detection
of Liver Metastases with Superparamagnetic Iron Oxide
in 15 Patients: Results of MR Imaging at 1.5 T,” Ameri-
can Journal of. Roentgenology, Vol. 152, No. 4, 1989, pp.
771-775.
[7] S. Rana, A. Gallo, R. S. Srivastava and R. D. K. Misra,
“On the Suitability of Nanocrystalline Ferrites as a Mag-
netic Carrier for Drug Delivery: Functionalization, Con-
jugation and Drug Release Kinetics,” Acta Biomateriala,
Vol. 3, 2007, p. 233.
[8] B. D. Ratner, “Biomaterials Science: An Introduction to
Materials in Medicine,” Academic Press, New York,
1996.
[9] A. S. Eggemann, A. K. Petford-Long, P. J. Dobson, J.
Wiggins, T. Bromwick, R. Dunin-Borkowski and T. Ka-
sama, “Synthesis and Characterisation of Silica Encapsu-
lated Cobalt Nanoparticles and Nanoparticle Chains,”
Journal of Magnetism and Magnetic Materials, Vol. 301,
No. 2, 2006, pp. 336-342.
[10] O. S. Nielsen, M. Horsman and J. Overgaard, “A Future
for Hyperthermia in Cancer Treatment,” European Jour-
nal of Cancer, Vol. 37, 2001, pp. 1587-1589.
doi:10.1016/S0959-8049(01)00193-9
[11] J. Van der Zee, “Heating the Patient: A Promising Ap-
proach?” Annals of Oncology, Vol. 13, No. 8, 2002, pp.
1173-1184. doi:10.1093/annonc/mdf280
[12] P. Wust, B. Hildebrandt, G. Sreenivasa, B. Rau, J. Gel-
Copyright © 2011 SciRes. MSA