Food and Nutrition Sciences
Vol.4 No.2(2013), Article ID:27720,7 pages DOI:10.4236/fns.2013.42022
Effect of Ice-Temperature and Spermidine on Chilling Sensitivity of Pepper
![]()
College of Food Science, Sichuan Agricultural University, Ya’an, China.
Email: *qinwen1967@yahoo.com.cn
Received December 30th, 2012; revised January 30th, 2013; accepted February 7th, 2013
Keywords: Spermidine; Ice-Temperature; Pepper; Chilling Insensitivity
ABSTRACT
The peppers which were treated at ice-temperature for 6 hours and 1 mmol/L spermidine for 10 min, followed by cold storage (4˚C ± 1˚C) respectively were investigated. The results indicated that the chilling injury was delayed and reduced by ice-temperature and spermidine treatments. Comparing with control group, ice-temperature and spermidine exerted significant effects on reduction of ascorbic acid (Vc) and chlororphyll contents, relatively high activity of superoxide dismutase (SOD), accumulation of malondialdehyde (MDA) as well as the increase of cell membrane penetrability. The results indicated that the spermidine and ice-temperature induced the activity of antioxidant enzyme SOD and maintained higher defence-related compound Vc as well as lower levels of membrane lipid peroxidation, which may be associated with chilling injury alleviation. There was a difference between ice-temperature treatment and spermidine treatment after 30 days. The results suggested that spermidine was more effective in reducing chilling sensitivity and prolonging storage of peppers.
1. Introduction
Polyamines (PAs), mainly diamine putrescine (Put), triamine spermidine (Spd), and tetramine spermine (Spm), are polycationic compounds of aliphatic nitrogenous base with low molecular weight and high activities that are present in all living organisms. Numerous researches in squash [1], citrus [2-3], water spinach and cabbage [4], pepper [5] suggested that polyamine had a close relationship with the chilling resistance of plants. Chilling injury results in significant increases in Put levels in many fruit, such as lemon, grapefruit, zucchini, eggplant and pepper, suggesting that PAs could protect plants from chilling injury due to their ability to preserve membrane integrity [6]. Exogenous polyamines have been proposed as a functional and potential application that play an important roles for regulating the stress response, adapting in adverse conditions as well as alleviating chilling injury. In recent years, exogenous polyamines have been applied to enhance the chilling resistance in a number of horticultural crops, including lychee [7], apple [8], blueberry [9], peach [10], cucumber [11]. It has been reported that exogenous polyamine treatments play a role in membrane stabilization by a significant reduction of the stress-induced electrolyte leakage and enhancement of stress-evoke activity of lipoxygenase [12]. Ice-temperature, the specific range of temperature between zero and freezing point which is below zero due to the electrolyte inside the cell, can restrain the metabolism and keep up them in the living state. The storage of fruits and vegetables at ice-temperature is the foremost and promising technology applied to inhibit the growth of microbes, to slow metabolism, to retard postharvest sensecence and to extent the shelf-life period of agricultural products without damaging the plant tissue. The icetemperature technique applied to the storage of strawberry [13], longan [14], pepper [15], lotus root [16], broccoli [17] have been reviewed. The researches showed that the ice-temperature technology had the superiority to keep postharvest products freshness and special flavor with high quality.
Pepper, native to tropical South America and sensitive to low temperature, is highly favored by consumers worldwide. The critical temperature of chilling injury is 6 degree or 9 degree Celsius according to different varieties. Therefore, peppers stored at low temperature especially below 9 degree are vulnerable to chilling injury. In recent researches, the application of heat treatment [18], exogenous substances such as H2O2 [19], 1-MCP and ethylene [20,21], NO [22], salicylic acid [23], polyamines et al. have been reported to reduce the development of chilling injury symptoms and maintain quality. However, there is lack of published studies on the application of spermidine and cold shock at the range of ice-temperature. Thus, the objective of this study was to determine whether the spermidine and ice-temperature shock are linked to reducing chilling injury, maintaining chlorophyll and vitamin C content, evaluating electrolyte leakage as well as changing malonaldehyde (MDA) content and antioxidant enzyme SOD during cold storage. This study was aim to lay a solid foundation for pepper storage and transportation.
2. Materials and Methods
2.1. Plant Material and Regent
Freshly harvested green bell peppers (Capsicum annuum L.) were obtained from local commercial orchard in Ya’an, China. Fruit were delivered to the laboratory immediately after harvest. Fruit were selected for uniform size and color and the absence of visual defects, then randomly divided into three lots for this study.
2.2. Ice-Temperature and Spermidine Treatments
Peppers were grouped into lots of 120 including 60 for evaluation of chilling injury and subjected to the following three treatments (Table 1): 1) control ( distilled water at room temperature for 10 min, T1); 2) treated at −1˚C ± 0.5˚C for 6 h (T2); 3) dipped in 1 mmol/L spermidine at room temperature for 10 min (T3). Following the treatments, fruit treated by spermidine and water were placed on the table and allowed to dry at room temperature before storage at 4˚C in a temperature-controlled chamber with relative humidity of 90%. Fruit were transferred into temperature-controlled chamber after storage at ice-temperature for 6 h. A 40 × 25 × 20 cm rectangle of foam was used to store peppers. Three replicates of 120 peppers each per treatment were conducted. Fruit were taken after treatments (time 0) and at 5 day intervals during storage for measurements of chilling injure, content of chlorophyll and vitamin C, activity of SOD, accumulation of MDA as well as electrolyte leakage.
Table 1. Different pretreatments for further analysis.

2.3. Determination of Quality Parameters
Chilling injury (CI) manifested as calyx browning, surface pitting with discoloration, fruit softening and decay in the end. For each fruit, CI was scored according to a 5-grade scale, where 0 = none; 1= slight (area of CI is below 25%); 2 = moderate (area of CI is between 25% and 50%); 3 = moderately severe (area of CI is between 50% and 75%); 4 = severe (area of CI is above 75%). CI index calculated using the following formula: CI index = [(CI level) × (number of fruit at the CI level)]/(total number of fruit in the treatment).
Vitamin C content was measured by 2,6-dichlorindophenol titration. 5 g of pulp were homogenized with 50 mL of 2% hydrochloric acid and then centrifuged for 15 min at 8000 g, 4˚C. Five milliliters of supernatant were mixed with 0.5 mL of 0.06 M potassium iodide, 2 mL of 5 g·L−1 starch solution and 2.5 mL distilled water. The reaction mixture was titrated by potassium iodate until the color turned to blue. 5 mL of 2% hydrochloric acid replacing the supernatant was used as control under the same condition. The content of vitamin C was calculated by volume difference of potassium iodate adjusted for equal amount of vitamin C between sample and control.
Chlorophyll content in pepper was quantified with 80% acetone as described by Xing et al. [24]. One gram of pulp sample was taken from the equators of 10 fruit at each time point during the storage. Chlorophyll was extracted with 10 mL of 80% (v/v) acetone, centrifuged, and light absorption by supernatant measured at wavelengths of 645 and 663 nm. The chlorophyll content was calculated using the following equation:
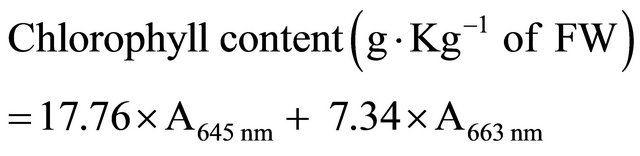
2.4. Evaluation of SOD Activity, Cell Membrane Permeability and MDA Accumulation
All extract procedures were conducted at 4˚C. 1 g of flesh tissue was ground with 5 ml of 50 mM sodium phosphate buffer (pH 7.8). The extract was then homogenised and centrifuged at 10,000 g for 20 min at 4˚C. The supernatants were used for enzyme assay. Superoxide dismutase (SOD) activity was determined photochemically by nitro-blue tetrazolium (NBT) according to the method described by Abbasi et al. [25] with slight modifications. The reaction mixture contained 50 mM sodium phosphate ( pH 7.8), 13 mM methionine, 0.75 mM NBT,20 µM riboflavin and 0.1 ml crude enzyme extract. The formation of blue formazan was monitored by recording the absorbance at 560 nm. One unit of SOD activity was defined as the amount of enzyme that causes a 50% inhibition of NBT reduction under assay conditions.
To determine the rate of cell membrane permeability, the method to determine electrolyte leakage described by Xing et al. [24] was used. The pepper tissue was cut with a borer into small discs (10 mm in diameter and 4 mm in thickness). Conductivity after incubation in 25 ml of 0.4 M mannitol solutions was measured with a DDS-11A conductivity meter after 3 h of incubation under constant shaking. The solutions were boiled at 100˚C for 30 min and cooled quickly before the total electrolyte leakages were measured. The relative electrolyte leakage was defined as a percentage of total: (initial/total) × 100.
TBARS were determined and expressed as malondialdehyde (MDA) equivalents, according to the method of Xing et al. [24] with slight modifications. Pulp tissue (2.0 g) from 10 pepper fruit were homogenized with 10 mL of 10% trichloroacetic acid and then centrifuged for 15 min at 8000 g, 4˚C. One milliliter of the supernatant was mixed with 3 mL of 0.5% thiobarbituric acid (TBA) dissolved previously in 10% trichloroacetic acid. The reaction mixture solution was heat-treated for 20 min at 100˚C, quickly cooled, and then centrifuged for 10 min at 8000 g to clarify precipitation. Absorbance at 523 nm was measured and subtracted from the nonspecific absorbance at 600 nm. The amount of MDA was expressed as µM·Kg−1 of fresh weight (FW).
2.5. Statistical Design
Experiments were performed using a completely randomized design. Experimental data are the mean ± SD of three replicates of the determinations for each sample. Data for analytical determinations were subjected to oneway analysis of variance (ANOVA). Sources of variation were storage and treatments. Mean comparisons were performed using HSD the Tukey’s test to examine if differences were significant at p < 0.05, extremely significant at p < 0.01. All statistical analyses were performed with SPSS v19.0 for Windows. Linear regressions were performed among several parameters using Origin Pro v 8.0 for Windows.
3. Results
3.1. Effect of Spermidine and Ice-Temperature on Chilling Injury
As shown in Figures 1(a) and (b), in the untreated control pepper fruit, CI symptoms occurred at 10 days after storage, while an initial CI symptom of pepper fruits treated by spermidine and ice-temperature was observed after 20-day-storage and 15-day-storage, respectively. The severity of CI was increased with the storage time, whereas spermidine and ice-temperature treatments before stored at 4˚C retarded chilling injury process and reduced the development and severity of CI symptoms.
The CI index in T2, T3 were significantly lower compared to the index in control fruit. For instance, the chilling injury rate was as high as 80% in control while almost half of peppers in T2, T3 were in good condition at the end of storage. Among all the treated fruit, spermidine was the most effective in alleviating CI of pepper fruit during storage at 4˚C.
3.2. Chlorophyll Content and Vc Content
The changes in chlorophyll content and Vc content in pepper fruit during the storage is shown in Table 2. The content in control fruit decreased gradually over time, while spermidine and ice-temperature treatments delayed the trend. During the first ten days, chlorophyll content in control exhibited a relatively lower decrease than those in T2, T3. While this trend was inbibited till the end of this experiment. At the end of storage, the chlorophyll content in T1 was just amount to 18.12% of original data. In contrast, chlorophyll content in T2, T3 were much higher than that in T1 (p < 0.1), however, the decrease of Vc content in T2, T3 were also conspicuous (p < 0.1) at the end of the experiment. Vc content showed trend similar to chlorophyll content. A steady decrease for Vc content was found in T1 during first 20 days then an abrupt decrease was observed up to the end of storage
 (a)
(a)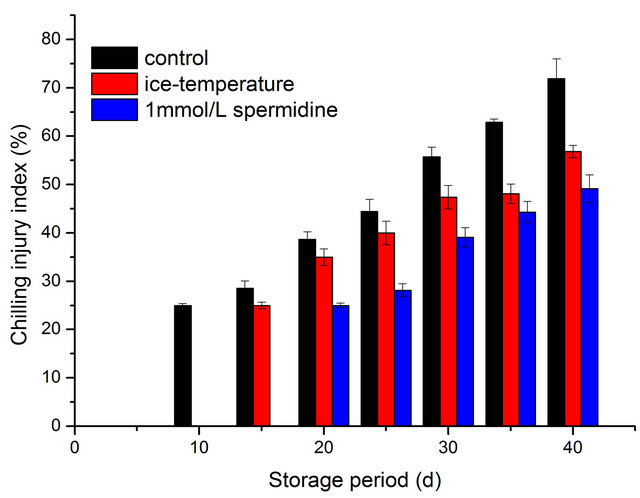 (b)
(b)
Figure 1. (a) Effect of different treatments on chilling injury rate of pepper during cold storage; (b) Effect of different treatments on chilling injury index of pepper during cold storage.
period. While the dramatic decline delayed by 10 days in T2, T3. At the end of the storage period, Vc content in T2, T3 were remarkably higher (2.9-fold, 2.1-fold, respectively) than that in T1 (p < 0.1).
Despite consistent decrease in chlorophyll content and Vc content of all treatments during the storage period but still spermidine and ice-temperature helped in retaining higher fruit chlorophyll and Vc when compared with control. The effectiveness of spermidine treatment was comparable to ice-tmeperature treatment, but was significantly more effective than that of control. The icetemperature appeared to result in a less decrease in chlorophyll content and Vc content.
3.3. Effect of Spermidine and Ice-Temperature on SOD Activity
SOD activity in both control and treated fruit increased firstly then decreased after storage for 20 days. Fruit pretreated with spermidine maintained remarkably higher SOD activity throughout the storage compared with the other two groups (Figure 2). The SOD activity peaked at 20 days and the maximum of SOD activities in T1, T2, T3 were 332.02 U·mg−1 protein, 349.2 U·mg−1 protein, 347.62 U·mg−1 protein, respectively. The changes of SOD in T2 and T3 exhibited a similar pattern during the storage until the last ten days. At the end of storage, the decrease of SOD activity in T1 was significantly higher than those in treated fruit, which was only amount to about 60.89 %, 56.67 % of those in T2, T3 (p < 0.01).
3.4. MDA Content and Electrolyte Leakage
MDA, the final product of lipid peroxidation, was regarded as the indicator of lipid peroxidation. Changes in MDA content was shown in Figure 3. In general, the
Table 2. Change of chlorophyll and vitamin C content of peppers under different conditions.

The fruit treated by different treatments at 4˚C was analysed. Each value corresponds to the mean ± SD of three replicates of ten fruit each. aSignificance: (*) significant at p ≤ 0.05; (**) Significant at p ≤ 0.01. Means within a row followed by different small letters indicate significant differences between treatments, L.S.D. test. Means within a column followed by different capital letters indicate significant differences between days of storage, L.S.D. test.
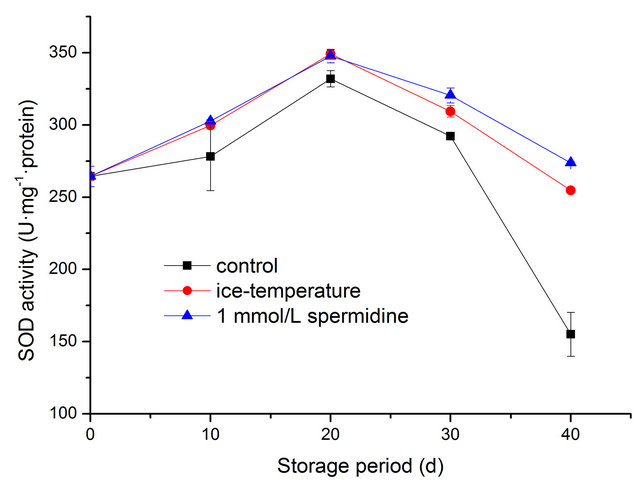
Figure 2. Effect of different treatments on SOD activity of pepper during cold storage.
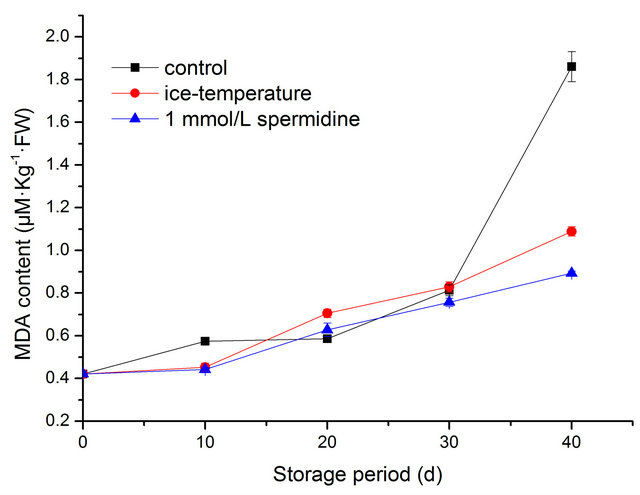
Figure 3. Effect of different treatments on MDA content of pepper during cold storage.
amount of MDA increased as the storage time increased. Spermidine and ice-temperature treatments significantly inhibited this increase of MDA content. At the end of the storage period, the MDA content of T2 and T3 was 1.088 µM·Kg−1·FW, 0.894 µM·Kg−1·FW, as opposed to 1.861 µM·Kg−1·FW in control samples. Compared with MDA content at the beginning of storage, control peppers showed a marked increase in MDA content by 342.4% over time. The results suggested that spermidine and icetemperature accounted for lower lipid peroxidation.
Membrane permeability changes during storage were analyzed by determining the relative electrolyte leakage. The leakage enhanced when the fruit suffered chilling injury. Changes in electrolyte leakage of pepper fruit were presented in Figure 4. The initial electrolyte leakage was 10.448%. In general, the electrolyte leakage increased persistently during storage both in samples and control group. During the first 10 days, the electrolyte leakage in T1 was much lower than that in T2 and T3 (p < 0.01). Thereafter, the electrolyte leakage in all samples were observed with a significant increase. The electrolyte leakage of T1, T2, T3 was 57.15%, 46.87%, 49.10%, respectively at the end of storage period. Compared with spermidine and ice-temperature fruits which remained non significant, the electrolyte leakage in control was considerably high (p < 0.01). Spermidine and ice-temperature treated fruit had a lower relative leakage rates than the control fruit, indicating that a higher membrane integrity was maintained.
4. Discussion
The application of spermidine and ice-temperature postponed the occurrence of chilling injury, alleviated CI symptoms of pepper fruit such as preventing pepper surface from pitting, diminishing the areas of chilling injury. Moreover, spermidine and ice-temperature maintained the pepper quality by delaying the decrease of chlorophyll content and Vc content.
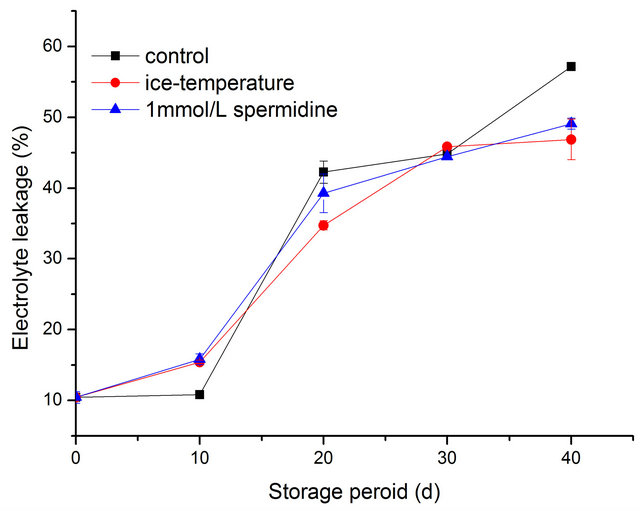
Figure 4. Effect of different treatments on cell membrane permeability of pepper during cold storage.
Various kinds of stress like low temperature, drought, salt result in metabolic disorder which is the consequence of oxidative oxidation resulting from overproduction of ROS, such as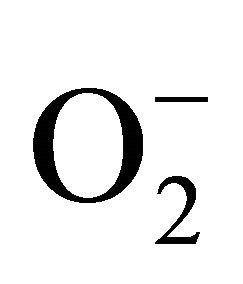 , singlet oxygen, H2O2 and hydroxyl radical, and breakdown of unsaturated fatty acids in membrane lipids [26]. Excess production of ROS contributes to the development and sensitivity of chilling injury. Therefore, protection from oxidative injury is crucial to cell survival under chilling stress and is thought to be a major mechanism of resistance to chilling stress. The onset of the tolerance has often been correlated with the accumulation of defense-related enzymes like SOD and compounds like ascorbic acid (Vc) [27]. Exogenous polyamines have been reported to be associated with the enhancement of reactive-oxygen-scavenging enzyme activities [28]. In our study, an increase in SOD activity was observed in pepper fruit during the first 20 days storage, following a sharp after that. Meanwhile, a remarkable increase in CI was presented in all treatments. At the end of storage, T3, in which SOD was the highest among these treatments, was also the one with the lowest level of chilling injury. The results suggested that the improvement of chilling tolerance in harvested peppers was related to the enhancement in activity of SOD. Similar results were also reported by Feng et al. [10]. Moreover, spermidine and ice-temperature pretreatments contributed to the weakening of chilling sensitive. Additionally, peppers pre-treated by spermidine and ice-temperature, especially ice-temperature, had relatively higher Vc content compared with control. It indicated that ascorbic acid may exert an effort on active oxygen scavenging. It has also been reported that exogenous polyamines not only resulted in accumulation of defense-related enzymes and compounds but also played a direct role in elimination of superoxide radicals generating through chemical and enzymatic pathways. Besides, triamine spermidine (Spd) and tetramine spermine (Spm) had higher efficiency of eliminating [29].
, singlet oxygen, H2O2 and hydroxyl radical, and breakdown of unsaturated fatty acids in membrane lipids [26]. Excess production of ROS contributes to the development and sensitivity of chilling injury. Therefore, protection from oxidative injury is crucial to cell survival under chilling stress and is thought to be a major mechanism of resistance to chilling stress. The onset of the tolerance has often been correlated with the accumulation of defense-related enzymes like SOD and compounds like ascorbic acid (Vc) [27]. Exogenous polyamines have been reported to be associated with the enhancement of reactive-oxygen-scavenging enzyme activities [28]. In our study, an increase in SOD activity was observed in pepper fruit during the first 20 days storage, following a sharp after that. Meanwhile, a remarkable increase in CI was presented in all treatments. At the end of storage, T3, in which SOD was the highest among these treatments, was also the one with the lowest level of chilling injury. The results suggested that the improvement of chilling tolerance in harvested peppers was related to the enhancement in activity of SOD. Similar results were also reported by Feng et al. [10]. Moreover, spermidine and ice-temperature pretreatments contributed to the weakening of chilling sensitive. Additionally, peppers pre-treated by spermidine and ice-temperature, especially ice-temperature, had relatively higher Vc content compared with control. It indicated that ascorbic acid may exert an effort on active oxygen scavenging. It has also been reported that exogenous polyamines not only resulted in accumulation of defense-related enzymes and compounds but also played a direct role in elimination of superoxide radicals generating through chemical and enzymatic pathways. Besides, triamine spermidine (Spd) and tetramine spermine (Spm) had higher efficiency of eliminating [29].
Pepper is so sensitive to low temperatures that they are vulnerable to chilling injury. Membranes are thought to be the primary sites for development of chilling injury. A unified theory to explain CI was found on low temperature induced membrane lipid phase transitions from a liquid-crystalline to a solid-gel state leading to a loss of membrane integrity and physiological dysfunction. Another theory was that CI results from direct effect of reduced temperatures on enzymes or indirect effect of membrane perturbations on intrinsic enzymes leading to accumulation of ROS and membrane lipid peroxidation [30]. The extent of lipid peroxidation depends on the degree of cold stress and is correlated with the extent of CI [31]. MDA increase was regarded as indicator of lipid peroxidation. In this study, a continuous increase in MDA content in all treated and control fruit was observed, whereas the application of spermidine and icetemperature significantly delayed the increase of MDA during the later storage duration (30 days later) leading to electrolyte leakage in consequence of membrane lipid peroxidation. Roberts et al. [32] found that exogenous polyamines could alter membrane fluidity as a conesquence of their combination with negatively charged phosphorlipid and other groups. Besides, exogenous polyamines and ice-temperature played an indirect role in regulating activities of membrane-bound enzymes exerting an effect on membrane stability and permeability. In this study, membrane permeability (revealed by electrolyte leakage) increased with storage duration, and after 20 days storage, the effectiveness of spermidine and icetemperature in electrolyte leakage began to show up compared with control. The results indicated that spermidine and ice-temperature had little impact on membrane permeability at the beginning of storage. As discussed above, spermidine and ice-temperature may be involved in a network of membrane metabolic pathways which regulate defense responses to chilling injury stress.
In conclusion, the use of spermidine and ice-temperature has been proved to enjoy the superiority when compared with the control fruit for chilling injury, chlorophyll content, Vc content as well as defense responses, however, the regulation of antioxidants as a result of spermidine and ice-temperature application is not clear. Besides, the protection mechanism of membrane permeability would need to be investigated in further studies.
REFERENCES
- G. F. Kramer and C. Y. Wang, “Correlation of Reduced Chilling Injury and Oxidative Damage with Increased Polyamine Levels in Zucchini Squash,” Plant, Vol. 76, No. 4, 1989, pp. 479-484. doi:10.1111/j.1399-3054.1989.tb05466.x
- G. A. Gonzalez-Aguilar, L. Zacarias, M. A. Perez Amador, et al., “Polyamine Content and Chilling Susceptibility Are Affected by Seasonal Changes in Temperature and by Conditioning Temperature in Cold-Stored ‘Fortune’ Mandarin Fruit,” Physiologia Plantarum, Vol. 108, No. 2, 2000, pp. 140-146. doi:10.1034/j.1399-3054.2000.108002140.x
- M. M. Kushad and G. Yelenosky, “Evaluation of Polyamine and Proline Levels during Low Temperature Acclimation of Citrus,” Plant Physiology, Vol. 84, No. 3, 1987, pp. 692-695. doi:10.1104/pp.84.3.692
- T. Hirofumi, H. Keiichi, M. Masashi, et al., “Comparison of Changes in Ethylene and Polyamines of Water Convolvulus and Chingensai Exposed to Chilling Stress,” Food Science and Technology Research, Vol. 5, No. 1, 1999, pp. 64-68. doi:10.3136/fstr.5.64
- G. A. Gonzάlez-Aguilar, L. Gayosso, R. Cruz, J. Fortiz, R. Bάez and C. Y. Wang, “Polyamines Induced by Hot Water Treatments Reduce Chilling Injury and Decay in Pepper Fruit,” Postharvest Biology and Technology, Vol. 18, No. 1, 2000, pp. 19-26. doi:10.1016/S0925-5214(99)00054-X
- D. Valero, D. Martínez-Romero and M. Serrano, “The Role of Polyamines in the Improvement of the Shelf Life of Fruit,” Trends in Food Science & Technology, Vol. 13, No. 6, 2002, pp. 228-234. doi:10.1016/S0924-2244(02)00134-6
- Y. M. Jiang and F. Chen, “A Study on Polyamine Change and Browning of Fruit during Cold Storage of Litchi,” Postharvest Biology and Technology, Vol. 5, No. 3, 1995, pp. 245-250. doi:10.1016/0925-5214(94)00021-J
- G. F. Kramer, C. Y. Wang and W. S. Conway, “Inhibition of Softening by Polyamines Application in ‘Golden Delicious’ and ‘McIntosh’ Apples,” Journal of the American Society for Horticultural Science, Vol. 116, No. 5, 1991, pp. 813-817.
- F. M. Basiouny, “Blueberry Fruit Quality and Storability Influenced by Postharvest Application of Polyamines and Heat Treatment,” Proceedings of the Florida State Horticultural Society, Vol. 109, 1996, pp. 269-272.
- Z. H. Feng, Y. L. Zhao, J. H. Li, G. Z. Yan, L. Wang and C. S. Wang, “Effect of Spermidine on Chilling-Sensitivity of ‘Okubao’ Peach,” Transactions of the Chinese Society for Agricultural Machinery, Vol. 40, No. 12, 2009, pp. 151-155.
- Y. L. Qiao, S. Q. Feng, L. P. Li, S. L. Zhang and Z. L. Liu, “Effect of Exogenous Polyamines Treatment on Chilling Injury and Storage Qualities of Cucumber,” Journal of Jilin Agricultural University, Vol. 27, No. 1, 2005, pp. 55-58.
- J. Kubiś, “Exogenous Spermidine Alters in Different Way Membrane Permeability and Lipid Peroxidation in Water Stressed Barley Leaves,” Acta Physiologiae Plantarum, Vol. 28, No. 1, 2006, pp. 27-33. doi:10.1007/s11738-006-0065-3
- W. R. Hu, Z. Q. Zhang, Y. M. Jiang and Z. L. Ji, “Study on the Parameter of Ice-Temperature Storage in Litchi (Litchi chinensis Sonn.),” Scientia Agricultural Sincia, Vol. 38, No. 4, 2005, pp. 797-802.
- H. L. Zhang, W. R. Hu, X. Q. Pang and Z. Q. Zhang, “Effect of Ice Temperature Technique and SO2 Releaser on Storage of Longan Fruits,” Acta Horticulturae Sinica, Vol. 33, No. 6, 2006, pp. 1325-1328.
- L. Guo, J. J. Cheng, Y. Ma, C. X. Zhao and S. Q. Tian, “Study on Storage of Green Pepper Controlled Freezing-Point,” Food Science, Vol. 25, No. 11, 2004, pp. 323- 325.
- L. G. Huang, H. N. Li, L. Zhang and Q. Z. Wang, “Influence of Ice-Temperature Storage on Lotus Roots,” Journal of Huazhong Agricultural University, Vol. 27, No. 2, 2008, pp. 317-320.
- N. Zhang, W. Q. Guan and Y. Zhang, “The Fresh-Keeping Effect of Ice-Temperature Combined with Plastic Film on Broccoli,” Food Science and Technology, Vol. 37, No. 1, 2012, pp. 42-44.
- S. H. Mirdehghan and M. Rahemi, “Effects of Hot Water Treatment on Reducing Chilling Injury of Pomegranate (Punica granatum) Fruit during Storage,” Acta Horticulturae, Vol. 3, No. 2005, pp. 887-892.
- W. Y. Chih, M. Murphy Terence, W. S. Wei and H. L. Chin, “H2O2 Treatment Induces Glutathione Accumulation and Chilling Tolerance in Mung Bean,” Functional Plant Biology, Vol. 29, No. 9, 2002, pp. 1081-1087. doi:10.1071/PP01264
- S. Alejandra, A. Lucía, M. Adela and C. Joaquín, “Reduction of Chilling Injury Symptoms in Persimmon Fruit cv. ‘Rojo Brillante’ by 1-MCP,” Postharvest Biology and Technology, Vol. 33, No. 3, 2004, pp. 285-291. doi:10.1016/j.postharvbio.2004.03.005
- A. Salvador, C. P. Carvalho, A. Monterde and J. M. Martinez-Jávega, “Note. 1-MCP Effect on Chilling Injury Development in ‘Nova’ and ‘Ortanique’ Mandarins,” Food Science and Technology International, Vol. 12, No. 6, 2006, pp. 165-170. doi:10.1177/1082013206063736
- S. P. Singh, Z. Singh and E. E. Swinny, “Postharvest Nitric Oxide Fumigation Delays Fruit Ripening and Alleviates Chilling Injury during Cold Storage of Japanese Plums (Prunus salicina Lindell),” Postharvest Biology and Technology, Vol. 53, No. 3, 2009, pp. 101-107. doi:10.1016/j.postharvbio.2009.04.007
- L. J. Wang, S. J. Chen, W. F. Kong, S. H. Li and D. D. Archbold, “Salicylic Acid Pretreatment Alleviates Chilling Injury and Affects the Antioxidant System and Heat Shock Proteins of Peaches during Cold Storage,” Postharvest Biology and Technology, Vol. 41, No. 3, 2006, pp. 244-251. doi:10.1016/j.postharvbio.2006.04.010
- Y. Xing, X. Li, Q. Xu, J. Yun, Y. Lu and Y. Tang, “Effects of Chitosan Coating Enriched with Cinnamon Oil on Qualitative Properties of Sweet Pepper (Capsicum annuum L.),” Food Chemistry, Vol. 124, No. 4, pp. 1443- 1450. doi:10.1016/j.foodchem.2010.07.105
- N. A. Abbasi, M. M. Kushad and A. G. Endress, “Active Oxygen-Scavenging Enzymes Activities in Developing Apple Flowers and Fruits,” Scientia Horticulturae, Vol. 74, No. 3, 1998, pp. 183-194. doi:10.1016/S0304-4238(98)00077-6
- J. M. Lyons, “Chilling Injury in Plants,” Annual Review of Plant Physiology, Vol. 24, 1973, pp. 445-466. doi:10.1146/annurev.pp.24.060173.002305
- R. A. Creelman and J. E. Mullet, “Biosynthesis and Action of Jasmonate in Plants,” Annual Review of Plant Physiology and Plant Molecular Biology, Vol. 48, 1997, pp. 355-381. doi:10.1146/annurev.arplant.48.1.355
- L. Jiang, “The Effect of Polyamines on Metabolism of Active Oxygen in Detached Leaves of Hhordeum vulgare var. nudum Hook. f.,” Acta Phytophysiologica Sinica, Vol. 19, No. 4, 1993, pp. 367-371.
- G. Drolet, E. B. Dumbroff, R. L. Legge and J. E. Thompson, “Radical Scavenging Properties of Polyamines,” Phytochemisty, Vol. 25, No. 2, 1986, pp. 367-371. doi:10.1016/S0031-9422(00)85482-5
- K. L. Parkin, A. Marangoni, R. L. Jackman, R. Y. Yada and D. W. Stanley, “Chilling Injury. A Review of Possible Mechanisms,” Journal of Food Biochemistry, Vol. 13, No. 2, 1989, pp. 127-153. doi:10.1111/j.1745-4514.1989.tb00389.x
- A. S. Lukatkin, “Contribution of Oxidative Stress to the Development of Cold Induced Damage to Leaves of Chilling-Sensitive Plants: Reactive Oxygen Species Formation during Plant Chilling,” Russian Journal of Plant Physiology, Vol. 49, No. 5, 2002, pp. 622-627. doi:10.1023/A:1020232700648
- D. R. Roberts, E. B. Dumbroff and J. E. Thompson, “Exogenous Polyamines Alter Membrane Fluidity in Bean Leaves—A Basis for Potential Misinterpretation of Their True Physiological Role,” Planta, Vol. 167, No. 3, 1986, pp. 395-401. doi:10.1007/BF00391345
NOTES
*Corresponding author.

