Chinese Medicine
Vol.5 No.2(2014), Article
ID:47016,9
pages
DOI:10.4236/cm.2014.52012
Design, Development and Phytochemical Evaluation of a Poly Herbal Formulation Linkus Syrup
Zeeshan Ahmed Sheikh1, Aqib Zahoor1*, Saleha Suleman Khan1, Khan Usmanghani1,2
1Department of Research and Development, Herbion Pakistan (Pvt.) Limited, 30/28 Korangi Industrial Area, Karachi, Pakistan
2Faculty of Pharmacy, Jinnah University for Women, Karachi, Pakistan
Email: *aqib.zahoor@herbion.com
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 23 April 2014; revised 26 May 2014; accepted 6 June 2014
ABSTRACT
The present work has been carried out on polyherbal formulation named as Linkus Syrup. The herbal formulation consists of Glycyrrhiza glabra, Hyssopus officinalis, Piper longum and Alpinia galangal. The Linkus syrup physico-chemical evaluation such as pH, density, identification of polysaccharide, tanning agents, ascorbic acid and shelf life was complied. The TLC and quantitative determination of alkaloid were quantified. Determination of biomarker has been validated for the analysis of vasicine. The study result revealed that Linkus syrup formulation was well standardized at different levels such as physic-chemical consistency and assay of bio marker compound.
Keywords:Syrup, Crude Herb, Anti-Tussive, Anti-Bronchitis, Anti-Pharyngitis, Sore Throat, Tracheitis, HPLC

1. Introduction
Cough is one of the most common symptoms for which patients seek medical attention from primary care physicians and pulmonologists [1] . These studies were carried out for the design, development and phytochemical evaluation of a poly herbal formulation named as Linkus syrup. The syrup consists of leaves of Adhatoda vasica used in the treatment of coughs, bronchitis, asthma, common cold, expectorant and antispasmodic (bronchodilator). The active components are vasicine (aka peganine) and vasicinone [2] . The root and rhizome of Glycyrrhiza glabra contain glycyrrhizin that inhibit growth and cytopathology of cytomegalovirus and are utilized in cough, catarrhal infections, asthma, bronchitis, coughs, arthritis and allergic complaints [3] -[5] . Hyssopus officinalis contains pinocamphone, camphone, pinocampheol, cineole used as an expectorant, bronchitis, respiratory infections, upper respiratory tract infections, coughs [3] [6] -[8] . The fruits and roots of Piper longum are attributed for diseases of respiratory tract, such as cough, bronchitis, asthma [9] and contain piperine, piperlongumine, piperlonguminine, methyl 3,4,5-trimethoxycinnamate [9] . Alpinia galanga has a bronchodilator and anti-inflammatory effect on bronchioles [10] [11] and the main components isolated from its rhizomes were galangoisoflavonoid, methyleugenol, galangin, acetoxychavicol acetate (ACA) and hydroxychavicol acetate In the standard German text, Hager’s Handbuch der Pharmazeutischen Praxis, Zingiber officinale has been listed as being used against nervousness, chronic inflammation of the intestine, coughing, conditions of the urinary tract and lower abdomen, rheumatism, and a sore throat [12] and the chemical constituents are zingerone, shogaols and gingerols, zingiberene, bisboline and cineol. The leaves and flower of Althea officinalis have demulcent, expectorant, diuretic, and emollient properties and generally used to treat ailments of the lungs and the urinary system [13] . The active constituents are Pectin, disaccharide sacharose, mucilage, Hypolaetin-8-glucoside, isoquercitrin, kaempferol, caffeic, p-coumaric acid, scopoletin, asparagines and many amino acids.
Cordia latifolia [14] is used to treat gastrointestinal, respiratory and dermatological disorders and the constituents cited were cordinoic acid, cordicilin, cordinol and cordicinol [15] . Zizyphus jujuba used for colds and coughs; they also have antifungal, antibacterial, antiulcer, anti-inflammatory, sedative and antiseptic properties [16] [17] and it possesses mauritine-A; amphibine-H, jubanine-A, jubanine-B; mucronine-D, and nummularine-B, sativanine-E, frangufoline, ziziphines jujubosides [17] . Onosma bracteatum leaves and stems are used to treat bronchitis and asthma [18] and main phytochemicals present are naphthazarins, naphthoquinones, alkannins and Shikonins [19] .
2. Material and Method
Herbs were bought through Supply Chain Department of the Herbion Pakistan (Pvt.) Limited, Karachi from the local market. All samples were stored in dark at 23˚C. The herbs utilized in Linkus syrup were morphologically compared with the authentic specimen available at QC department of Herbion Pakistan Pvt. Ltd., they details are as follows, Adhatoda vasica reference No. B18, Glycyrrhiza glabra reference No.M4, Piper longum reference No. F1, Viola odorata reference No. B2, Hyssopus officinalis reference No.Z1, Alpinia galangal reference No. A2, Cordia latifolia reference No. S9, Althea officinalis reference No. K3, Zizyphus vulgaris reference No.U3, Onosma bracteatum reference No. B1. These herbs have also been authenticated and verified by Prof. Dr. Iqbal Azhar, Department of Pharmacognosy, Faculty of Pharmacy, University of Karachi. and chemically for biomarker of some herbs. All the other chemicals and reagents were of pure analytical grade and obtained from local supplier.
2.1. Development of Linkus Syrup
With extensive literature review and traditional knowledge research and development department of Herbion Pakistan Pvt. Limited was successfully developed the formulation for cough and cold which named as Linkus Syrup.
2.1.1. Selection and Evaluation of Herbs
Table 1 represents the quantity and pharmacological action of herbs used in this preparation.
All herbs were tested for their prescribed part, macro and microscopic descriptions, identification, chemical assays, foreign matter, total ash, Acid insoluble ash, alcohol and water soluble part of extract. These properties with specifications were listed in Table 2.
2.1.2. Preparation of Extracts
Each herb was extracted individually in vacuum jacketed extractor with water five times to the amount of herb taken. Stir for 10 minutes and then start heating. Heat to boil. The temperature should be from 100˚C - 110˚C. Then slow down the temperature to 80˚C - 90˚C. Amount of the herb, extraction time and percent yield are mentioned in the Table 3.
2.1.3. Preparation of Linkus Syrup
625 liter deionized water was took in manufacturing tank. Heated and boil for 40 to 45 minutes. When boiling
Table 1. The herbs used, quantity and pharmacological actions.
*amount of herb used in the preparation of 10 ml syrup.
Table 2. Specification and analytical method of raw material used in Linkus syrup by quality operation.
Table 3. Extraction of Linkus syrup herbs.
was started, add 1750 kg sugar slowly with constant stirring. Continue heating with constant stirring till it became homogenize syrup. Separately dissolved Methyl Paraben, Propyl Paraben, Citric acid and Glycerin in 83 liter deionized Water, mixed, heat and boiled thoroughly. Mix this solution to the homogenized syrup and stirred for 20 minutes. Took 416 Liters of Purified water in jacketed kettle and add to it 73.75 kg combine extract. Heat till it was fully dissolved and then filter the solution. add the filtered solution to homogenized syrup prepared previously, stirred and mix for 20 min. After mixing, allow the syrup to cool down by chilled water circulation. Circulation was continued until the temperature of the syrup reached to “room temperature”. Mixed the flavors like Peppermint Oil, Clove Oil and Propylene glycol, then made up the volume of syrup to 1250 liters with deionized water. Afterward allow final mixing by stirring for half an hour. This process represented here in Table 4.
3. Phytochemical Evaluations
Brown color syrup was prepared with characteristic odor, having pH in between 3.0 - 6.0, and density 1.25 - 1.35 g/cm3. Identification of polysaccharides, tanning agent, and ascorbic acid were performed by test tubes method. While the identification of vasicine was done by TLC.
3.1. Physical Evaluation
3.1.1. Appearance
Brown color viscosity syrup with characteristic odor, Determination was conducted visually.
3.1.2. рН Determination
Dissolved 10 ml of syrup in 100 ml of water. Filtered and determine pH potentiometrically Standard: 3.0 - 6.0
3.1.3. Density
Determination was conducted by SP XII method 3 and it was according to Standard: 1.25 to 1.35 g/cm3.
3.1.4. Qualitative Identification Reactions
Placed 5 ml of the preparation to a flat-bottomed 50 ml flask. Added 20 ml of 96% spirit and mixed; precipitated suspension was formed (polysaccharides). Left the obtained solution for 1 hour for layer separation and filter carefully through the folded paper filter (Solution А).
Added 3 drops of Ferric chloride solution to 3 ml of the solution A. After shaking greenish-yellow coloring appears (tanning agents). To 3 ml of A solution added 3 drops 0.1% spirit solution of sodium 2.6-dichlorfenolindofenolite solution had a reddish-violet coloring at mixing (ascorbic acid).
Table 4. Manufacturing of Linkus syrup.
Preparation of 0.1% Alcohol Solution of Sodium 2, 6-Dichlorphenolindophenolat Placed 0.1 g of sodium 2, 6-dichlorphenolindophenolyat in 100 ml of measuring retort and dissolve it in 30 ml of 96% ethyl alcohol and then made up the volume with 96% ethyl alcohol. Shelf life of solution was 1 month
3.1.5. Qualitative Identification by TLC
Applied 0.05 ml of solution I to chromatographic plate “Silica gel 60 F254” (size 10 × 20 cm manufactured by Merck) in the form of spot. Dry the plate in hot air for 5 minutes and placed it in the chamber preliminary saturated for 1 hour with a mixture of the following solvent: 96% alcohol : chloroform : hydrous ammonia (30:19:1).
When the front of solvents was reached the plate edge, took it out from the chamber and dried in a drying oven for З minutes at 100˚C. Then apply Dragendorff reagent to the plate. 1 reddish pink color spot (vasicine) was shown on the plate in UV light at wave length 254 nm Rf (0.4).
The physicochemical and organoleptic parameters so generated for standardization were further determined by HPLC and UV spectrophotometers as mentioned by using colorimeter.
3.2. Quantitative Determination of Total Alkaloids
About 12 g (exact weight, with accuracy to 0.0001 g) (about 10 ml) of preparation was placed in a 100 ml dividing funnel. Added 1 ml of hydrous ammonia, mixed then adds 20 ml of water and mixed again. Added 25 ml of chloroform to the obtained solution in the funnel. Shake carefully for 3 minutes. After full division of layers. Filtered lower chloroform layer through the paper filter with anhydrous sodium sulfate (about 10 g) in 500 ml round-bottomed flask.
Repeated extraction procedure 4 times more using 25 ml portions (5 times in total). Collected chloroform layers in 500 ml round bottom flasks... Combined chloroform extraction steamed to dryness on a water bath under vacuum. The dry residue was dissolved in 3 ml of 96% alcohol (Solution 1).
Transferred 1 ml of solution 1 to 50 ml volumetric flask. Added 1 ml of hydrous ammonia and made up the mixture volume with 96% alcohol to the mark. To 2 ml of the obtained solution add 10 ml of 96% alcohol and mixed.
Absorbance was measured on 302 nm by using 96% spirit as blank solution.
Total alkaloids were calculated as vasicine in percentage (X) through below formula:
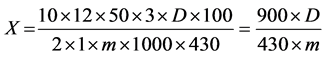
where:
D = Optical density of the tested solution;
m = weight of preparation in grams;
430 = Specific parameter of vasicine absorption.
3.3. Determination of Vasicine by HPLC
3.3.1. Sample Preparation
About 12 g (exact weight) of preparation placed in a 100 ml dividing funnel. Added directly 20 ml of water, and 3 ml of hydrous ammonia, then carefully mixed the contents. The organics were extracted with 50 ml of chloroform (3 times in total). Combined chloroform extraction evaporated to dryness under vacuum. Dissolved the dry residue in 20 ml of mobile phase and transferred quantitatively into a 25 ml volumetric flask. The solution makeup to the mark with mobile phase. Filtered the obtained solution through a filter with pore size 0.45 µm and used for chromatography.
3.3.2. Vasicine Standard Solution Preparation
Placed about 50 mg (exact weight) of RSO vasicine standard solution in a 50 ml volumetric flask and dissolved in mobile phase.
The volume of the solution was brought up to the mark with mobile phase. Placed 1 ml of the obtained solution in a 25 ml volumetric flask and diluted with mobile phase to the mark. Filtered the obtained solution if necessary. Used fresh solution.
Chemical analysis HPLC was performed using Agilent 1260 HPLC system (Agilent Technologies, Santa Clara, CA). Chromatographic separation and detection of samples were performed.
Chromatography conditions—liquid chromatograph with UV detector.
Column—chromatography column µ BondapakTM C18 10 µm 125˚A 3.9 × 300 mm or equivalent.
Mobile phase—degassed mixture consisting of acetonitrile: 0.1 M phosphate buffer: glacial acetic acid (15: 85: 1 v/v).
Flow rate—0.7 ml/min.
Wave length—300 nm.
Column temperature—room temperature.
Vasicine content in Linkus Syrup should not be less than 0.3 mg per 10 ml was calculated by the following formula:

f—0.838 conversion factor of vasicine Hydrochloride to vasicine ASMP—Mean value of peak area of tested solution samples ASTD—Mean value of peak area of standard solution samples WSMP—Preparation weight, mg WSTD—Standard weight, mg P—Percent purity of standard sample, % D—Density of syrup
3.3.3. Preparation of Phosphate Buffer
5.68 gm of sodium di-hydrogen phosphate and 3.63 gm potassium di-hydrogen phosphate transferred to 1000 ml volumetric flask, added 100 ml of water and mixed. Brought the solution volume to the mark with water [20] . Other parameters and norms of developed Linkus syrup were summarized in Table 5.
After evaluating the above parameters the preparation of Linkus syrup was established.
4. Results and Discussion
Standardization of biologically active material in the poly herbal formulation and quantitative analysis cover identification of chemical components where as quantitative assay measure the identification and level of bio
Table 5. Different physico-chemical parameters.
marker in the extract establish the standard of that particular compound for validation. Linkus is a poly herbal formulation manufactured by Herbion Pakistan (Pvt.) Limited. In order for the standardization of the procedure of the finished product and there in the five different herbal drugs of different batches were analyzed for physico-chemical parameters. The drugs in the Linkus are Glycyrrhiza glabra, Hyssopus officinalis, Piper longum and Alpinia galangal. The formulation organoleptic characteristics such as identification by TLC, assay of alkaloid content, foreign matter, total ash, acid insoluble ash, alcohol; soluble extraction and water soluble extraction are adequately presented in Table 2. Thereafter the extraction of different herbal drugs is given in Table 3, giving details of quantity used, quantity of thick extract, extraction time taken and percentage yield of the extract. Preparation of manufacturing of the syrup is depicted in Table 3. Physico-chemical evaluation such as appearance, pH determination plays a significant role in the quality assessment. The identification with quantitative reaction and preparation of 0.1% alcohol solution exhibit the indication polarity concern of plant materials. TLC was done to augment the determination of alkaloids components and there in the identification of vascine was located. All these values show that the physic-chemical parameters adopted the values were within the limits and repeated experiments confirmed the verification. HPLC was performed to confirm the quantitative and qualitative presence of vasicine for validation. The present standardization under taken reveal compliance with all the physic-chemical and analytical procedures, therefore it is concluded that Linkus syrup is well standardized product at the base line parameters.
If these analytical profiles compared with the work of Modi et al. where Zeal cough syrup employing physico-chemical test and bioassay marker compounds agree with quality and potency [21] . In another study Desai and co worker validated the, manufacturing procedure of Vasu cough syrup with the poly herbal formulation for compliance for their reproducibility [22] . The work done by Khadair and associates validation analysis of hederacoside C, the marker of Ivy plant and percentage recovery was given as 99.6% [23] . In a patent of cough syrup the extract of Adhatoda vasica along with Hedychium and Curcuma cough syrup has been validated in greater details [24] . Therefore the Linkus syrup standardization fall under the specific guide lines of quality herbal medicine and in line with different as mentioned earlier follow the prerequisite for global harmonization.
5. Conclusion
In this study for medicinal plants component, Glycyrrhiza glabra, Hyssopus officinalis, Piper longum and Alpinia galangal were formulated for the indication of anti-tussive activity. The physico chemical and organoleptic parameter data so generated were recorded for standardization. TLC was carried out on the aqueous extract and Rf value (0.4) of vasicine as raddish pink spot on exposure to Draggendroff and UV was realized. Quantitative and spectrophotometric estimation of alkaloid was found between the range of 0.035% to 0.07%. Determination of vasicine as biomarker by HPLC in linkus syrup determined as 0.3 mg per 10 mL. The analytical profile of linkus syrup and there in vasicine have been validated to proved that linkus syrup in different batches confirmed the verification. Further study on Linkus syrup is being carried out to determine the efficacy as anti-tussive agent and will be reported elsewhere. The following animal model of cough was used to investigate the antitussive potential of Linkus. Healthy Wistar albino rats were treated orally with the recommended doses of Linkus. After 15 minutes, they were individually placed in a closed Plexiglass chamber (20 × 10 × 10 cm) and exposed to citric acid inhalation (0.1 g/ml) for 7 minutes. The cough reflexes were counted for the last 5 minutes and compared with those of the control animals. The results obtained showed good responses.
References
- Irwin, R.S., Boulet, L.-P., Cloutier, M.M., et al. (1998) Managing Cough as a Defense Mechanism and as a Symptom: A Consensus Panel Report of the American College of Chest Physicians. Chest, 114, 133S-181S. http://dx.doi.org/10.1378/chest.114.2_Supplement.133S
- Silva, T.R., Pongravoon, U. and Malmfors, T. (2000) Adhatoda vasica: A Critical Review of Ethanopharmacological and Toxicological Data. Journal of Ethnopharmacology, 72, 1-20. http://dx.doi.org/10.1016/S0378-8741(00)00225-7
- Bown, D. (1995) Encyclopedia of Herbs and Their Uses. Dorling Kindersley, London.
- Numazaki, K., Umetsu, M., Chiba, S. (1994) Effect of Glycyrrhizin in Children with Liver Dysfunction Associated with Cytomegalovirus Infection. The Tohoku Journal of Experimental Medicine, 172, 147-153. http://dx.doi.org/10.1620/tjem.172.147
- Tsukiyama, R. (2002) Antibacterial Activity of Licochalcone A against Spore-Forming Bacteria. Antimicrobial Agents and Chemotherapy, 46, 1226-1230. http://dx.doi.org/10.1128/AAC.46.5.1226-1230.2002
- Chevalier, A. (1996) The Encyclopedia of Medicinal Plants Dorling Kindersley, London.
- Grieve, A. (1984) Modern Herbal. Penguin.
- Leung, A.Y. and Foster S. (1996) Encyclopedia of Natural Ingredients. 2nd Edition, John Wiley & Sons, New York.
- CSIR (1989) The Wealth of India: A Dictionary o f Indian Raw Materials and Industrial Products. Council of Scientific and Industrial Research, New Delhi, 8, 96-99.
- Kasirajan, B., Maruthamuthu, R., Gopalakrishnan, V., Arumugam, K., Asirvatham, H., Murali, V., et al. (2007) A Database for Medicinal Plant Use in Treatment of Asthma. Bioinformation, 2, 105-106. http://dx.doi.org/10.6026/97320630002105
- Khare, C.P. (2007) Indian Medicinal Plants—An Illustrated Dictionary. 1st Edition, Springer Science, New York, 9-15.
- Blaschek, W., Hager, H., Hansel, R., Wolf, H.U., Surmann, P., Nyrnberg, E., et al. (1990) Hager’s Handbuch der Pharmazeutischen Praxis. 5th rev. ed., Springer Verlag, Berlin.
- Petkewich, R. and Peter. R. (2006) What’s That Stuff Marshmallow. Chemical & Engineering News, 84, 41. http://dx.doi.org/10.1021/cen-v084n016.p041
- Hernandez, T., Canales, M., Teran, B., Avila, O., Duran, A., Garcia, A.M., Hernandez, H., Angeles-Lopez, O., Fernandez Ariza, M. and Avila, G. (2007) Antimicrobial Activity of the Essential Oil Extracts of Cordia curassavica. Journal of Ethnopharmacology, 111, 137-141. http://dx.doi.org/10.1016/j.jep.2006.11.002
- Sabira, B., Perwaiz, S., Bina, S.S., Khan, S., Fayyaz, S. and Ramzan, M. (2011) Chemical constituents of Cordia latifolia and Their Nematicidal Activity. Chemistry & Biodiversity, 8, 850-861. http://dx.doi.org/10.1002/cbdv.201000058
- Jiang, J.G., Huang, X.J., Chen, J. and Lin, Q.S. (2007) Comparison of the Sedative and Hypnotic Effects of Flavonoids, Saponins, and Polysaccharides Extracted from Semen Zizyphus jujuba. Natural Product Research, 21, 310-320. http://dx.doi.org/10.1080/14786410701192827
- Mahajan, R.T. and Chopda, M.Z. (2009) Phytopharmacology of Zizyphus jujuba Mill—A Plant Review. Pharmacognosy Reviews, 3, 320-329.
- Kirtikar, K.R. and Basu, B.D. (1999) Indian Medicinal Plants, Vol. 3. 2nd Edition, International Book Distributors, 1699.
- Kumar, N., Kumar, R. and Kishore, K. (2013) Onosma L.: A Review of Phytochemistry and Ethnopharmacology. Pharmacognosy Reviews, 7, 140-151. http://dx.doi.org/10.4103/0973-7847.120513
- Srivastava, S., Verma, R.K., Gupta, M.M., Singh, S.C. and Kumar, S., (2001) HPLC Determination of Vasicine and Vasicinone in Adhatoda vasica with Photo Diode Array Detection. Journal of Liquid Chromatography & Related Technologies, 24, 153-159. http://dx.doi.org/10.1081/JLC-100001478
- Modi, J., Soni, H., Pandya, K., Patel, G. and Patel, N. (2014) A Detail Phyto-Chemical Evaluation of Herbo-Mineral Formulation Used in Respiratory Diseases. Journal of Pharmacognosy and Phytochemistry, 2, 36-42.
- Desai, L., Oza, J. and Khatri, K. (2012) Prospective Process Validation of Polyherbal Cough Syrup Formulation. Journal of Advanced Pharmaceutical Technology & Research, 2, 225-231.
- Khdair, A., Mohammad, M.K., Tawaha, K., Al-Hamarsheh, E., AlKhatib, H.S., Al-khalidi, B., Bustanji, Y., Najjar, S. and Hudaib, M. (2010) A Validated RP HPLC-PAD Method for the Determination of Hederacoside C in Ivy-Thyme Cough Syrup. International Journal of Analytical Chemistry, 2010, Article ID: 478143, 5p.
- Herbal Formulation Comprising Extracts of Adhatoda, Hedychium and Curcuma as Cough Syrup, WIPO Patent Application WO/2005/077393 Kind Code: A1.
NOTES

*Corresponding author.


