International Journal of Astronomy and Astrophysics
Vol.2 No.4(2012), Article ID:26118,6 pages DOI:10.4236/ijaa.2012.24030
Cyanopolyynes as Organic Molecular Wires in the Interstellar Medium
1Centre for Environmental Sciences and Department of Chemistry Faculty of Sciences, Universidad de Chile, Santiago, Chile
2Department of Chemistry, Faculty of Basic Sciences Universidad Metropolitana de Ciencias de la Educación, Santiago, Chile
Email: correo@raulmorales.cl
Received September 22, 2012; revised October 27, 2012; accepted November 18, 2012
Keywords: Cyanopolyynes Structures; Dipole Moments; Inner Molecular Resistances; Organic Molecular Wires; ISM Radial Column Density
ABSTRACT
Cyanopolyynes (H[C≡C]n-CN or HC2n+1N, where n = 1, 2, 3, …, n) are commonly observed in the interstellar medium (ISM) as well as in the envelopes of carbon-rich stars. These linear molecular structures can be well described with a one-dimensional conduction model, which considers the scattering processes of electrons through the charge transfer conduction bridge of the H[C≡C]n-molecular wire containing the CN group as an electron-acceptor terminal unit. Therefore, our results using this model enable a better understanding of the longest molecules observed in interstellar space and provide new insight into why these particular cyanopolyynes reach a maximum length, such as is observed from astronomical experimental spectral data and cosmological chemical models. Dipole moments and geometrical parameters of these cyanopolyynes were obtained from ab initio molecular orbital calculations using the restricted Hartree-Fock approach and 6-311G* basis set, in order to obtain the inner resistance as a new parameter of chemical reaction feasibility for this molecular series. Using this last molecular parameter, we have been able to analyze the possibility of identifying long molecular species that can be found under local thermodynamic equilibrium in some ISM such us HC25H, HC27H, and HC29N, which have not been observed at present.
1. Introduction
New chemical models applied to the circumstellar envelope surrounding carbon-rich stars or particular interstellar mediums (ISM) are being developed to determine the presence and abundance of different molecular species [1-5]. Furthermore, the production of large molecules in heterogeneous astronomical environments is one of several theoretical astrochemical conjectures used to explain how complex molecular systems such as fullerenes in the ISM can originate from long linear molecular compounds such as cyanopolyynes.
All of the smaller members of the series have been detected in the ISM, particularly in circumstellar envelopes surrounding carbon-rich stars, corresponding to cold circumstellar shells, and they all have a conjugated, unsaturated carbon chain terminated at one end by an H atom and at the other by the CN group.
The smaller members of these series are well known at the laboratory, but under hard experimental conditions and extreme detection sensitivity, only its well known the rotational spectra up to HC17N, as the longest members of the series [6]. However, HC7N to HC11N were first discovered in the ISM by means of theoretical predictions of their rotational spectra and the subsequent detection of these spectra by radio telescopes. While HC11N, the largest linear interstellar molecule was the last-discovered member of these cyanopolyynes series [7,8].
Since 1978, several spectral observations from various interstellar regions have reported these types of oligomeric structures, particularly polyynic wires involving the CN group [9,10]. However, cosmological and theoretical conjectures derived from observational considerations have systematically demonstrated the absence of molecular wires longer than five units.
These molecular structures, from a physicochemical point of view, can be seen as a special case of molecular systems characterized by a basic triple bond unit that is repeated in a linear sequence generating several oligomeric structures, generally between one and five units as a maximum length with a CN terminal group. Therefore, the molecular structure can be defined by an electron-donor (D) terminal group, an intermediate group constituted by an unsaturated carbon molecular bridge (W) and an electron-acceptor (A) terminal group, where an electronic charge transfer process from D to A through the linear molecular structure determines several particular characteristics of these D-W-A systems.
Whenever this kind of molecular wires has been a subject of long data research, particularly associated to the concept of molecular electronic device [11-14], previous studies on polyenic systems in our laboratory were based on the electronic conduction properties of conjugated oligomeric compounds of the D-W-A type [14-17]. These studies have permitted us to focus our attention on these interstellar cyanopolyynes.
Therefore, in the present work, we have started a physicochemical model that allows us to derive electronic conduction properties from the dipole moments, a classical parameter used in structural molecular spectroscopy studies, in order to understand why long molecular organic wire concentrations are not favored from a structural point of view. Thus, our molecular results are coherent to observational evidence based on the radial column density distribution of cyanopolyynes observed in the circumstellar envelope of carbon-rich stars such as the IRC+10216 system [4] analyzed in the present work. Furthermore, our analysis extended to the ISM under local thermodynamic equilibrium (LTE) anticipates eventual observational evidence of very low ranges of radial column densities of long species such as HC25H, HC27H, and HC29N, which have not been observed at present.
2. The One-Dimensional Conduction Model
A few years ago, we developed a one-dimensional conduction model for D-W-A molecular systems [15,17]. Our model consider a scattering process of electrons through the internal charge transfer conduction bridge (W) to determine the molecular resistance and the molecular resistivity by means of the linear and non-linear contributions derived from the dipole moments and the p-molecular orbital bridge length (L) as fundamental physicochemical parameters.
This one-dimensional conduction model is based on a novel comprehension of the role of these oligomeric compounds, where the p-conduction channel of the molecular wires can be seen as a one-dimensional charge migration channel induced by the electron-acceptor group, assuming a typical scattering process of electrons as found in a metal wire system [18]. Thus, the inner conductance of this molecular wire determines the molecular resistance, and the final charge distribution determines the dipole moment. This model can be applied to any oligomeric molecular system that preserves the orientation of the dipole moment through the main axis of the molecular wire [17]. The molecular wire length and the dipole moment due to the ground-state chargetransfer process induced by the CN group are two fundamental parameters of the ground electronic state of these cyanopolyynes.
Therefore, the dipole moment of every nth oligomer can be represented as [17]
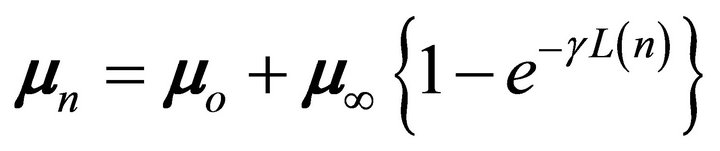 (1)
(1)
where 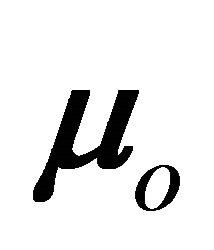 is the dipole moment of the first compound of the oligomeric series (HCN) without a bridge unit (n = 0),
is the dipole moment of the first compound of the oligomeric series (HCN) without a bridge unit (n = 0), 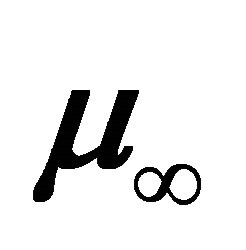 is a molecular constant of the oligomeric series at the limit value for L®¥,
is a molecular constant of the oligomeric series at the limit value for L®¥, 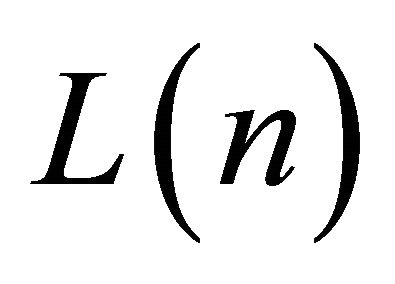 is the molecular wire length of the nth oligomer (-[C≡C]n-), and g is the wire conduction constant of this oligomeric molecular series.
is the molecular wire length of the nth oligomer (-[C≡C]n-), and g is the wire conduction constant of this oligomeric molecular series.
By considering a scattering process of the electronic flow through the molecular wire length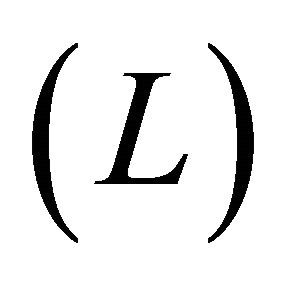 , we obtain the atomic scale conductance
, we obtain the atomic scale conductance 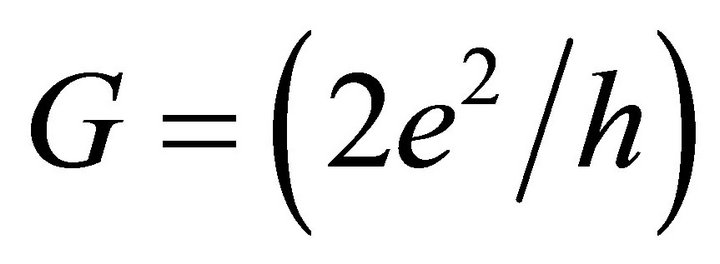
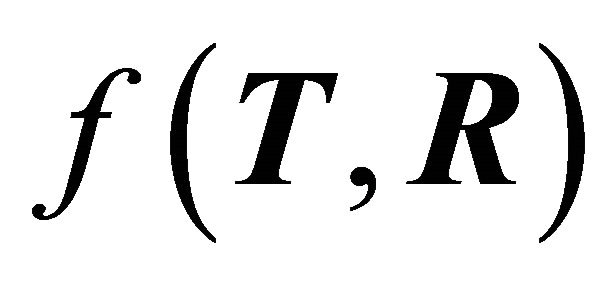 , where e is the electron charge, h is Planck’s constant, T and R are the transmission and reflection probability factors, respectively, and
, where e is the electron charge, h is Planck’s constant, T and R are the transmission and reflection probability factors, respectively, and 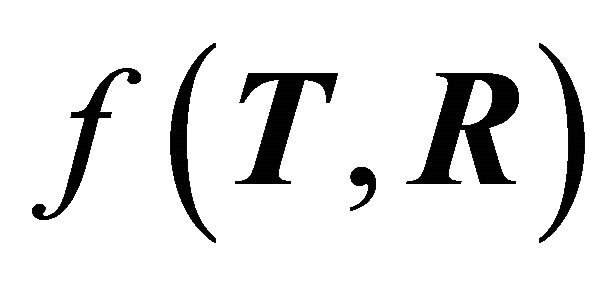 is a function of T and R given by
is a function of T and R given by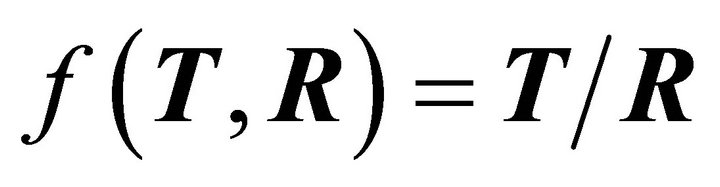 , where T + R = 1.
, where T + R = 1.
If under this approach [17] we consider an electronic scattering transmission of the type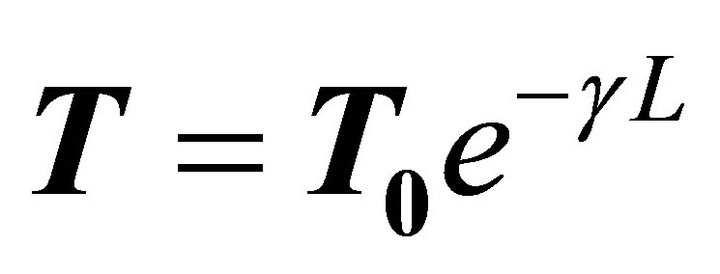 , with T0 = 1, we obtain the molecular resistance
, with T0 = 1, we obtain the molecular resistance 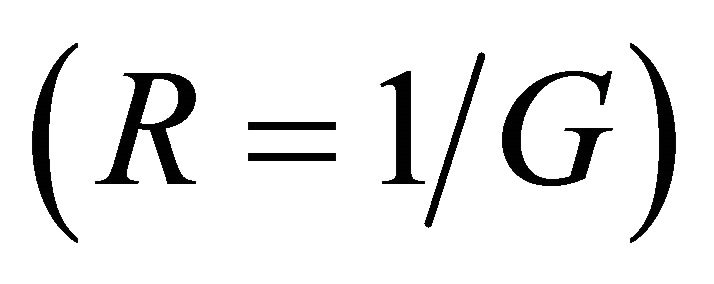 according to:
according to:
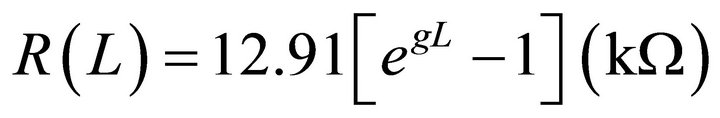 (2)
(2)
where 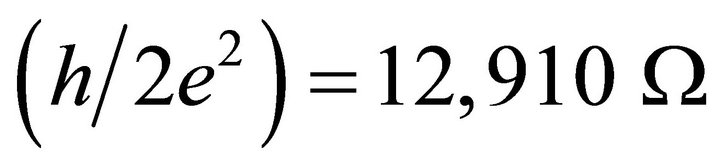 or 12.91 kW. However, to determine the linear molecular resistivity
or 12.91 kW. However, to determine the linear molecular resistivity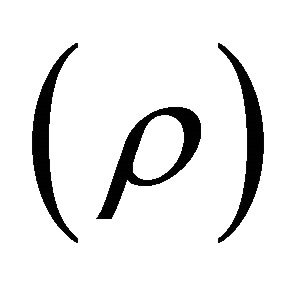 , this last equation can be expanded as a Maclaurin series,
, this last equation can be expanded as a Maclaurin series,
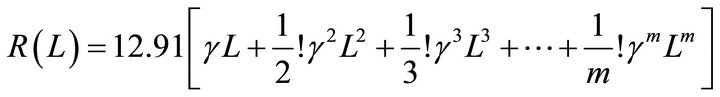 (3)
(3)
where the first term defines the linear contribution to the molecular resistance, given by , and the linear molecular resistivity follows as
, and the linear molecular resistivity follows as 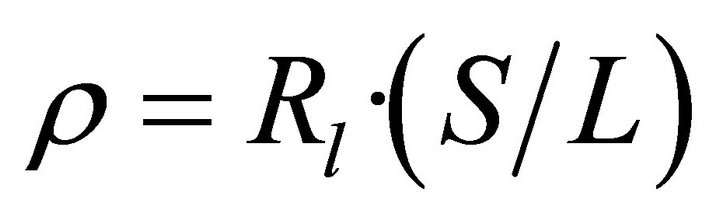 , where S is the molecular wire cross-section estimated to be 4.5 Å2 [14].
, where S is the molecular wire cross-section estimated to be 4.5 Å2 [14].
3. Results and Discussion
It is well known fact by experimentalist the inherent difficulties that these cyanopolyynes present in order to reach dipole moment measurements in vacuum. Additionally, molecular instability or hard experimental conditions have been observed in the laboratory synthesis of the larger members of the cyanopolyynes. However, these oligomeric compounds, from a geometrical and charge distribution perspective, can be well described by means of density-functional molecular-orbital calculations. Therefore, we used the SPARTAN’06 Quantum Mechanics Program (PC/x86), which employs the restricted Hartree-Fock (RHF) approach and the 6-311G* basis set, to determine the best molecular geometry optimization and the electronic charge distribution as a function of the length of the molecular wire for the oligomeric series under study.
The molecular wire lengths and dipole moments of the cyanopolyynes, calculated using this theoretical approach are shown in Table 1. Figure 1 shows a plot of the dipole moment versus the molecular wire length for the

Table 1. Molecular wire length and dipole moments of the cyanopolyynes.

Figure 1. Dipole moments of the cyanopolyynes (H-[C≡C]n- CN), n = 0 to 29, in the RHF approach using a 6-311G* basis set.
cyanopolyynes. We can observe a functional dependence for both parameters according to the behavior expected in Equation (1). Therefore, the expected functional dependence between mn and L has permitted us to make a best fit for the curve, where the final parameters of Equation (1) are the following: g = 0.125 ± 0.001 (Å−1), with m0 = 3.215 ± 0.006 (Debye), m¥ = 3.077 ± 0.006 (Debye), and r2 = 0.99996.
First, one can see how the dipole moment changes as a function of the molecular length. The dipole moments of the long species converge to 6.292 ± 0.012 Debye. Only insignificant changes are expected after HC29N. This phenomenon shows the inherent and limited property of the CN group to be an electron acceptor group in this molecular wire. Reflection due to electronic charge transfer becomes more important than transmission in terms of the wire length. In other words, the CN group is not strong enough to stabilize a long molecular array.
The molecular wire g-conduction constant exhibits a behavior similar to that of other oligomeric compounds [17]. Now, based on the g-conduction constant derived from Figure 1, we can determine the electronic resistance as a function of the length of the molecular wire according to Equation (2). Table 2 depicts the molecular resistance of these cyanopolyynes under study. Furthermore, if we use the linear component of the molecular resistance, as can be seen in Equation (3), these cyanopolyynes have a linear resistivity of 72.6 mW cm, which is similar to other molecular wires previously reported [17] (see Table 3).
From our results, we can appreciate how this molecular wire series follows the one-dimensional conduction model very well. The molecular parameters, such as the geometry and dipole moments, appear to be quite accurate with the RHF approach and the 6-311G* basis set.
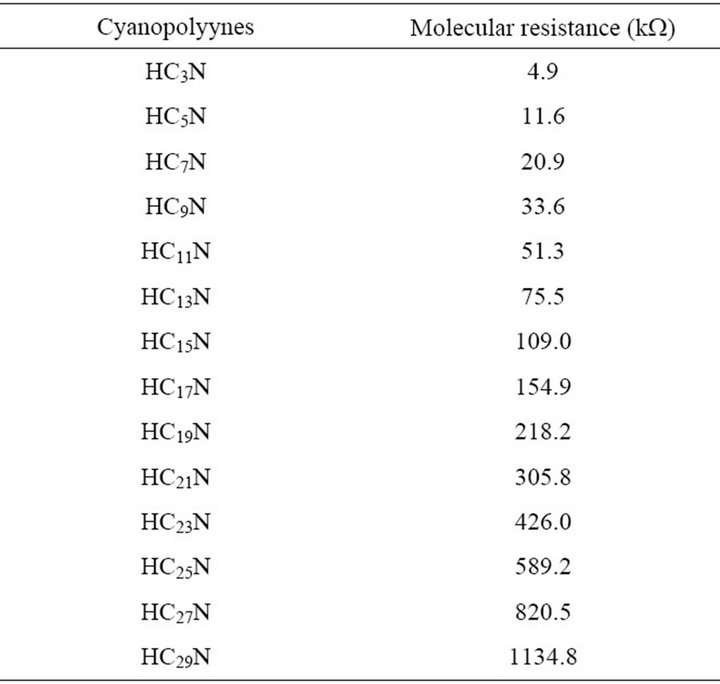
Table 2. Molecular resistances of the cyanopolyynes.
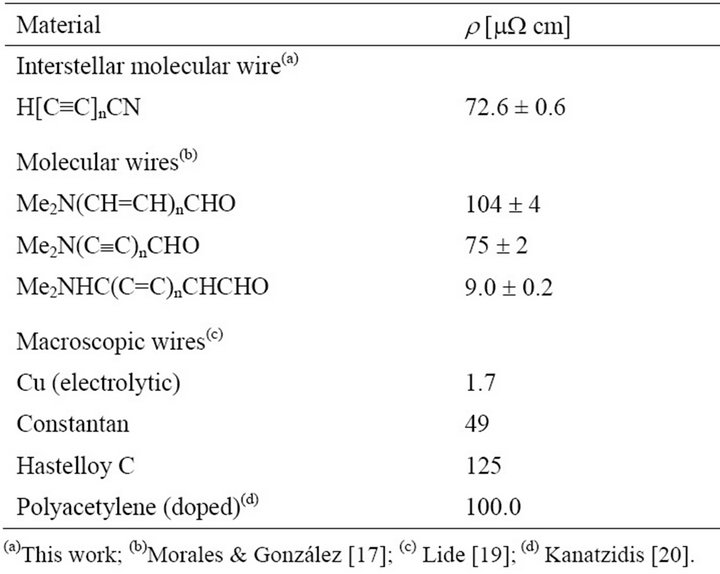
Table 3. Molecular wires linear resistivities.
Therefore, the molecular resistance can be used as a new criterion for chemical reactions feasibility of these linear molecular species. This is due to the fact that all species are synthesized from the same original reactants under the same conditions of LTE in bulk delimited by low-temperature dense-cloud regions. Thus, the molecular resistance to the internal charge transfer emerges as an indicator of the polarity strength of the ground state during the molecular formation process. Consequently, every new extension of the molecular wire of the cyanopolyynes due to -C≡Cunit gradually presents an additional resistance to the internal charge transfer process and, subsequently, their reaction feasibility necessarily decreases, due to the weak attraction force of the cyano electron-acceptor group.
Over 50 different molecules, including several oligomeric species, have been detected in IRC+10216 or CW Leonis, which is a carbon-rich star that is embedded in a thick dust envelope. This stellar source has been well studied both observationally and theoretically, and chemical models have carefully reproduced detailed radial distributions of these cyanopolyynes.
Thus, the report by Millar et al. [4] is an extension of the new standard model adapted to large molecules in a carbon-rich environment, including 3851 reactions and 407 gas-phase species. According to these authors the positive ion-molecule and neutral-neutral chemistry leading to the production of cyanopolyyne species was extended from that of the interstellar model so that cyanopolyynes as complex as HC23N were included. Reactions involving the radical CN and hydrocarbons were involved in the formation of cyanopolyynes, as they are in dense clouds, but reactions between the radical C2H and smaller cyanopolyynes were far more important in the IRC+10216 envelope chemistry. Therefore, the role of the CN group in the chemical reactions associated with cyanopolyyne synthesis is determined by the electronic feasibility of the charge transfer between the molecular wire and the electron acceptor group. Thus, we can expect a relationship between the oligomeric species density distribution and the molecular resistance to the internal charge transfer of the molecular wires that determines the final probability of the molecular array density under LTE.
In particular, observed and theoretical radial column densities for the cyanopolyynes in IRC + 10216 are well represented by different complex astrophysical models. Therefore, we have used the best theoretical data determined at present by the Millar et al. model [4], which include HC3N to HC23N, and we have correlated these density data to our molecular resistances as can be seen in Figure 2. A very good logarithmic correlation between both data sets can be observed, given an appropriate basement to our hypothesis respect to the chemical reaction feasibility in terms of the molecular wire length.
Other significant fact emerges from the broad scale of cyanopolyyne densities and the molecular wire lengths observed, where this linear correlation presents a new practical method for predicting new species at very low concentrations. Thus, we have extended the observed trend by means of an extrapolation to the HC25N, HC27N, and HC29N molecular resistances in order to obtain the radial column densities expected for these molecules.
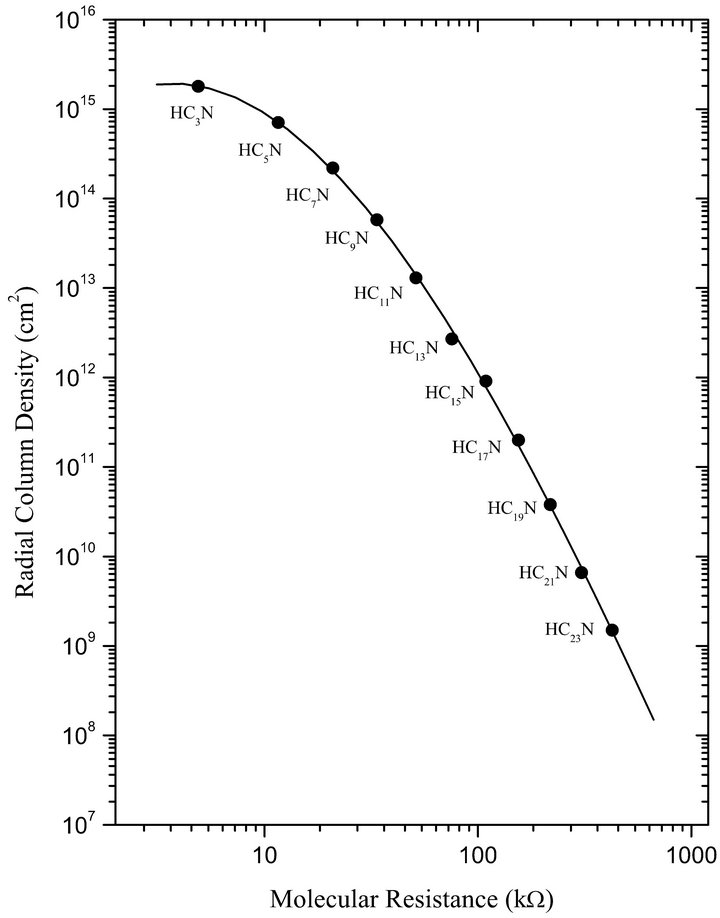
Figure 2. Theoretical radial column densities for cyanopolyynes (H-[C≡C]n-CN) in the circumstellar envelope of the carbon-rich star IRC + 10216 [4] versus molecular resistances calculated in the present work.
The extrapolated values are 2.8 × 108, 5.07 × 107, and 9.02 × 106 (cm−2), respectively. These expected values can give new inside to observational registration based on large exposition time required for low concentrations, only possible to obtain from Atacama Large Millimeter Array (ALMA), the new international observatory project.
4. Conclusions
Our results present a new line of analysis respect to long molecular wires when chemical models are being used in a complex network of reaction schemes under LTE as well as in radial column density estimations of ISM. Effectively, our predictions about how the radial column densities of long molecular wires decline with increasing molecular resistance introduces a new criterion for radio searches of long molecules, which is important given the large amount of integration time that will be necessary for observing these particular molecular systems.
Our work also shows how the dipole moments of an oligomeric series increase with chain length up to reach a certain saturation point, as can be seen in this cyanopolyynes series. Therefore it is not possible to distinguish between long molecular wires which dipole moments are similar enough according to the error measurements. In our case, the HC29N define a natural detection limit of the present series.
This work motivates studies of other molecular wires in the ISM that are currently in progress in our laboratory.
5. Acknowledgements
The authors would like to acknowledge the Centre for Environmental Sciences of the University of Chile for financial support.
REFERENCES
- E. Herbst and E. F. van Dishoeck, “Complex Organic Interstellar Molecules,” Annual Review of Astronomy and Astrophysics, Vol. 47, No. 1, 2009, pp. 427-480. doi:10.1146/annurev-astro-082708-101654
- J. R. Pardo, J. Cernicharo and J. R. Goicoechea, “Observational Evidence of the Formation of Cyanopolyynes in CRL 618 through the Polymerization of HCN,” Astrophysical Journal, Vol. 628, No. 1, 2005, pp. 275-282. doi:10.1086/430774
- P. M. Woods, T. J. Millar, E. Herbst and A. A. Zijlstra, “The Chemistry of Protoplanetary Nebulae,” Astronomy and Astrophysics, Vol. 402, No. 1, 2003, pp. 189-199. doi:10.1051/0004-6361:20030215
- T. J. Millar, E. Herbst and R. P. A. Bettens, “Large Molecules in the Envelope Surrounding IRC+10216,” Monthly Notices of the Royal Astronomical Society, Vol. 316, No. 1, 2000, pp. 195-203.
- M. A. Cordiner and T. J. Millar, “Density-Enhanced Gas and Dust Shells in a New Chemical Model for IRC+ 10216,” Astrophysical Journal, Vol. 697, No. 1, 2009, pp. 68-78. doi:10.1088/0004-637X/697/1/68
- M. C. McCarthy, W. Chen, M. J. Travers and P. Thaddeus, “Microwave Spectra of 11 Polyyne Carbon Chains,” Astrophysical Journal, Vol. 129, No. 2, 2000, pp. 611-623.
- M. B. Bell, P. A. Feldman, S. Kwok and H. E. Matthews, “Detection of HC11N in IRC+10216,” Nature, Vol. 295, 1982, pp. 389-391. doi:10.1038/295389a0
- M. B. Bell, P. A. Feldman, M. J. Travers, M. C. McCarthy, C. A. Gottlieband and P. Thaddeus, “Detection of HC11N in the Cold Dust Cloud TMC-1,” Astrophysical Journal Letters, Vol. 483, No. 1, 1997, pp. L61-L64. doi:10.1086/310732
- H. W. Kroto, C. Kirby, R. M. Walton, L. W. Avery, N. W. Broten, J. M. MacLeod and T. Oka, “The Detection of Cyanohexatriyne, H(CC3)CN, in Heiles’ Cloud 2,” Astrophysical Journal Letters, Vol. 219, No. 3, 1978, pp. L133-L137.
- D. Smith, “The Ion Chemistry of Interstellar Clouds,” Chemical Reviews, Vol. 92, No. 7, 1992, pp. 1473-1485. doi:10.1021/cr00015a001
- F. L. Carter, “Molecular Electronic Devices,” Marcel Dekker Inc., New York, 1986.
- C. Joachim and S. Roth, “Atomic and Molecular Wires,” Kluwer Academic Publishers, Dordrecht, 1997. doi:10.1007/978-94-011-5882-4
- D. K. James and J. M. Tour, “Molecular Wires,” Topics in Current Chemistry, Vol. 257, 2005, pp. 33-62. doi:10.1007/b136066
- C. Hernández and R. G. E. Morales, “Bridge Effect in Charge-Transfer Photoconduction Channels. 1. Aromatic Carbonyl Compounds,” Journal of Physical Chemistry, Vol. 97, No. 45, 1993, pp. 11649-11651. doi:10.1021/j100147a016
- R. G. E. Morales and C. González-Rojas, “Dipole Moments of Polyenic Oligomeric Systems. Part I. A OneDimensional Molecular Wire Model,” Journal of Physical Organic Chemistry, Vol. 11, No. 12, 1998, pp. 853- 856. doi:10.1002/(SICI)1099-1395(199812)11:12<853::AID-POC74>3.0.CO;2-Y
- C. González and R. G. E. Morales, “Molecular Resistivities in Organic Polyenic Wires. I. A One-Dimensional Photoconduction Charge Transfer Model,” Chemical Physics, Vol. 250, No. 3, 1999, pp. 279-284. doi:10.1016/S0301-0104(99)00335-3
- R. G. E. Morales and C. González-Rojas, “Dipole Moments of Polyenic Oligomeric Systems. Part II. Molecular Organic Wire Resistivities: Polyacetylenes, Allenes and Polylines,” Journal of Physical Organic Chemistry, Vol. 18, No. 9, 2005, pp. 941-944. doi:10.1002/poc.931
- R. Landauer, “Electrical Resistance of Disordered OneDimensional Lattices,” Philosophical Magazine, Vol. 21, No. 172, 1970, pp. 863-867. doi:10.1080/14786437008238472
- D. R., Lide, “CRC Handbook of Chemistry and Physics,” 85th Edition, CRC Press Inc., Boca Ranton, 2004, pp. 12-234.
- M. G. Kanatzidis, “Polymeric Electrical Conductors,” Chemical Engineering News, Vol. 68, No. 49, 1990, pp. 36-54. doi:10.1021/cen-v068n049.p036

