Journal of Water Resource and Protection
Vol. 5 No. 9 (2013) , Article ID: 37430 , 6 pages DOI:10.4236/jwarp.2013.59091
Fatty Acids Composition and Biodiesel Characterization of Dunaliella salina
Department of Botany and Microbiology, Faculty of Science, Alexandria University, Alexandria, Egypt
Email: *emfakhr@hotmail.com
Copyright © 2013 Eman M. Fakhry, Dahlia M. El Maghraby. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received June 16, 2013; revised July 2, 2013; accepted August 9, 2013
Keywords: Biodiesel Characterization; Biomass; Dunaliella salina; Fatty Acids
ABSTRACT
This study discusses the perspectives regarding the green alga Dunaliella salina Toed for biodiesel manufacturing purposes. The alga was cultivated under controlled lab conditions. Biomass concentration at early stationary grown microalga was 2.6 mg/L dry weight, while the algal oil was about 27.1% of the biomass. Algal oil was esterified and analyzed using GLC technique. Fourteen fatty acid methyl esters were identified. The amount of saturated and unsaturated fatty ester fractions was 35% and 65% respectively. The physicochemical properties of fatty acids comprising biodiesel were discussed. However, culture optimization coupled with genetic improvement will definitely represent contributions to bring about innovation in oil hyper-producing D. salina that will ultimately meet with success.
1. Introduction
Owing to the rising price of fuel and the worry about global warming that is associated with burning fossil fuels as well as the depleting nature of fossil fuel resources [1], biofuel is gaining a significant value as an attractive fuel. Microalgae can offer numerous different types of renewable biofuels. These comprise methane produced by anaerobic digestion of the algal biomass [2]; photobiologically produced biohydrogen [3,4] and biodiesel resultant from microalgal oil [1,5]. In this situation, the employing of microalgae as a sustainable source of energy seemed extremely attractive to the energetic market [6,7]. Microalgae exhibit several important aspects for innovative research on renewable energy. Easy and inexpensive nutrient system, faster growth rate, high biomass productivity and smart biochemical profile recommend strong application as a bioenergy foundation [8].
Biodiesel is a mixture of monoalkyl esters of long chain fatty acids derived from a renewable lipid feedstock [9]. It is composed of 90% - 98% triglycerides, and smaller amounts of monoand diglycerides and free fatty acids, besides residual amounts of phospholipids, phosphatides, carotenes, tocopherols, sulphur compounds and water [10]. Algal oil can be converted into biodiesel through a process called transesterification. Transesterification is a chemical reaction between triglycerides and alcohol in the presence of a catalyst to produce monoesters that are termed as biodiesel [11-13]. The success of algal biodiesel depends on the structure of the economy and energy replacement. From an economic perspective, developed countries are more likely to adopt clean energies because they have more flexible economies [14].
Depending on species, many microalgae have been described as lipid-rich strains for their hydrocarbons and other complex oils. However, not all algal oils are satisfactory for the production of biodiesel [15,16]. Many microalgal species can be promoted to accumulate high lipid contents [17], although average lipid contents vary between 1% and 70%, some species may reach 90% (w/w DW) under certain conditions [18-20]. Studies are considered necessary to achieve a fundamental understanding of the relationship between the combination of environmental parameters and algal mass production with high lipid content. Maximum biomass and lipid content of Botryococcus braunii are attained by photoperiod of 12 h light: 12 h dark, temperature of 23˚C, and 0.8755% NaCl, and 30 - 60 W/m2 irradiance of light intensity [21].
Dunaliella salina Toed is a motile unicellular wall-less green alga that accumulates β-carotene and has a cell size of about 6 - 14 μm. It grows in a wide range habitat. It has a high tolerance to salt, temperature and light. This microorganism is quite easy to cultivate and has a relatively high growth rate and lipid content [22].
The objective of this investigation is to determine the significance of Dunaliella salina as a prospective source for potential future biodiesel. Fatty acids profile of the alga as a tool for testing the resulting biodiesel was studied. The following biodiesel properties were discussed: kinematic viscosity, cetane number, oil stability and oil density.
2. Materials and Methods
2.1. Organism and Cultivation Conditions
Dunaliella salina is a green halophilic alga that sharing among the most basic and essential components of aquatic environment. It was obtained from the algal culture collection of the phycology laboratory, Botany and Microbiology Department, Faculty of Science, University of Alexandria. This organism was originally isolated from the brine water of salty lagoon at El Max district, Alexandria, Egypt.
Axenic samples of known volume were inoculated into sterilized 1 L Erlenmeyer flasks containing 400 ml MH medium [23]. These cultures were maintained in a photoincubator set at 25˚C ± 1˚C, a light intensity of 60 μmol photon s−1∙m−2 (as supplied by cool-white fluorescence tubes) and a 12:12 h dark: light photoperiod with manual shaking twice per day for 16 days.
2.2. Biomass Estimation
Algal growth was characterized based on cell counts. 1 ml of algal culture was removed from the replicate flasks at regular intervals throughout the experiment. It preserved with iodine solution prior to counting using a haemocytometer slide under bright-field contrast microscopy.
The cultures were harvested by centrifugation at 5000 rpm for 15 min. Pellets were then washed twice with distilled water and dehydrated at 30˚C until a constant weight was achieved. The dry weight of the algal biomass was estimated from the average of at least three representative samples.
2.3. Lipid Extraction
At early stationary phase after 12 days of experimentation, lipids were extracted by using chloroform: methanol (2:1) solvent mixture according to the methods of Bligh and Dyer [24]. The extracted lipid was separated into two layers, the upper layer methanol together with water was removed and the chloroform layer including lipid was collected. The residues were subjected to repeated extraction twice. The entire extracts were mixed together forming crude oil extract and the chloroform was evaporated.
2.4. Fatty Acid Methyl Ester Analysis
Via direct methylation, FAMEs were prepared as described by Christie [25]. The sample was esterified in 1% sulfuric acid in absolute methanol. The mixture left overnight at 50˚C then water containing sodium chloride is added and the required esters are extracted with hexane to separate the layers. The hexane layer is washed with water containing potassium bicarbonate and dried over anhydrous sodium sulfate. The solvent was evaporated using rotary evaporator. The composition of FAME was quantified and identified on a Shimadzu gas-liquid chromatography equipped with a flame ionization detector with packing column material Hp-5. The carrier gas was nitrogen and the short speed was 5 mm/min. Fatty acid methyl esters of the micro-alga were deduced by comparing their retention times with those of standards. Quantification was based on the internal standard method.
2.5. Statistical Analysis
All analyses were carried out in triplicate, and the standard deviations (SD) were determined.
3. Results and Discussion
3.1. Algal Biomass and Oil Content
Figure 1 shows that the number of cells after 12 days of experimentation achieved a maximum value of 7.1 × 106 cells/ml. This value corresponding to 2.6 mg/L dry weight and a relatively high oil content of 27.1% (Table 1). For the purpose of biodiesel production, high oil content and rapid growth rates are major factors in selecting an algal strain [18]. Rodolfi et al. [26] reported that alga with high biomass productivity may manifest in a relative low lipid productivity and vice versa.
3.2. Fatty Acid Compositional Profiles
The fatty acids composition of D. salina was evaluated at early stationary growth phase. Table 2 revealed that the percentage of unsaturated FAMEs (65.24 %) is remarkable compared to that of saturated FAMEs (34.76%). The amount of unsaturated fractions is between 4.85% and 15.24%, while that of saturated is between 1.67% and
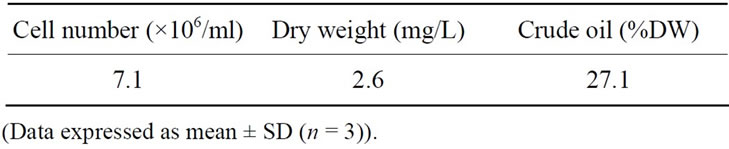
Table 1. Cell density and oil content of Dunaliella salina at early stationary phase.
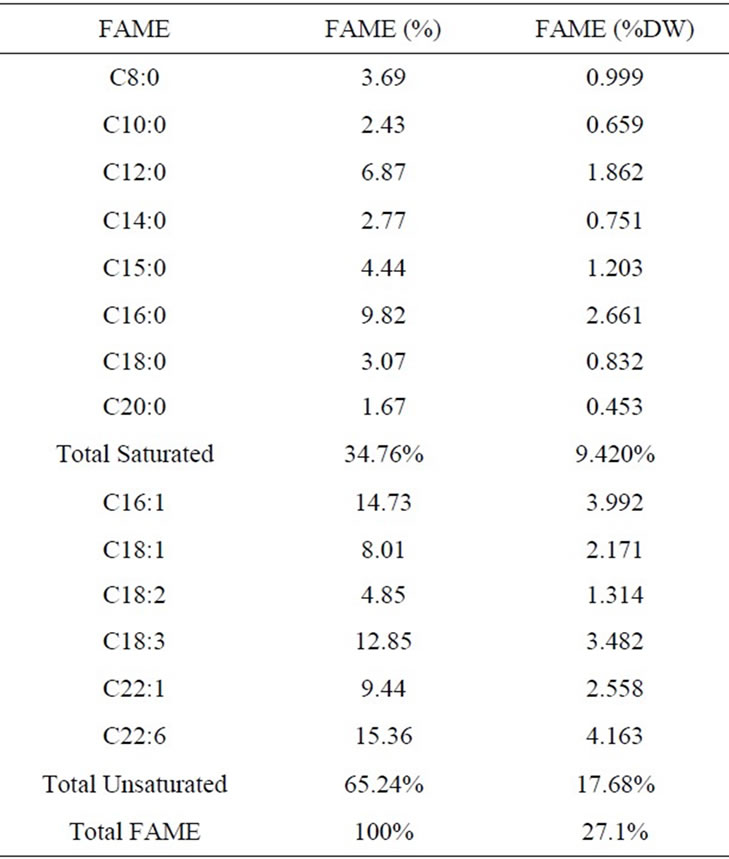
Table 2. The fatty acid profile of Dunaliella salina.
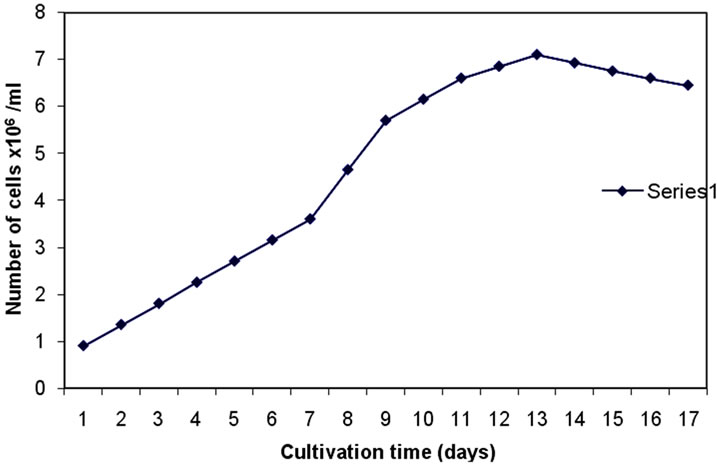
Figure 1. Number of cells of Dunaliella salina over a 16-day cultivation.
9.82%. Normally, the fatty acid compositional profiles of algal strains are influenced by specific growth conditions such as nutrient levels, temperatures, and light intensities [27]. This makes it more difficult to define a single compositional profile for algal-based biodiesel [28]. As well, clear differences in carbon chain length and degree of unsaturation are important algal oil characteristics for biodiesel production and may influence its properties and performance [29].
3.3. Physical Properties of Algal Biodiesel
Table 2 summarized that for D. salina the chain length of fatty acids comprising biodiesel were between C8 and C22. The frequently detected ones were Docosahexaenoic, Palmitic, Palmitoleic and Linolenic acid. Table 3 listed the properties of biodiesel fuel described by its kinematic viscosity, cetane number, oil stability and oil density. These properties are affirmed by the type and concentration of the resultant fatty acid esters [11,30,31]. When reviewing these properties, it is useful to consider the standard specifications that have been established by various fuel standard-setting organizations particularly ASTM (American Society for Testing and Materials) and CEN (European Committee for Standardization).
Viscosity is a significant fuel property with respect to in-use performance of biodiesel since it influenced the operation of the fuel injection equipment. Viscosity increases with increasing fatty acid chain length and degree of saturation. The increase in viscosity affects the fluidity of the fuel producing several failures in the engines besides it may lead to less accurate operation of the fuel injectors [28,32]. To solve this drawback, a chemical reaction to reduce the viscosity of the oil (transesterification) was designed. This process transforms the triglycerides (TG) contained in the oil into a mixture of alkylesters (biodiesel); being the latest the more feasible alternative to diesel fuels [33]. The values of viscosity in this study are in the range of 2.4 - 5.8 mm2/s, which are into the minimum (1.9 mm2/s) and maximum (6.0 mm2/s) range required by the ASTM [34].
The cetane number is a commonly used indicator for the determination of diesel fuel quality, especially the ignition quality. It measures the readiness of the fuel to auto-ignite when injected into the engine [35]. High cetane numbers guarantee good cold start behavior and a smooth run of the engine. In contrast, fuels with low cetane numbers tend to increase gaseous and particulate exhaust emissions because of incomplete combustion [36]. The ideal mixture of fatty acids has been suggested to be C16:1, C18:1 and C14:0 in the ratio 5:4:1. Such a biodiesel would have the properties of very low oxidative potential [37,38]. In the current investigation, the ratio was about 5:3:1. In view of that, the C18:1 acid concentration of D. salina has to be improved by controlling culture conditions.
The twice amount of unsaturated to saturated FAMEs shown in Table 2 are helpful in determining the inbuilt oxidative stability of the biodiesel fuels produced from the tested alga. In this study, biodiesel is rich in unsaturated fatty acid methyl esters (C16:1, C18:1, C18:2, C18:3, C22:1 and C22:6) having over 32.2% polyunsaturated FAMEs; thus, it would be expected to have reduced oxidative stability. In general, oxidative stability is influenced by unsaturation, since higher unsaturation leads to poorer stability [28,39].
Density is an important biodiesel parameter, with impact on fuel quality, because injection systems, pumps,
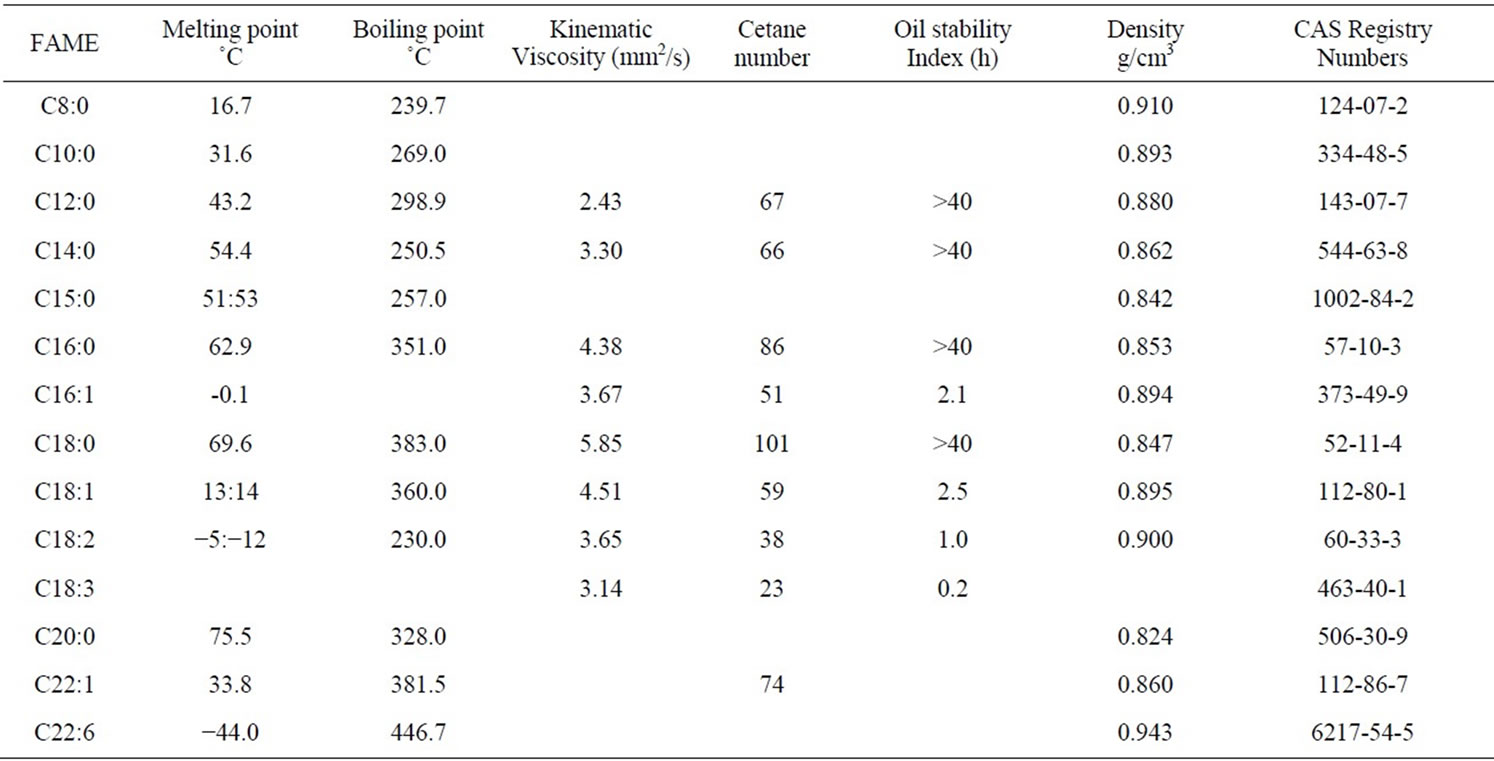
Table 3. Biodiesel properties of Dunaliella salina [37,42,43].
and injectors must deliver an amount of fuel precisely adjusted to provide proper combustion [40]. Variation in the density of FAMEs of the tested D. salina was recorded in Table 3. The present study assumed that the density depends upon the raw materials used for biodiesel fuel production and the biodiesel methyl ester profile. Gouw and Vlugter [41] recommended that the density of fatty acids is inversely proportional to their number of carbon atoms. Ramírez-Verduzco et al. [37] proposed that an increase in the number of carbon atoms may causes a decrease in the density, and to some extent the decrease is insignificant when the number of carbon atoms is larger. Also, the authors suggested that the density can be estimated from parameters related to the chemical structure of FAMEs in particularly molecular weight and the number of double bonds.
4. Conclusion
Based on the profile of fatty acid composition, D. salina was evaluated as oil producer. The alga was cultivated batch-wise under optimum laboratory conditions. Cell density of 7.1 × 106 cells/ml culture, equivalent to a dry weight of 2.6 mg/L produces 27.1% oil. Fatty acid profiles were identified as C8:0, C10:0, C12:0, C14:0, C15:0, C16:0, C18:0, C20:0, C16:1, C18:1, C18:2, C18:3, C22:1 and C22:6. The ratio of unsaturated to saturated fatty acids was approximately 2:1. The properties of biodiesel fuel described by its kinematic viscosity, cetane number, oil stability and oil density were discussed. So, it is assumed that D. salina is suitable for biodiesel production. However, manipulating culture conditions and exploring the concept of genetic engineering before energy products from this microalga can enter the market were recommended.
5. Acknowledgements
The authors would like to thank Professor Nagwa Gamal-ElDin Mohammady, Department of Botany and Microbiology, Faculty of Science, Alexandria University for her great support and valuable discussion.
REFERENCES
- M. Gavrilescu and Y. Chisti, “Biotechnology—A Sustainable Alternative for Chemical Industry,” Biotechnology Advances, Vol. 23, No. 7-8, 2005, pp. 471-499. doi:10.1016/j.biotechadv.2005.03.004
- P. Spolaore, C. Joannis-Cassan, E. Duran and A. Isambert, “Commercial Applications of Microalgae,” Journal of Bioscience and Bioengineering, Vol. 101, No. 2, 2006, pp. 87-96. doi:10.1263/jbb.101.87
- A. S. Fedorov, S. Kosourov, M. L. Ghirardi and M. Seibert, “Continuous H2 Photoproduction by Chlamydomonas Reinhardtii Using a Novel Two-Stage, Sulfate-Limited Chemostat System,” Applied Biochemistry and Biotechnology, Vol. 121, No. 1-3, 2005, pp. 403-412. doi:10.1385/ABAB:121:1-3:0403
- I. K. Kapdan and F. Kargi, “Bio-Hydrogen Production from Waste Materials,” Enzyme and Microbial Technology, Vol. 38, No. 5, 2006, pp. 569-582. doi:10.1016/j.enzmictec.2005.09.015
- A. Banerjee, R. Sharma, Y. Chisti and U. C. Banerjee, “Botryococcus Braunii: A Renewable Source of Hydrocarbons and Other Chemicals,” Critical Reviews in Biotechnology, Vol. 22, No. 3, 2002, pp. 245-279. doi:10.1080/07388550290789513
- G. Huang, F. Cheng, D. Wei, X. Zhang and G. Chen, “Biodiesel Production by Microalgal Technology,” Applied Energy, Vol. 87, No. 1, 2010, pp. 38-46. doi:10.1016/j.apenergy.2009.06.016
- L. Brennan and P. Owende, “Biofuels from Microalgae— A Review of Technologies of Production, Processing and Extraction of Biofuels and Co-Products,” Renewable and Sustainable Energy Reviews, Vol. 14, No. 2, 2010, pp. 557-577. doi:10.1016/j.rser.2009.10.009
- M. M. Phukan, S. R. Chutia, B. K. Konwar and R. Kataki, “Microalgae Chlorella as a Potential Bio-Energy Feedstock,” Applied Energy, Vol. 88, No. 10, 2011, pp. 3307- 3312. doi:10.1016/j.apenergy.2010.11.026
- A. Demirbas, “Progress and Recent Trends in Biodiesel Fuels,” Energy Conversion and Management, Vol. 50, No. 1, 2009, pp. 14-34. doi:10.1016/j.enconman.2008.09.001
- K. Bozbas, “Biodiesel as an Alternative Motor Fuel: Production and Policies in the European Union,” Renewable and Sustainable Energy Reviews, Vol. 12, No. 2, 2008, pp. 542-552. doi:10.1016/j.rser.2005.06.001
- G. Knothe, J. Van Gerpen and J. Krahl, “The Biodiesel Handbook,” AOCS Press, Champaign, 2005, p. 302. doi:10.1201/9781439822357
- J. M. Marchetti, V. U. Miguel and A. E. Errazu, “Possible Methods for Biodiesel Production,” Renewable and Sustainable Energy Reviews, Vol. 11, No. 6, 2007, pp. 1300- 1311. doi:10.1016/j.rser.2005.08.006
- Y. C. Sharma and B. Singh, “Development of Biodiesel: Current Scenario,” Renewable and Sustainable Energy Reviews, Vol. 13, No. 6-7, 2009, pp. 1646-1651. doi:10.1016/j.rser.2008.08.009
- D. H. Lee, “Algal Biodiesel Economy and Competition among Bio-Fuels,” Bioresource Technology, Vol. 102, No. 1, 2011, pp. 43-49. doi:10.1016/j.biortech.2010.06.034
- I. A. Guschina and J. L. Harwood, “Lipids and Lipid Metabolism in Eukaryotic Algae,” Progress in Lipid Research, Vol. 45, No. 2, 2006, pp. 160-186. doi:10.1016/j.plipres.2006.01.001
- N. G. Mohammady, “Characterization of the Fatty Acid composition of Nannochloropsis Salina as a Determinant of Biodiesel Properties,” Zeitschrift fur Naturforshung, Vol. 66c, No. 7/8, 2011, pp. 328-332. doi:10.5560/ZNC.2011.66c0328
- J. Sheehan, T. Dunahay, J. Benemann and P. Roessler, “A Look Back at the US Department of Energy’s Aquatic Species Program: Biodiesel from Algae,” NREL/TP- 580-24190, National Renewable Energy Laboratory, 1998. doi:10.2172/15003040
- Y. Chisti, “Biodiesel from Microalgae,” Biotechnology Advances, Vol. 25, No. 3, 2007, pp. 294-306. doi:10.1016/j.biotechadv.2007.02.001
- Y. Li, B. Wang, N. Wu and C. Q. Lan, “Effects of Nitrogen Sources on Cell Growth and Lipid Production of Neochloris oleoabundans,” Applied Microbiology and Biotechnology, Vol. 81, No. 4, 2008, pp. 629-636. doi:10.1007/s00253-008-1681-1
- Y. Li, M. Horsman, N. Wu, C. Q. Lan and N. DuboisCalero, “Biofuels from Microalgae,” Biotechnology Progress, Vol. 24, No. 4, 2008, pp. 815-820.
- J. Qin, “Bio-Hydrocarbons from Algae: Impacts of Temperature, Light and Salinity on Algae Growth,” Rural Industries Research and Development Corporation Report, Adelaide SA Australia: Barton ACT: RIRDC, 2005.
- H. Tang, N. Abunasser, M. E. D. Garcia, M. Chen, K. Y. Simon Ng and S. O. Salley, “Potential of Microalgae Oil from Dunaliella tertiolecta as a Feedstock for Biodiesel,” Applied Energy, Vol. 88, No. 10, 2011, pp. 3324-3330. doi:10.1016/j.apenergy.2010.09.013
- L. A. Loeblich, “Photosynthesis and Pigments Influenced by Light Intensity and Salinity in the Holophilic Dunaliella salina (Chlorophyta),” Journal of the Marine Biological Association of the United Kingdom, Vol. 62, No. 3, 1982, pp. 493-508. doi:10.1017/S0025315400019706
- E. G. Bligh and W. J. Dyer, “A Rapid Method of Total Lipid Extraction and Purification,” Canadian Journal of Biochemistry and Physiology, Vol. 37, No. 8, 1959, pp. 911-917. doi:10.1139/o59-099
- W. W. Christie, “Gas Chromatography Mass Spectrometry Methods for Structural Analysis of Fatty Acids,” Lipids, Vol. 33, No. 4, 1998, pp. 343-351. doi:10.1007/s11745-998-0214-x
- L. Rodolfi, G. C. Zittelli, N. Bassi, G. Padovani, N. Biondi and G. Bonini, “Microalgae for Oil: Strain Selection, Induction of Lipid Synthesis and Outdoor Mass Cultivation in a Low-Cost Photobioreactor,” Biotechnology and Bioengineering, Vol. 102, No. 1, 2009, pp. 100-112. doi:10.1002/bit.22033
- Q. Hu, M. Sommerfeld, E. Jarvis, M. Ghirardi, M. Posewitz and M. Seibert, “Microalgal Triacylglycerols as Feedstocks for Biofuel Production: Perspectives and Advances,” The Plant Journal, Vol. 54, No. 4, 2008, pp. 621- 639. doi:10.1111/j.1365-313X.2008.03492.x
- S. K. Hoekman, A. Broch, C. Bins, E. Ceniceros and M. Natarajan, “Review of Biodiesel Composition, Properties, and Specifications,” Renewable and Sustainable Energy Reviews, Vol. 16, No. 1, 2012, pp. 143-169. doi:10.1016/j.rser.2011.07.143
- M. J. Griffiths and S. T. L. Harrison, “Lipid Productivity as a Key Characteristic for Choosing Algal Species for Biodiesel Production,” Journal of Applied Phycology, Vol. 21, No. 5, 2009, pp. 493-507. doi:10.1007/s10811-008-9392-7
- G. Knothe, “Designer Biodiesel: Optimizing Fatty Ester Composition to Improve Fuel Properties,” Energy and Fuels, Vol. 22, No. 2, 2008, pp. 1358-1364. doi:10.1021/ef700639e
- C. J. Chuck, C. D. Bannister, J. G. Hawley, M. G. Davidson, I. La Bruna and A. Paine, “Predictive Model to Assess the Molecular Structure of Biodiesel Fuel,” Energy and Fuels, Vol. 23, No. 4, 2009, pp. 2290-2294. doi:10.1021/ef801085s
- M. Balat, “Modeling Vegetable Oil Viscosity,” Energy Source Part A, Vol. 30, No. 20, 2008, pp. 1856-1869. doi:10.1080/15567030701457392
- A. Tabernero, E. M. Martín del Valle and M. A. Galán, “Evaluating the Industrial Potential of Biodiesel from a Microalgae Heterotrophic Culture: Scale-Up and Economics,” Biochemical Engineering Journal, Vol. 63, 2011, pp. 104-115. doi:10.1016/j.bej.2011.11.006
- T. Gouw, J. Vlugter and C. Roelands, “Physical Properties of Fatty Acid Methyl Esters: VI. Viscosity,” Journal of the American Chemists Society, Vol. 43, No. 7, 1966, pp. 433-434.
- M. Balat and H. Balat, “Progress in Biodiesel Processing,” Applied Energy, Vol. 87, No. 6, 2010, pp. 1815- 1835. doi:10.1016/j.apenergy.2010.01.012
- L. F. Ramírez-Verduzco, J. E. Rodríguez-Rodríguez and A. R. Jaramillo-Jacob, “Predicting Cetane Number, Kinematic Viscosity, Density and Higher Heating Value of Biodiesel from Its Fatty Acid Methyl Ester Composition,” Fuel, Vol. 91, No. 1, 2012, pp. 102-111. doi:10.1016/j.fuel.2011.06.070
- G. Knothe, “Dependence of Biodiesel Fuel Properties on the Structure of Fatty Acid Alkyl Esters,” Fuel Processing Technology, Vol. 86, No. 10, 2005, pp. 1059-1070. doi:10.1016/j.fuproc.2004.11.002
- P. M. Schenk, S. R. Thomas-Hall, E. Stephens, U. C. Marx, J. H. Mussgnug, C. Posten, O. Kruse and B. Hankamer, “Second Generation Biofuels: High-Efficiency for Biodiesel Production,” Bioenergy Research, Vol. 1, No. 1, 2008, pp. 20-43. doi:10.1007/s12155-008-9008-8
- M. J. Ramos, C. M. Fernandez, A. Casas, L. Rodriguez and A. Perez, “Influence of Fatty Acid Composition of Raw Materials on Biodiesel Properties,” Bioresource Technology, Vol. 100, No. 1, 2009, pp. 261-268. doi:10.1016/j.biortech.2008.06.039
- M. Jorge Pratas, S. V. D. Freitas, M. B. Oliveira, S. C. Monteiro, A. S. Lima and J. A. P. Coutinho, “Biodiesel Density: Experimental Measurements and Prediction Models,” Energy and Fuels, Vol. 25, No. 5, 2011, pp. 2333- 2340. doi:10.1021/ef2002124
- T. H. Gouw and J. C. Vlugter, “Physical Properties of Fatty Acid Methyl Esters. I. Density and Molar Volume,” Journal of the American Chemists Society, Vol. 41, No. 2, 1964, pp. 142-145.
- B. R. Moser and S. F. Vaughn, “Efficacy of Fatty Acid Profile as a Tool for Screening Feedstocks for Biodiesel Production,” Biomass and Bioenergy, Vol. 37, 2012, pp. 31-41. doi:10.1016/j.biombioe.2011.12.038
- B. R. Moser, “Biodiesel Production, Properties and Feedstocks. In Vitro Cellular and Developmental Biology,” Plants, Vol. 45, No. 3, 2009, pp. 229-266.
NOTES
*Corresponding author.

