American Journal of Plant Sciences
Vol.07 No.14(2016), Article ID:71039,16 pages
10.4236/ajps.2016.714175
Isolation and Molecular Characterization of Elite Indigenous Rhizobia Nodulating Phaseolus bean (Phaseolus vulgaris L.)
Yusuph Namkeleja1, Kelvin Mtei2, Patrick A. Ndakidemi1
1Department of Sustainable Agriculture, Biodiversity and Ecosystem Management, The Nelson Mandela African Institution of Science and Technology, Arusha, Tanzania
2Department of Water and Environmental Science and Engineering, The Nelson Mandela African Institution of Science and Technology, Arusha, Tanzania
Copyright © 2016 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: June 8, 2016; Accepted: September 27, 2016; Published: September 30, 2016
ABSTRACT
Nowadays application of biological nitrogen fixation (BNF) through rhizobia inoculums is highly promoted as a solution to solve the problem of poor soil fertility in areas where legumes are cultivated. This is due to the fact that, rhizobia enhance nitrogen fixation, induce disease resistance, reduce heavy metal in the soil, facilitate bioavailabity of iron in soil and is environmental friendly. To get rhizobia strains which are suitable for inoculants production, isolation and molecular characterization of elite rhizobia are highly needed. Molecular characterization acts as a spark plug for discovery of many microbes including Rhizobia. Multi Locus Sequence Analysis (MLSA), 16S rRNA gene sequence analysis, DNA-DNA hybridization and SDS-PAGE analysis of the whole-cell proteins are the molecular techniques mostly used in characterizing rhizobia. But before deciding to use or not to use rhizobia inoculants in certain areas, knowing the population size of indigenous rhizobia found in that area is very important, because this is a major factor which determines inoculums responses as well gives clues on which areas need or do not need inoculation. The Most Probable Number (MPN) method is mostly used in enumerating rhizobia population of the soil. Given that, in most of the developing countries, including Tanzania, Biological Nitrogen Fixation (BNF) technology is not fully flourished; more efforts in isolation, molecular characterization of elite rhizobia and estimation of indigenous rhizobia population in various areas are required.
Keywords:
Biological Nitrogen Fixation, Inoculums, Rhizobium-Legume Symbiosis, Phaseolus Bean

1. Introduction
Phaseolus bean (Phaseolus vulgaris L.) is the third most important legume crop grown worldwide superseded by soy bean and peanuts [1] - [4] . Legumes exhibit high variability in their ability to fix atmospheric nitrogen whereby soybean and cowpea outperform Phaseolus bean. Although Phaseolus bean is a poor fixer of atmospheric nitrogen [5] [6] and its yield in Tanzania is below its production potential (<3 t∙ha−1), the crop is still widely grown in most regions of the country [7] - [9] . It is the major and the most important staple food legumes in Eastern, Central and Southern Africa [1] [9] - [11] . It is among the most important sources of dietary protein to human [11] . Its production in tropics and subtropics regions is deterred by poor soil fertility [9] [12] - [15] . This is due to the fact that this crop is mostly grown by the resource-poor small scale farmers [9] [16] who are not able to purchase expensive inorganic fertilizers.
The demand for increased food production due to increase of the world population and increased number of malnutrition cases has given a room for nitrogenous chemical fertilizers to be used in most cropping systems [17] . Likewise, in Tanzania, there is no practical feasibility of avoiding the use of inorganic fertilizers while meeting sustainable food needs, taking into consideration of annual population increase rate of 2.7% (National Bureau of Statistics, 2012). Despite the excessive use of nitrogenous chemical fertilizers, there is no remarkable improvement in crop yield [18] [19] . Nevertheless, excessive use of inorganic fertilizers is known to have undesirable effects on agriculture, food, biodiversity and environment at large [20] [21] . This situation created the need for finding other best alternatives for resolving the problem of poor soil fertility. Among alternatives thought, it is the use of Biological Nitrogen Fixation (BNF). BNF in large percent is done by a gram negative soil bacteria called rhizobia which are capable of fixing free atmospheric nitrogen (N2) into ammonia (NH3), a form which can be utilized by plants [22] [23] .
Nowadays, more emphasizes are on the use of BNF technology in agricultural systems in order to overcome the problems associated with depletion of soil fertility as well as reducing excessive use of inorganic fertilizers [24] - [26] . BNF by rhizobia is considered as an inexpensive and environmental friendly alternative to improve crop yield in comparison to its counterpart chemical nitrogen fertilizers [27] - [34] . It improves crop production by enhancing nitrogen fixation, preventing plant disease and chelating iron [35] - [37] . Inoculation of legumes with rhizobia inoculants is the mostly used BNF technology in agriculture as evidenced in previous studies [38] - [41] , but in most African countries including Tanzania, this technology is not well established [42] . BNF technology may be very successful if isolation and characterization of indigenous rhizobia found in the soil are done properly as may foster the discovery of strong strains for inoculants production [41] . Also it has been emphasized that prior to application of rhizobium inoculants in the field, it is necessary to determine population size of indigenous rhizobia found in that soil as indigenous rhizobia population influences inoculums responses [43] . The lower the population size of the indigenous rhizobia, the higher the responses of inoculums if other factors remain constant [43] - [45] . Singleton and Tavares [44] have reported that, it is not possible to enhance N2-fixation when indigenous soil rhizobia populations were above threshold number of 102 cells of bacteria per gram of soil. Therefore, there is a need of doing isolation, molecular characterization of elite rhizobia strains and determining population of indigenous rhizobia nodulating Phaseolus beans in various places. This will help to identify the suitable rhizobia strains for inoculants production aiming in enhancing crop yield and identifying right places where inoculation is needed.
2. Rhizobia-Legume Symbiosis
Rhizobia-legume symbiosis is the most studied plant-bacteria mutualism [46] . Rhizobia are soil bacteria that are well known for their symbiotic relationship with legumes even though they are also found in soil devoid of legumes [23] [47] - [49] . Rhizobia are grouped into two major groups which are fast grower and slow growers [50] . Fast grower takes 3 - 5 days to grow on the media and when grown in media containing bromothymol blue (BTB) indicator, they undergo alkaline reaction. While slow growing rhizobia takes about 7-10 days to grow on the media and show acidic reaction on BTB [51] . Most of rhizobia which nodulate Phaseouls beans are from genus Rhizobium, and species belongs to genus Rhizobium are fast growers [50] . Rhizobia range from symbiotic which nodulate legumes to non symbiotic which are unable to nodulate legumes at all. Symbiotic rhizobia are divided in two groups which are mutualists and parasites. Mutualist rhizobia supply their hosts with nitrogen at a reasonable carbon cost while parasite rhizobia infect legume plants but they fix little or no nitrogen inside their nodules [48] . This review is based on mutualist rhizobia.
Mutualist rhizobia convert atmospheric nitrogen to ammonia and provide organic nitrogenous compounds to the plants [22] [37] . Through symbiotic relationship between rhizobia and legume, rhizobia provide combined form of nitrogen to plant while plant provides shelter and energy to rhizobia [23] [47] - [49] . Some findings explained that, the drivers of rhizobia-legume interaction are dryness and infertility of the soils [51] . That means the rhizobia nodulate legume in order to get shelter from hostile environment and legume accepts rhizobia in order to get access to combined nitrogen hence be able to survive and colonize the area which are dry with low nitrogen (infertile areas). But the real origin of rhizobia-legume symbiosis is not known up to moment [23] . What is a known is that, symbiosis between rhizobia and legumes is genetically controlled. Rhizobia have a nodulation gene (nodABC) which encode enzyme responsible for core structure of the signal molecule (Nod factor, NF) needed to induce nodule formation in host plant [23] . Initial interaction consists of stimulation of biochemical activity in the rhizobial strains by flavonoid and isoflavonoid molecules in the plant root exudates [53] . These compounds stimulate the activity of nod (nodulation) genes, the gene whose products are required to enable nodulation of the cognate legume host [22] .
3. Significance of Legume-Rhizobia Symbiosis
Rhizobia are found to have many benefits in agriculture industry. Some of the documented benefits includes; promotion of plant growth through nitrogen fixation, solubilization of insoluble phosphate, controlling crop diseases, chelation of iron and bioremediation of heavy metals [26] [35] - [37] [54] [55] .
3.1. Nitrogen Fixation
All living organisms need nitrogen for their survival [22] . It is the nutrient most required for plant growth and is a key for good yield of agriculturally important crops [56] . It is a primary nutrient for plant growth and survival due to the fact that nitrogen is a source of cells proteins, enzymes and chlorophyll [57] . Nitrogen is efficiently utilized by many organisms (all eukaryotes) when it is in form of ammonium NH4 or nitrate NO3 [22] [54] . But high amount of nitrogen that is found in the earth is atmospheric dinitrogen gas (N2) which cannot be directly used by the plants. Some Bacteria (including rhizobia) and Archaea are the only organisms that can reduce atmospheric nitrogen to ammonia through a process known as biological nitrogen fixation [22] [41] .
Legumes including Common bean acquire nitrogen by living symbiotically with rhizobia which are capable of fixing atmospheric nitrogen (N2) in root nodules and supplying it to the plant [56] . Genera Rhizobium, Mesorhizobium, Allorhizobium, Sinorhizobium, Arzorhizobium and Bradyrhizobium which all together form Rhizobia, play a vital role in converting free nitrogen in the soil into ammonia [54] . Also some species of bacteria that belongs to genus Bacillus like B. cereus, B. fusiformis, B. marisflavi and B. alkalidiazotrophicus are involved in nitrogen fixation [58] but rhizobia remains as the most effective nitrogen fixing bacteria [23] [59] - [61] . It was reported that, in tropics and subtropics, legume-rhizobia symbiosis of is a key player in nitrogen cycle and is a major contributor of nitrogen to terrestrial biosphere [41] . Additionally, Lindström and Mousavi [23] reported that BNF by rhizobia is the most efficient system with a mean annual fixation rate of 55,140 kgN per hectare, compared with 0.330 kg N per hectare for other biological systems. It∙has been estimated that symbiotic interaction between microbes and legumes worldwide reduce about 100 million metric tons of atmospheric nitrogen per year hence saving about US$ 8 billion per year in fertilizer [62] . Moreover, BNF is considered as relatively inexpensive in comparison to its counterpart chemical nitrogen fertilizers as Jonah, Chemining’wa [30] expounded that, the cost of inoculants required for one hectare is about US$ 4 while the cost of chemical nitrogen fertilizers required for the same area (one hectare) is about US$ 45. Therefore, there is a need of providing knowledge on the significance of BNF and enhancing availability and use of inoculants through isolation and characterization of rhizobia in various legumes. If BNF is efficiently utilized, it will enhance legumes production as well as reducing cost of production.
3.2. Disease Control
Apart from nitrogen fixation, rhizobia are able to induce plant resistance against disease [55] [63] . They are reported to have the ability of controlling crop diseases through elimination of the plant’s enemies including microbial pathogens, insects and weeds [37] [64] . They induce defensive enzymes and chemicals that suppress pathogens and enabling a process called Induced Systematic Resistance (ISR) to plants [65] . Findings from Ehteshamul-Haque and Ghaffar [66] proved that Rhizobium meliloti can inhibit growth of pathogens like Macrophomina phaseolina, Rhizoctonia solani and Fusarium solani while Bradyrhizobium japonicum can inhibit pathogens like M. phaseolina and R. solani in leguminous (soybean, mung bean) and non-leguminous (sunflower and okra) plants. Pawar, Pawar [37] , reported that Rhizobium secrete antifungal compounds which selectively inhibit the growth of pathogenic fungi only and not rhizospheric bacteria. Due to these findings there is a need of continued searching for rhizobia strain for Phaseolus bean that will be efficient in nitrogen fixation as well as in providing resistance to diseases. This will be possible through isolation and characterizations of indigenous rhizobia with dual purpose characteristics. By doing that, it will improve production of both legumes and non-legumes crops because rhizobia will reduce pathogens infections to all crops.
3.3. Solubilization of Insoluble Phosphate
Phosphorus (P) is one of the most essential macronutrients for the growth and development of plants [66] - [69] . The natural source of phosphorous (P) in the soil is organic and mineral phosphates. But both of them organic and mineral phosphates in large quantity are found inform of insoluble phosphate which cannot be utilized by the plant, a situation making soluble phosphate (a form that can be utilized by the plants) to be always scarce in the soil [67] [70] [71] . Due to the scarcity nature and importance of soluble phosphate for plant growth and crop production, leads to the excess use of chemical P fertilizers which have economic and environmental burdens [67] . To reduce excessive use of industrial chemical P fertilizers, rhizobia-legume symbiosis considered as among the most powerful solution. Rhizobium-legume symbiosis enhances utilization of naturally available insoluble phosphate to make phosphorous available for plants. Research evidence has established that Rhizobium is among the most powerful phosphate solubilizer [67] [68] . Rhizobium leguminosarum biovar viceae and Rhizobium meliloti are the few examples of rhizobium species which are capable of solubilizing insoluble phosphate [72] [73] . In that context determining and isolation of rhizobia species which are suitable for inoculants production are activities of special vitality for enhancing crop production and environmental protection, isolation and characterization of rhizobia strains.
3.4. Chelation of Iron and Bioremediation of Heavy Metals
Iron is the essential element for plant growth [74] as it responsible for formation of chlorophyll. Most iron in the soil is in the form of ferric ion (F3+) in which plant can’t utilize it hence leads to scarcity of bioavailable iron in soil and plant surface [74] [75] . Under iron-limiting conditions, rhizobia tend to produce low-molecular-weight compounds called siderophores to acquire enough ferric ion [74] [76] . Siderophore (iron bearer) have ability to convert ferric iron F3+ into soluble F3+ and they transfer soluble F3+ and delivering it to the plant roots surfaces where becomes reduced to Fe2+ and absorbed by plants [77] [78] . Apart from iron, Rhizobacteria also have ability to chelate other heavy metals such as cadmium, lead, nickel, arsenic, aluminium, magnesium zinc, copper, cobalt, and controlling its mobility and availability to the growing plant through release of chelating agents, acidification, phosphate solubilization and redox changes [79] . For instance study done by Saleh and Saleh [80] indicated that dual inoculation with Arbuscular mycorrhizal (AM) fungus and Rhizobium on the host plant cowpea (Vigna sinensis) lead to increased tolerance against high concentration of Zinc and Cadmium. Therefore rhizobia-legume symbiosis is important in reducing heavy metals in the soil and it has been proposed as a proper tool for bioremediation of heavy metals in the soil.
4. Determining Indigenous Rhizobia Population
Inoculation of legumes with introduced rhizobia strain is a common agricultural practice intended to promote nitrogen fixation and increasing crop yield [43] . But for Rhizobium inoculants to be efficient in fixing nitrogen as well as increasing crop yield, population size of indigenous rhizobia strains play an importance role [31] . The size of the indigenous rhizobia population is the most powerful environmental factors that determine the competitive success of inoculated rhizobia versus indigenous rhizobia strain found in the area [81] . It has been found that the likelihood of a response to inoculation with Rhizobium strains decreased as the numbers of indigenous rhizobia increased [43] [45] . Singleton and Tavares [44] found the same inverse correlation between rhizobia inoculants with native rizhobia population. According to Singleton and Tavares [44] , introduced rhizobia strains (inoculants) are always outcompeted with the native rhizobia strains, thus, it is not possible to enhance N2-fixation when indigenous soil rhizobia populations were above threshold number (102 cells of bacteria per gram of soil) and had some effective strains. Amijee and Giller [10] reported that response of Phaseolus vulgaris to Rhizobium inoculants is not common in areas with large number of indigenous rhizobia. Furthermore, Meade, Higgins [31] explained that the number of indigenous rhizobia present in the soil before inoculation affect the concentration of inoculants required in that area. Study done by Weaver and Frederick [82] indicated that for rhizobia inoculums to be able to form 50% or more of the nodules must be applied in a rate of 1000 times higher than the size of the indigenous population in the soil. Also increase in economic yield due to inoculation is a function of indigenous rhizobia present in the soil [45] . That means economic yield due to inoculums application increase as a number of indigenous rhizobia per gram of soil decrease and vice versa (Figure 1). It is important to note that the indigenous rhizobia population is not the only factor that determines inoculums responses as there are other factors such as soil N availability, physiochemical constraints (like soil pH and salinity) and climatic conditions such as temperature, moisture content and oxygen stresses [12] [43] [44] [83] . However, the population of indigenous rhizobia is among of the most important factor. For that reason, many findings recommended that for successful use of rhizobia
Figure 1. Comparison of the fit of observed to estimated inoculation responses by use of a hyperbolic equation to describe the relationship between numbers of indigenous rhizobia and legume inoculation responses. Only 26 of the 29 total species-site combination are presented. Source: [45] .
inoculants, there should be prior knowledge on the size of the indigenous rhizobia population [31] [44] [84] . Therefore, in order to know which areas need inoculation and at which quantity, knowing population of indigenous rhizobia found in that area is of importance.
Method for Determining Population of Indigenous Rhizobia in the Soil
To make easy determination of indigenous rhizobia population found in the soil the Most Probable Number (MPN) method are usually used. MPN is the widely used approach in enumerating number of rhizobia found in the soils [85] - [87] . The MPN method of population estimate is based on the equation:
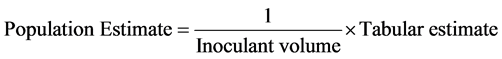
Additionally, computer software called Most Probable Number Enumeration System (MPNES) is employed to perform the same task (Woomer et al., 1990).
5. Isolation of Elite Rhizobia Strains
Rhizobia strains isolation is done by taking nodules from the host legume, sterilizing them by using ethanol and sodium hypochlorite, followed by crushing it in sterile petri dish by using blunt tipped sterilized forceps and lastly streaking drop of the nodule suspension on the media. Yeast-Mannitol agar (YMA) and peptone glucose agar are used as growth media while bromothymol blue (BTB) or Congo Red (CR) are used as indicators [50] [51] . In most cases, YMA containing CR or BTB indicators are used as evidenced in many studies [40] [62] [88] . The use of indicator media reported to camouflage real morphologies and distort growth rate of the rhizobia [51] . Because of this drawback, some isolation is done on YMA plates without indicator media. Rhizobia isolation which does not involve use of indicator media, aims at investigating uniformity of colonies growth across the plates, uniformity indicate pure culture while non- uniformity indicate contamination.
Isolation of rhizobia is a valuable process to maximize agricultural production [89] - [91] . It helps to get strong strain for nitrogen fixation, because effectiveness in nitrogen fixation by soil rhizobia population do vary widely between species [44] and the number of rhizobia that are not yet known is big and exceed the known one [22] [92] . Therefore, isolation of elite rhizobia is a stepping stone towards discovering effective strain that will be more efficient in fixing nitrogen for various legumes. Isolation of rhizobia strain from nonspecific (promiscuous) legumes gives a wide chance of identifying new effective strain for such legumes. Common bean (Phaseolus vulgaris L.) is amongst the promiscuous legume hosts [1] [93] and several rhizobia species have been reported to nodulate this legume, although not always effective in terms of fixing N2 [56] . Since common bean is a most important legume crop in most African countries as earlier mentioned, there is a need of isolating elite rhizobia strains nodulating it in areas where its production is practiced but yield potential has never been realized.
6. Molecular Characterization of Rhizobia
The use of molecular techniques in the characterization of microorganisms steered discovery of several new bacterial phylogenetics that were previously unknown [41] [94] [95] . Molecular characterization came in practice because classification based on morphological and physiological characters did not reflect true evolutionary relationship [22] . Among the molecular techniques used in characterization of the bacteria includes; Multi Locus Sequence Analysis (MLSA), Sequence analysis of 16S rDNA, 16S rRNA gene sequence analysis, DNA-DNA hybridization and SDS-PAGE analysis of the whole-cell proteins [62] [96] .
The sequence analysis of 16S rRNA and DNA-DNA hybridization has been used frequently in bacteria taxonomy [1] . 16S rRNA gene sequencing enables classification to genus level, while DNA-DNA hybridization helps classification to species level [97] . The 16S rRNA gene is mostly used in characterization and classification of bacteria because it contains conserved small sub-unit which does not undergo mutation easily with time [22] [97] . It is found in almost all bacteria, and is large (1500 bp) enough for informatics purposes [17] [98] . But it has been confirmed that, the use of 16S rRNA gene sequencing alone as phylogenetic marker for characterising bacteria is difficult and may give wrong results [1] . This is due to the reason that 16S rRNA gene have low divergence between closely related species [99] [100] hence cannot distinguish between recently distinguished species [101] . Also it is susceptible to genetic recombination and horizontal gene transfer and it found in multiple copies in genome of some bacteria [3] [102] . To solve this problem, the DNA-DNA hybridization came as a solution to the weaknesses of 16S rRNA gene sequence analysis.
DNA-DNA hybridization is able to differentiate between closely related species [96] [103] , but it has been reported that, its results tend to vary between laboratories where DNA-DNA hybridization was done, a situation which may leads to conflicting result for the same sets of strains [1] [104] . Nowadays, the Multi Locus Sequence Analysis (MLSA) is considered as the best in species identification and delineation than 16S rRNA gene sequence analysis and DNA?DNA hybridization [105] . Despite the drawbacks they have, 16S rRNA gene sequence analysis and DNA-DNA hybridization are still the most frequently used molecular techniques in taxonomy of bacteria as evidenced in many studies. The use of such molecular techniques to characterize elite rhizobia strains will help to identify exactly what species of rhizobia are effective for nodulating and fixing nitrogen and performing other functions in Phaseolus bean, thus making easy in inoculants production. Unfortunately in Tanzania there are very few studies concerning molecular characterization of rhizobia isolated from the natural environments, hence more researches in this area are needed.
7. Conclusion
Isolation, molecular characterization of elite rhizobia strains nodulating Phaseolus bean and enumeration of indigenous rhizobia population in soil where beans are cultivated or expected to be cultivated are important for increasing productivity. It may help to broaden knowledge on rhizobiology as well as to prosper the discovery of unknown effective and efficient strain of rhizobia and ultimately to enhance inoculums production. More efforts and interest must be directed in this area so as to rescue the productivity of legumes considering that legumes are the major source of dietary protein and carbohydrates for human being.
Acknowledgements
We thank Nelson Mandela African Institution of Science and Technology and the Government of the United Republic of Tanzania and N2Africa project for their support in this work.
Cite this paper
Namkeleja, Y., Mtei, K. and Ndakidemi, P.A. (2016) Isolation and Molecular Characterization of Elite Indigenous Rhizobia Nodulating Phaseolus bean (Phaseolus vulgaris L.). American Journal of Plant Sciences, 7, 1905-1920. http://dx.doi.org/10.4236/ajps.2016.714175
References
- 1. Aserse, A.A., et al. (2012) Phylogeny and Genetic Diversity of Native Rhizobia Nodulating Common Bean (Phaseolus vulgaris L.) in Ethiopia. Systematic and Applied Microbiology, 35, 120-131.
http://dx.doi.org/10.1016/j.syapm.2011.11.005 - 2. Faghire, M., et al. (2012) Identification at the Species and Symbiovar Levels of Strains Nodulating Phaseolus vulgaris in Saline Soils of the Marrakech Region (Morocco) and Analysis of the otsA Gene Putatively Involved in Osmotolerance. Systematic and Applied Microbiology, 35, 156-164.
http://dx.doi.org/10.1016/j.syapm.2012.02.003 - 3. Van Berkum, P., Beyene, D. and Eardly, B.D. (1996) Phylogenetic Relationships among Rhizobium Species Nodulating the Common Bean (Phaseolus vulgaris L.). International Journal of Systematic Bacteriology, 46, 240-244.
http://dx.doi.org/10.1099/00207713-46-1-240 - 4. Cao, Y., et al. (2014) Diversity and Distribution of Rhizobia Nodulated with Phaseolus vulgaris in Two Ecoregions of China. Soil Biology and Biochemistry, 78, 128-137.
http://dx.doi.org/10.1016/j.soilbio.2014.07.026 - 5. Giller, K. (1990) Assessment and Improvement of Nitrogen Fixation in Tropical Phaseolus vulgaris L. Soil Use and Management, 6, 82-84.
http://dx.doi.org/10.1111/j.1475-2743.1990.tb00809.x - 6. Mnasri, B., et al. (2014) Rhizobium azibense sp. nov., a Nitrogen Fixing Bacterium Isolated from Root-Nodules of Phaseolus vulgaris. International Journal of Systematic and Evolutionary Microbiology, 64, 1501-1506.
http://dx.doi.org/10.1099/ijs.0.058651-0 - 7. Hillocks, R., et al. (2006) Phaseolus Bean Improvement in Tanzania, 1959-2005. Euphytica, 150, 215-231.
http://dx.doi.org/10.1007/s10681-006-9112-9 - 8. Kisetu, E., Silayo, S.A. and Tsere, G.S. (2013) Use of Predictive Screening Parameters in Selected Common Bean Genotypes to Assess Their Salt Tolerance Ability Using NaCl Concentration. Advanced Journal of Agricultural Research, 1, 51-60.
- 9. Ndakidemi, P., et al. (2006) Yield and Economic Benefits of Common Bean (Phaseolus vulgaris) and Soybean (Glycine Max) Inoculation in Northern Tanzania. Australian Journal of Experimental Agriculture, 46, 571-577.
http://dx.doi.org/10.1071/EA03157 - 10. Amijee, F. and Giller, K.E. (1998) Environmental Constraints to Nodulation and Nitrogen Fixation of Phaseolus vulgaris L. in Tanzania. I. A Survey of Soil Fertility, Root Nodulation and Multi-Locational respoNses to Rhizobium Inoculation. African Crop Science Journal, 6, 159-169.
http://dx.doi.org/10.4314/acsj.v6i2.27812 - 11. Ribeiro, R.A., et al. (2013) Novel Rhizobium Lineages Isolated from Root Nodules of the Common Bean (Phaseolus vulgaris L.) in Andean and Mesoamerican Areas. Research in Microbiology, 164, 740-748.
http://dx.doi.org/10.1016/j.resmic.2013.05.002 - 12. Giller, K., et al. (1998) Environmental Constraints to Nodulation and Nitrogen Fixation of Phaseolus vulgaris L in Tanzania II Response to N and P Fertilizers and Inoculation with Rhizobium. African Crop Science Journal, 6, 171-178.
http://dx.doi.org/10.4314/acsj.v6i2.27813 - 13. Mwandemele, O.D. and Nchimbi, S.F. (1990) Country reports-Tanzania. In: Smithson, J.B., Ed., Proceedings of Workshop on Bean Varietal Improvement in Africa, Maseru, 163-178.
- 14. Okalebo, J., et al. (2007) Available Technologies to Replenish Soil Fertility in East Africa. In: Bationo, A., Waswa, B., Kihara, J. and Kimetu, J., Eds., Advances in Integrated Soil Fertility Management in Sub-Saharan Africa: Challenges and Opportunities, Springer, Netherlands, 45-62.
http://dx.doi.org/10.1007/978-1-4020-5760-1_3 - 15. Pereira, R.M., et al. (2006) Molecular Characterization of Bacterial Populations of Different Soils. Brazilian Journal of Microbiology, 37, 439-447.
http://dx.doi.org/10.1590/S1517-83822006000400007 - 16. Graham, P. (1981) Some Problems of Nodulation and Symbiotic Nitrogen Fixation in Phaseolus vulgaris L.: A Review. Field Crops Research, 4, 93-112.
http://dx.doi.org/10.1016/0378-4290(81)90060-5 - 17. Schroder, J.J. (2014) The Position of Mineral Nitrogen Fertilizer in Efficient Use of Nitrogen and Land: A Review. Natural Resources, 5, 936-948.
http://dx.doi.org/10.4236/nr.2014.515080 - 18. Niazi, T. (2004) Rural Poverty and the Green Revolution: The Lessons from Pakistan. The Journal of Peasant Studies, 31, 242-260.
http://dx.doi.org/10.1080/0306615042000224294 - 19. Sanfo, S. and Gérard, F. (2012) Public Policies for Rural Poverty Alleviation: The Case of Agricultural Households in the Plateau Central Area of Burkina Faso. Agricultural Systems, 110, 1-9.
http://dx.doi.org/10.1016/j.agsy.2012.02.006 - 20. Davidson, E.A. (2009) The Contribution of Manure and Fertilizer Nitrogen to Atmospheric Nitrous Oxide Since 1860. Nature Geoscience, 2, 659-662.
http://dx.doi.org/10.1038/ngeo608 - 21. Suprapta, D.N. (2012) Potential of Microbial Antagonists as Biocontrol Agents against Plant Fungal Pathogens. Journal of International Society for Southeast Asian Agricultural Sciences, 18, 1-8.
- 22. Giller, K.E. (2001) Nitrogen Fixation in Tropical Cropping Systems. CABI.
http://dx.doi.org/10.1079/9780851994178.0000 - 23. Lindstrom, K. and Mousavi, S.A. (2010) Rhizobium and Other N-Fixing Symbioses. eLS.
- 24. Tairo, E.V. and Ndakidemi, P.A. (2013) Bradyrhizobium japonicum Inoculation and Phosphorus Supplementation on Growth and Chlorophyll Accumulation in Soybean (Glycine max L.). American Journal of Plant Sciences, 4, 2281-2289.
http://dx.doi.org/10.4236/ajps.2013.412282 - 25. Nyoki, D. and Ndakidemi, P.A. (2014) Effects of Bradyrhizobium japonicum Inoculation and Supplementation with Phosphorus on Macronutrients Uptake in Cowpea (Vigna unguiculata (L.) Walp). American Journal of Plant Sciences, 5, 442-451.
http://dx.doi.org/10.4236/ajps.2014.54058 - 26. Loganathan, M., Garg, R., Saha, S., Bag, T.K. anf Rai, A.B. (2010) Selection of Antagonistic Rhizobacteria against Soil Borne Pathogens. Journal of Mycopathological Research, 48, 227-232.
- 27. Gaurav, S., Chatterjee, S. and Chandra, M. (2009) Efficient Nitrogen Fixing Rhizobial Isolate Infecting Vigna radiata L. Asian Journal of Agricultural Sciences, 1, 62-65.
- 28. Ghimire, A. (2002) A Review on Organic Farming for Sustainable Agriculture. Department of Agriculture Extension and Rural Sociology, Institute of Agriculture and Animal Science Rampur, Chitwan, 6.
- 29. Haru, A. and Ethiopia, W. (2012) Influences of Inoculation Methods and Phosphorus Levels on Nitrogen Fixation Attributes and Yield of Soybean (Glycine max L.) at Haru, Western Ethiopia. American Journal of Plant Nutrition and Fertilization Technology, 2, 45-55.
http://dx.doi.org/10.3923/ajpnft.2012.45.55 - 30. Jonah, N., Chemining’wa, G.N., Muthomi, J.W. and Shibairo, S.I. (2012) Effect of Rhizobium Inoculation and Nitrogen Fertilizer Application on Growth, Nodulation and Yield of Two Garden Pea Genotypes. Journal of Animal & Plant Sciences, 15, 2147-2156.
- 31. Meade, J., Higgins, P. and O’Gara, F. (1985) Studies on the Inoculation and Competitiveness of a Rhizobium leguminosarum Strain in Soils Containing Indigenous Rhizobia. Applied and Environmental Microbiology, 49, 899-903.
- 32. Mfilinge, A., Mtei, K. and Ndakidemi, P.A. (2015) Economic Benefit of Rhizobial Inoculation and Fertilisation with Phosphorus and Potassium on Selected Bush Bean Varieties [Phaseolus vulgaris L.] in Northern Tanzania. African Journal of Applied Agricultural Sciences and Technologies, 2, 57-71.
- 33. Nyoki, D. and Patrick, A.N. (2013) Economic Benefits of Bradyrhizobium japonicum Inoculation and Phosphorus Supplementation in Cowpea (Vigna unguiculata (L) Walp) Grown in Northern Tanzania. American Journal of Research Communication, 1, 173-189.
- 34. Tairo, E.V. and Ndakidemi, P.A. (2013) Yields and Economic Benefits of Soybean (Glycine max L.) as Affected by Bradyrhizobium japonicum Inoculation and Phosphorus Supplementation. American Journal of Research Communication, 1, 159-172.
- 35. Bhattacharyya, P. and Jha, D. (2012) Plant Growth-Promoting Rhizobacteria (PGPR): Emergence in Agriculture. World Journal of Microbiology and Biotechnology, 28, 1327-1350.
http://dx.doi.org/10.1007/s11274-011-0979-9 - 36. Kala, T.C., Christi, R.M. and Bai, N.R. (2011) Effect of Rhizobium Inoculation on the Growth and Yield of Horsegram (Dolichos biflorus Linn). Plant Archives, 11, 97-99.
- 37. Pawar, V.A., et al. (2014) Effect of Rhizobium on Seed Germination and Growth of Plants. Journal of Academia and Industrial Research (JAIR), 3, 84.
- 38. Bull, C., Shetty, K. and Subbarao, K. (2002) Interactions between Myxobacteria, Plant Pathogenic Fungi, and Biocontrol Agents. Plant Disease, 86, 889-896.
http://dx.doi.org/10.1094/PDIS.2002.86.8.889 - 39. Chisholm, S.T., Coaker, G., Day, B. and Staskawicz, B.J. (2006) Host-Microbe Interactions: Shaping the Evolution of the Plant Immune Response. Cell, 124, 803-814.
http://dx.doi.org/10.1016/j.cell.2006.02.008 - 40. Deshwal, V.K. and Chaubey, A. (2014) Isolation and Characterization of Rhizobium leguminosarum from Root Nodule of Pisum sativum L. Journal of Academia and Industrial Research (JAIR), 2, 464.
- 41. Lindstrom, K., Murwira, M., Willems, A. and Altier, N. (2010) The Biodiversity of Beneficial Microbe-Host Mutualism: The Case of Rhizobia. Research in Microbiology, 161, 453-463.
http://dx.doi.org/10.1016/j.resmic.2010.05.005 - 42. Bala, A., Karanja, N., Murwira, M., Lwimbi, L., Abaidoo, R. and Giller, K. (2011) Production and Use of Rhizobial Inoculants in Africa. N2Africa.
- 43. Thies, J.E., Singleton, P.W. and Bohlool, B.B. (1991) Influence of the Size of Indigenous Rhizobial Populations on Establishment and Symbiotic Performance of Introduced Rhizobia on Field-Grown Legumes. Applied and Environmental Microbiology, 57, 19-28.
- 44. Singleton, P. and Tavares, J. (1986) Inoculation Response of Legumes in Relation to the Number and Effectiveness of Indigenous Rhizobium Populations. Applied and Environmental Microbiology, 51, 1013-1018.
- 45. Thies, J.E., Singleton, P.W. and Bohlool, B.B. (1991) Modeling Symbiotic Performance of Introduced Rhizobia in the Field by Use of Indices of Indigenous Population Size and Nitrogen Status of the Soil. Applied and Environmental Microbiology, 57, 29-37.
- 46. Subramanian, S.B., Yan, S., Tyagi, R., Surampalli, R. and Zhang, T. (2009) Biofertilizers/ Bioinoculants. In: Tyagi, R.D., Surampalli, R.Y., Yan, S., Zhang, T.C., Kao, C.M. and Lohani, B.N., Eds., Sustainable Sludge Management: Production of Value Added Products, American Society of Civil Engineers (ASCE), 203-230.
http://dx.doi.org/10.1061/9780784410516.ch09 - 47. Denison, R.F. and Kiers, E.T. (2004) Why Are Most Rhizobia Beneficial to Their Plant Hosts, Rather than Parasitic? Microbes and Infection, 6, 1235-1239.
http://dx.doi.org/10.1016/j.micinf.2004.08.005 - 48. Denison, R.F. and Kiers, E.T. (2004) Lifestyle Alternatives for Rhizobia: Mutualism, Parasitism, and Forgoing Symbiosis. FEMS Microbiology Letters, 237, 187-193.
http://dx.doi.org/10.1111/j.1574-6968.2004.tb09695.x - 49. West, S.A., Kiers, E.T., Simms, E.L. and Denison, R.F. (2002) Sanctions and Mutualism Stability: Why Do Rhizobia Fix Nitrogen? Proceedings of the Royal Society of London B: Biological Sciences, 269, 685-694.
http://dx.doi.org/10.1098/rspb.2001.1878 - 50. Somasegaran, P. and Hoben, H.J. (2012) Handbook for Rhizobia: Methods in Legume-Rhizobium Technology. Springer Science & Business Media, Berlin.
- 51. Bala, A., Abaidoo, R. and Woomer, P. (2010) Rhizobia Strain Isolation and Characterisation Protocol. www.N2Africa.org
- 52. Schrire, B., Lewis, G. and Lavin, M. (2005) Biogeography of the Leguminosae. In: Lewis, G.P., Schrire, B.D., MacKinder, B. and Lock, M., Eds., Legumes of the World, Kew Publishing, 21-54.
- 53. Ndakidemi, P.A. and Dakora, F.D. (2003) Legume Seed Flavonoids and Nitrogenous Metabolites as Signals and Protectants in Early Seedling Development. Functional Plant Biology, 30, 729-745.
http://dx.doi.org/10.1071/FP03042 - 54. Loganathan, M., Garg, R., Venkataravanappa, V., Saha, S. and Rai, A.B. (2014) Plant Growth Promoting Rhizobacteria (PGPR) Induces Resistance against Fusarium wilt and Improves Lycopene Content and Texture in Tomato. African Journal of Microbiology Research, 8, 1105-1111.
http://dx.doi.org/10.5897/AJMR2013.5653 - 55. Zaidi, A., Khan, M., Ahemad, M. and Oves, M. (2009) Plant Growth Promotion by Phosphate Solubilizing Bacteria. Acta Microbiologica et Immunologica Hungarica, 56, 263-284.
http://dx.doi.org/10.1556/AMicr.56.2009.3.6 - 56. Dall’Agnol, R.F., et al. (2014) Rhizobium paranaense sp. nov., an Effective N2-Fixing Symbiont of Common Bean (Phaseolus vulgaris L.) with Broad Geographical Distribution in Brazil. International Journal of Systematic and Evolutionary Microbiology, 64, 3222-3229.
http://dx.doi.org/10.1099/ijs.0.064543-0 - 57. Matiru, V.N. and Dakora, F.D. (2004) Potential Use of Rhizobial Bacteria as Promoters of Plant Growth for Increased Yield in Landraces of African Cereal Crops. African Journal of Biotechnology, 3, 1-7.
http://dx.doi.org/10.5897/AJB2004.000-2002 - 58. Sorokin, I.D., et al. (2008) Bacillus alkalidiazotrophicus sp. nov., a Diazotrophic, Low Salt-Tolerant Alkaliphile Isolated from Mongolian Soda Soil. International Journal of Systematic and Evolutionary Microbiology, 58, 2459-2464.
http://dx.doi.org/10.1099/ijs.0.65655-0 - 59. Botha, W.J., Jaftha, J.B., Bloem, J.F., Habig, J.H. and Law, I.J. (2004) Effect of Soil Bradyrhizobia on the Success of Soybean Inoculant Strain CB 1809. Microbiological Research, 159, 219-231.
http://dx.doi.org/10.1016/j.micres.2004.04.004 - 60. Doignon-Bourcier, F., Willems, A., Coopman, R., Laguerre, G., Gillis, M. and de Lajudie, P. (2000) Genotypic Characterization of Bradyrhizobium Strains Nodulating Small Senegalese Legumes by 16S-23S rRNA Intergenic Gene Spacers and Amplified Fragment Length Polymorphism Fingerprint Analyses. Applied and Environmental Microbiology, 66, 3987-3997.
http://dx.doi.org/10.1128/AEM.66.9.3987-3997.2000 - 61. Lupwayi, N.Z., Claytona, G.W., O’Donovanb, J.T. and Grantc, C.A. (2011) Soil Microbial Response to Nitrogen Rate and Placement and Barley Seeding Rate under No Till. Agronomy Journal, 103, 1064-1071.
http://dx.doi.org/10.2134/agronj2010.0334 - 62. Hassen, A.I., Bopape, F.L. and Trytsman, M. (2014) Nodulation Study and Characterization of Rhizobial Microsymbionts of Forage and Pasture Legumes in South Africa. World, 2, 93-100.
- 63. Van Loon, L. (2007) Plant Responses to Plant Growth-Promoting Rhizobacteria. European Journal of Plant Pathology, 119, 243-254.
http://dx.doi.org/10.1007/s10658-007-9165-1 - 64. Lugtenberg, B. and Kamilova, F. (2009) Plant-Growth-Promoting Rhizobacteria. Annual Review of Microbiology, 63, 541-556.
http://dx.doi.org/10.1146/annurev.micro.62.081307.162918 - 65. Ramamoorthy, V., Viswanathan, R., Raguchander, T., Prakasama, V. and Samiyappan, R. (2001) Induction of Systemic Resistance by Plant Growth Promoting Rhizobacteria in Crop Plants against Pests and Diseases. Crop Protection, 20, 1-11.
http://dx.doi.org/10.1016/S0261-2194(00)00056-9 - 66. Ehteshamul-Haque, S. and Ghaffar, A. (1993) Use of Rhizobia in the Control of Root Rot Diseases of Sunflower, Okra, Soybean and Mungbean. Journal of Phytopathology, 138, 157-163.
http://dx.doi.org/10.1111/j.1439-0434.1993.tb01372.x - 67. Son, H.-J., Park, G.-T., Cha, M.-S. and Heo, M.-S. (2006) Solubilization of Insoluble Inorganic Phosphates by a Novel Salt-and pH-Tolerant Pantoea agglomerans R-42 Isolated from Soybean Rhizosphere. Bioresource Technology, 97, 204-210.
http://dx.doi.org/10.1016/j.biortech.2005.02.021 - 68. Rodríguez, H. and Fraga, R. (1999) Phosphate Solubilizing Bacteria and Their Role in Plant Growth Promotion. Biotechnology Advances, 17, 319-339.
http://dx.doi.org/10.1016/S0734-9750(99)00014-2 - 69. Chen, Y., Rekha, P.D., Arun, A.B., Shen, F.T., Lai, W.-A. and Young, C.C. (2006) Phosphate Solubilizing Bacteria from Subtropical Soil and Their Tricalcium Phosphate Solubilizing Abilities. Applied Soil Ecology, 34, 33-41.
http://dx.doi.org/10.1016/j.apsoil.2005.12.002 - 70. Goldstein, A.H. (1994) Involvement of the Quinoprotein Glucose Dehydrogenase in the Solubilization of Exogenous Phosphates by Gram-Negative Bacteria. In: Torriani-Gorini, A., Yagil, E. and Silver, S., Eds., Phosphate in Microorganisms: Cellular and Molecular Biology, ASM Press, Washington DC, 197-203.
- 71. Richardson, A.E., Barea, J.-M., McNeill, A.M. and Prigent-Combaret, C. (2009) Acquisition of Phosphorus and Nitrogen in the Rhizosphere and Plant Growth Promotion by Microorganisms. Plant and Soil, 321, 305-339.
http://dx.doi.org/10.1007/s11104-009-9895-2 - 72. Halder, A., Mishra, A.K., Bhattacharyya, P. and Chakrabartty, P.K. (1990) Solubilization of Rock Phosphate by Rhizobium and Bradyrhizobium. The Journal of General and Applied Microbiology, 36, 81-92.
- 73. Halder, A. and Chakrabartty, P. (1993) Solubilization of Inorganic Phosphate by Rhizobium. Folia Microbiologica, 38, 325-330.
http://dx.doi.org/10.1007/BF02898602 - 74. Compant, S., Duffy, B., Nowak, J., Clément, C. and Barka, E.A. (2005) Use of Plant Growth-Promoting Bacteria for Biocontrol of Plant Diseases: Principles, Mechanisms of Action, and Future Prospects. Applied and Environmental Microbiology, 71, 4951-4959.
http://dx.doi.org/10.1128/AEM.71.9.4951-4959.2005 - 75. Loganathan, M., Rai, A.B., Singh, A. and Saha, S. (2014) Plant Growth Promoting Rhizobacteria in Vegetable Disease Management. In: Kharwar, R.N., Upadhyay, R.S., Dubey, N.K. and Raghuwanshi, R., Eds., Microbial Diversity and Biotechnology in Food Security, Springer, Berlin, 373-382.
- 76. Berraho, E., Lesueur, D., Diem, H.G. and Sasson, A. (1997) Iron Requirement and Siderophore Production in Rhizobium ciceri during Growth on an Iron-Deficient Medium. World Journal of Microbiology and Biotechnology, 13, 501-510.
http://dx.doi.org/10.1023/A:1018553022960 - 77. Nakamura, H., Nakamura, K. and Yodoi, J. (1997) Redox Regulation of Cellular Activation. Annual Review of Immunology, 15, 351-369.
http://dx.doi.org/10.1146/annurev.immunol.15.1.351 - 78. von Wirén, N., Khodr, H. and Hider, R.C. (2000) Hydroxylated Phytosiderophore Species Possess an Enhanced Chelate Stability and Affinity for Iron(III). Plant Physiology, 124, 1149-1158.
http://dx.doi.org/10.1104/pp.124.3.1149 - 79. Sayyed, R., Patel, P.R. and Reddy, M.S. (2013) Role of PGPR in Bioremediation of Heavy Metal Ions and Plant Growth-Promotion of Wheat and Peanut Grown in Heavy Metal Contaminated Soil. In: Recent Advances in Biofertilizers and Biofungicides (PGPR) for Sustainable Agriculture, Proceedings of 3rd Asian Conference on Plant Growth-Promoting Rhizobacteria (PGPR) and other Microbials, Manila, Philippines, 21-24 April 2013, Asian PGPR Society for Sustainable Agriculture.
- 80. Saleh, M. and Saleh, A.-G. (2006) Increased Heavy Metal Tolerance of Cowpea Plants by Dual Inoculation of an Arbuscular Mycorrhizal Fungi and Nitrogen-Fixer Rhizobium Bacterium. African Journal of Biotechnology, 5, 133-142.
- 81. Thies, J.E., Bohlool, B.B. and Singleton, P.W. (1992) Environmental Effects on Competition for Nodule Occupancy between Introduced and Indigenous Rhizobia and among Introduced Strains. Canadian Journal of Microbiology, 38, 493-500.
http://dx.doi.org/10.1139/m92-081 - 82. Weaver, R. and Frederick, L. (1974) Effect of Inoculum Rate on Competitive Nodulation of Glycine max L. Merrill. II. Field Studies. Agronomy Journal, 66, 233-236.
http://dx.doi.org/10.2134/agronj1974.00021962006600020015x - 83. Hungria, M. and Vargas, M.A. (2000) Environmental Factors Affecting N2 Fixation in Grain Legumes in the Tropics, with an Emphasis on Brazil. Field Crops Research, 65, 151-164.
http://dx.doi.org/10.1016/S0378-4290(99)00084-2 - 84. Peterson, H.L. and Loynachan, T.E. (1981) The Significance and Application of Rhizobium in Agriculture. In: Giles, K.L. and Atherly, A.G., Eds., Biology of the Rhizobiaceae, International Review of Cytology, Supplement 13, Academic Press, New York.
- 85. Ndakidemi, P.A., et al. (2014) Estimates of Rhizobia Population in Soils from Northern Tanzania Using the Most Probable Number (MPN) Counts. African Journal of Agricultural Science and Technology (AJAST), 2, 137-150.
- 86. Olsen, P., Sanda, E. and Keyser, H. (1996) The Enumeration and Identification of Rhizobial Bacteria in Legume Inoculant Quality Control Procedures. NifTAL Center, Paia, 96 p.
- 87. Woomer, P., Bennett, J. and Yost, R. (1990) Overcoming the Inflexibility of Most-Probable-Number Procedures. Agronomy Journal, 82, 349-353.
http://dx.doi.org/10.2134/agronj1990.00021962008200020035x - 88. Zahran, H., et al. (2013) Identification of Rhizobial Strains Nodulating Egyptian Grain Legumes. International Microbiology, 16, 157-163.
- 89. Berrada, H. and Fikri-Benbrahim, K. (2014) Taxonomy of the Rhizobia: Current Perspectives. British Microbiology Research Journal, 4, 616-639.
http://dx.doi.org/10.9734/BMRJ/2014/5635 - 90. Berrada, H., Nouioui, I., Iraqui Houssaini, M., El Ghachtouli, N., Gtari, M. and Fikri Benbrahim, K. (2012) Phenotypic and Genotypic Characterizations of Rhizobia Isolated from Root Nodules of Multiple Legume Species Native of Fez, Morocco. African Journal of Microbiology Research, 6, 5314-5324.
- 91. Simon, Z., Mtei, K., Gessesse, A. and Ndakidemi, P.A. (2014) Isolation and Characterization of Nitrogen Fixing Rhizobia from Cultivated and Uncultivated Soils of Northern Tanzania. American Journal of Plant Sciences, 5, 4050-4067.
http://dx.doi.org/10.4236/ajps.2014.526423 - 92. Wolde-Meskel, E., Terefework, Z., Frostegard, A. and Lindstrom, K. (2005) Genetic Diversity and Phylogeny of Rhizobia Isolated from Agroforestry Legume Species in Southern Ethiopia. International Journal of Systematic and Evolutionary Microbiology, 55, 1439-1452.
http://dx.doi.org/10.1099/ijs.0.63534-0 - 93. Valverde, A., Igual, J.M., Peix, A., Cervantes, E. and Velázquez, E. (2006) Rhizobium lusitanum sp. nov. a Bacterium That Nodulates Phaseolus vulgaris. International Journal of Systematic and Evolutionary Microbiology, 56, 2631-2637.
http://dx.doi.org/10.1099/ijs.0.64402-0 - 94. Macrae, A. (2000) The Use of 16S rDNA Methods in Soil Microbial Ecology. Brazilian Journal of Microbiology, 31, 77-82.
http://dx.doi.org/10.1590/S1517-83822000000200002 - 95. Whitman, W.B., Coleman, D.C. and Wiebe, W.J. (1998) Prokaryotes: The Unseen Majority. Proceedings of the National Academy of Sciences of the United States of America, 95, 6578-6583.
http://dx.doi.org/10.1073/pnas.95.12.6578 - 96. Yao, Z.Y., Kan, F.L., Wang, E.T., Wei, G.H. and Chen, W.X. (2002) Characterization of Rhizobia That Nodulate Legume Species of the Genus Lespedeza and Description of Bradyrhizobium yuanmingense sp. nov. International Journal of Systematic and Evolutionary Microbiology, 52, 2219-2230.
http://dx.doi.org/10.1099/00207713-52-6-2219 - 97. Peter, J., Young, W. and Haukka, K.E. (1996) Diversity and Phylogeny of Rhizobia. New Phytologist, 133, 87-94.
http://dx.doi.org/10.1111/j.1469-8137.1996.tb04344.x - 98. Patel, J.B. (2001) 16S rRNA Gene Sequencing for Bacterial Pathogen Identification in the Clinical Laboratory. Molecular Diagnosis, 6, 313-321.
http://dx.doi.org/10.2165/00066982-200106040-00012 - 99. Germano, M.G., Menna, P., Mostasso, F.L. and Hungria, M. (2006) RFLP Analysis of the rRNA Operon of a Brazilian Collection of Bradyrhizobial Strains from 33 Legume Species. International Journal of Systematic and Evolutionary Microbiology, 56, 217-229.
http://dx.doi.org/10.1099/ijs.0.02917-0 - 100. Vinuesa, P., Silva, C., Lorite, M.J., Izaguirre-Mayoral, M.L., Bedmar, E.J. and Martínez-Romero, E. (2005) Molecular Systematics of Rhizobia Based on Maximum Likelihood and Bayesian Phylogenies Inferred from rrs, atpD, recA and nifH Sequences, and Their Use in the Classification of Sesbania Microsymbionts from Venezuelan Wetlands. Systematic and Applied Microbiology, 28, 702-716.
http://dx.doi.org/10.1016/j.syapm.2005.05.007 - 101. Reller, L.B., Weinstein, M.P. and Petti, C.A. (2007) Detection and Identification of Microorganisms by Gene Amplification and Sequencing. Clinical Infectious Diseases, 44, 1108-1114.
http://dx.doi.org/10.1086/512818 - 102. Haukka, K., Lindstrom, K., Peter, J. and Young, W. (1996) Diversity of Partial 16S rRNA Sequences among and within Strains of African Rhizobia Isolated from Acacia and Prosopis. Systematic and Applied Microbiology, 19, 352-359.
http://dx.doi.org/10.1016/S0723-2020(96)80062-2 - 103. Stackebrandt, E. and Goebel, B. (1994) Taxonomic Note: A Place for DNA-DNA Reassociation and 16S rRNA Sequence Analysis in the Present Species Definition in Bacteriology. International Journal of Systematic Bacteriology, 44, 846-849.
http://dx.doi.org/10.1099/00207713-44-4-846 - 104. Rosselló-Mora, R. (2006) DNA-DNA Reassociation Methods applied to Microbial Taxonomy and Their Critical Evaluation. In: Stackebrandt, E., Ed., Molecular Identification, Systematics, and Population Structure of Prokaryotes, Springer, Berlin, 23-50.
http://dx.doi.org/10.1007/978-3-540-31292-5_2 - 105. Martens, M., Dawyndt, P., Coopman, R., Gillis, M., De Vos, P. and Willems, A. (2008) Advantages of Multilocus Sequence Analysis for Taxonomic Studies: A Case Study Using 10 Housekeeping Genes in the Genus Ensifer (Including Former Sinorhizobium). International Journal of Systematic and Evolutionary Microbiology, 58, 200-214.
http://dx.doi.org/10.1099/ijs.0.65392-0


