Natural Science
Vol.08 No.06(2016), Article ID:67839,11 pages
10.4236/ns.2016.86033
The Approximate Analytical Solution of Non-Linear Equation for Simultaneous Internal Mass and Heat Diffusion Effects
Mayathevar Renugadevi1, Saminathan Sevukaperumal2, Lakshmanan Rajendran3*
1Department of Mathematics, P. M. T. College, Usilampatti, India
2Department of Mathematics, J. J. College of Arts and Science, Pudukkottai, India
3Department of Mathematics, Sethu Institute of Technology, Kariapatti, India

Copyright © 2016 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 17 May 2016; accepted 26 June 2016; published 29 June 2016
ABSTRACT
For the first time a mathematical modelling of porous catalyst particles subject to both internal mass concentration gradients as well as temperature gradients, in endothermic or exothermic reactions has been reported. This model contains a non-linear mass balance equation which is related to rate expression. This paper presents an approximate analytical method (Modified Adomian decomposition method) to solve the non-linear differential equations for chemical kinetics with diffusion effects. A simple and closed form of expressions pertaining to substrate concentration and utilization factor is presented for all value of diffusion parameters. These analytical results are compared with numerical results and found to be in good agreement.
Keywords:
Chemical and Biological Systems, Modified Adomian Decomposition Method, Nonlinear Reaction Diffusion, Porous Catalyst Particles, Mass and Diffusion Effect

1. Introduction
In many engineering and industrial applications, catalytic processes in chemical reactors are often considered to be very useful. This induces particular attention to the study of catalytic reactions at the single-particle level [1] . Moreover, the reaction behavior of porous catalyst particles had been studied over nearly a quarter of a century [2] - [4] . Majority of chemical reactions are accompanied by heat transfer effects; they either release or absorb heat. This can lead to appreciable increase (or decrease) of temperature toward the particle centre [5] - [7] . Since chemical reaction rates vary rapidly increase with temperature, this effect could radically change the behavior of the catalyst particles. Analysis of chemical kinetics with diffusion effects usually leads to solving strongly nonlinear differential equations. Detailed reviews of mathematical models describing reactions in porous catalyst particle can be found in [8] . Assuming a flat geometry for the particle and that conductive heat transfer is negligible compared to convective heat transfer. The approximate behavior of the functional forms is sufficiently similar for various geometric forms [9] [10] so that the spherical particle is a approximation [11] [12] for most cases encountered, such as cylindrical pellets, or irregular granules. When the chemical reaction is accompanied by a heat effect, not only a mass concentration gradient, but also appreciable temperature gradients can exist within the particle. Weisz and Hicks [13] solved the non-linear mass balance equation using numerical method.
However, to the best of our knowledge, there was no rigorous analytical solution for the concentration of reactant of catalyst having been reported. The purpose of this communication is to derive simple analytical expression for concentration and utilization factor for all possible values of reaction/diffusion parameters using the modified Adomian decomposition method.
2. Mathematical Formulation of the Problem
The dimensionless mass transport equation of porous catalyst particle is [13]
 (1)
(1)
where
 (2)
(2)
 (3)
(3)
where y is the dimensionless concentration, x is the dimensionless radius of the spherical catalyst pellet, c is the dimensionless concentration of reactant, K is thermal conductivity, H is molar heat of reaction. The parameter  expresses the sensitivity of the reaction rate to temperature;
expresses the sensitivity of the reaction rate to temperature;  is the maximum temperature variation
is the maximum temperature variation  which could exist within the particle relative to the boundary temperature. The boundary conditions are
which could exist within the particle relative to the boundary temperature. The boundary conditions are
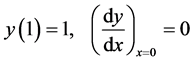 (4)
(4)
The utilization factor 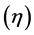 is given by
is given by
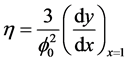 (5)
(5)
3. Analytical Expression of the Concentration Using Modified Adomian Decomposition Method (MADM)
In the recent years, much attention is devoted to the application of the Adomian decomposition method to the solution of various scientific models [14] . The MADM yields, without linearization, perturbation, transformation or discretisation, an analytical solution in terms of a rapidly convergent infinite power series with easily computable terms. The decomposition method is simple and easy to use and produces reliable results with few iterations. The rate of convergence of modified Adomian decomposition method is higher than standard Adomian decomposition method [15] - [17] . Using this method (see Appendix A), we can obtain the analytical expression of concentration (see Appendix B), of the substrate as follows:
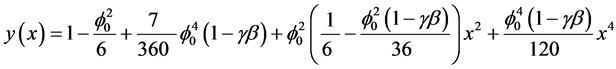 (6)
(6)
Using Equations (5) and (6), we can obtain the effectiveness factor
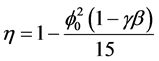 (7)
(7)
The Equation (6) and (7) represent the new and simple analytical expression of concentration of substrate and effectiveness factor provided
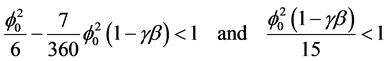 (8)
(8)
4. Numerical Simulation
The diffusion Equation (1) for the boundary condition (Equation (4)) is also solved numerically. We have used the function pdex1 in MATLAB software to solve numerically the initial-boundary value problem for the nonlinear differential equation. This numerical solution is compared with our analytical results in Figure 1 and Figure 2. Upon comparison, it gives a satisfactory agreement for all values of the dimensionless parameters ,
,  and
and . The Matlab program is also given in Appendix C.
. The Matlab program is also given in Appendix C.
5. Discussion
The nonlinear system for coupled heat and mass transfer in a spherical non-isothermal catalyst pellet is solved analytically. The concentration of substrate depends on the following three factors 



Figure 1(a) and Figure 1(b) show the dimensionless concentration of substrate y for various dimensionless pellet raidus x. The concentrations were computed for various values of the dimensionless parameter. From Figure 1(a) and Figure 1(b), it is evident that the value of concentration 







The normalized numerical simulation of three dimensional substrate concentration y versus dimensionless pellet radius x is shown in Figures 2(a)-(c). The time independent concentration y is represented in Figures 2(a)-(c). For fixed value of



Figure 1. Plot of dimensionless concentration y versus dimensionless pellet radius x. The concentrations were computed for various values of the dimensionless parameter 






Figure 2. The normalized dimensionless concentration y versus dimensionless pellet radius x and dimensionless parameters





the concentration of 






The variation in effectiveness factor for various values of 



6. Conclusion
In this work, we have discussed the mathematical model of catalyst particle in a porous medium through which reactants diffuses. We have obtained the approximate analytical expression for the steady state concentration of substrate for all values of 



Figure 3. Plot of the utilization factor 



Figure 4. Plot of the utilization factor 


Acknowledgements
The authors express their gratitude to the reviewers for their valuable comments to improve the quality of the manuscript. This work was supported by the Department of Science and Technology (DST) (No. SB/SI/PC- 50/2012), New Delhi, India. The authors are thankful to the Head of the Department of Mathematics, Principal and Chairman of Sethu Institute of Technology, Kariapatti for their encouragement.
Cite this paper
Mayathevar Renugadevi,Saminathan Sevukaperumal,Lakshmanan Rajendran, (2016) The Approximate Analytical Solution of Non-Linear Equation for Simultaneous Internal Mass and Heat Diffusion Effects. Natural Science,08,284-294. doi: 10.4236/ns.2016.86033
References
- 1. Kierzenka, J. and Shampine, L.F. (2001) A BVP Solver Based on Residual Control and the MATLAB PSE. ACM Transactions on Mathematical Software, 27, 299-316.
- 2. Damkohler, G. (1943) Ubertemperatur in kontaktkornern (Excess Temperature in Catalyst Grains). Zeitschrift f¨ur Physikalische Chemie, 193, 16-28.
- 3. Kim, D.N. and Kim, Y.G. (1981) An Experimental Study of Multiple Steady States in a Porouscatalyst Due to Phase Transition. Journal of Chemical Engineering of Japan, 14, 311-317.
http://dx.doi.org/10.1252/jcej.14.311 - 4. Kim, D.H. and Kim, Y.G. (1981) Simulation of Multiple Steady States in a Porous Catalyst Due to Phase Transition. Journal of Chemical Engineering of Japan, 14, 318-322.
http://dx.doi.org/10.1252/jcej.14.318 - 5. Jaguste, D.N. and Bhatia, S.K. (1991) Partial Internal Wetting of Catalyst Particles: Hysteresis Effects. AIChE Journal, 37, 650-660.
http://dx.doi.org/10.1002/aic.690370503 - 6. Makinde, O.D. (2004) Exothermic Explosions in a Slab: A Case Study of Series Summation Technique. International Communications in Heat and Mass Transfer, 31, 1227-1231.
http://dx.doi.org/10.1016/j.icheatmasstransfer.2004.08.020 - 7. Makinde, O.D. (2005) Strong Exothermic Explosions in a Cylindrical Pipe: A Case Study of Series Summation Technique. Mechanics Research Communications, 32, 191-195.
http://dx.doi.org/10.1016/j.mechrescom.2004.02.008 - 8. Landau, L.D. and Lifshits, Y.M. (1964) Statistical Physics. Nauka, Moscow. (In Russian)
- 9. Weisz, P.B. and Prater, C.D. (1954) Advances in Catalysis. Academic Press, New York, Vol. 6, p. 143.
- 10. Wicke, E. and Brötz, W. (1949), Diffusion, Strömung und Reaktionsgeschwindigkeit im Innern poröser Kontaktkörper. Chemie Ingenieur Technik, 21, 219-226.
http://dx.doi.org/10.1002/cite.330211104 - 11. Aris, R. (1957) On Shape Factors for Irregular Particles-1: The Steady State Problem, Diffusion and Reaction. Chemical Engineering Sciences, 6, 262-268.
http://dx.doi.org/10.1016/0009-2509(57)85028-3 - 12. Weisz, P.B. and Goodwin, R.B. (1963) Combustion of Carbonaceous Deposits within Porous Catalyst Particles I, Diffusion-Controlled Kinetics. Journal of Catalyst, 2, 397-404.
http://dx.doi.org/10.1016/0021-9517(63)90104-0 - 13. Weisz, P.B. and Hicks, J.S. (1962) The Behaviour of Porous Catalyst Particles in View of Internal Mass and heat Diffusion Effects. Chemical Engineering Science, 17, 265-275.
http://dx.doi.org/10.1016/0009-2509(62)85005-2 - 14. Adomian, G. (1984) Convergent Series Solution of Nonlinear Equations. Journal of Computational and Applied Mathematics, 11, 225-230.
http://dx.doi.org/10.1016/0377-0427(84)90022-0 - 15. Hasan, Y.Q. and Zhu, L.M. (2008) Modified Adomian Decomposition Method for Singular Initial Value Problems in the Second-Order Ordinary Differential Equations. Surveys in Mathematics and Its Applications, 3, 183-193.
- 16. Hosseini, M.M. (2006) Adomian Decomposition Method with Chebyshev Polynomials. Applied Mathematics and Computation, 175, 1685-1693.
http://dx.doi.org/10.1016/j.amc.2005.09.014 - 17. Wazwaz, A.M. (1999) A Reliable Modification of Adomian Decomposition Method. Applied Mathematics and Computation, 102, 77-86.
http://dx.doi.org/10.1016/S0096-3003(98)10024-3
Appendix A. Basic Concept of Modified Adomian Decomposition Method [16]
Consider the nonlinear differential equation in the form

with initial condition

where 


So, the problem (A.1) can be written as,

The inverse operator 

Applying 


By operating 

The Adomian decomposition method introduce the solution 



where the components 




By substituting (A.8) and (A.9) into (A.7),

Through using the Adomian decomposition method, the components 

which gives

From (A.9) and (A.12), we can determine the components

Appendix B: General Solution of Equation (1) Using the Adomian Decomposition Method
In this appendix, we derive the general solution of nonlinear Equation (1) by using the Adomian decomposition method. We write the Equation (1) in the operator form,

where


where A and B are the constants of integration. We let,

and

where

In view of Equations (B. 3 - B. 5), Equation (B. 2) gives

We identify the zeroth component as

Using the boundary condition (4) we get,

and the remaining components can be obtained using the recurrence relation

where 




The remaining polynomials can be generated easily, and so,


Adding (B. 8), (B. 12) and (B. 13) we get the Equation (6) in the text.
Appendix C: The Matlab Program to Find the Numerical Solution of Equation (1)
function pdex1
m = 2;
x = linspace(0,1);
t = linspace(0,100);
sol = pdepe(m,@pdex1pde,@pdex1ic,@pdex1bc,x,t);
u = sol(:,:,1);
surf(x,t,u)
title('Numerical solution computed with 20 mesh points.')
xlabel('Distance x')
ylabel('Time t')
figure
plot(x,u(end,:))
title('Solution at t = 2')
xlabel('Distance x')
ylabel('u(x,2)')
% --------------------------------------------------------------
function [c,f,s] = pdex1pde(x,t,u,DuDx)
c = 1;
f = DuDx;
Q=1;
B=1.5;
r=1;
s =-(Q^2)*u*exp(r*B*(1-u)/(1+B*(1-u)));
% --------------------------------------------------------------
function u0 = pdex1ic(x)
u0 = 1;
% --------------------------------------------------------------
function [pl,ql,pr,qr] = pdex1bc(xl,ul,xr,ur,t)
pl = 0;
ql = 1;
pr = ur-1;
qr = 0;
Nomenclature
CA Concentration of reactant A inside the catalyst pellet (mole/cm3)
CA,s Concentration of reactant A at the surface of catalyst pellet (mole/cm3)

E Activation energy (kJ∙mol−1)
rA Arrhenius reaction rate (s−1)
Rg Universal gas constant (8.3145 J∙k−1∙mol−1)
T Temperature inside the catalyst pellet (K)
Tref Reference temperature (K)
Ts Temperature at the surface of catalyst pellet (K)
x Dimensionless radius of the spherical catalyst pellet (none)
y Dimensionless concentration along radial direction of catalyst pellet (none)
Greek Symbols




Submit your manuscript at: http://papersubmission.scirp.org/
NOTES
*Corresponding author.




