Materials Sciences and Applications
Vol.3 No.1(2012), Article ID:16971,10 pages DOI:10.4236/msa.2012.31007
Study of Dielectric and Piezoelectric Properties in the Ternary System Pb0.98Ca0.02[{(Zr0.52Ti0.48)0.98(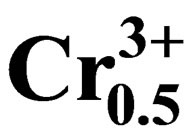 ,
,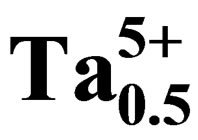 4998550415 height=53.4849992752075 />)0.02}1–zPz]O3 Doping Effects
4998550415 height=53.4849992752075 />)0.02}1–zPz]O3 Doping Effects
![]()
1Laboratoire de Chimie Appliquée, Université de Biskra, Biskra, Algérie; 2Département de Science de la Matière, Université de Ouargla, Ouargla, Algérie.
Email: {hamzioui_louanes, aboutarfaia}@yahoo.fr
Received November 7th, 2011; revised December 8th, 2011; accepted December 29th, 2011
Keywords: PZT; Piezoelectricity; Electronic Materials; Dielectric Properties; Methods Physico-Chemical of Analysis
ABSTRACT
The effects of P2O5 oxide on microstructure, dielectric and piezoelectric properties of Pb0.98Ca0.02[{(Zr0.52Ti0.48)0.98(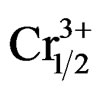 ,
,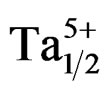 )0.02}1–zPz]O3 ternary ceramics were investigated. Specimens with various contents of P2O5 from 0 to 12 wt% were prepared by a conventional oxide mixing technique. The effect of P2O5 doping with regard to the development of the crystalline phase, density, microstructure, dielectric, ferroelectric and piezoelectric characteristics has been investigated. It has been found that the sintering temperature of piezoelectric Pb0.98Ca0.02[{(Zr0.52Ti0.48)0.98(
)0.02}1–zPz]O3 ternary ceramics were investigated. Specimens with various contents of P2O5 from 0 to 12 wt% were prepared by a conventional oxide mixing technique. The effect of P2O5 doping with regard to the development of the crystalline phase, density, microstructure, dielectric, ferroelectric and piezoelectric characteristics has been investigated. It has been found that the sintering temperature of piezoelectric Pb0.98Ca0.02[{(Zr0.52Ti0.48)0.98(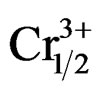 ,
,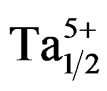 )0.02}1–zPz]O3 can be reduced by phosphorus addition without compromising the dielectric properties. A sintered density of 94 % of the theoretical density was obtained for 4 wt% P2O5 addition after sintering at 1050˚C for 4 h. Ceramics sintered at 1050˚C with 4 wt% P2O5 achieve excellent properties, which are as follows: kp = 0.73, ρ = 0.09 × 10+4 (Ω·cm), εr = 18800, tanδ = 0.0094 and Tc = 390˚C.
)0.02}1–zPz]O3 can be reduced by phosphorus addition without compromising the dielectric properties. A sintered density of 94 % of the theoretical density was obtained for 4 wt% P2O5 addition after sintering at 1050˚C for 4 h. Ceramics sintered at 1050˚C with 4 wt% P2O5 achieve excellent properties, which are as follows: kp = 0.73, ρ = 0.09 × 10+4 (Ω·cm), εr = 18800, tanδ = 0.0094 and Tc = 390˚C.
1. Introduction
Lead-based perovskite-type solid solutions consisting of the ferroelectric and relaxor materials have attracted a growing fundamental and practical interest because of their excellent dielectric, piezoelectric and electrostrictive properties which are useful in actuating and sensing applications [1,2]. However, the sintering of PZT at high temperatures gives rise to a lead loss, which drastically degrades the device performance. Generally, a lead loss at high temperatures can be prevented by atmospherecontrolled sintering of PZT. However, such composition requires sintering at a high temperature (>1250˚C) in a controlled atmosphere to contain lead volatilization so as to avoid a shift in composition. To get around the problem, different sintering aids have been tried by various workers [3-5]. However, for practical applications, such sintering aids need proper selection so that the electrical and piezoelectric properties of the ceramics do not degrade.
The dielectric constants increased with the addition of NiO, Fe2O3, Gd2O3, Nb2O5 or WO3 and decreased with Cr2O3 or MnO2 addition [6-12]. Duran et al. studied the effect of MnO addition on the sintering and piezoelectric properties of Sm-modified lead titanate ceramics. The maximum density observed was 96.8% of the theoretical densit for 1% MnO addition at a sintering temperature of 1150˚C [13]. The main role of dopants is generally improved physical and mechanical properties of these materials. This work aims at, to study the influence of P2O5 on the properties dielectric and piezoelectric of a ceramics material of general formula: Pb0.98Ca0.02[(Zr0.52Ti0.48)0.98 ( ,
,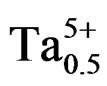 )0.02]O3 and of structure perovskite.
)0.02]O3 and of structure perovskite.
2. Experimental Procedure
The compositions used for the present study were Pb0.98 Ca0.02[{(Zr0.52Ti0.48)0.98(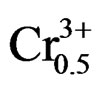 ,
,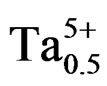 )0.02}1–zPz]O3 with z varying as 0, 2, 4, 6, 8, 10 and 12 wt% respectively. The samples were prepared by a conventional oxide mixing technique. The appropriate amounts of PbO (99.9%), TiO2 (99.9%), ZrO2 (99.0%), Ta2O5 (99.9%), CaO (99.9%), Cr2O3 (99.9%) and P2O5 (99.9%) powders were weighed and mixed by ball milling with partially stabilized zirconia balls as media in isopropyl alcohol for 6 h. After drying, the mixture was calcined in a covered alumina crucible at 800˚C for 4 h. The calcined powders were again ball milled for 24 h. The resulting powders were uniaxially compacted into pellets of 10 mm in diameter at a pressure of 5 MPa, followed by isostatically pressing at 150 MPa. To investigate their sintering behavior, the specimens were sintered in a sealed alumina crucible at temperatures ranging from 1000˚C to 1180˚C for 2 h. To limit PbO loss from the pellets, a PbO-rich atmosphere was maintained by placing an equimolar mix ture of PbO and ZrO2 inside the crucible. The weight loss of a well-sintered specimen was less than 0.5 wt%, thus a 0.5 wt% excess PbO was added to compensate for the lead loss during sintering. The bulk density was measured using the Archimedean method. The sintered compounds are carefully ground, then analyzed by the scanning electron microscopy (SEM) is a technical for estimating the size distribution, the average size of grains after sintering and qualitatively assess the presence of porosity. The micrographics are made using a Microscope JMS 6400. To investigate the electrical properties, the sintered disks were lapped on their major faces, and then sliver electrodes were deposited with a low temperature paste at 700˚C for 30 min. The piezoelectric samples were poled in a silicone oil bath at 100˚C by applying 20 kV/cm for 20 min. then cooling them under the same electric field. They were aged for 24 h prior to testing. The temperature dependence of dielectric properties was measured at temperatures ranging from room temperature to 420˚C with a heating rate of 2 ˚C/min using an impedance analyzer—HP4192A, Hewlett-Packard, Palo Alto, CA. The electromechanical coupling factor, kp, was determined by the resonance and antiresonance technique using another impendence analyzer (SI1260 Impedance/Gain-Phase Analyzer, Solartron, UK). (kp = [2.51(fa – fr)/fr)]1/2, where fr and fa are the resonance and anti-resonance frequencies, respectively [14]. Variation of the dielectric constant εr, resistivity and also the angle of the losses were examined by using a measuring bridge type RLC (bridge Schering) depending on temperature, concentration, the frequency.
)0.02}1–zPz]O3 with z varying as 0, 2, 4, 6, 8, 10 and 12 wt% respectively. The samples were prepared by a conventional oxide mixing technique. The appropriate amounts of PbO (99.9%), TiO2 (99.9%), ZrO2 (99.0%), Ta2O5 (99.9%), CaO (99.9%), Cr2O3 (99.9%) and P2O5 (99.9%) powders were weighed and mixed by ball milling with partially stabilized zirconia balls as media in isopropyl alcohol for 6 h. After drying, the mixture was calcined in a covered alumina crucible at 800˚C for 4 h. The calcined powders were again ball milled for 24 h. The resulting powders were uniaxially compacted into pellets of 10 mm in diameter at a pressure of 5 MPa, followed by isostatically pressing at 150 MPa. To investigate their sintering behavior, the specimens were sintered in a sealed alumina crucible at temperatures ranging from 1000˚C to 1180˚C for 2 h. To limit PbO loss from the pellets, a PbO-rich atmosphere was maintained by placing an equimolar mix ture of PbO and ZrO2 inside the crucible. The weight loss of a well-sintered specimen was less than 0.5 wt%, thus a 0.5 wt% excess PbO was added to compensate for the lead loss during sintering. The bulk density was measured using the Archimedean method. The sintered compounds are carefully ground, then analyzed by the scanning electron microscopy (SEM) is a technical for estimating the size distribution, the average size of grains after sintering and qualitatively assess the presence of porosity. The micrographics are made using a Microscope JMS 6400. To investigate the electrical properties, the sintered disks were lapped on their major faces, and then sliver electrodes were deposited with a low temperature paste at 700˚C for 30 min. The piezoelectric samples were poled in a silicone oil bath at 100˚C by applying 20 kV/cm for 20 min. then cooling them under the same electric field. They were aged for 24 h prior to testing. The temperature dependence of dielectric properties was measured at temperatures ranging from room temperature to 420˚C with a heating rate of 2 ˚C/min using an impedance analyzer—HP4192A, Hewlett-Packard, Palo Alto, CA. The electromechanical coupling factor, kp, was determined by the resonance and antiresonance technique using another impendence analyzer (SI1260 Impedance/Gain-Phase Analyzer, Solartron, UK). (kp = [2.51(fa – fr)/fr)]1/2, where fr and fa are the resonance and anti-resonance frequencies, respectively [14]. Variation of the dielectric constant εr, resistivity and also the angle of the losses were examined by using a measuring bridge type RLC (bridge Schering) depending on temperature, concentration, the frequency.
3. Results and Discussion
3.1. Sintered Density
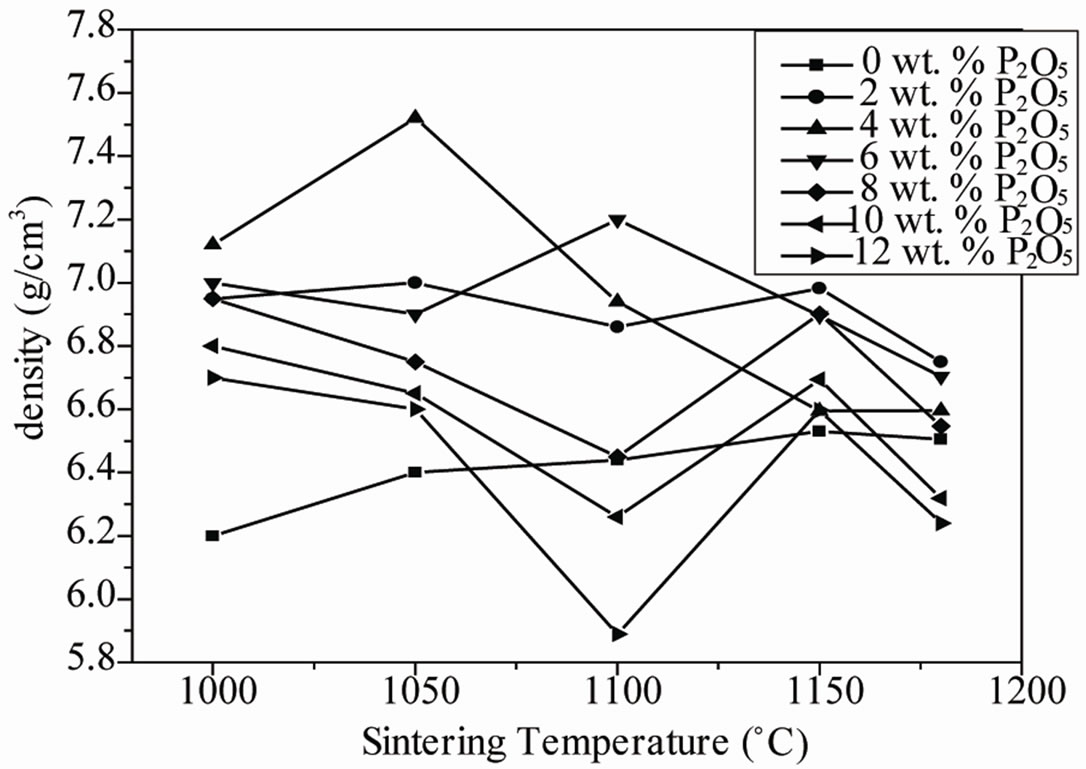
Figure 1(a) shows the variation of density with sintering temperature and the amount of P2O5 addition. This curves show the similar variation trend with increasing sintering temperature. The density of specimens sintered at 1050˚C showed the maximum value of 7.52 g·cm−3 at 4 wt% P2O5 and then was decreased after the maximum value. This variation is mainly attributed to the formation of liquid phase of excess PbO that improves densification of
 (a)
(a)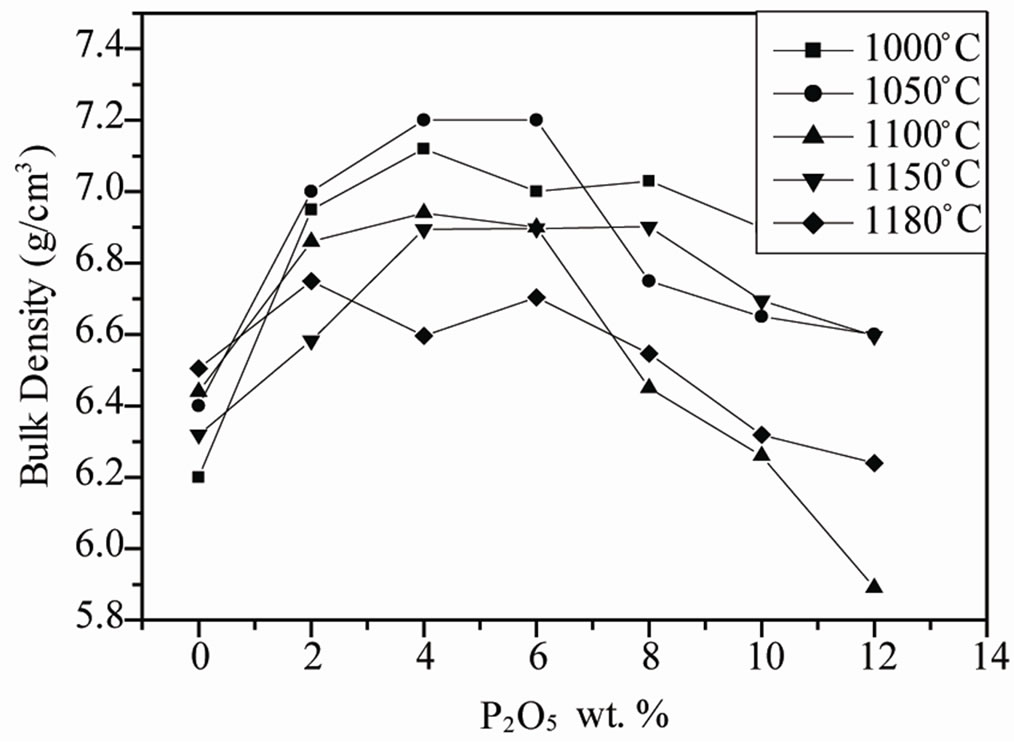 (b)
(b)
Figure 1. (a) Variation of the density of PZT-CCT samples with sintering temperature at different P2O5 addition; (b) Variation of bulk density of sintered PZT-CCT samples with P2O5 addition at different sintering temperature.
the ceramics. However, a large amount of this liquid phase leads to low density which may result from the formation of voids [15]. The densities of the sintered pellets are shown in Figure 1(b). From this figure, it is evident that initially the bulk density (as a percentage of theoretical density) of the pellets decreases with 1 wt% P2O5 addition, then also with an increasing amount of P2O5 addition up to 4.0 wt%, the bulk density increased and then greater than 4.0 wt% P2O5 addition, the density again decreased. This behavior can be explained by decomposition of P2O5 (melting point around 570˚C) can aid in liquid-phase sintering. Initially, the density of the pellets decreased on addition of 4 wt% P2O5, because the amount of liquid formed was probably too low to get an appreciable densification. With increasing P2O5, the liquid-phase sintering dominated and was optimum for 4 wt% P2O5 addition leading to maximum densification. With a higher amount of P2O5 addition, the densification again decreased because there should be an optimum amount of liquid above which densification is inhibited due to the formation of a thick coating of the liquid around the grains [16]. Recently, Saha et al. [17] reported that a small amount of phosphorous addition (as P2O5) can improve the sintering of the PZT ceramics.
3.2. Study of Morphological
Figure 2 shows SEM microstructures of the fracture surfaces of samples sintered at various temperatures. The
 (a)
(a) (b)
(b) (c)
(c)
Figure 2. SEM micrographs of Pb0.98Ca0.02[(Zr0.52Ti0.48)0.98 (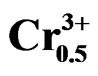 ,
, )0.02]O3 specimens doped with 4 wt% P2O5 and sintered at (a) 1000˚C, (b) 1050˚C and (c) 1100˚C.
)0.02]O3 specimens doped with 4 wt% P2O5 and sintered at (a) 1000˚C, (b) 1050˚C and (c) 1100˚C.
distributions in the grain shape and size of the samples are rather uniform. All the samples showed an intergranular fracture mechanism indicating that the grain boundaries are mechanically weaker than the grains. These samples appear very dense and of a homogeneous granular structure, the three samples seem homogeneous and there do not seem to be grains of the pyrochlore phase which are identifiable by their pyramidal form. It is noted that the average grain size increases with increasing sintering temperature for 2 μm at 1000˚C up to 2.671 μm at 1050˚C then this size decreases slightly to 1100˚C to reach the value of 2.51 μm; and the broader the granulo-metric distribution “Figure 2(b)”, the more the size of the grains gets bigger. With increasing P2O5, the liquid-phase sintering dominated and was optimum for 4 wt% P2O5 addition leading to maximum densification. The increase in grain size may have led to the decrease of oxygen vacancies in PZT [18]. Figure 2 also shows that the porosity decreases with the increase of sintering temperature (it reaches a minimum at 1050˚C), which is consistent with the increase of the densification of specimens.
3.3. Phase Structure
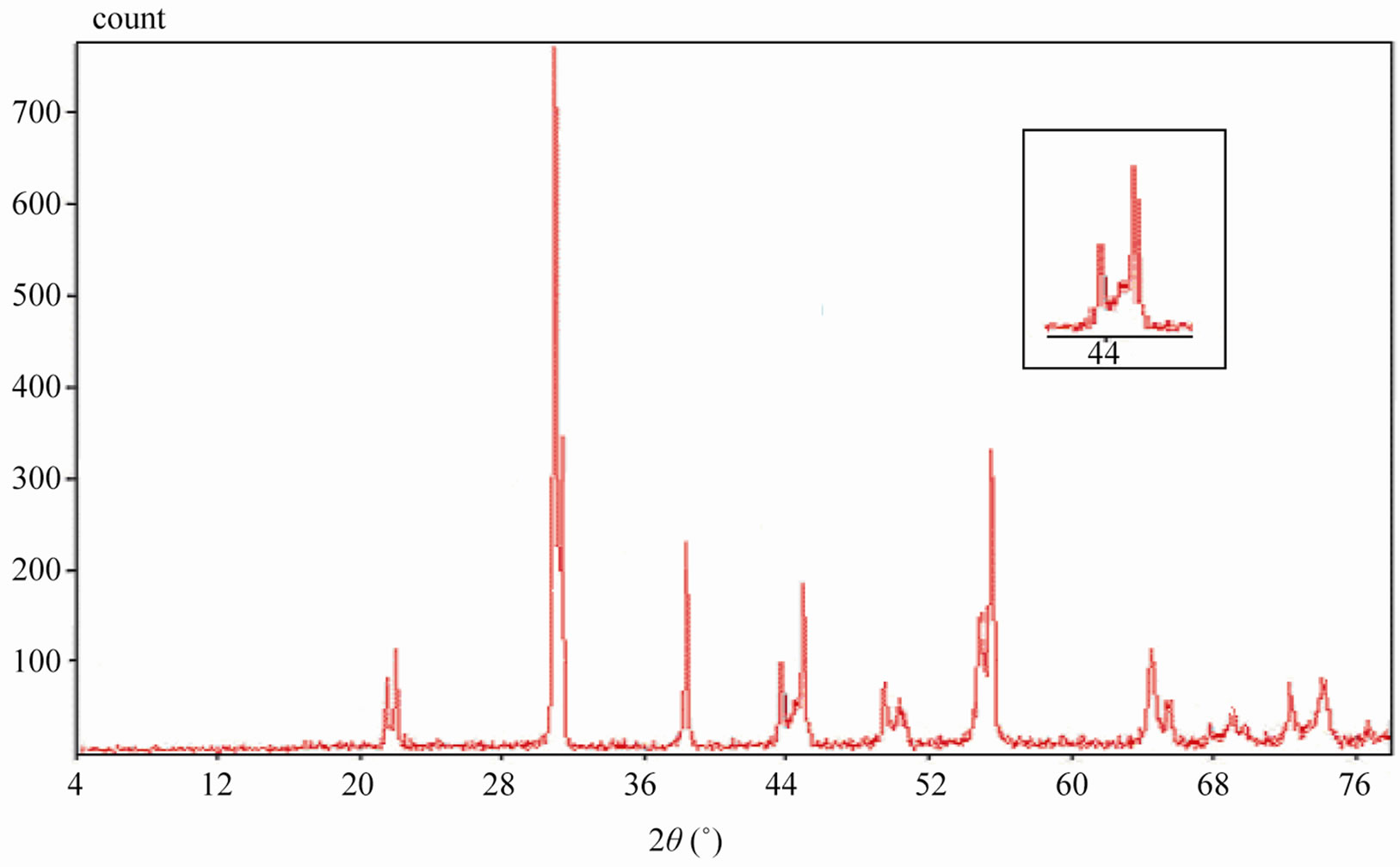
Sintered powders were examined by X-ray diffractometry to ensure phase purity and to identify the phases of the materials. The results of X-rays on the samples sintered at 1050˚C are illustrated in Figure 3. The ceramics with various P2O5 contents all exist as pure perovskite phase. The ceramics exist as tetragonal phase which is indicated by the single (002)T and (200)T peak at z = 0.00 and z = 0.02. As P2O5 content increases from 4 to 8 wt%, the ceramics coexist as tetragonal and rhombohedral phase revealed by the coexistence of (002)T and (200)R peaks in the 2θ range from 43.8˚ to 45.3˚. The ceramics with z = 0.08 and z = 0.10 exist as tetragonal phase revealed by the splitting of (002)T and (200)T peaks in the 2θ range from 43.5˚ to 45.4˚.
3.4. Dielectric Properties
The electrical properties were strongly dependent on the phase of the specimens. Figure 4 shows the dielectric constant εr and dielectric loss tanδ of the ceramics sintered at 1050˚C as a function of P2O5 content measured at 1 kHz. The temperature of the maximum dielectric constant (Tm) increased and the dielectric constant peak sharpened with increasing P2O5 concentration. Dielectric loss tanδ slowly deceases at first (4 wt%), and begins to increase when P2O5 content is up to 8 wt%.
Figure 5 shows dielectric loss (tanδ) of ceramics sintered at 1050˚C as a function of P2O5 content it is noticeable that there is a variation of the dielectric losses
 (a)
(a)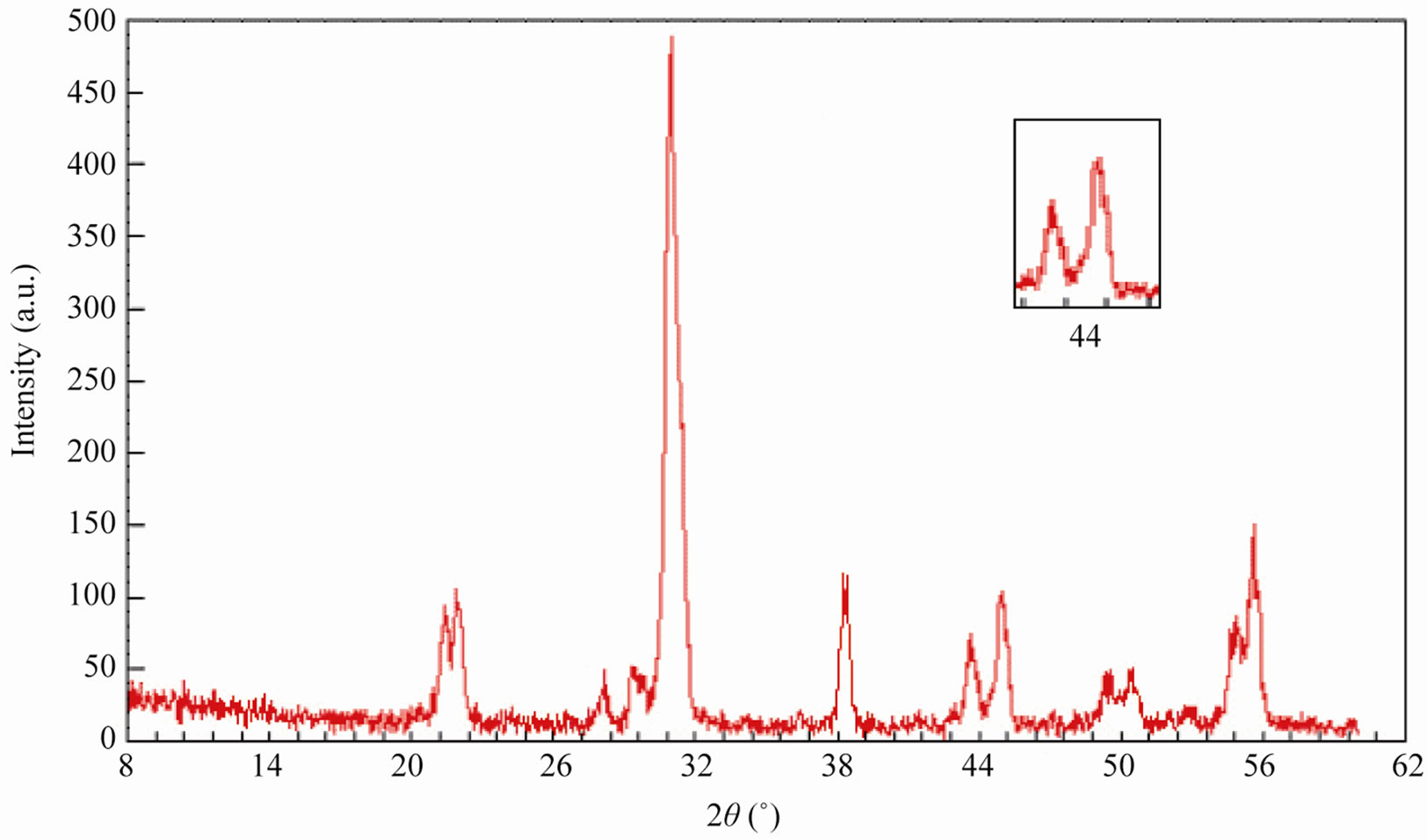 (b)
(b)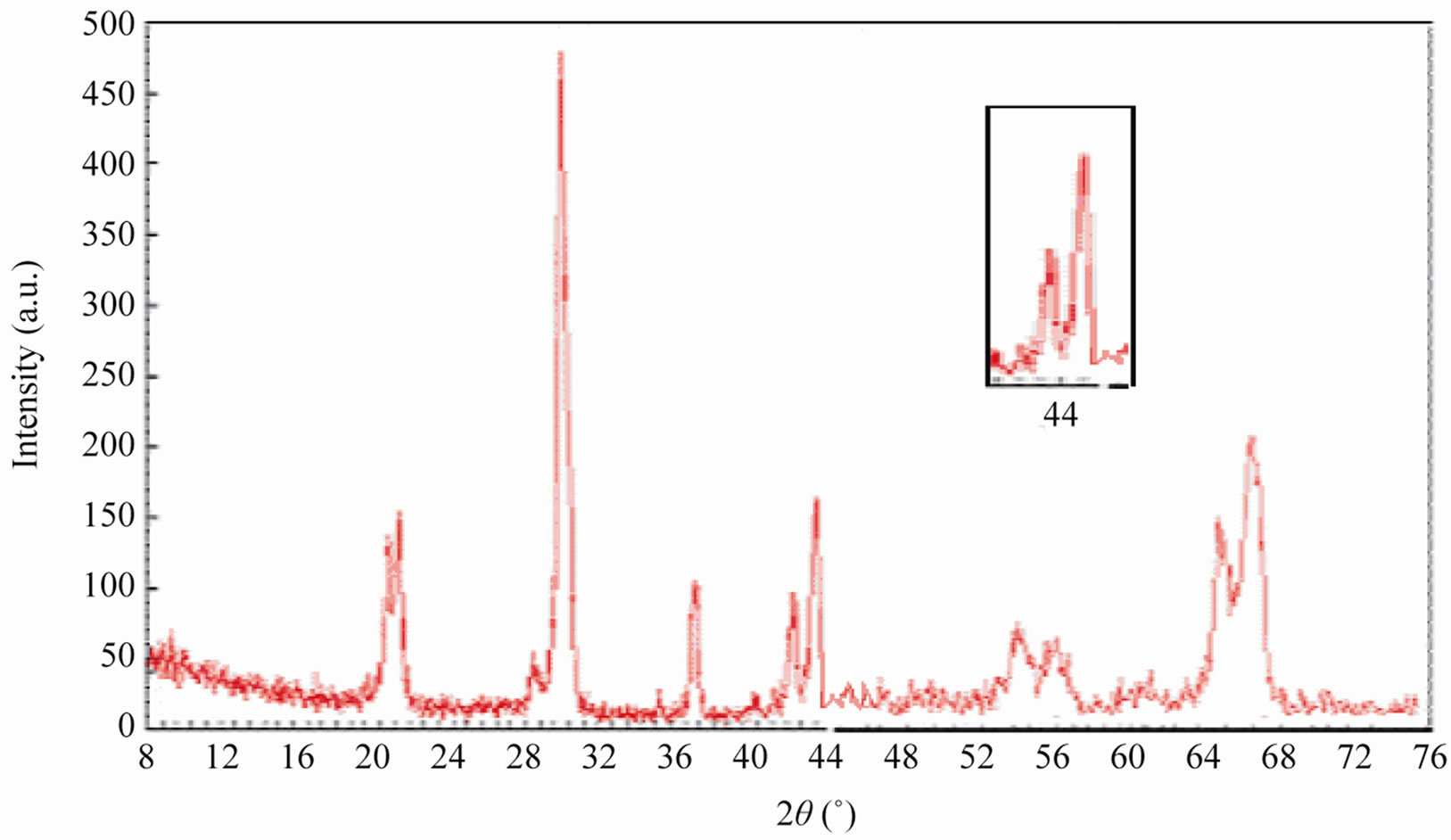 (c)
(c)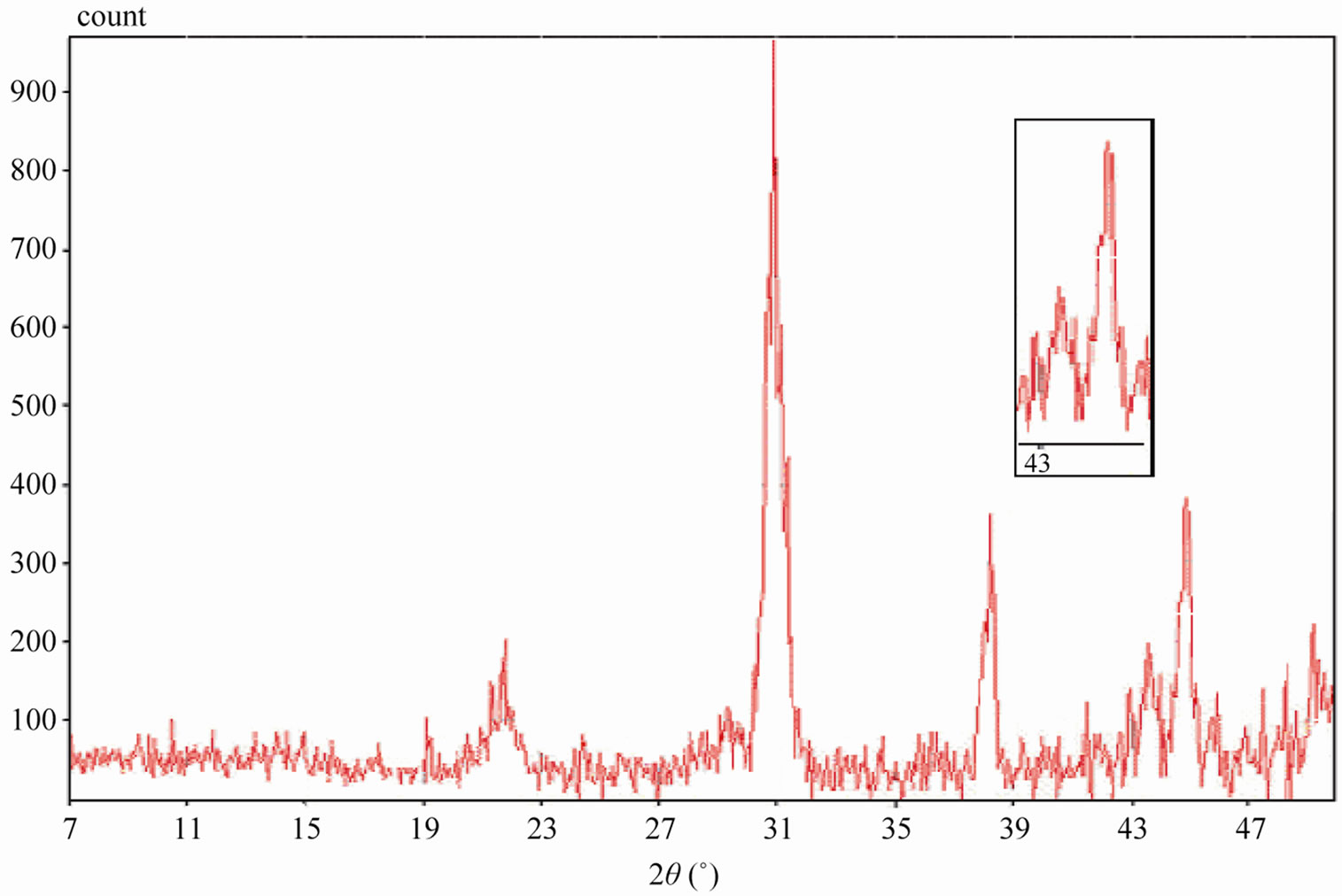 (d)
(d)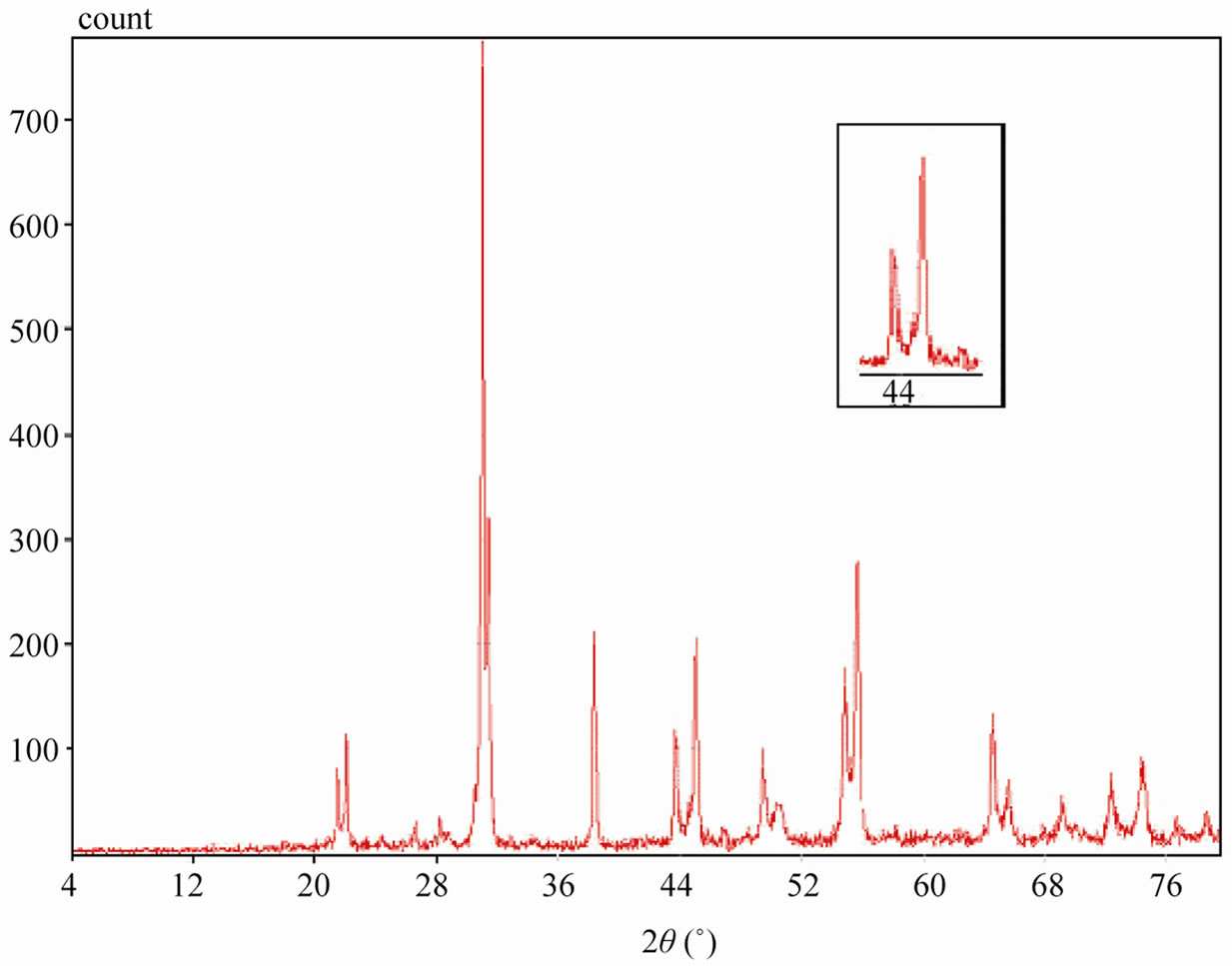 (e)
(e)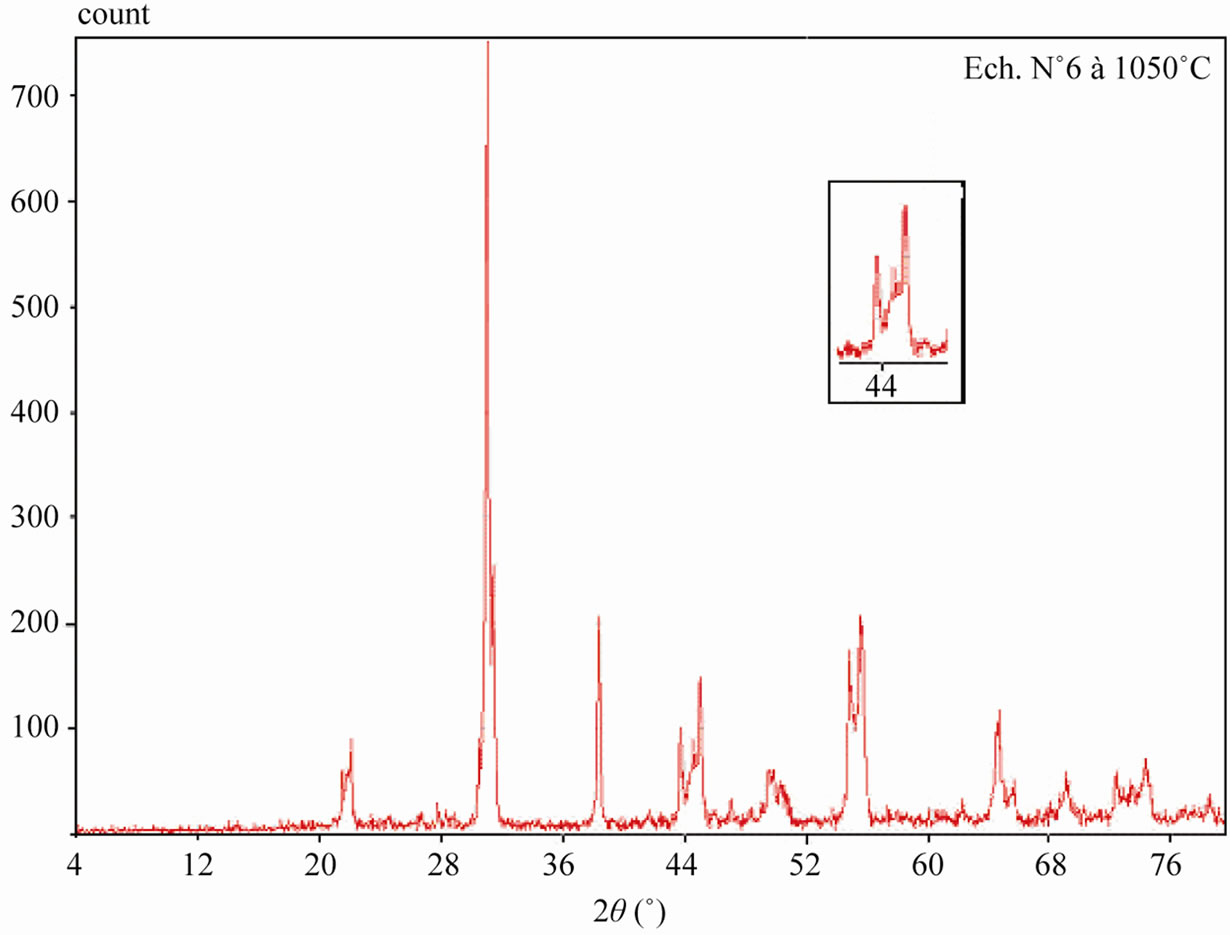 (f)
(f)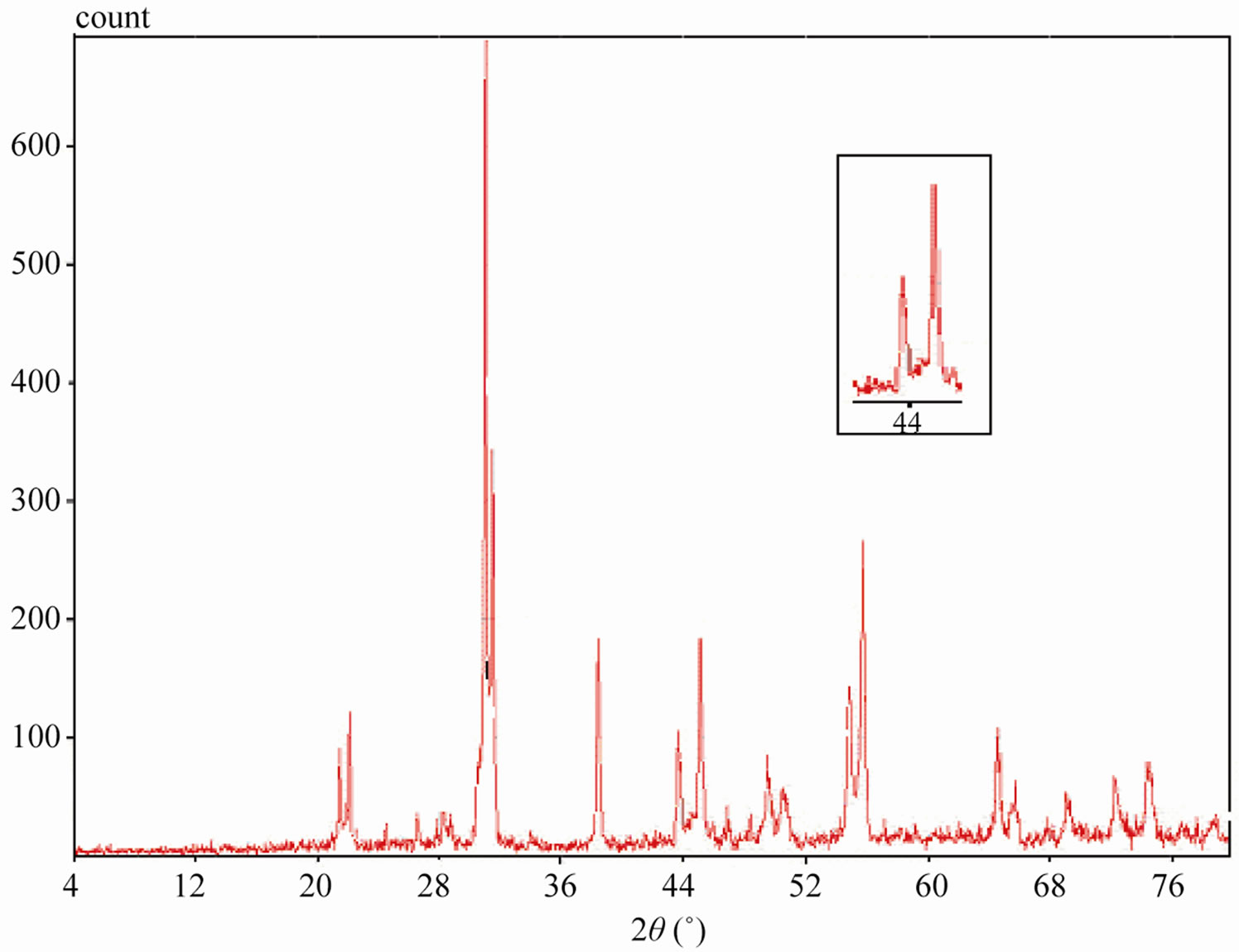 (g)
(g)
Figure 3. XRD patterns of sintered PZT-CCT ceramics with varying P2O5 addition: (a) 0 wt%; (b) 2 wt%; (c) 4 wt%; (d) 6 wt%; (e) 8 wt%; (f) 10 wt% and (g) 12 wt% sintered at 1050˚C.
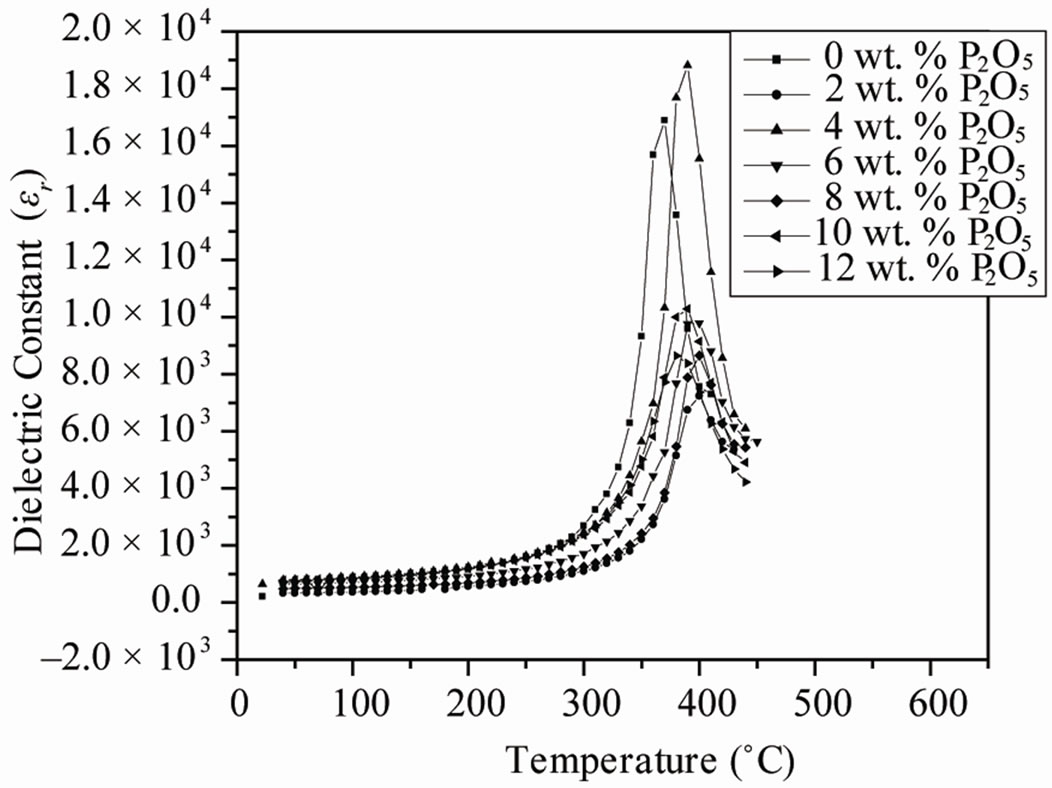
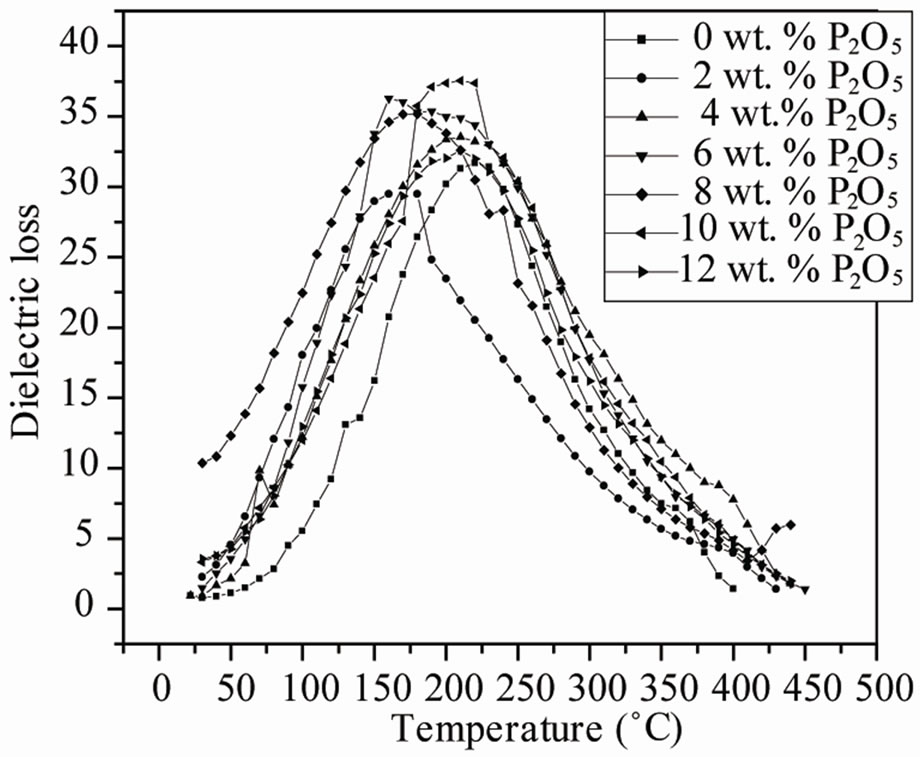
Figure 4. Temperature dependence of the dielectric constant εr and dielectric loss tanδ for perovskite Pb0.98Ca0.02 [{(Zr0.52Ti0.48)0.98(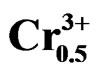 ,
,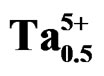 )0.02}1–xPx]O3 ceramics.
)0.02}1–xPx]O3 ceramics.
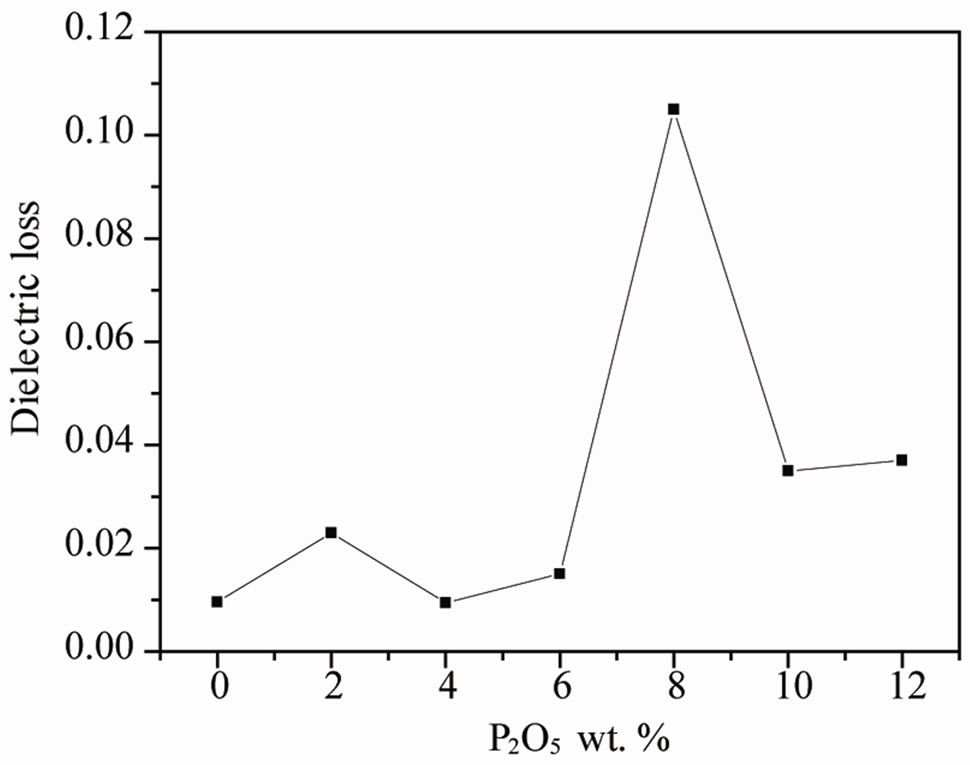
Figure 5. tanδ of ceramics sintered at 1050˚C as a function of concentration of P2O5.
with increasing concentration of P2O5. Dielectric loss (tanδ) slowly deceases at first, and begins to increase when P2O5 content is up to 6 wt%. This indicates that the compound doped with 4 wt% of P2O5 is denser than other doped.
The effects of P2O5 on the dielectric constant and dissipation factor measured at 1 kHz are shown in Figure 6. The position of anomalies in the εr and tanδ curves, corresponding to FR(HT)-PC phase transition, is dependent on the P2O5 content. The dielectric peak temperatures (Tc) shift slightly to lower temperatures with increasing P2O5 content. It is well known that Tc varies with substitution. Therefore, the decrease in Tc can be explained by assumption that P2O5 substituted in the perovskite structure.
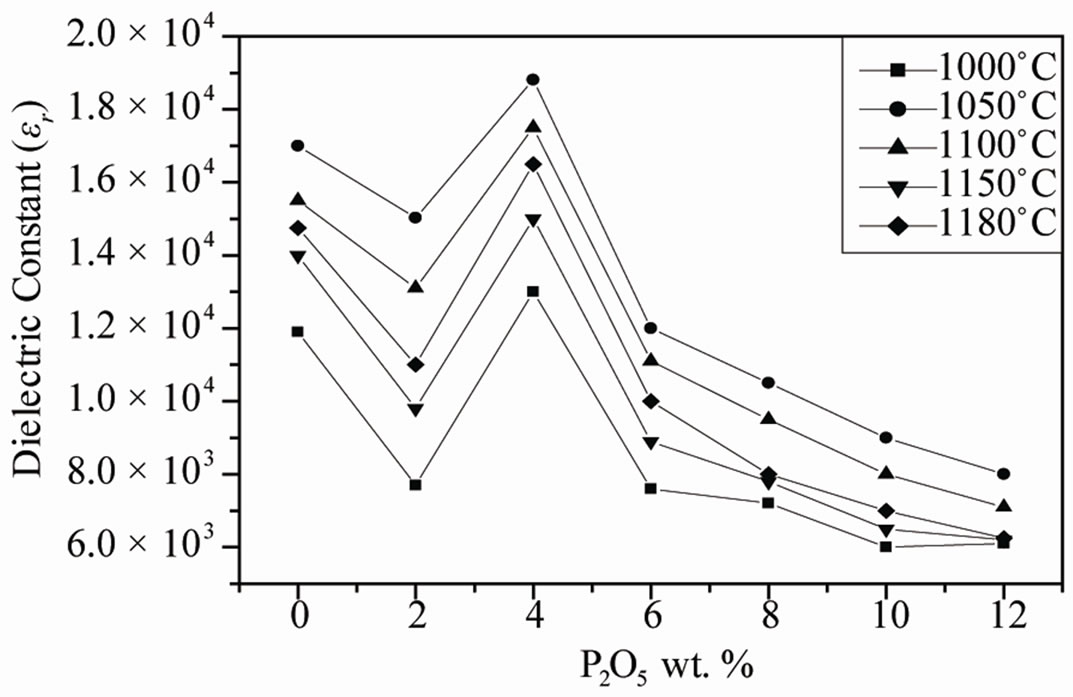
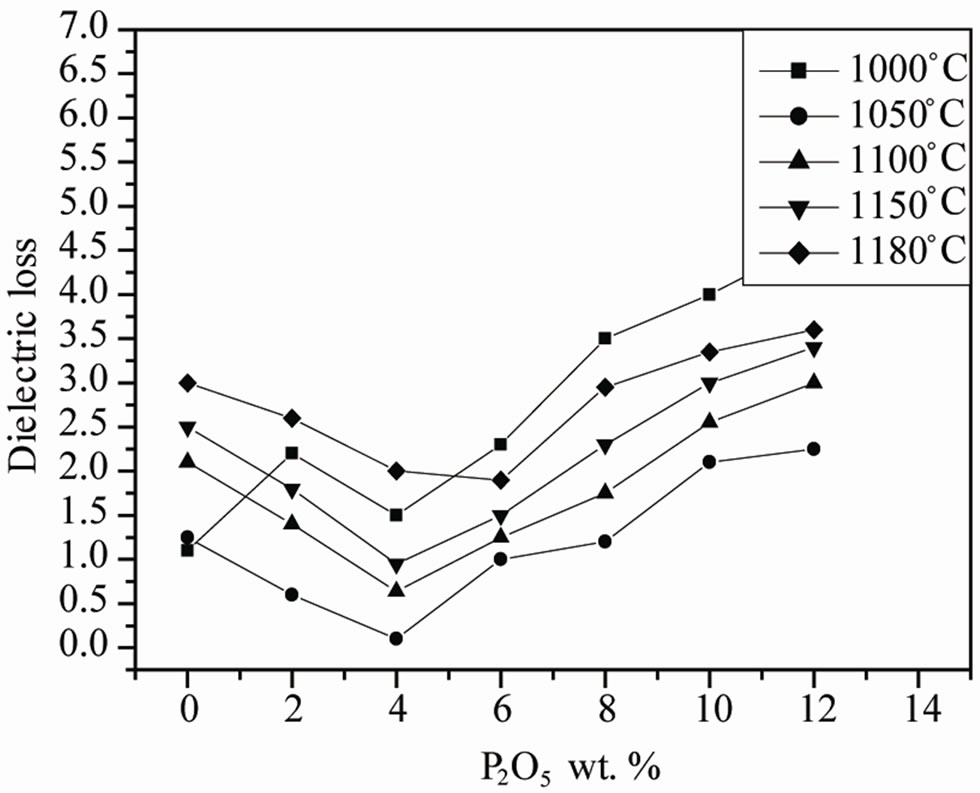
Figure 6. Evolution of the dielectric constant and dielectric loss tanδ as a function of P2O5 additive at different sintering temperature.
The maxima in εr curves become broadened when the P2O5 content exceeds 4.0 wt%. The broadening or diffuseness of dielectric peaks occurs mainly due to the statistical composition fluctuations because large amount of the second phases appear in PZT ceramics. A statistical treatment based on a postulated Gaussian distribution of local Curie temperature is associated with the composition fluctuations [19].
The variation of the dielectric constant as a function of frequenc from 1 kHz to 100 kHz for Pb0.98Ca0.02[{(Zr0.52 Ti0.48)0.98(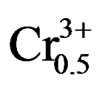 ,
,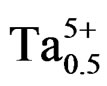 )0.02}1–xPx]O3 is illustrated in Figure 7. With the increase of frequency, dielectric constant varies moderately before 30 kHz, and then goes up abruptly. According to these results, we can consider the compound doped by 4 wt% P2O5 as a ceramic soft (depolarize easily) and can be used at high frequencies in the transmission technology. Some authors explain the maximum value of the dielectric constant at room temperature and values of low frequency by the existence of different types of polarization [20,21].
)0.02}1–xPx]O3 is illustrated in Figure 7. With the increase of frequency, dielectric constant varies moderately before 30 kHz, and then goes up abruptly. According to these results, we can consider the compound doped by 4 wt% P2O5 as a ceramic soft (depolarize easily) and can be used at high frequencies in the transmission technology. Some authors explain the maximum value of the dielectric constant at room temperature and values of low frequency by the existence of different types of polarization [20,21].
3.5. Electromechanical Properties
The electrical properties were strongly dependent on the
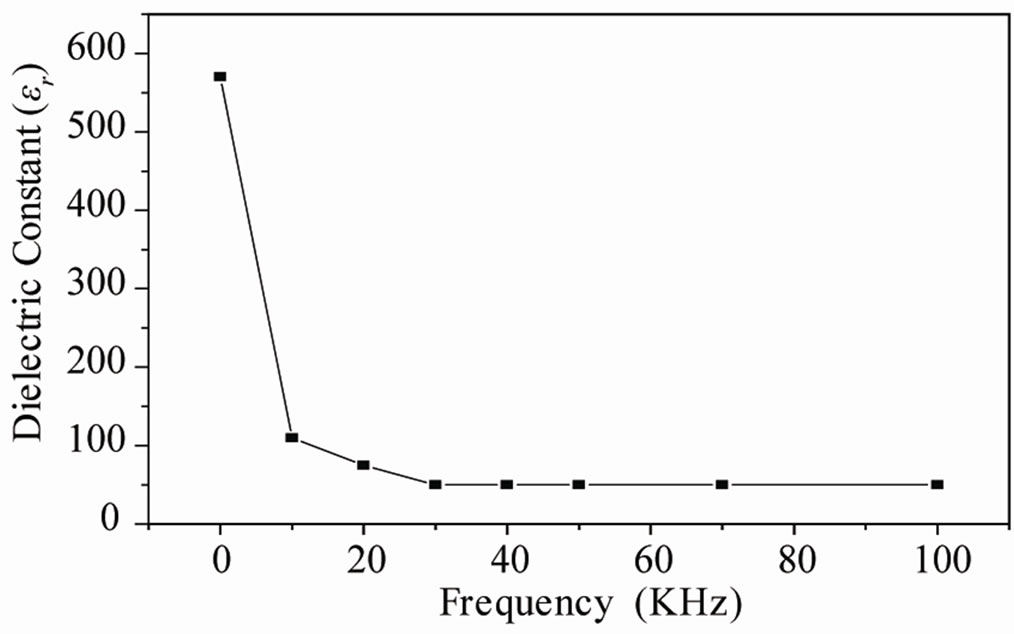
Figure 7. Evolution of the dielectric constant depending on the frequency for the sample doped by 4 wt% of P2O5.
phase of the specimens. The room-temperature electromechanical coupling factor (kp) of Pb0.98Ca0.02[{(Zr0.52 Ti0.48)0.98(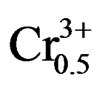 ,
,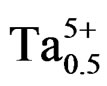 )0.02}1–zPz]O3 ceramics sintered at 1050˚C as a function of P2O5 content is plotted in Figure 8. It can be observed that both of the kp curve possess a peak with increasing P2O5 content. The kp of P2O5 ceramics at z = 0 is 59.0%. With increasing P2O5 content (4 wt%), the kp of PZTMN ceramics reach their maximum values of 72.3%.
)0.02}1–zPz]O3 ceramics sintered at 1050˚C as a function of P2O5 content is plotted in Figure 8. It can be observed that both of the kp curve possess a peak with increasing P2O5 content. The kp of P2O5 ceramics at z = 0 is 59.0%. With increasing P2O5 content (4 wt%), the kp of PZTMN ceramics reach their maximum values of 72.3%.
3.6. Study of Resistivity and Conductivity
The resistivity and conductivity of PZTMN sintered at 1050˚C as a function of temperature are plotted in Figures 9 and 10, independently. It can be seen that both resistivity decrease monotonically with increasing temperature. The above variation of conductivity is different from resistivity shown in Figure 9. The curves of this figure show that there is a relationship between the relative change in temperature and the two electrical factors (conductivity and resistivity). More temperature increases, the resistivity of each sample decreases more and more (Figure 9(a)). It decreases for the sample of doped 4 wt%.
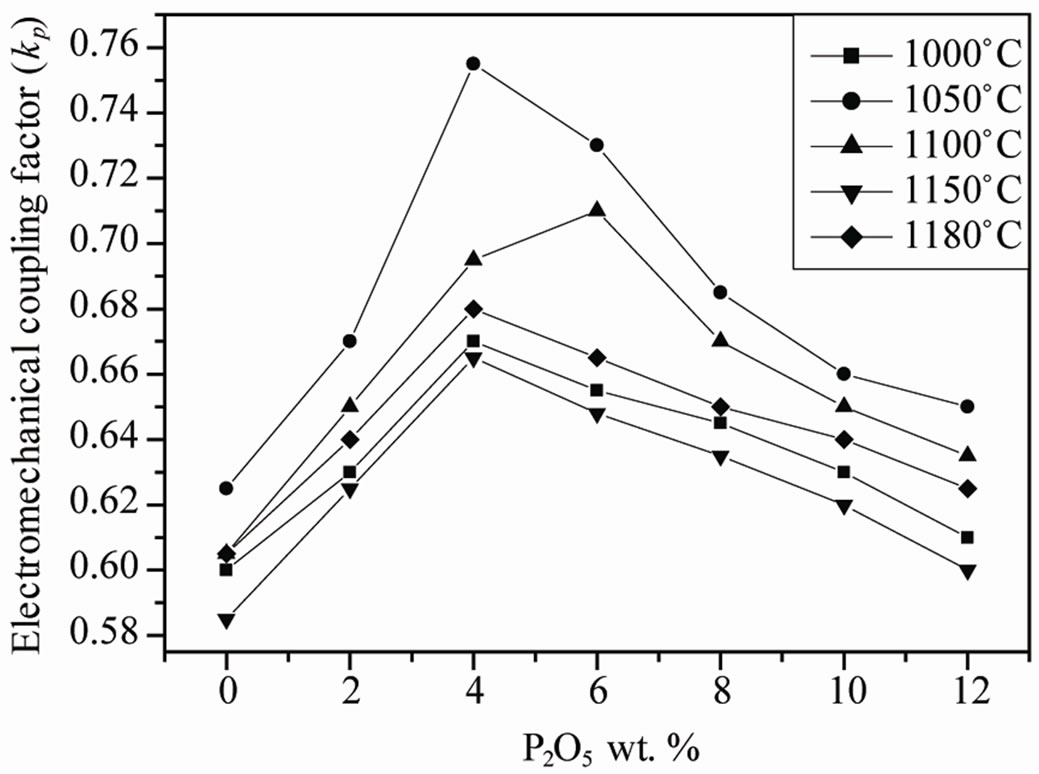
Figure 8. Evolution of kp as a function of P2O5 additive at different sintering temperature.
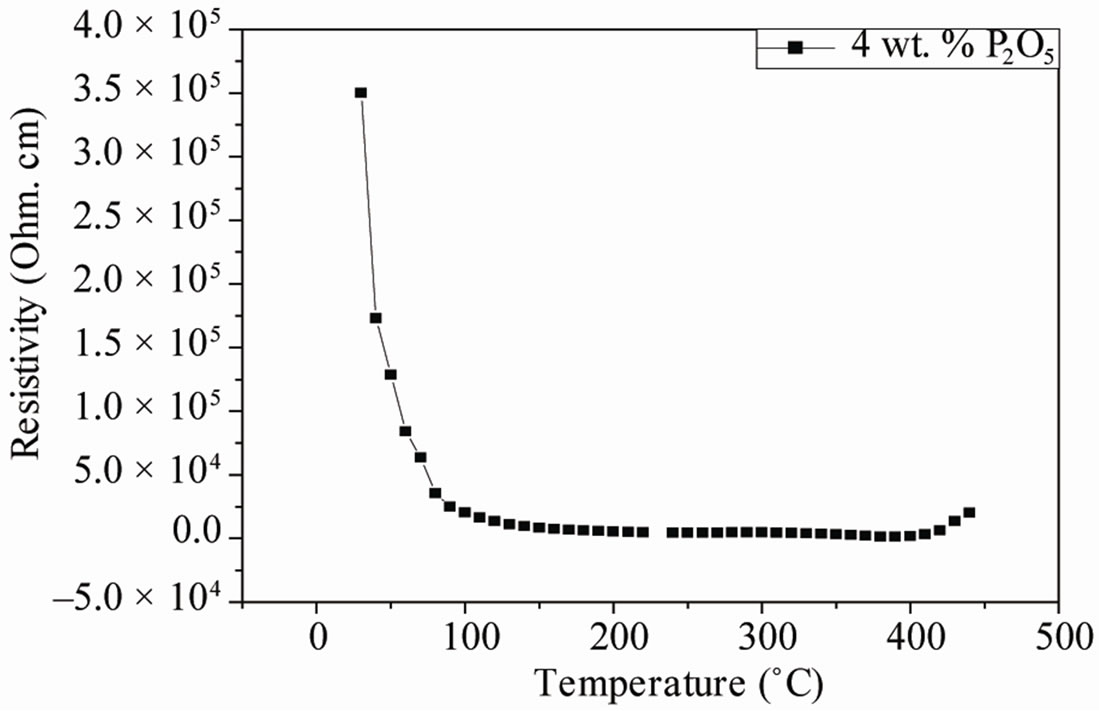
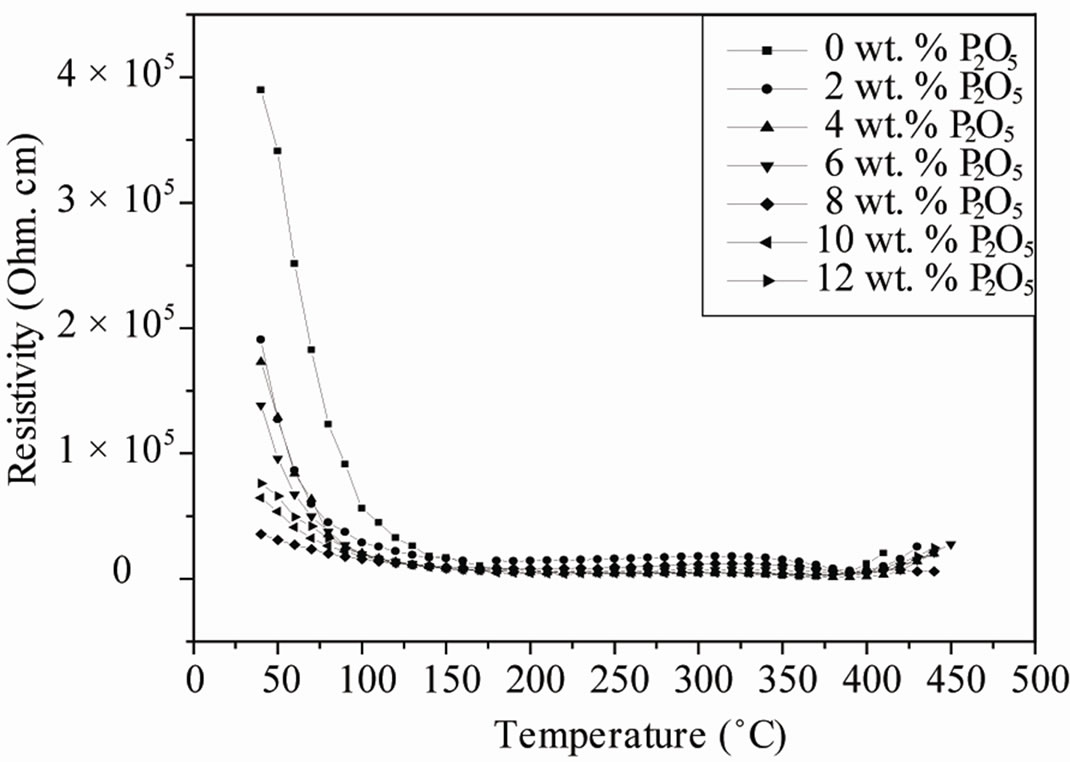
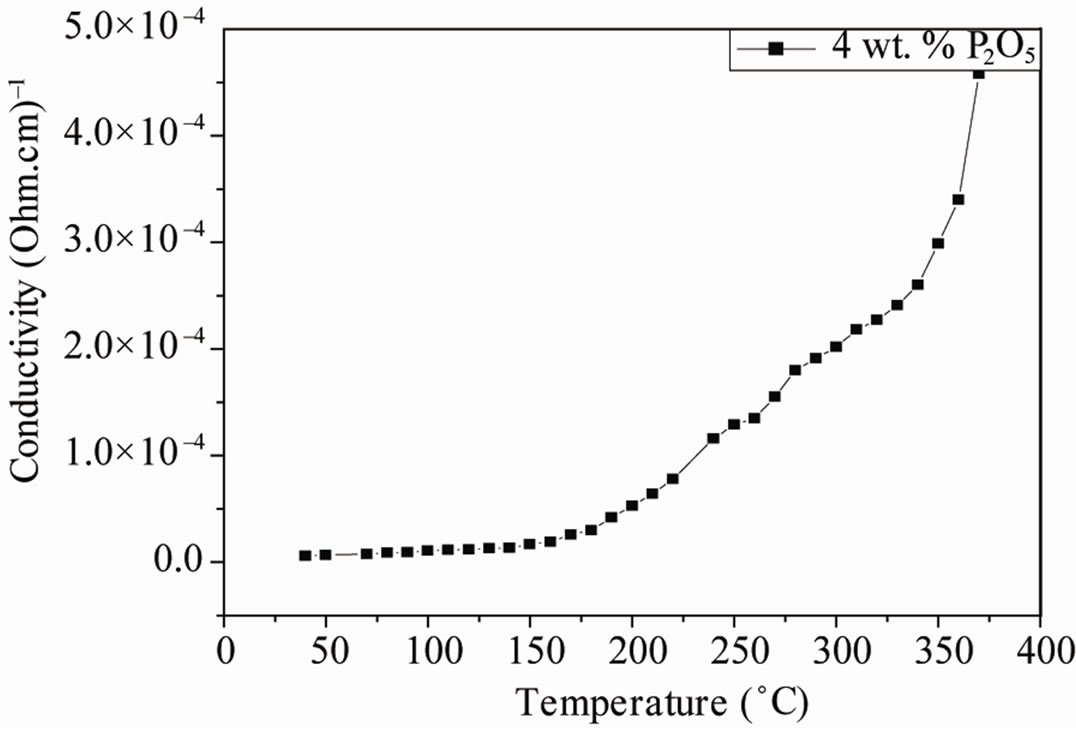
Figure 9. Variation in resistivity and conductivity sintered at 1050˚C as a function of temperature.
P2O5 of 35 × 10+4 (Ω·cm) when T = 50˚C until it reaches the value 0.09 × 10+4 (Ω·cm) to 350˚C (Figure 9 (b)). This is due to the fact that high-temperature thermal energy may be sufficient to break some connections or ionic and covalent causes some mobility of ions. However, the electrical conductivity varies in the opposite direction of resistivity; it grows with the increase of temperature (Figure 9(c)). It can reach a maximum value of 7.5 × 10–4 (Ω·cm)–1 at a temperature of 350˚C.
4. Conclusions
The compounds of the solution solid zirconate-titanate
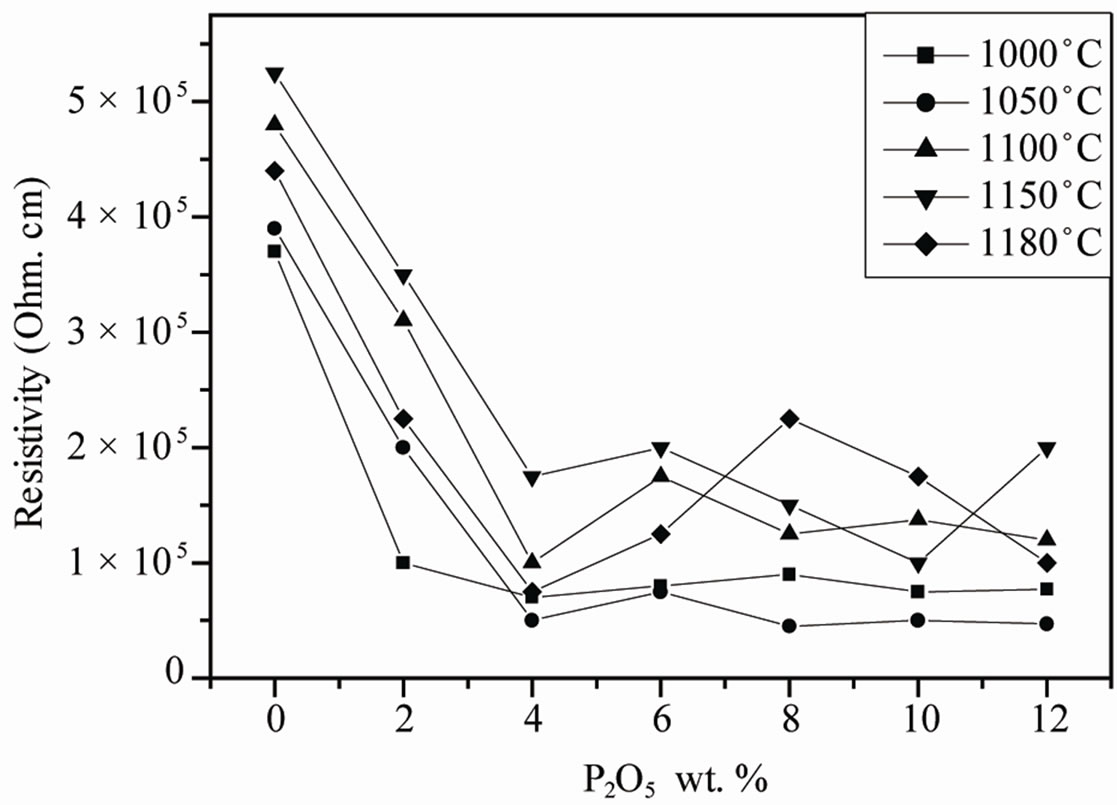
Figure 10. Evolution of the resistivity as a function of P2O5 additive at different sintering temperature.
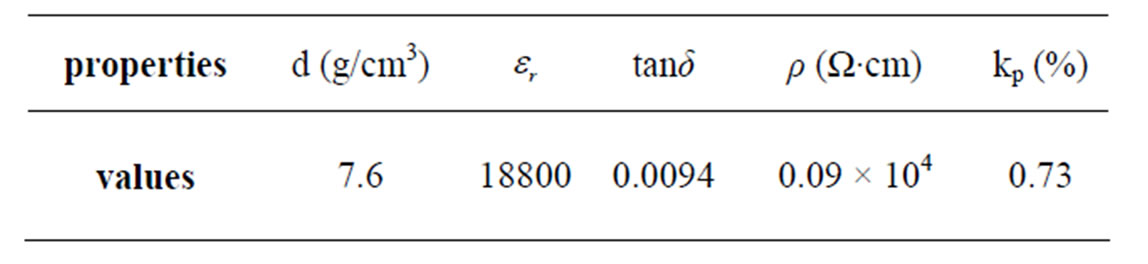
Table 1. Dielectric and piezoelectric properties of PZTMN doped with 4 wt% of P2O5 and sintered at 1050˚C.
lead, noted PZT, general formula Pb0.98Ca0.02[{(Zr0.52 Ti0.48)0.98(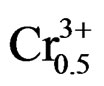 ,
,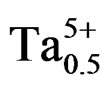 )0.02}1–zPz]O3 as z vary from 0.00 to 0.12 by setup of 0.02, it has been prepared from a mixture of oxides by the method ceramics. The effect of sintering temperature on density and porosity was studied to achieve the optimum sintering temperature corresponding to the maximum density and minimum value of porosity, because this temperature (1050˚C) corresponds to a better quality product. Low-temperature densification of PZT can be achieved by the incorporation of a small amount of P2O5 as a sintering aid without sacrificing the dielectric properties.
)0.02}1–zPz]O3 as z vary from 0.00 to 0.12 by setup of 0.02, it has been prepared from a mixture of oxides by the method ceramics. The effect of sintering temperature on density and porosity was studied to achieve the optimum sintering temperature corresponding to the maximum density and minimum value of porosity, because this temperature (1050˚C) corresponds to a better quality product. Low-temperature densification of PZT can be achieved by the incorporation of a small amount of P2O5 as a sintering aid without sacrificing the dielectric properties.
The preferable sintering temperature was about 1050˚C and presented the maximum bulk density of 7.62 g/cm3. The study of dielectric properties and piezoelectric of this compound in the solid as a function of temperature allows us to have high values of dielectric constant and planar electromechanical coupling factor at z = 0.04. The results of measurement of dielectric and piezoelectric properties of this material are reported in the Table 1.
REFERENCES
- G. H. Haertling, “Ferroelectric Ceramics: History and Technology,” Journal American Ceramic Society, Vol. 82, No. 4, 1999, pp. 797-818. doi:10.1111/j.1151-2916.1999.tb01840.x
- K. Uchino, “Ferroelectric Device,” Marcel Dekker, New York, 2000.
- S. Y. Cheng, S. L. Fu, C. C. Wei and G. M. Ke, “The Properties Low-Temperature Fixed Piezoelectric Ceramics,” Journal of Materials Science, Vol. 21, No. 2, 1986, pp. 571-576. doi:10.1007/BF01145525
- H. G. Lee, J. H. Choi and E. S. Kim, ” Low-Temperature Sintering and Electrical Properties of (1−x)Pb(Zr0.5Ti0.5)- O3-xPb(Cu0.33Nb0.67)O3 Ceramics,” Journal of Electroceramics, Vol. 17, No. 2-4, 2006, pp. 1035-1040. doi:10.1007/s10832-006-0384-1
- R. Mazumder, A. Sen and H. S. Maiti, “Impedance and Piezoelectric Constants of Phosphorous-Incorporated Pb- (Zr0.52Ti0.48)O3 Ceramics,” Materials Letters, Vol. 58, No. 25, 2004, pp. 3201-3205. doi:10.1016/j.matlet.2004.06.011
- G. Robert, M. D. Maeder, D. Damjanovic and N. Setter, “Synthesis of Lead Nickel-Niobate Zirconate Titanate Solid Solutions by a B-Site Precursor,” Journal American Ceramic Society,” Vol. 84, No. 12, 2001, pp. 2863-2868. doi:10.1111/j.1151-2916.2001.tb01107.x
- L. Pdungsap, S. Boonyeun, P. Winotai, N. Udomkan and P. Limsuwan, “Effects of Gd3+ Doping on Structural and Dielectric Properties of PZT (Zr:Ti = 52:48) Piezoceramics,” The European Physical Journal B, Vol. 48, No. 3, 2005, pp. 367-372. doi:10.1140/epjb/e2005-00407-9
- S. J. Yoon, A. Joshi and K. Uchino, “Effect of Additives on the Electromechanical Properties of Pb(Zr,Ti)O3-Pb- (Y2/3W1/3)O3 Ceramics,” Journal of the American Ceramic Society, Vol. 80, No. 4, 2005, pp. 1035-1039. doi:10.1111/j.1151-2916.1997.tb02942.x
- G. A. Smolenskii and A. I. Agranovskaya, “Dielectric Polarization of a Number of Complex Compounds,” Soviet Physics Solid State, Vol. 1, No. 10, 1960, pp. 1429-1437.
- F. Kulcsar, “Electromechanical Properties of Lead Titanate Zirconate Ceramics Modified with Tungsten and Thorium,” Journal American Ceramic Society, Vol. 48, No. 1, 1965, pp. 48-54. doi:10.1111/j.1151-2916.1965.tb11796.x
- N. Abdessalem and A. Boutarfaia, “Effect of Composition on the Electromechanical Properties of Pb[ZrxTi(0.9-x)- (Cr1/5, Zn1/5, Sb3/5)0.1]O3 Ceramics,” Ceramics International, Vol. 33, No. 2, 2007, pp. 293-296. doi:10.1016/j.ceramint.2005.08.008
- J. S. Kim and K. H. Yoon, “Physical and Electrical Properties of MnO2-Doped Pb(ZrxTi1−x)O3 Ceramics,” Journal of Materials Science, Vol. 29, No. 3, 1994, pp. 809-815. doi:10.1007/BF00445997
- P. Duran, J. F. Fernandez and C. Moure, “Effect of MnO Additions on the Sintering and Piezoelectric Properties of Samarium-Modified Lead Titanate Ceramics,” Journal of Materials Science Letters, Vol. 10, No. 15, 1991, pp. 917- 919. doi:10.1007/BF00724781
- Z. He, J. Ma, R. Z. Hang, “Investigation on the Microstructure and Ferroelectric Properties of Porous PZT Ceramics,” Ceramics International, Vol. 30, No. 7, 2004, pp. 1353-1356. doi:10.1016/j.ceramint.2003.12.108
- R. Sumang and T. Bongkarn, “The Effect of Excess PbO on Crystal Structure, Microstructure, Phase Transition and Dielectric Properties of (Pb0.75 Sr0.25)TiO3 Ceramics,” Taylor & Francis Group LLC, Vol. 403, No. 1, 2010 , pp. 82-90. doi:10.1080/00150191003748949
- P. Goel, S. Sharma, K. L. Yadav and A. R. James, “Structural and Dielectric Properties of Phosphorous-Doped PLZT Ceramics,” Pramanas, Vol. 65, No. 6, 2005, pp. 1127-1132. doi:10.1007/BF02705288
- A. K. Saha, D. Kumar, O. Parkash, A. Sen and H. S. Maiti, “Effect of Phosphorus Addition on the Sintering and Dielectric Properties of Pb(Zr0.52Ti0.48)O3,” Materials Research Bulletin, Vol. 38, No. 7, 2003, pp. 1165-1174. doi:10.1016/S0025-5408(03)00112-0
- O. Ohtaka, R. Von Der Mühll and J. Ravez, “Low-Temperature Sintering of Pb(Zr,Ti)O3 Ceramics with the Aid of Oxyfluoride Additive: X-Ray Diffraction and Dielectric Studies,” Journal American Ceramic Society, Vol. 78, No. 3, 1995, pp. 805-808. doi:10.1111/j.1151-2916.1995.tb08251.x
- W. Heywang, “Ferroelektrizität in Perowskitischen Systemen und Ihre Technischen Anwendungen,” Zeitschrif Angewandte Physik, Vol. 19, 1965, pp. 473-481.
- S. Babu, D. Singh and A. Govindan, “Electrical Properties of Calcium Modified PZT System,” International Journal of Computer Science et Technologie, Vol. 2, No. 1, 2011, pp. 128-131.
- IEEE Standard on Piezoelectricity, IEEE Standard 176- 1978, Institute of Electrical and Electronic Engineers, New York, 1978.
NOTES
*Corresponding author.

