Advances in Chemical Engineering and Science
Vol.05 No.03(2015), Article ID:57591,5 pages
10.4236/aces.2015.53030
Spontaneous Changes of Solid 5,7-di-tert-butylspiro(2,5)octa-4,7-diene-6- one in Storage Conditions at Room Temperature
Alexandre A. Volodkin1, Gennady E. Zaikov1, Lydia N. Kurkovskaja1, Sergey M. Lomakin1, Irina M. Levina1, Elena V. Koverzanova2
1Federal State Budgetary Establishment of a Science of Institute of Biochemical Physics of N. M. Emanuelja of Russian Academy of Sciences, Moscow, Russia
2Federal State Budgetary Establishment of a Science of Institute of Chemical Physics of N. N. Semenov of Russian Academy of Sciences, Moscow, Russia
Email: chembio@sky.chph.ras.ru
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 3 March 2015; accepted 27 June 2015; published 30 June 2015
ABSTRACT
On an example of spontaneous changes 5,7-di-tert-butylspiro (2,5) octa-4,7-diene-6-one at room temperature, a new example of condensations in a solid station is opened. The single crystal of 2-(3’, 5'-di-tert-butyl-4'-hydroxy-phenyl)-ethyloxy-p-cresole is produced as result of a structure transformation 5,7-di-tert-butylspiro (2,5) octa-4,7-diene-6-one in a solid phase at room temperature except a single crystal at transformation 5,7-di-tert-butylspiro (2,5) octa-4,7-diene-6-one a new substance is formed too. Spectrums 1Н and 13С NMR differ from similar spectrums of initial compound and the single crystal. New substance represents an admixture of isomeric compounds, capable reversible to be transformed in initial 5,7-di-tert-butylspiro(2,5) octa-4,7-diene-6-one and 2-(3’, 5'-di-tert-butyl-4'-hydroxyphenyl)-ethyloxy-p-cresole.
Keywords:
Solid-Phase Reactions, 5,7-Di-tert-butylspiro(2,5)octa-4,7-diene-6-one, 2-(3’, 5'-di-tert- butyl-4'-hydroxyphenyl)-ethyloxy-p-cresole, NMR and IR Spectroscopy

1. Introduction
Examples solid-phase reactions at influence of pressure and other external factors in which result the free energy of a system are changed. A spontaneous chemical process in conditions of absence of external factors are possible only in the presence of superfluous free an energy in an initial chemical combination [1] . Isothermal changes in biological objects are a basis of ability to live of organisms, and their role in organic chemistry development is obvious. The organic solid-phase reactions proceeding at ambient temperature thereupon can be of interest. This reaction by us is opened in the course of research from a of 5,7-di-tert-butylspiro (2,5) octa-4,7-diene-6-one in storage conditions in a isolated conditions for a long time. One of signs of this reaction is formation from a powder of the single crystal which has the constitution of 2-(3’, 5'-di-tert-butyl-4'-hydroxyphenyl)-ethy-loxy-p- cresole according to spectrums 1Н, 13С and 17О NMR and IR-spectrum. Hence, in the course of powder storage 5,7-di-tert-butylspiro(2,5)octa-4,7-diene-6-one spontaneous condensations proceed and re-alkylation of tert- butyl groups with simultaneous introduction methyl group in a benzene ring. Except the single crystal at transformation 5,7-di-tert-butylspiro(2,5)octa-4,7-diene-6-one, the new substance is formed. Spectrums 1Н and 13С NMR differ from similar spectrums of initial compound and the single crystal. This substance represents an admixture of isomeric compounds, capable reversible to be transformed in initial 5,7-di-tert-butylspiro(2,5) cta- 4,7-diene-6-one and 2-(3’, 5'-di-tert-butyl-4'-hydroxyphenyl)-ethyloxy-p-cresole.
2. Experimental Part
The spectrums NMR registered on the device “Avance-500 Bruker” rather TMS. IR-spectra removed on spectrometer “PERKIN-ELMER 1725-X. Chromato-mass spectrometer analysis made on complex of devices including gas chromatograph “Trace-1310’ and Mass spectrums registered mass spectrometer detector” ISQ at ionisation by electronic impact with energy 70 eV. As result of a analysis 2-(3’, 5'-di-tert-butyl-4'-hydroxyphenyl)- ethyloxy-p-cresole. (2) mass numbers with m/z are identified: 232.26, 217.24, 203.23, 189.20, 175.20, 161.18, 147.16, 133.15, 115.12, 107.11, 91.12, 77.10, 57.13, 41.12.
5,7-Di-tert-butylspiro(2,5)octa-4,7-diene-6-one (1) synthesized from toluene sulphonate 3,5-di-tert-butyl-4- hydroxyphenylethane-2-ol, according to [2] . Yield of 95%, m.p..105˚C - 106˚C (from hexane). Spectrum 1Н NMR (CDCl3 δ, ppm): 1.26 (s., 18 H, tBu); 1.52 (s. 4 H, CH2CH2); 6.12 (s. 2H,). Spectrum 13С NMR (CDCl3 δ, ppm) 18.83 (CH2); 25.24 (C-CH3); 28.74 (C-CH3); 34.19 (C); 143.78 (С=C-Н); 147.39 (C=С); 185 (C=O). IR - (ν/cm−1): 1639, 1602 (С=С-С=О).
2-(3’, 5'-di-tert-butyl-4 '-hydroxyphenyl)-ethyloxy-p-cresole. (2). Compound 1 in a powder (4.5 g) in weighing bottle and abandoned at ambient temperature ~ 6 months. The formed single crystal in mass of 2.2 g separated, m.p. 95˚C - 96˚C. Spectrum 1Н NMR (CDCl3, d, ppm., J/Hz): 1.43 (s., 18 Н, tBu); 2.45 (s., 3Н, СН3); 2.90 (t., 2Н, СН2-СН2-Ar, J = 7.4); 4.21 (t., 2Н, СН2-СН2-Ar, J = 7.4); 5.13 (s., Н, OH); 6.93 (s., 2Н, Ar); 7.31 (d., 2Н, Ar ’, J = 8.3); 7.74 (d.2Н, Ar ’, J = 8.3). Spectrum 13С NMR (CDCl3, d, ppm): 21.1 (СН3); 29.7 (С-СН3); 33.71 (C); 34.78 (СH2), 70.60 (СН2); 124.9 (С=С-Н); 126.2 (C’= С-H); 127.3 (C' =С’-Н); 129.2 (C=C-С=С-OH); 132.77 (C=С-С=С-ОH); 135.6 (C’ =С’-С’ =С’-ОH); 144.0 (HО-С=С); 152.19 (H-О-С’=С’). Spectrum 17О NMR (CDCl3, d, ppm) 162.4 (C-O-C). IR-Spectrum (, n/cm−1): 3598 (OH), 1176 (C-O-C).
After branch of the single crystal, residual of reactionary mass in form of substance (2a, 2b) took and analyzed; m.p. 109˚C - 111˚C. Spectrum 1Н NMR (CDCl3 δ, ppm., J/Hz): 1.31 (s., 36Н, tBu); 1.84 (s., 6 Н, СН3); 2.78 (t.,2Н, СН2, J =12); 3.11 (d. 2Н, СН2, J =12); 4.27 (.s.24 H); 5.00 (s 2Н, OH); 6.93 (s., 4Н, Ar); 7.21 (d. 2Н, J = 12). Spectrum 13С NMR (CDCl3 δ, ppm): 22.37 (СН3); 29.79 (С-СН3); 33.60 (СН2); 36.98 (C); 56.70 (СН2); 124.97 (С-Н); 128.30 (С=С-Н); 135.17 (С=С-O); 151.67 (О=С-С=С); 171.62 (C-OH); 178.60 (С=О).
Thermolisis of substance (2а and 2b) in heptane of solution. Solution of 0.5 g a substance (2а, 2b) in 15 ml n-heptane boiled ≈2 h, distilled off solvent, fractionated a reaction mixture by sublimation. Crystals 5,7-Di- tert-butylspiro (2,5) octa-4,7-diene-6-one (1) separated and identified by the NMR method, m.p.105˚C - 106˚C. From a residual crystallization from hexane received 2-(3’, 5 -di-tert-butyl-4'-hydroxyphenyl)-ethyloxy-p-cre- sole. (2), m.p.95˚C - 96˚C. The spectrum 1Н NMR is identical to the spectrum compound 2, received earlier.
3. Results and Discussions
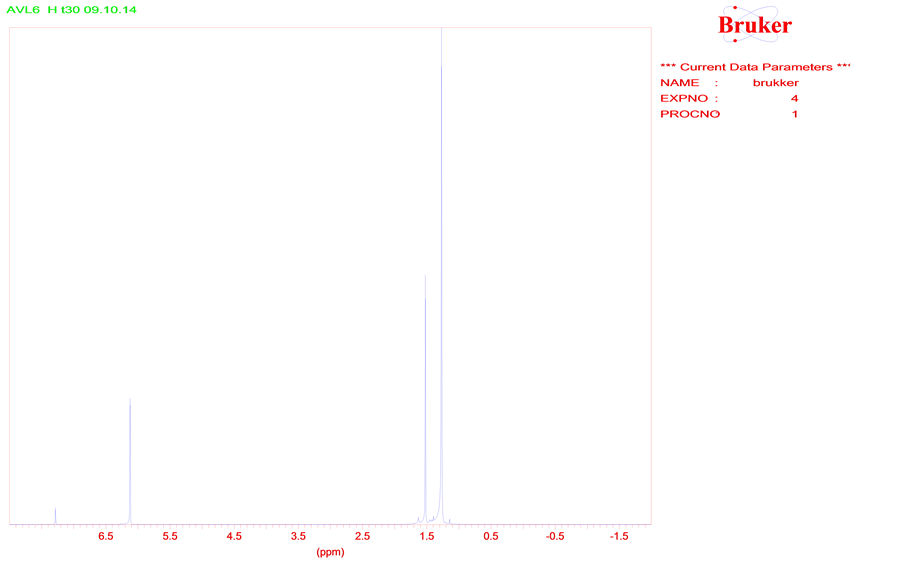
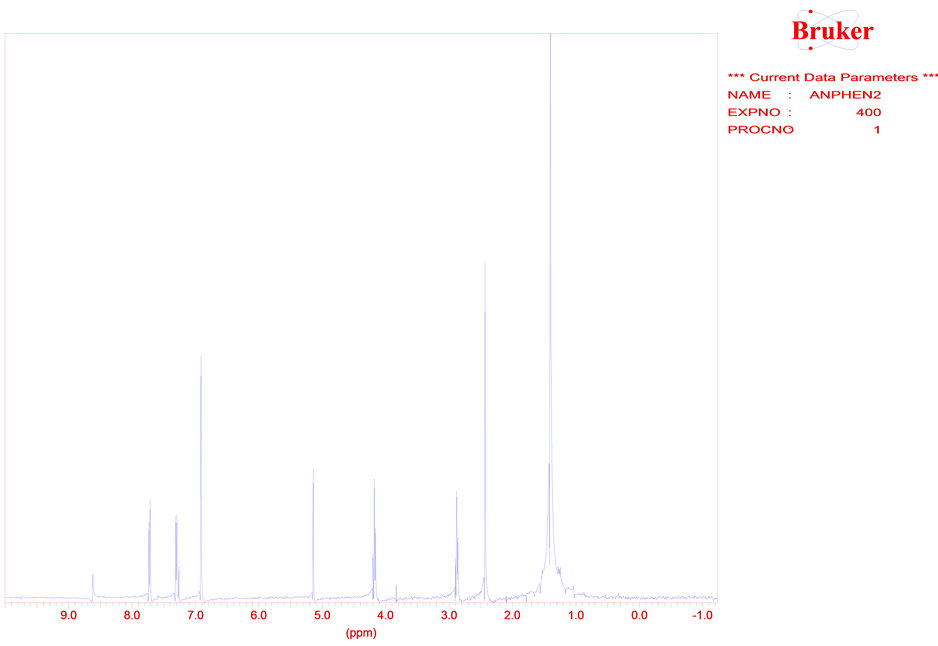
Slowly chemical processes are proceed in a storage conditions of a powdery 5,7-di-tert-butylspiro(2,5)octa- 4,7-diene-6-one at temperature ≈20˚C. There are a results of spectrums 1Н and 13С NMR initial compound 1 (Figure 1) and the single crystal formed of powder 1 (Figure 2).
Dates of IR-spectra of compounds 1 and 2 confirmed the fact of structural changes in molecule 1. The spec-
Figure 1. The spectrum 1Н NMR of 5,7-di-tert-butylspiro(2,5)octa-4,7-diene-6-one (1).
Figure 2. The spectrum of 1Н NMR of the single crystal of 2-(3’, 5'-di-tert-butyl-4 '-hydroxyphenyl)-ethy-loxy-p-cresole. (2).
trum 1Н NMR compound 1 consists of three “singlets” from protons of tert-butyl groups (s., 1.26 ppm), protons CH2-CH2 group (s., 1.52 ppm) and protons of hexatomic cycle (s., 6.12 ppm) that will be coordinated with the data [3] . Positions of signals from atoms of carboneum in the spectrum 13С NMR correspond to structure 1. The signal of carboneum in a spirane cycle is displayed at 34.19 ppm, carbonyl group carboneum is at 185 ppm.
From the spectrum NMR of compound 2 follows that a molecule consists of two aromatic cycles bridged by bunch from group СН2-СН2 of atoms.
Signals of 34.78 and 70.60 ppm fall into to carbon atoms of system atom CH2-CH2 of structure 2, that is confirmed by correlation in co-ordinates С-Н. Dates of the spectrum of 13С NMR (21.06 ppm) and 1Н NMR (2.45 ppm.) specify on СН3 group in pair position of benzene ring of compound 2. There are at the spectrum 1Н NMR signal (singlet) of 6.93 ppm (m-protons of aromatic cycle), two doublet signals of 7.30 and 7.74 ppm (4 protons of fragment from p-cresole). This result confirms a stage of eliminating of tert-butyl groups in the course of formation of the single crystal 2. From interpretation of spectrums 1Н and 13С NMR remains opened a question of the mechanism of formation CH3 group in the course of the transformation of structure 1 to structure 2. The signal 1Н NMR from phenolic hydroxyl is present (5.1 ppm.) and IR-spectrum (3598 cm−1). Frequency of 1175 cm−1 is characteristic for C-O-C com-munication. Presence at a molecule of oxygen at a ether group is confirmed by the spectrum 17О NMR. At the ionisation by electronic impact with energy 70 eV at the mass spectrum chromatogram there are values of mass numbers 232.4, 203.3, 107 m/z which can be carried to structure (3) and to ions with masses: m/z = 203.3 and m/z = 107 units.
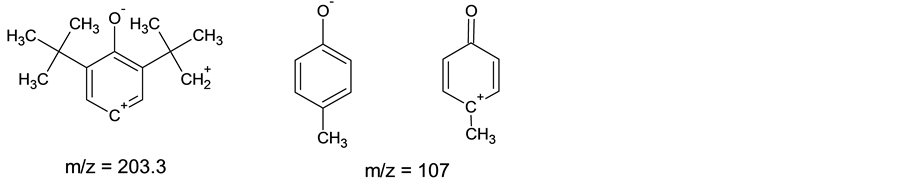
This data allows to assume a structure of the single crystal 2-(3’, 5'-di-tert-butyl-4'-hydroxy-phenyl)-ethy- loxy-p-cresole.and process of formation compound 2 (Schema 1).
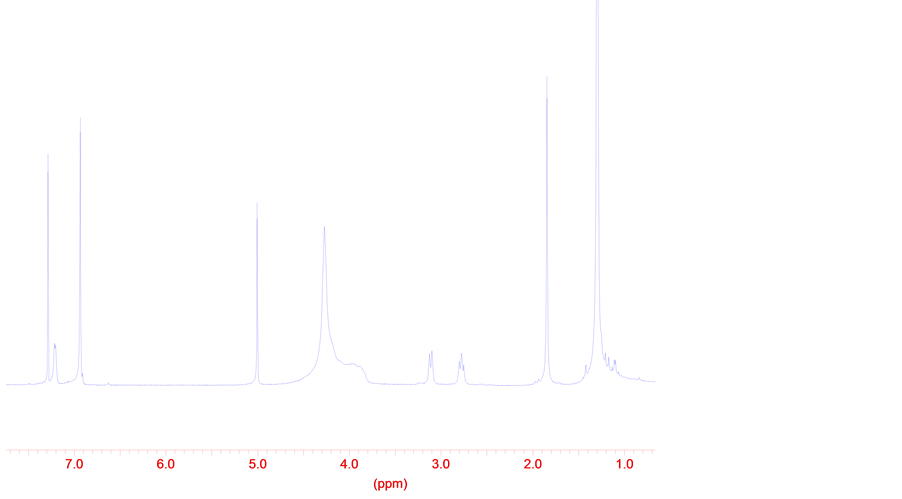
As result of acid hydrolysis of the compound 2 is formed p-cresole that confirms structure 2. Simultaneously in the solid phase a process of dimerization proceed, that follows from spectrums of 1Н and 13С NMR reactionary mass 2а, (Figure 3), formed of a powder of the compound 1 (Schema 2).
In course of a reversible dimerization a few isomers are formed with identical on enthalpy and entropy, for example, compounds with structures 2а or 2b, which can be compounds of a substance of reactionary mass.
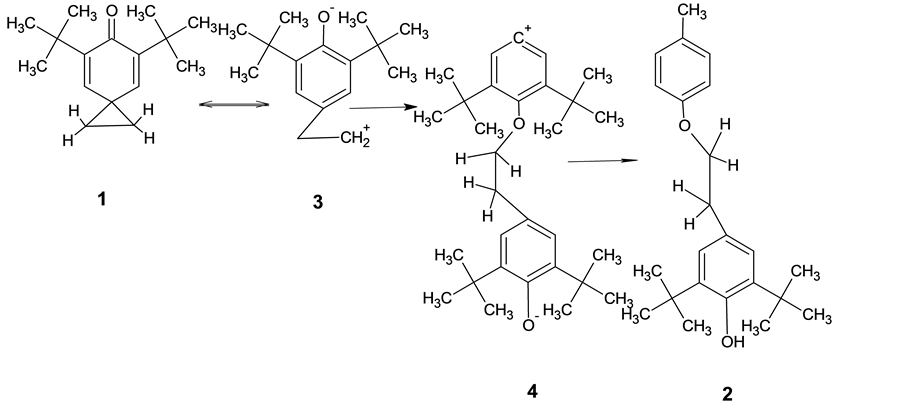
Schema 1. The mechanism of formation of compound 2.
Figure 3. The spectrum 1Н NMR of reactionary mass 2а after formation 5,7-di-tert-butylspi- ro(2,5)octa-4,7-diene-6-one (1) in isolated state at room temperature within 6 months.

Schema 2. The mechanism of formation of compounds 2a and 2b.
The structure 2a consists of two a hexatomic cycles. One of which is a aromatic, containing two tertiary butyl group, phenolic OH group and СН2-СН2 group. Formation OH group (s., 5.0 ppm) probably may by as result at moving of proton from group СН2-СН2 to a second molecula of structure 1.The result of a substituent with vinyl communication confirms by the spectrum 1Н NMR of compound 2а (doublet signal at 7.21 ppm). The signal of 1.84 ppm will be co-ordinated with a structure 2b and a group CH3. Decoding of widened signal in a field of 4.5 ppm., by quantity of integrated protons to equal quantity of protons in structure 1, is a subject of subsequent researches. It is obvious that 2а and 2b represent only a part of possible mesomeric structures, which are in dynamic equilibrium that is actually and explains appearance in spectrum 1Н NMR of the widened signal.
For purpose investigation of not ordinary aproperty of 5,7-di-tert-butylspiro(2,5)octa-4,7-diene-6-one are calculated structures, discussed in the present work: 5,7-di-tert-butylspiro(2,5)octa-4,7-diene-6-one (1), crystal 2-(3’, 5'-di-tert-butyl-4 '-hydroxyphenyl)-ethyloxy-p-cresole (2), and also others (2а, 2b), specified on schemes 1 and 2.
The results (Table 1) of the calculation of structures (method of Hartrii-Foka, UHF) confirm possibility specified above a transformation of initial 5,7-di-tert-butylspiro (2,5)octa-4,7-diene-6-one (1).
Table 1. The energy of formation, enthalpy and entropy of 5,7-di-tert-butylspiro(2,5)octa-4,7-diene-6-one (1), 2-(3’, 5'-di- tert-butyl-4'-hydroxyphenyl)-ethyloxy-p-cresole (2), and structures 2а, 2b.
From comparison of values energy formations (−H˚f) compound 1, dimers 2а and 2b follows that energy of formation decreases (for 2а Δ(H˚f = −36.2 kcal∙mol−1, for 2b Δ(H˚f = −45.3 kcal∙mol−1). Values of enthalpies (H˚) practically do not differ, there is reduction of entropy, for 2а ΔS˚ = 38.9 unit, for 2b ΔS˚ = 33.0 unit. From results of comparison of values energy formations 2а, 2b and their structures follows, that the structure 2b with spiro system and hydrogen atom is more preferable.
4. Conclusion
On an example of spontaneous changes 5,7-di-tert-butylspiro(2,5)octa-4,7-diene-6-one at room temperature, a new example of condensations in a solid station is opened. Spontaneous reactions of transformation in a solid phase proceed. One of signs of this reaction is formation from a powder of the single crystal which according to spectrums 1Н, 13C and 17О the NMR and IR-spectrum has the constitution of 2-(3’, 5'-di-tert-butyl-4'-hydrox- yphenyl)-ethyloxy-p-cresole. As a result of transformation 5,7-di-tert-butylspiro (2,5) octa-4,7-diene-6-one, a new substance is formed too. Spectrums 1Н and 13С NMR differ from similar spectrums of the starting compound and the single crystal. This substance represents an admixture of isomeric compounds, capable to be transformed in initial 5,7-di-tert-butylspiro (2,5) octa-4,7-diene-6-one and 2-(3’, 5'-di-tert-butyl-4'-hydroxyl- phenyl)-ethyloxy-p-cresole.
References
- Shchegolikhin, A.N. (2011) Dynamics Research Solid State in Organic Crystals. Thesis PhD, Institute of Biochemical Physics of N.M.Emanuelja of Russian Academy of Sciences, Moscow, 142.
- Ershov, V.V. and Belostotskaja, I.S. (1965) Di-Tert-Butylspirocyclodienones. Herald of Russian Academy of Sciences, Series chemistry, 1301. [In Russian]
- Chamot, D. and Pirkle, W. (1969) Direct Nuclear Magnetic Resonance Observation of Stereochemically Distinctions. Journal of the American Chemical Society, 91, 1569-1571. http://dx.doi.org/10.1021/ja01034a070