Advances in Bioscience and Biotechnology
Vol.5 No.5(2014), Article ID:44488,10 pages DOI:10.4236/abb.2014.55055
A Review of the Factors Affecting the Incidence of Malignant Hyperthermia in the Greater Kansas City Area
Charles H. Williams1*, George P. Hoech2, Mark G. Zukaitis3
1Sunrise Beach, USA
2Kansas City, USA
3Gastonia, USA
Email: *chwilliams2135@sbcglobal.net
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 25 January 2014; revised 9 March 2014; accepted 26 March 2014
Abstract
Malignant Hyperthermia (MH) is a rare genetic disease. However, it is devastating when it occurs in a patient. MH is usually triggered by inhalational anesthetics and/or depolarizing muscle relaxants. Public awareness of MH has increased with the presentation of an episode on the television program, “House”, and the availability of web-based information. For over 20 years, the MH susceptible pig has been used in experiments by our group as an animal model for MH in humans. The incidence of Malignant Hyperthermia in the Greater Kansas City Area has declined dramatically since the introduction of Sevoflurane in 1992 as the anesthetic of choice (over 60% usage rate) in most surgical procedures. Historically, Malignant Hyperthermia was reported to occur at a rate of 1:50,000 during surgical procedures [1] . In the Greater Kansas City Area, Malignant Hyperthermia (MH) occurred at a rate of 1:53,636 during the 1965-1985 time period, as there were 38 MH cases in 35 patients [2] . During the past ten years (1996-2006), there were only 2 cases of MH, representing an incidence rate of 1:597,240. That decrease is an 11.13 fold (or 89%) decrease which is very significant. Despite the reduced incidence of Malignant Hyperthermia, two recent cases of MH that result in deaths in Wisconsin and Florida make it imperative that MH is recognized early and appropriate treatment initiated without delay. We have expanded our analysis of the futile cycle mechanism that underlies the MH syndrome. MH is equivalent to the rapid discharge of a battery by a short circuit.
Keywords:Sevoflurane; Malignant Hyperthermia

1. Introduction
Malignant Hyperthermia (MH) is a rare genetic disease. However, it is devastating when it occurs in a patient. MH is usually triggered by inhalational anesthetics and/or depolarizing muscle relaxants. Public awareness of MH has increased with the presentation of an episode on the television program, “House”, and the availability of web-based information.
For over 20 years, the MH susceptible pig has been used in experiments by our group as an animal model for MH in humans.
The incidence of Malignant Hyperthermia in the Greater Kansas City Area has declined dramatically since the introduction of Sevoflurane in 1992 as the anesthetic of choice (over 60% usage rate) in most surgical procedures. Historically, Malignant Hyperthermia was reported to occur at a rate of 1:50,000 during surgical procedures [2] . In the Greater Kansas City Area, Malignant Hyperthermia (MH) occurred at a rate of 1:53,636 during the 1965-1985 time period, as there were 38 MH cases in 35 patients [3] . During the past ten years (1996-2006), there were only 2 cases of MH, representing an incidence rate of 1:597,240. That decrease is an 11.13 fold (or 89%) decrease which is very significant.
2. Background
In 1984, Charles H. Williams, PhD (CHW) was contacted by Maruishi Pharmaceutical Co., Osaka, Japan, via Biodynamics, East Millstone, NJ, to study a new anesthetic agent that they were interested in developing. Sevoflurane was first synthesized in 1971 by a chemist at Baxter Labs, Chicago, USA [4] . Maruishi Pharm. had recently licensed the use of sevoflurane from Baxter Labs. A detailed and complete study of sevoflurane in 8 Malignant Hyperthermia Susceptible (MHS) pigs and 6 normal pigs was performed by CHW [5] [6] . We studied the onset time (time to induction of anesthesia), blood pressure, heart rate, Electrocardiogram (EKG), cardiac output, central venous pressure, core and rectal temperature, plus a complete blood analysis and blood gases, much as would be done in a modern hospital intensive care unit. We also measured the amount of sevoflurane in the inspired air, the expired air, and also the oxygen and carbon dioxide present in the re breathing circuit [7] .
Our conclusion was that sevoflurane was a very good anesthetic agent that had a rapid onset. The pig was anesthetized quickly, and recovered very rapidly when sevoflurane was discontinued which allowed the pig to wake up very quickly. These data were presented at the New York State Anesthesiologists meeting at the Hilton: New York City in October 1989 [6] .
Maruishi then developed sevoflurane for the Japanese market and commenced clinical studies in humans. CHW provided a notarized letter that the scientific results reported in 1984 were true science and was not influenced by the paid contract to perform the studies.
In 1990, Maruishi purchased the complete rights to sevoflurane from Baxter Labs for a total of 8 million dollars payable in four payments of two million dollars each year over a four year period [8] . They purchased the patent rights to the one-step synthesis procedure. This outright purchase prevented anyone, including Baxter Labs from making sevoflurane by the one step process and selling it in any market. Subsequently, Maruishi Pharm. entered into an agreement with Abbott Labs, Chicago and Baxter Labs, to have Abbott Labs as the sole distributor of sevoflurane in the USA [8] . A recent court case reveals that sevoflurane brought in 200 million dollars in sales in the USA in 2000 to Abbott Labs. Sevoflurane represents 57.7% of all anesthetic sales in the USA [8] .
As would be expected, Baxter Labs was distressed to discover it had sold the rights to the #1 anesthetic in the USA. As a competitive strategy, Baxter Labs bought Ohmeda (Ohio Medical), a defunct US anesthesia company, which had developed a 2-step process for making sevoflurane and obtained a patent that would then allow the manufacture of sevoflurane by a 2-step process that did not utilize the 1-step patent process Baxter had sold to Maruishi. In litigation with Maruishi and Abbott, the court ruled that Baxter could not sell their generic version of sevoflurane until December 2005 [8] . Subsequently, several problems have appeared from the use of generic versions of sevoflurane [9] .
3. Data Collection
Anesthesia data was requested from 22 Hospital and Surgicenters in the Greater Kansas City Area for the year 2006. Five Hospitals did not respond (Table 1 and Figure 1).
Table 1. Greater Kansas City Area data 2006.
Note: Seventeen out of 22 surgical centers/departments in the Greater Kansas City Area responded with data for this study.
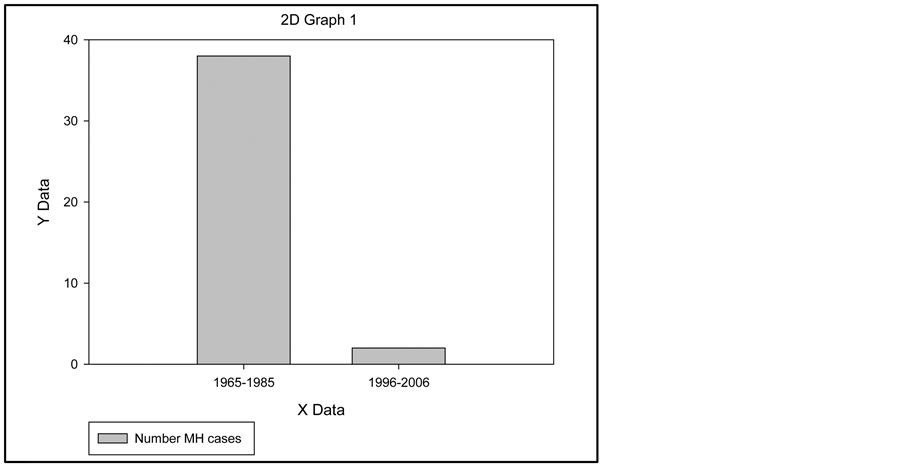
Figure 1. Number of MH cases in the Greater Kansas City Area.
During 2006, 119,448 general anesthetics were administered in the 17 surgical suites surveyed. The data we obtained were: 1) Total inhalation general anesthetics: 119, 448 × 10 yrs = 1,194,480. 2) MH incidence: 2:1,194,480 = 1:597,240. We extrapolated the 2006 number of potent inhalational anesthetics for this period of 10 years which gives a total of 1,194,448 anesthetics. The incidence of MH is 1:597, 240. The two MH cases in the Kansas City area during 1996-2006 were diagnosed early, treated with dantrolene sodium, survived the MH episode, and had a full recovery.
The administration of anesthetics used was: 1) Sevoflurane: 72,061; 2) Sevoflurane + Succinylcholine: 13,673; 3) Percent Sevoflurane + Succinylcholine: 18.9%; 4) Other Inhalational Agent + Succinylcholine: 13,727.
A total of 72,061 Sevoflurane anesthetics were administered accounting for 60 per cent of the general anesthetics. All other potent inhalation anesthetics totaled 47,387 accounting for the remaining 40 per cent. 19 percent of each group received succinylcholine. We extrapolated the surgical case data back for a period of 10 years to obtain data for the MH cases.
We conducted a MedLine search of the literature on MH cases with Sevoflurane and found 29 reported cases of MH during the use of sevoflurane anesthesia. Succinylcholine was used as the muscle relaxant in all of these MH cases; all patients were treated with Dantrolene and survived.
Outside the Greater Kansas City Area, the MH case of SPN of Lacrosse, WI was brought to our attention [10] . The reporting article did not state whether succinylcholine was utilized and which potent inhalational agent was used. SPN was treated with Dantrolene, but did not survive.
By 1972, CHW had developed the premise that depolarizing muscle relaxants were a primary cause of MH in Malignant Hyperthermia Susceptible (MHS) pigs along with Halothane anesthesia [11] . That premise is illustrated in Figure 2.

Figure 2. FISHER atomic model of d-Tubocurarine, Pancuronium, and Succinylcholine. Note the 10.4 Angstrom spacing of the quaternary ammonium groups which is believed to be necessary for pharmacologic activity.
Hoech, et al. further developed this premise by using Metubine Iodide to protect the MHS pig from Halothane induced MH [12] . A pharmacologic dosage was required to achieve this protective effect. Buzello, et al. extended this notion by showing that Vecuronium, a non-depolarizing muscle relaxant was safe to use in MHS pigs [13] . However, a clinical dose of Vecuronium did not protect the MHS pigs from a triggering dose of Halothane. CHW, et al. also showed that Atracurium, another non-depolarizing muscle relaxant did not trigger MH in MHS pigs [14] . Nor did a clinical dose of Atracurium protect the MHS pigs from a triggering dose of Halothane. Organon 9426 (Rocuronium), another non-depolarizing muscle relaxant, did not trigger MH in MHS pigs [15] , but did have the unusual property of completely blocking the development of MH in MHS pigs from use of Halothane at a clinical dosage level [15] .
We have developed the idea that a sodium channel genetic defect is the cause of MH. This premise has been presented in a full length publication [1] .
4. Discussion
Since the introduction of Sevoflurane into clinical anesthesia practice about 1992, there has been an 11.13 fold (or 89%) decrease in MH cases in the Greater Kansas City Area surgical suites during the 1996-2006 period. We attribute the decrease to four factors: 1) use of Sevoflurane as the anesthetic of choice. 2) decline in the use of succinylcholine. 3) diligent intake questioning as to the patient’s Family History, Medical History, and physical findings, and if any factor is suggestive of MHS, succinylcholine and potent inhalational agents are customarily avoided; 4) the prophylactic use of Dantrolene in select cases, which has probably prevented clinical MH cases. (The relevant items in the Family History, Medical History, and pertinent physical findings are described in the MHAUS Adverse Metabolic Reaction to Anesthesia Report Form [16] .) As a result of these pro-active changes in the practice of anesthesia, there have only been two MH cases in the Greater Kansas City Area during the past 10 years. This contrasts markedly with the thirty-eight MH cases during a 20 year period (1965-1985). If you divide the 38 cases in 20 years (1.9/year) by 2 cases in ten years (0.2/year), the incidence of MH has declined to 11% of its previous rate of occurrence due to the present procedures, with no fatal incidents.
Our experimental studies of sevoflurane showed that over two hours of sevoflurane anesthesia is required to trigger MH in highly susceptible MHS pigs. The MH syndrome that developed was very mild and easily reversed by dantrolene. All of our experimental MHS pigs were recovered, held for one week to clear all drugs, and rechallenged with Halothane to prove a second time that they were MH susceptible [6] .
We believe that the complete elimination of succinylcholine from the surgical suite could further reduce the incidence of MH. All of the reported cases of MH in human patients were accompanied by succinylcholine usage as the muscle relaxant during sevoflurane anesthesia. Our experimental work with MHS pigs clearly demonstrated that succinylcholine was capable of triggering MH by itself without the use of halothane [11] . We also demonstrated that halothane was capable of triggering MH in MHS pigs. These facts alone strongly suggest that the elimination of these two agents should further decrease the incidence of MH in human patients. Physicians who use these agents are playing a game of chance with their patient’s life because sporadic cases escape detection and show up as a fully developed MH case. Physicians who continue to use succinylcholine should complete a critical risk vs. benefit analysis of each patient before using succinylcholine. If succinylcholine is used, the treating physician should be prepared for the possibility of a MH reaction and have appropriate remedial treatments available.
The selective identification and treatment of MHS patients with alternative drugs may have played a role in decreasing the number of MH cases in the Greater Kansas City Area, however, because we do not have a rapid, accurate test for MH susceptibility in human patients that can be applied prior to surgery, there will be MHS patients that slip through the personal history screening process.
We have not observed a large shift in the population of the Greater Kansas City Area that could account for the decreased incidence of MH cases.
Two recent fatal cases of MH in young people, SPN from La Crosse, WI and SJK, [17] -[21] of Boca Raton, FL highlight the fact that MH in human patients remains a life threatening situation that needs our full attention.
Anesthesia Record SPN, La Crosse, WI dated 12 Jan 2005:
1) Preop Evaluation: The only comment of note was patient’s preoperative anxiety.
2) Pulse: Initial pulse rate on anesthesia record was 82/min. After 1400 hrs. the pulse rate rose gradually to 120/min. at 1625 hours. The last recorded pulse rate on the record was 132/min. at 1625 hours. Labetelol 5 mg. at 1304 hrs. and 7.5 mg. at 1430 hrs. was given to decrease BP and caused slight decreases in pulse rate.
3) End Tidal CO2 rose from 48 Torr at 1200 hrs. After two hours of anesthesia ETCO2 was in mid 50s and after four hours in upper 60s. At 4 hrs. 15 min. ETCO2 was 80 Torr, at 4 hrs. 30min. ETCO2 was 110 Torr.
4) Airway: Anesthesia was administered via a LMA (Laryngeal Mask Airway). It is not evident whether respirations were controlled, spontaneous or assisted. I could not determine respiratory rate, Tidal volume or minute volume.
5) Temperature was monitored via axillary temperature probe. Temperature started at 32˚C and in the first hour rose to 34˚C, rising to 36˚C at 1600 hours (I could find no record of the core temperature being measured.).
6) Timeliness of MH diagnosis: The presumed triggering agent Sevoflurane was discontinued at 1629 hours. MH cart was brought to operating room at the same 1629 hours. The warming blanket was turned off at 1540 hours. There was not a timely diagnosis of the MH metabolic process.
Unfortunately, SPN died after several hours of resuscitative treatment. (Drs. Henry Rosenberg and Al Rothstein stated in the APSF Newsletter Summer 2006 Vol. 21, No.1, p. 32 regarding this patient: “By the time the anesthesia team determined that MH was occurring and Dantrolene should be given, the train had left the station and was racing down the tracks.”)
The temperature monitoring via axillary (armpit) did not provide an accurate and reliable temperature readout of core or rectal temperature. We have published data from our MHS pig experiments showing that the skin ceases to radiate heat whenever MH develops [22] . The heat produced in MH comes from muscle mass so temperature monitoring must be internal rather than external [23] . This observation is further supported by the fact that esophageal temperature was 110.8˚F after the patient was intubated with a # 8 O.T in the SPN case (Figure 3).
SJK, case number: 08-0366, West Palm Beach, FL. [18] “[SJK] was given succinylcholine, she developed muscle rigidity and her body temperature rose. She was transferred to the nearest emergency department. She developed disseminated intravascular coagulation and multi-organ failure and died at the hospital.”
5. Conclusions
The Malignant Hyperthermia Susceptible pig has been a superb animal model for human Malignant Hyperthermia. See CHW’s extensive publications on this subject [23] -[28] . Given that extensive human testing of the MH response is not possible, the animal model remains the best tool for evaluating the suitability of various anesthetic treatments of human patients. The available data suggest that MH human patients have the same negative response to succinylcholine and halothane as the animal model.
We now have a well tested anesthetic, Sevoflurane, which has been used for many years in millions of people that is a weak trigger for Malignant Hyperthermia. This is borne out by only 29 MH cases in the literature and a 19 fold (or 89%) decrease of MH incidents in the Greater Kansas City Area.
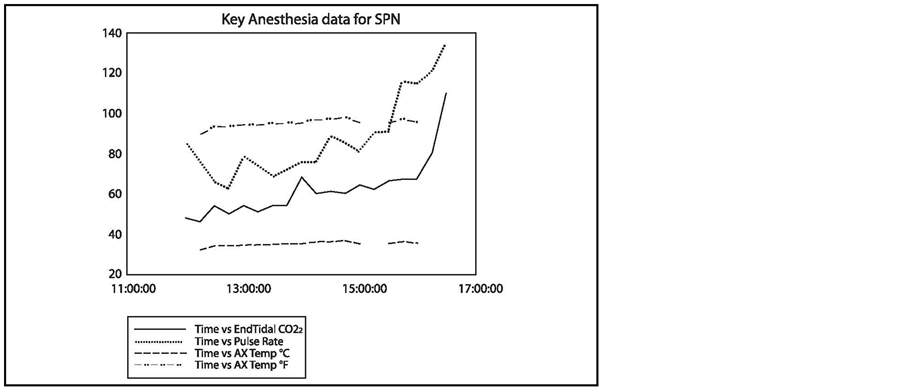
Figure 3. Key Anesthesia data for SPN. Data were obtained from the anesthesia record of SPN.
We have three muscle relaxants, Vecuronium, Atracurium, and ORGANON 9426 (Rocuronium) that are known to be non triggering agents in the MHS pig and the available data suggest that they are safe to use in humans. Rocuronium is the only muscle relaxant that can also prevent MH in pigs when challenged with Halothane or succinylcholine.
6. Futile Cycle Analysis
The rapid rate of heat production in muscle is driven by a futile cycle, most likely by a rapid membrane depolarization of the 90 mv membrane potential, by a defective sodium channel. The genetically defective sodium channel is evidenced by an action potential at the myoneural junction that is higher in amplitude and of longer duration than a normal action potential [29] . This electrical activity is equivalent to a short circuit in a battery that produces copious amounts of heat and thereby drives the metabolism to a maximum rate of over 10× normal.
Acknowledgements
Note: This manuscript was presented at the Amer Physiol Soc Fall meeting at Hilton Head, SC as 20.3 on board # 67 on 26 Sept 2008. The abstract is in The Physiologist: 51(6): 54, Dec 2008.
The graphs were prepared with Sigma Plot v.11 and translated by Advantage Systems Inc., Camdenton, MO to the current format.
We thank Kate Andersen, University of Missouri Medical School Library, Columbia, MO for the computer search online for the Malignant Hyperthermia-Sevoflurane published works.
We thank Gregory D. Williams, JD and Nathan C. Williams, JD for editorial revision of this manuscript.
We thank Dolores B. Williams, BS for typing and revising the manuscript.
References
- Williams, C.H. (2014) Malignant Hyperthermia: A Runaway Thermogenic Futile Cycle at the Sodium Channel Level. Advances Bioscience Biotechnology, 5, 197-200. http://dx.doi.org/10.4236/abb.2014.53025
- Britt, B.A. (1987) A History of Malignant Hyperthermia. In: Malignant, H., Ed., BA Britt, Martinus Nijhoff Publishing, Boston, xi-xxii.
- Zukaitis, M.G., Hoech, Jr., G.P. and Robinson, J.D. (1986) Malignant Hyperthermia—Pre and Post Dantrolene: A Survey of the Greater Kansas City Area from 1965-1985: 15 Sept 1965. January 1986 Meeting of the Rocky Mountain Traumatoligic Society in Aspen,
- Wallin, R.F. and Napoli, M.D. (1971) Sevoflurane (Fluro-methyl-1,1,1,3,3,3,-hexafluro-2-propyl ether) a New Inhalational Agent. Fed Proceed, 30, 442.
- Williams, C.H., et al. (1989) Cardiac Performance and Hemodynamics in Malignant Hyperthermia Susceptible and Normal Pigs during Sevoflurane Anesthesia. Attachment #1.
- Williams, C.H., Stovall, R.M., Dozier, S.E., Watts, M.T., Bayless, P.A., Marvasti, M.A., Farias, M., Ekery, D.D., Daley, I.W. and Satoh, N. (1988) Cardiac Performance and Hemodynamics in Malignant Hyperthermia Susceptible and Normal Pigs during Sevoflurane Anesthesia. Poster Presentation at the New York State Society of Anesthesiologists Meeting, Hilton, New York City, 10 December 1989,
- Watts, M.T., Escarzaga, M. and Williams, C.H. (1992) GTas-Chromatographic Headspace Analysis of Sevoflurane. Journal of Chromatography B: Biomedical Sciences and Applications, 577, 289-298. http://dx.doi.org/10.1016/0378-4347(92)80250-T
- Abbott Laboratories v. Baxter International Inc. (2002) WL 467147 (N.D.Ill. 2002).
- Kharasch, E.D. (2007) Sevoflurane: The Challenges of Safe Formulation. APSF Newsletter Fall, 22, 48.
- Rosenberg, H. and Rothstein, A. (2006) Malignant Hyperthermia Death Holds Many Lessons. APSF Newsletter: Summer, 21, 32-34.
- Williams, C.H., et al. (1977) Proceedings of the Focus on Malignant Hyperthermia Symposium, Wausau, 13 September, 3 Chapters. EO Henschel, Appleton Century Crofts, New York.
- Hoech, Jr., G.P., Roberts, J.T., Williams, C.H., Waldman, S.D., Simpson, S.T. and Trim, C.M. (1980) Prevention of Porcine Malignant Hyperthermia with Metocurine. In: Thermoregulatory Mechanisms and Their Therapeutic Implications. 4th International Symposium Pharmacology of Thermoregulation, Oxford, 137-141, Karger:Basel.
- Buzello, W., Williams, C.H., Chandra, P., Watkins, M.L. and Dozier, S.E. (1985) Vecuronium and Porcine Malignant Hyperthermia. Anesthesia & Analgesia, 64, 575-279. http://dx.doi.org/10.1213/00000539-198505000-00012
- Williams, C.H., Dozier, S.E., Ilias, W.K. and Fulfer, R.T. (1985) Porcine Malignant Hyperthermia: Testing of Atracurium in MH Susceptible Pigs. Anesthesia & Analgesia, 64, 112.
- Williams, C.H., Chandra, P., Serda, R. and Martinez, M. (1992) Malignant Hyperthermia Induction in Susceptible Swine Following Exposure to Organon 9426. Abstracts S350. Anesthesia & Analgesia, 74, S1-S365.
- (2006) MHAUS AMRA Report Version 9.1.
- (2008) Milwaukee Journal Sentinel: JS Online: Only 20, Athlete Had Zest for Life (Steven Nook). By Amy Rabideau Silvers.
- Stephanie Kuleba, Case # 080366, Palm Beach Medical Examiner’s Office.
- (2008) The Informed Patient: A Fresh Focus on a Rare Risk of Anesthesia. The Wall Street Journal: 4/30/2008, Laura Landro.
- Medical Examiner’s Office (2008) West Palm Beach, Stephanie J. Kulebea, Case # 080366:21 May 2008.
- (2008) Making Everlasting Memories, Worldwide Memoralization—Stephanie Jude Kuleba: Guest Book: 10/24/2008.
- Williams, C.H., Houchins, C. and Shanklin, M.D. (1975) Energy Metabolism in Pigs Susceptible to the Fulminant Hyperthermia-Stress Syndrome. British Medical Journal, 3, 411-413, Errata 604.
- Cark, M.G., Williams, C.H., Pfeifer, W.F., Bloxham, D.P., Holland, P.C., Taylor, C.A. and Lardy, H.A. (1973) Accelerated Substrate Cycling of Fructose-6-Phosphate in the Muscle of Malignant Hyperthermic Pigs. Nature, 245, 99-101. http://dx.doi.org/10.1038/245099a0
- Williams, C.H. (1974) The Development of an Animal Model for the Fulminant Hyperthermia-Porcine Stress Syndrome. Proceedings of the Focus on Malignant Hyperthermia Symposium, Wausau, 13 September 1974, 117-140.
- Williams, C.H., et al. (1977) The Fulminant Hyperthermia-Stress Syndrome: Genetic Aspects, Hemodynamic and Metabolic Measurements in Susceptible and Normal Pigs. Proceedings of 2nd International Symposium on Malignant Hyperthermia, Denver, 1-3 April 1977, 161-173.
- Williams, C.H., Farias, M., Marvasti, M.A., Knight, A.B., Ekery, D.D., Daley, I.W. and Zayed, I. (1990) Malignant Hyperthermia Induction in Susceptible Swine Following Exposure to Arduan. Anesthesia & Analgesia, 70, S433. http://dx.doi.org/10.1213/00000539-199002001-00433
- Williams, C.H., et al. (1986) Metabolic Rate in Malignant Hyperthermia Pigs. In: Swine in Biomedical Research, Vol. 1, Ed., ME Tumbleson, Plenum, New York, 573-582.
- Williams, C.H. (1988) Experimental Malignant Hyperthermia. Springer Verlag, New York.
- Steiss, J.E., Bowen, J.M. and Williams, C.H. (1981) Electromyographic Evaluation of Malignant Hypermia Susceptible Pigs. American Journal of Veterinary Research, 42, 1175-1178.
Attachments
Sevoflurane abstract: Williams, CH et al. 1985 submitted to IARS, 27 July 1989. However, it was rejected for presentation at the Fall 1989 meeting.


NOTES

*Corresponding author.


