Advances in Bioscience and Biotechnology
Vol.1 No.2(2010), Article ID:2087,7 pages DOI:10.4236/abb.2010.12016
Optimization of media constituents for the production of lipase in solid state fermentation by Yarrowia lipolytica from palm Kernal cake (Elaeis guineensis)
![]()
1Center for Biotechnology, Department of Chemical Engineering, College of Engineering, Andhra University, Visakhapatnam, India;
2Department of Biotechnology, GITAM Institute of Technology, GITAM University, Gandhinagar, Rushikonda, Visakhapatnam, India;
3M.R.College of Pharmacy, Phool Baugh, Vizianagaram, India;
4Department of Biotechnology, Anil Neerukonda Institute of Technology and Sciences (ANITS), Sangivalasa, Bheemunipatnam, Visakhapatnam, India.
Email: saratbabuimandi@gmail.com; saratbabu_imandi@yahoo.com; sitakaranam@gmail.com; ghrao77@gmail.com
Received 9 March 2010; revised 20 March 2010; accepted 21 March 2010.
Keywords: Lipase; Palm Kernal cake; Optimization; Plackett-Burman design; Yarrowia lipolytica
ABSTRACT
The production of extra cellular lipase in Solid State Fermentation (SSF) using Yarrowia lipolytica NCIM 3589 with Palm Kernal cake (Elaeis guineensis) has been studied. Different parameters such as incubation time, inoculum level, initial moisture content, carbon level and nitrogen level of the medium were optimized. Screening of various process variables has been accomplished with the help of Plackett-Burman design. The maximum lipase activity of 18.58 units per gram of dry fermented substrate (U/gds) was observed with the substrate of Palm Kernal cake in four days of fermentation.
1. INTRODUCTION
Lipases (triacylglycerol acylhydrolases, EC 3.1.1.3) are one of the most important classes of industrial enzymes. They hydrolyse triglycerides into diglycerides, monoglycerides, glycerol and fatty acids. In recent years, there has been an increasing interest in the study of lipases mainly due to their potential applications as medicines (digestive enzymes), food additives (flavour modifying enzymes), clinical reagents (glyceride-hydrolysing enzymes) and cleaners (detergent additives) [1]. Additionally, a promising application field for lipases is in the biodegradation of plastics such as polyhydroxyalkanoates (PHA) and polycaprolactone (PCL) [2,3]. Lipases would be economically manufactured in solid state fermentation.
Solid state fermentation (SSF) is defined as the fermentation of solids in the absence of free water; however, the substrate must possess enough moisture to support the growth and metabolism of microorganisms. Recently, several reports have been published indicating the application of this culture in upgrading the food and industrial wastes and in the production of fine chemicals and enzymes. The utilization of by-products and wastes from food and industrial sources has several advantages over submerged fermentation such as superior productivity, simple techniques, reduced energy requirements, low wastewater output, improved product recovery and the reduction in production costs [4]. In SSF, any type of substrate, including industrial wastes, could be used to enhance the production of enzymes because of their richness in fatty acids, triacylglycerols and/or sugars. The use of cheap raw materials would diminish the operating costs of the process. Moreover, total capital investment for lipase production has been reported to be significantly lower in solid state fermentation than in submerged fermentation [5]. Most studies on lipolytic enzymes production with bacteria, fungi and yeasts have been performed in submerged fermentation; however, there are only few reports on lipase synthesis in solid state fermentation. In recent years, considerable research has been carried out using agricultural wastes, which are renewable and abundantly available to produce valueadded products. For example babassu oil cake [6], olive cake and sugar cane bagasse [7], gingelly oil cake [8], wheat bran [9], rice bran [10] and Jatropha curcas seed cake [11] have been used as the substrates for lipase production.
The global production of palm kernel cake (PKC), a by-product of oil extraction from the nut of the palm tree, Elaeis guineensis, is ever increasing due to the tremendous growth of the oil palm industry in many parts of Asia and Africa [12]. World producers of palm kernel include Malayasia, Indonesia, Nigeria, Colombia, India, and Thailand. In Malaysia alone, about 3 million tonnes of palm kernel were produced in 2001 producing about 1.4 million tonnes of palm kernel oil together with 1.6 million tonnes of PKC as its byproduct. Palm trees are widely cultivated in the state of Andhra Pradesh, India. The low cost and availability of PKC have recently generated much interest in its potential use in fish diets. In recent years, the cost of imported feed ingredients used in commercial aqua feeds in many developing countries in Asia has continued to rise due to increased global demand and fluctuation in foreign currency exchange. The rising cost of imported ingredients such as fish meal, soyabean meal, corn flour and wheat flour greatly cuts into the profit margins of local fish farmers to such an extent that many local aquaculture enterprises are no longer profitable. This is especially true for the culture of lower-value fish species such as catfish, tilapia and carps. There is currently a great interest within the animal feed industry to reduce costs by using locally available feed ingredients like Palm kernel cake.
The present study aimed at developing appropriate bioprocesses for the utilization of Palm kernel cake. Palm kernel contains 16% protein, 15% crude fiber, 48% carbohydrates and 4% ash. This composition is appropriate to support good microbial growth and enzyme production. Use of Palm kernel cake as a substrate for solid state fermentation thus appears logical and worth exploring. SSF has been employed to grow microbes and to produce extracellular lipase [9,13]. Even though large number of agricultural/food residues have been worked out as suitable substrates for SSF [14], there is no report on SSF of Palm kernel cake so far to the best of our knowledge.
Hence an attempt is made in this paper to utilize the PKC as a substrate for the production of lipase by solid state fermentation. It was under taken to optimize the key process variables, including incubation time, inoculum level, initial moisture content, carbon level, and nitrogen level of the medium for the production of lipase using this oil cake under SSF.
2. MATERIALS AND METHODS
2.1. Substrate
PKC, obtained from a local palm oil distillery (Jeevan Enterprises) unit of Vizianagaram, India, was used as the substrate. It was dried at 60˚C for 72 h to reduce the moisture content to around 5%, and ground to the desired size (2 mm).
2.2. Chemical Analysis of PKC
Palm kernel cake is the major byproduct of oil palm kernel oil extraction. PKC, when powdered, gives meal or flour. As such there is no significant alteration in the content of nutrients when the cake is converted to meal or flour. Palm kernel meal contains 20% protein which is the lowest among the oil seed meals. Carbohydrates are the major constituents of palm kernel meal than most of the oil seed meals. It is characterized by higher fiber content. The contents of nitrogen free extract (NFE) and crude fiber depend on the method of oil extraction from palm kernel. PKC contains a good amount of minerals: 0.69% phosphorus, 0.42% potassium, 0.29% calcium, and 0.017% iron [15]. The chemical composition of PKC was presented in Table 1.
2.3. Microorganism and Growth Conditions
Yarrowia lipolytica NCIM 3589, obtained from National Chemical Laboratory, Pune, India, was used throughout the study. The culture was maintained on MGYP slants having the composition (%): malt extract 0.3, glucose 1.0, yeast extract 0.3, peptone 0.5 and agar agar 2.0. The pH of the medium was adjusted to 6.4–6.8 and culture was incubated at 30˚C for 48 h. Sub-culturing was carried out once in 2 weeks and the culture was stored at 4˚C.
2.4. Inoculum and Media Preparation
The Yarrowia strain was cultivated in a medium containing peptone 5 g, yeast extract 3 g and sodium chloride 3 g per liter of distilled water. The cells were cultivated in this medium at 30˚C on a shaker at 200 rpm for 24 h [16]. Ten grams of substrate was weighed into a 250 ml Erlenmeyer flask and to this a supplemental salt solution was added to the desired moisture level. The composition of the salt solution was as follows (% w/w): KH2PO4: 0.1; MgSO4.7H2O: 0.05; CaCl2: 0.01; NaCl: 0.01; H3BO3: 0.00005; CuSO4.5H2O: 0.000004; KI: 0.00001; FeCl3.4H2O: 0.00002; ZnSO4.7H2O: 0.00004; MnSO4.H2O: 0.00004 [17]. Out of various compounds tried, glucose and urea were ultimately selected as carbon and nitrogen sources respectively as per the Plackett-Burman design. The contents were thoroughly mixed and autoclaved at 121˚C (15 psi) for 20 min.
2.5. Solid State Fermentation and Extraction of Lipase
The sterilized substrate along with media as shown in the above section was inoculated with 2 ml of inoculum.
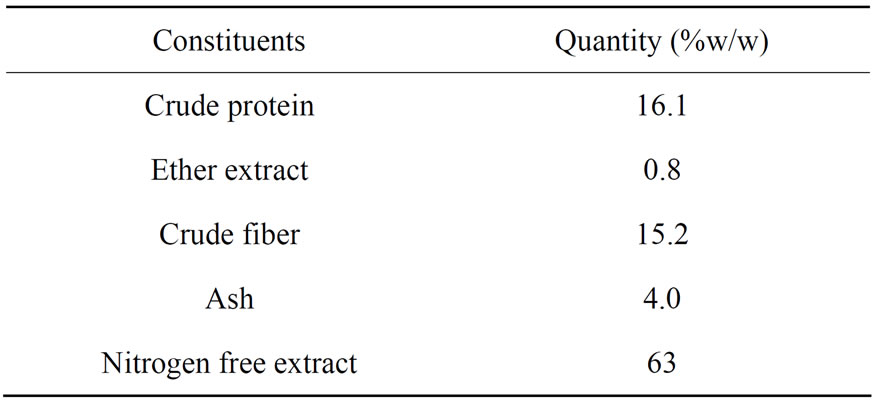
Table 1. Chemical composition of PKC [17].
The contents were mixed thoroughly and incubated in a slanting position at 30˚C. All the experiments were carried out in duplicate and samples were withdrawn after 4 days of incubation. The crude enzyme from the fermented material was recovered by simple extraction method. For this, the fermented substrate was mixed thoroughly with 100 ml of distilled water and the contents were agitated for 1 h at the room temperature in a rotary shaker at 150 rpm. At the end of extraction, the liquid was filtered off through Whatman No.1 filter paper and the resulting clear filtrate was used for lipase assay.
2.6. Lipase Assay
Lipase activity was assayed by the colorimetric method of Winkler and Stuckmann, [18] by measuring the micromoles of 4-nitrophenol released from 4-nitrophenyl palmitate. One unit of lipase activity was defined as the enzyme amount that releases 1μmol of p-nitrophenol per minute under assay conditions. Enzyme activity was expressed as units/gram of the initial dry substrate (U/gds).
3. EXPERIMENTAL DESIGN AND OPTIMIZATION
3.1. Plackett-Burman Design
The purpose of this optimization step was to identify which ingredients of the medium had a significant effect on lipase activity. The Plackett–Burman [19] statistical experimental design is a versatile method for screening the important variables. The total number of experiments to be carried out is K + 1, where K is the number of variables. Each variable is represented at two levels, high and low denoted by (+) and (–) respectively. The statistical software package STATISTICA 6.0 (Stat-Ease Inc., Tulsa, OK, USA) was used for analyzing the experimental data.
The effect of each variable on lipase activity was calculated by using the following equation:
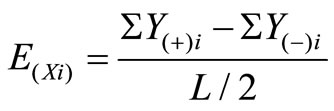 (1)
(1)
where E(Xi) is the effect of levels of the tested variables, Y(+)i and Y(–)i are the lipase activity from the experimental runs in which the variables being tested are added to the medium at their maximum and minimum levels respectively and L is the number of experiments carried out. When the value of concentration effect (E(Xi)) of the tested variable is positive, the influence of the variable is greater at the high concentration, and when it is negative, the influence of the variable is greater at the low concentration.
4. RESULTS AND DISCUSSION
4.1. Effect of Incubation Time
The incubation time is an important factor for the production of extracellular lipase by the microorganisms [20]. The amount of lipase activity was observed daily during a period of five days. The maximum lipase activity was observed on fourth day as shown in Table 2. At longer incubation periods, the lipase activity decreased which might be due to the depletion of nutrients, accumulation of toxic end products, and the change in pH of the medium, or loss of moisture. Several researchers have reported different incubation periods for optimal lipase production. Maximum lipase activity was achieved after 48 h of incubation by Ul-Haq et al., [21] with Rhizopus oryzae. Cordova et al., [7] reported the maximum lipase activity by R.pullis after 24 h of incubation using the mixture of olive oil cake and sugar cane bagasse as substrate. In another study, the maximum lipase activity by Aspergillus niger occurred after 5 days of incubation [9]. Benjamin and Pandey, [22] obtained maximum production of lipase by Candida rugosa after 3 days of incubation.
4.2. Effect of Inoculum Level
Different levels of the inoculum were tried to study their effect on lipase activity (Figure 1) so as to find an

Table 2. Effect of incubation time on lipase activity.
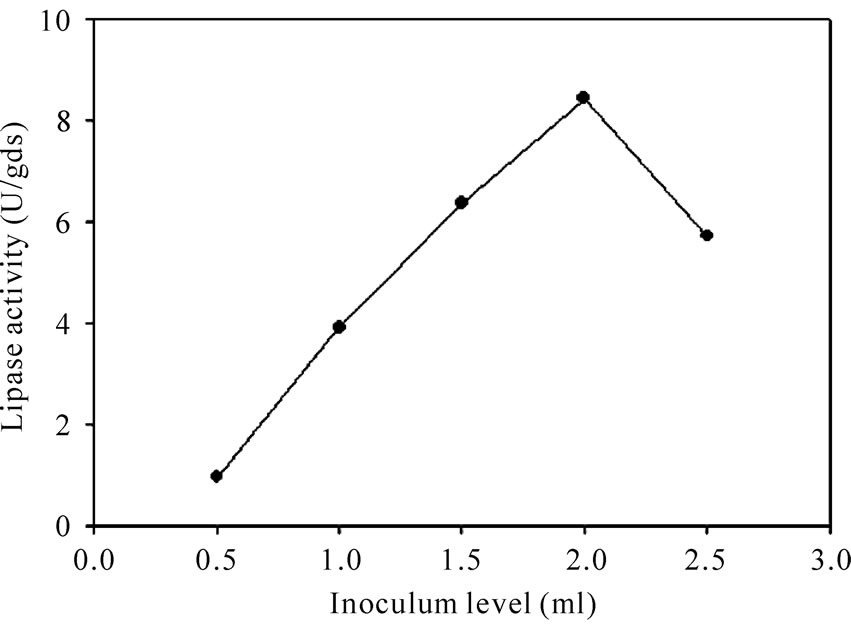
Figure 1. Effect of inoculum level on lipase activity.
optimum inoculum level in the fermentation process. A lower inoculum may give insufficient biomass causing reduced product formation, whereas a higher inoculum may produce too much biomass leading to the poor product formation [23]. In this study, the maximum lipase activity (8.43 U/gds) was obtained with 2 ml (20% v/w) inoculum level. Various investigators have used different levels of inoculum for lipase production employing different microorganisms. Maximum lipase production by Rhizopus oligosporus was achieved by UlHaq et al., [21] with 1 ml of inoculum. An inoculum concentration of 1.07 × 108 spores/10 g of substrate were found to be optimal for lipase production by Aspergillus niger [8]. Diaz et al., [13] used an inoculum concentration of 3 × 107 spores/g of dry substrate for maximum lipase production by R. homothallicus.
4.3. Effect of Initial Moisture Content
Moisture content of substrate plays a vital role for the microbial growth and for effecting biochemical activities in SSF [24]. The maximum lipase activity was obtained at 70% v/w initial moisture content as presented in Figure 2. Lipase production was decreased at very higher moisture content which may be attributed to the decrease in porosity and hence decrease in gaseous exchange leading to suboptimal growth and less enzyme production as indicated by Silman et al., [25]. Less lipase activity was observed at lower moisture content due to the reduction in the solubility of nutrients of the substrate, lowers the degree of swelling, and creates higher water tension as suggested by Perez-Guerra et al., [26]. Mahanta et al., [11] reported initial moisture content at 50% of substrate as ideal for lipase production using Jatropha curcas seed cake as the substrate.
4.4. Identification of Important Nitrogen Source Using Plackett-Burman Design
A total of ten nitrogen sources were screened through twelve experimental runs. The experimental plan and

Figure 2. Effect of initial moisture content on lipase activity.
corresponding lipase production were shown in Table 3. The pareto graph (Figure 3) was used to show the effect of all nitrogen sources (both organic and inorganic) on lipase production. A p-value of less than 0.05 for the six variables viz., urea, peptone, malt extract, yeast extract, (NH4)2SO4, and NH4H2PO4 indicates that they are significant. From the statistical analysis, it was also found that lipase production was affected by the above six nitrogen sources as evident from their F-values and p-values as shown in Table 4. In addition, the coefficient of determination (R2) of the model was found to be 0.9992 which explains the 99.92% variability of the data. Urea had the confidence level more than 95% in comparison to the other variables and thus considered to be highly significant for lipase production. Here one dummy variable (DV) was employed to evaluate the standard errors of the experiment. Further studies were conducted by taking urea at different concentrations. Figure 4 shows that 1.5 (%w/w) of urea yielded maximum lipase activity. However, at higher levels the
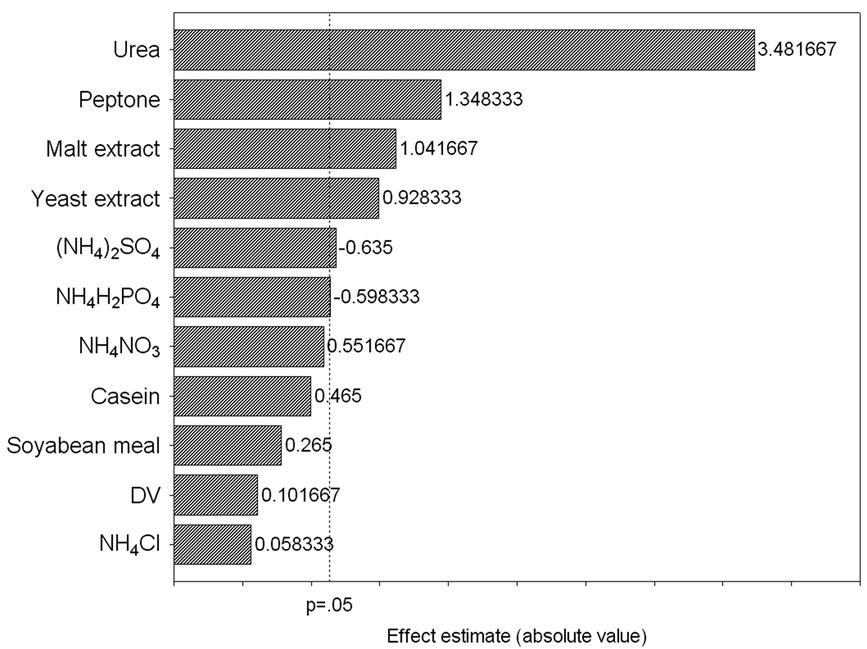
Figure 3. Pareto graph showing effect of various nitrogen sources on lipase production based on the observation of Plackett–Burman design.
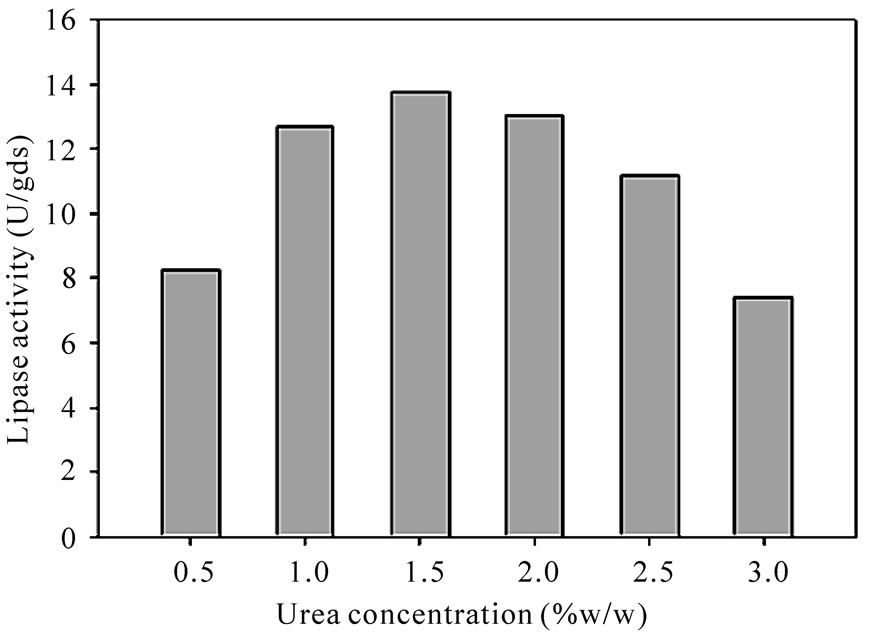
Figure 4. Effect of urea concentration on lipase activity.
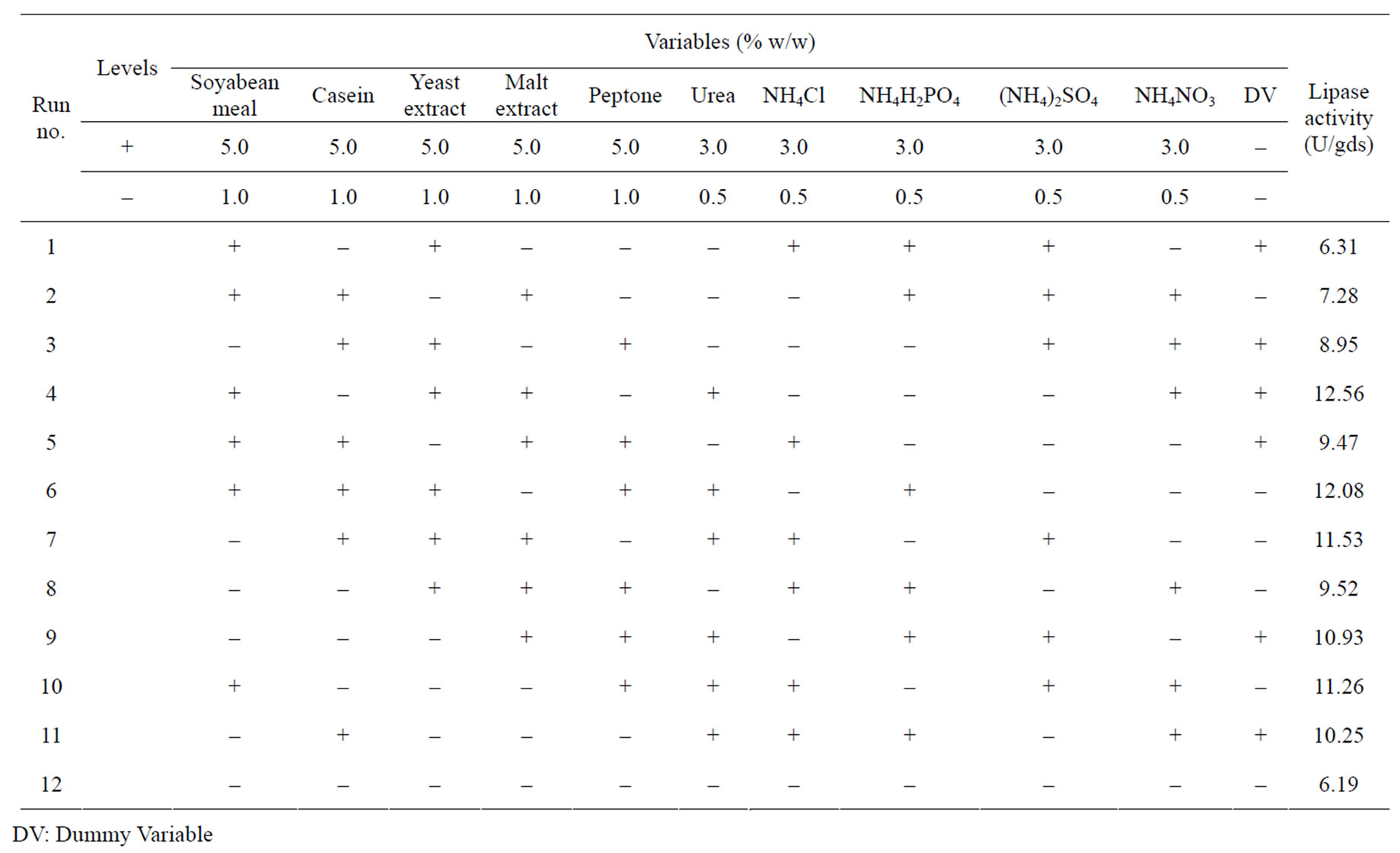
Table 3. Plackett–Burman experimental design matrix for screening of various nitrogen sources for lipase production.
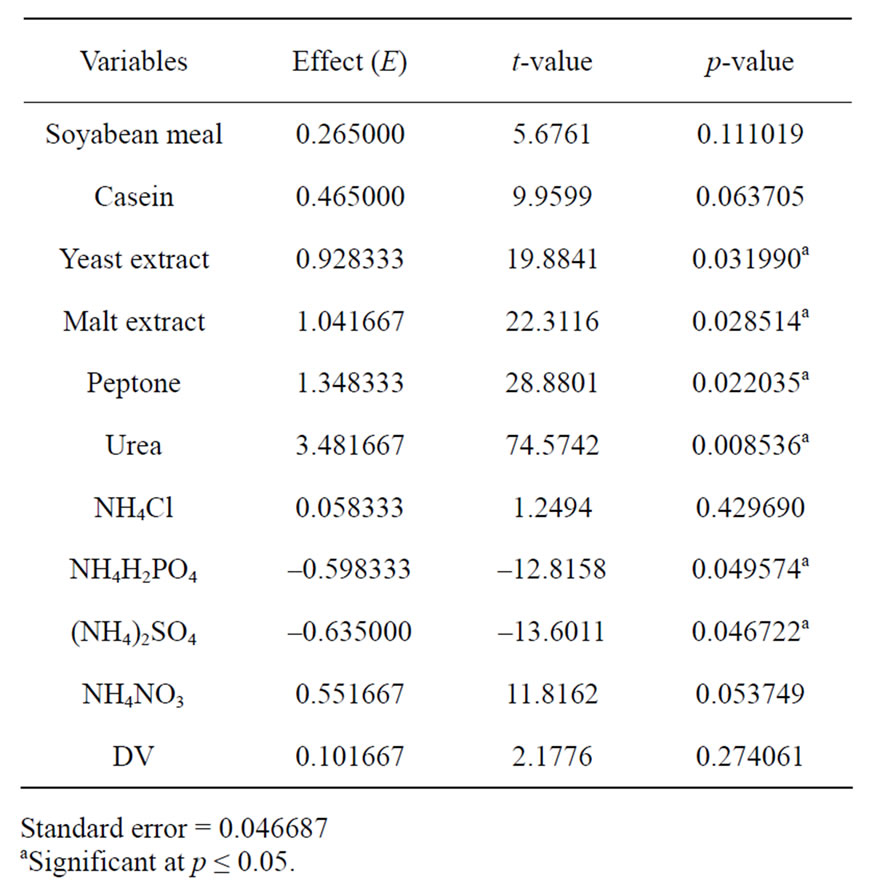
Table 4. Effects of different nitrogen sources for lipase production from the results of Plackett-Burman design.
production was reduced due to the inhibitory effects of urea. Banjamin and Pandey [22] reported that urea was found to be best nitrogen source for lipase production with C.rugosa.
4.5. Identification of Important Carbon Source Using Plackett-Burman Design
Five carbon sources were screened by eight experimental runs. The experimental plan and corresponding lipase production were shown in Table 5. The pareto graph (Figure 5) was used to show the effect of all the carbon sources on lipase production. A p-value less than 0.05 for the three variables viz., glucose, sucrose, and fructose indicates that they are significant. From the analysis, it was also found that lipase production was affected by the above three carbon sources as evident from their F-values and p-values as shown in Table 6. In addition, the coefficient of determination (R2) of the model was 0.9998 which explains the 99.98% variability of the data. Glucose had a confidence level of above 95% in comparison to other variables and thus considered to be highly significant for lipase production. Two dummy variables (DV1 & DV2) were employed to evaluate the standard errors of the experiment. Figure 6 presents the results of different concentrations of glucose on lipase activity.
5. CONCLUSIONS
The Palm kernel cake supported good microbial growth and enzyme production as evident by its chemical composition. Marine yeast Yarrowia lipolytica NCIM 3589 was used for the fermentation. The presence of PKC
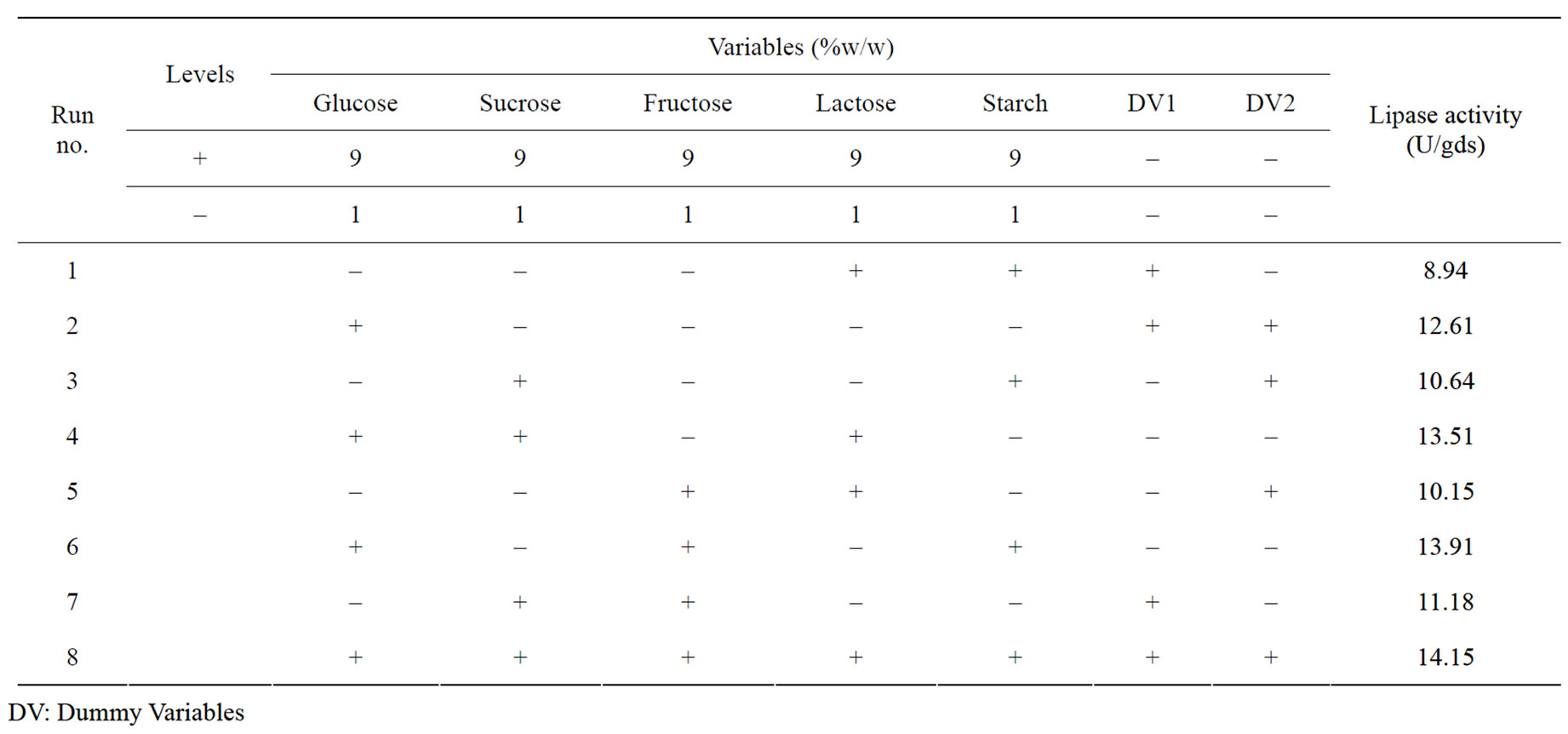
Table 5. Plackett–Burman experimental design matrix for screening of different carbon sources for lipase production.
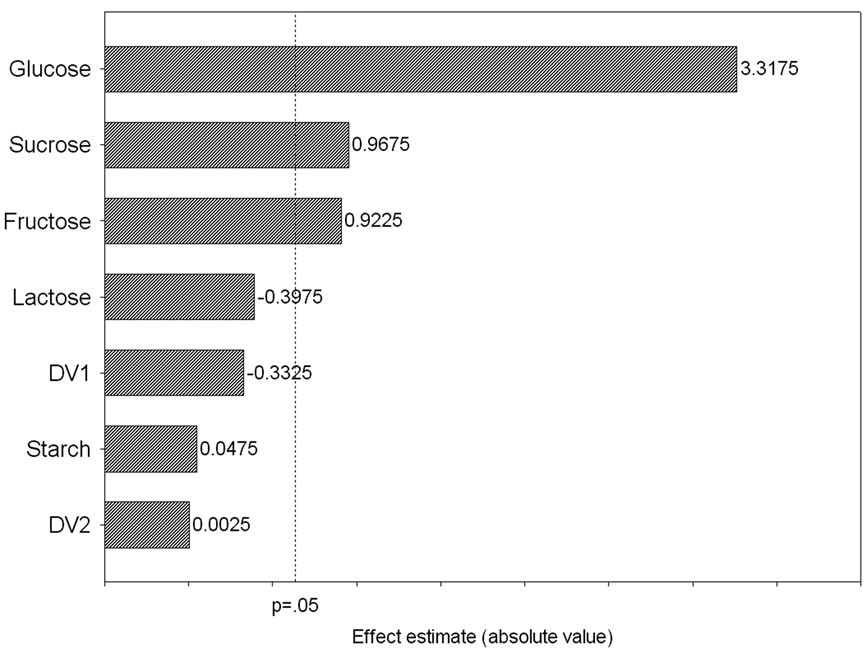
Figure 5. Pareto graph showing effect of various carbon sources on lipase production basedon the observation of Plackett-Burman design.
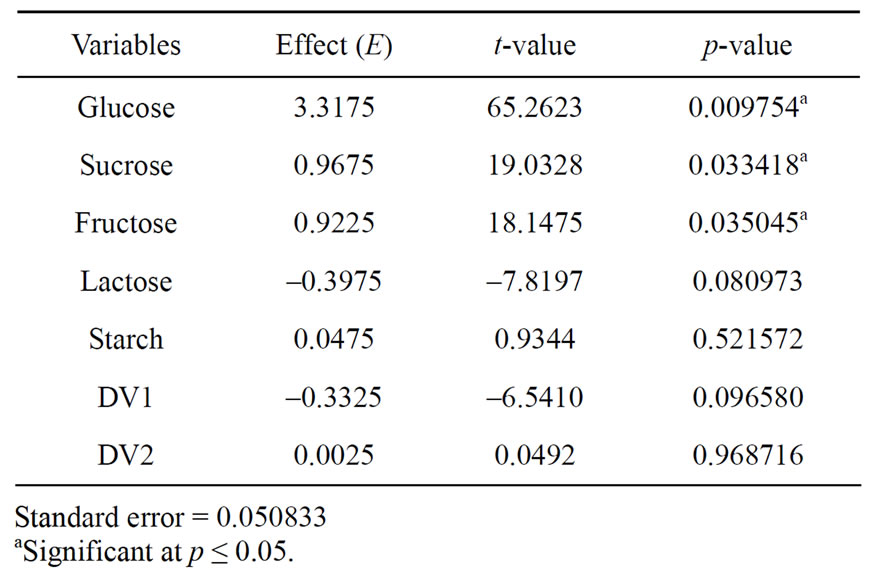
Table 6. Effects of different carbon sources for lipase production from the results of Plackett-Burman design.
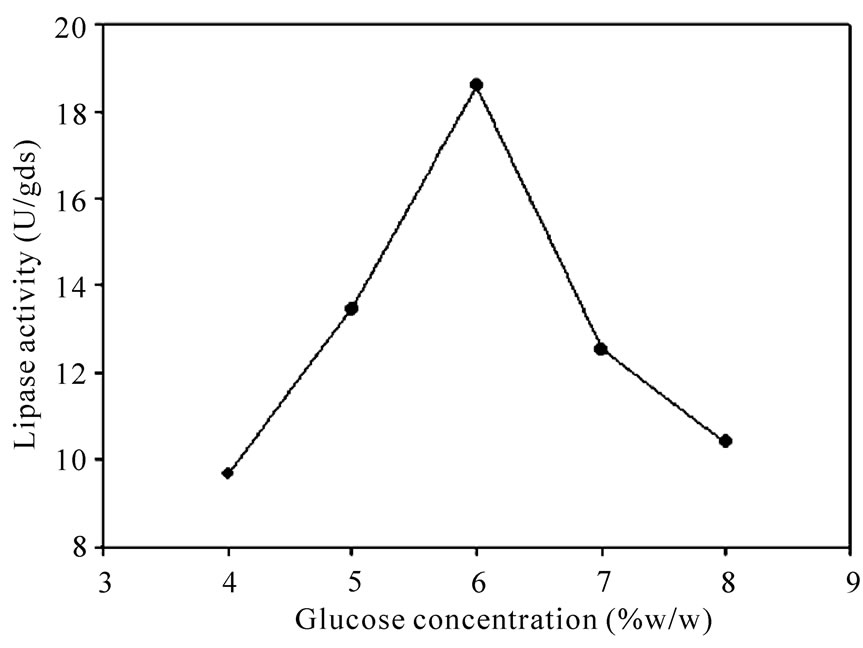
Figure 6. Effect of glucose concentration on lipase activity.
with 70% moisture content yielded the maximum lipase activity (18.58 U/gds) in four days. The high lipase activity achieved in conjunction with the abundantly available palm kernel cake in the state of Andhra Pradesh, India, paved a way for the industrial exploitation of this substrate under solid state fermentation using the indigenous Yarrowia lipolytica NCIM 3589 as a suitable micro organism.
REFERENCES
- Sharma, R., Chisti, Y. and Banerjee, U.C. (2001) Production, purification, characterization, and applications of lipases. Biotechnology Advances, 19(8), 627-662.
- Jaeger, K.E., Steinbuchel, A. and Jendrossek, D. (1995) Substrate specificities of bacterial polyhydroxyalkanoate depolymerases and lipases: Bacterial lipases hydrolyze poly(w-hydroxyalkanoates). Applied Environmental and Microbiology, 61(8), 3113-3118.
- Mochizuki, M., Hirano, M., Kanmuri, Y., Kudo, K. and Tokiwa, Y. (1995) Hydrolysis of polycaprolactone by lipase: effects of draw ratio on enzymatic degradation. Journal of Applied Polymer Science, 55(2), 289-296.
- Ashok, P. (2003) Solid state fermentation. Biochemical Engineering Journal, 13(2-3), 81-84.
- Castilho, L.R., Polato, C.M.S., Baruque, E.A., Sant Anna Jr, G.L. and Freire, D.M.G. (2000) Economic analysis of lipase production by Penicillium restrictum in solid-state and submerged fermentations. Biochemical Engineering Journal, 4(33), 239-247.
- Gombert, A.K., Pinto, A.L., Castilho, L.R. and Freire, D.M.G. (1999) Lipase production by Penicillium restrictum in solid-state fermentation using babassu oil cake as substrate. Process Biochemistry, 35(1-2), 85-90.
- Cordova, J., Nemmaoui, M., Ismaili-Alaoui, M., Morin, A., Roussos, S., Raimbault, M. and Benjilali, B. (1998) Lipase production by solid state fermentation of olive cake and sugar cane bagasse. Journal of Molecular Catalysis B: Enzymatic, 5(1-4), 75-78.
- Kamini, N.R., Mala, J.G.S. and Puvanakrishnan, R. (1998) Lipase production from Aspergillus niger by solid-state fermentation using gingelly oil cake. Process Biochemistry, 33(5), 505-511.
- Mahadik, N.D., Puntambekar, U.S., Bastawde, K.B., Khire, J.M. and Gokhale, D.V. (2002) Production of acidic lipase by Aspergillus niger in solid state fermentation. Process Biochemistry, 38(5), 715-721.
- Rao, P.V., Jayaraman, K. and Lakshmanan, C.M. (1993) Production of lipase by Candida rugosa in solid state fermentation. 1: Determination of significant process variables. Process Biochemistry, 28(6), 385-389.
- Mahanta, N., Gupta, A. and Khare, S.K. (2008) Production of protease and lipase by solvent tolerant Pseudomonas aeruginosa PseA in solid-state fermentation using Jatropha curcas seed cake as substrate. Bioresource Technology, 99(6), 1729-1735.
- PORLA (2000) Palm Oil Statistics. Ministry of Primary Industries, Kuala Lumpur.
- Diaz, M.J.C., Rodríguez, J.A., Roussos, S., Cordova, J., Abousalham, A., Carriere, F. and Baratti, J. (2006) Lipase from the thermotolerant fungus Rhizopus homothallicus is more thermostable when produced using solid state fermentation than liquid fermentation procedures. Enzyme and Microbial Technology, 39(5), 1042-1050.
- Raimbault, M. (1998) General and microbiological aspects of solid substrate fermentation. Electronic Journal of Biotechnology, 1(3), 174 189.
- Salunkhe, D.K., Chavan, J.K., Adsule, R.N. and Kadam, S.S. (1999) World oil seeds chemistry, Technology, and Utilization. Van Nostrand Reinhold, NewYork.
- Imandi, S.B., Bandaru, V.V.R., Somalanka, S.R., Bandaru, S.R. and Garapati, H.R. (2008) Application of statistical experimental designs for the optimization of medium constituents for the production of citric acid from pineapple waste. Bioresource Technology, 99(10), 4445-4450.
- Imandi, S.B. (2008) Studies on the production of lipase in solid state fermentation using Artificial Neural Networks and Genetic Algorithms. Ph.D. Thesis, Andhra University, Visakhapatnam, India.
- Winkler, U.K. and Stuckmann, M. (1979) Glycogen, hyaluronate and some other polysaccharides greatly enhance the formation of exolipase by Serratia marcescens. Journal of Bacteriology, 138(3), 663-670.
- Plackett, R.L. and Burman, J.P. (1946) The Design of Optimum Multifactorial Experiments. Biometrika, 33(4), 305-325.
- Shirazi, S.H., Rahman, S.R. and Rahman, M.M. (1998) Production of Extra cellular lipases by Saccharomyces cerevisiae. World Journal of Microbiology and Biotechnology, 14(4), 595-597.
- Ul-Haq, I., Idrees, S. and Rajoka, M.I. (2002) Production of lipases by Rhizopus oligosporus by solid-state fermentation. Process Biochemistry, 37(6), 637-641.
- Benjamin, S. and Pandey, A. (1997) Coconut cake: a potent substrate for production of lipase by Candida rugosa in solid state fermentation. Acta Biotechnologica, 17(3), 241-251.
- Mudgetti, R.E. (1986) Manual of industrial biotechnology. American Society for Microbiology, Washington DC.
- Babu, I.S. and Rao, G.H. (2007) Lipase production by Yarrowia lipolytica NCIM 3589 in solid state fermentation using mixed substrate. Research Journal of Microbiology, 2(5), 469-474.
- Silman, R.W., Conway, M.F., Anderson, R.A. and Bagley, E.B. (1979) Production of aflatoxin in corn by large scale solid state fermentation process. Biotechnology and Bioengineering, 21(10), 1799-1808.
- Pérez-Guerra, N., Torrado-Agrasar, A., López-Macias, C. and Pastrana, L. (2003) Main characteristics and applications of solid substrate fermentation. Electronic Journal of Environmental, Agricultural and Food Chemistry, 2(3), 343-350.

