Open Journal of Applied Sciences
Vol.05 No.07(2015), Article ID:58295,16 pages
10.4236/ojapps.2015.57038
Biosorption of Ni2+ and Cd2+ from Aqueous Solutions Using NaOH-Treated Biomass of Eupenicillium ludwigii: Equilibrium and Mechanistic Studies
Abdulkawi Ali Al-Fakih
Faculty of Science, Department of Medical Microbiology, Ibb University, Ibb, Yemen
Email: Fakeeh16@yahoo.com
Copyright © 2015 by author and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 30 June 2015; accepted 21 July 2015; published 24 July 2015
ABSTRACT
The removal of Ni2+ and Cd2+ ions by Eupenicillium ludwigii biomass was studied in a batch system. The optimum pH for the biosorption was 6 for Ni2+ and 5 for Cd2+. Temperature changes in the range from 15˚C to 40˚C affected the biosorption capacity, and the nature of the reaction was found to be endothermic for both metal ions. HCl was the best desorbing agent for the desorption of both metals. Chemical modifications of the biomass demonstrated that carboxyl and amine groups played an important role in Ni2+ and Cd2+ biosorption. Ion exchange mechanism was also suggested in the biosorption process.
Keywords:
Nickel, Cadmium, Heavy Metals, Biosorption, Eupenicillium ludwigii

1. Introduction
Heavy metals pose a significant threat to the environment and public health because of their toxicity, accumulation in the food chain, and persistence in nature [1] . Nickel (Ni) and cadmium (Cd) are of major concern because of their higher usage in developing countries and potential pollution impact. These metals are released into the environment by many processes such as electroplating, metal finishing, mining, leather tanning, wood preservation, pulp processing, steel manufacturing, and so on [1] [2] . The exposure to Ni and Cd leads to adverse health effects to human, so their removal from industrial effluents is an extremely significant step in the protection of the environment and human health. Conventional methods for removing heavy metals from polluted effluents are ineffective or extremely expensive especially when the metals in solution are in the range of 1 - 100 mg∙L−1, and often restricted because of secondary problems with metal-bearing sludge, which are extremely difficult to be disposed [1] [3] [4] . The ability of inactive, dead microbial biomasses to remove metal ions has received considerable attention for the development of an efficient, clean, and cheap technology for wastewater treatment at metal concentrations as low as 1 mg∙L−1 [5] . This technology is called biosorption, which is an innovative, eco-friendly, and low cost effective method for the removal of toxic heavy metals from wastewaters. Among biological materials, fungal biomasses are widely used in biosorption technology, as they are able to remove heavy metals from aqueous solutions in substantial quantities, abundantly available from industrial fermentations, and they show biosorption efficiency and affinity for more metal ions [6] . The potential of fungi for biosorption of heavy metals from aqueous solutions is well documented [7] -[9] . In general, the fungal cell wall has many potential binding sites, including amino, carboxyl, phosphate, sulfhydryl, and other functional groups [10] . The aim of the present study is to determine the potential of the dead biomass of Eupenicillium ludwigii (Eup. ludwigii) to remove Ni2+ and Cd2+ ions in a batch mode and the function of many environmental factors such as pH, time contact, initial metal ions concentration, temperature, biomass dose, and shaking rate. The effect of pretreatment on biosorption capacity of the biomass is also investigated.
2. Materials and Methods
2.1. Fungus and Growth Conditions
Fungal isolate (Eup. ludwigii) was isolated from a soil located in Shebein El-koom, Menoufia Governorate, Egypt, and identified by Regional Center for Mycology and Biotechnology (Al-Azhar University, Cairo, Egypt). The medium used for the growth of fungus was Potato Dextrose (PD) medium (Oxoid, England). For production of the biomass, the fungus was cultivated on a rotary shaker (Jeio Tech SI-900 R, Korea) at 125 rpm and 28˚C for 3 days in 500 ml Erlenmeyer flask containing 250 ml of PD medium.
2.2. Preparation of Fungal Biomass for Biosorption Studies
After 3 days of growth, the fungal biomass was harvested using a plastic sieve, followed by washing with generous amounts of double distilled water (ddH2O) to remove residual growth medium, and drained to remove excess water by gentle pressing through Whatman filter paper No. 1. This biomass will be referred to as untreated biomass.
2.3. NaOH Treatment
According to Gharieb et al. [9] with some modifications, NaOH treatment was performed by boiling viable fungal biomass in 0.5 N NaOH (1:10, w/v) for 15 min. The resulting biomass was washed extensively with ddH2O until the pH of the washing solution was close to neutral range (6.8 - 7.2). This biomass will be referred to as treated biomass. Both untreated and NaOH-treated biomasses were dried in an oven for 24 h at 60˚C. Then, dried biomasses were powdered in a mortar with a pestle, sieved through a sieve with 125 μm openings, and stored in a desiccator for future use.
2.4. Preparation of Metal Solution
All chemicals used in the present study were of analytical grade. Stock metal solutions of Ni2+ and Cd2+ (1000 mg∙L−1) were prepared separately by dissolving NiSO4.6H2O and 3CdSO4∙8H2O, respectively in ddH2O. For experiments with various metal concentrations, the stock solutions were diluted further with ddH2O. The pH value of each test metal solution was adjusted to desirable value with 0.1 M HCl or 0.1 M NaOH.
2.5. Biosorption Experiments
The biosorption of Ni2+ and Cd2+ from aqueous solutions were carried out in batch systems. 0.04 g of dried fungal biomass (untreated or NaOH-treated) was added to 100 ml Erlenmeyer flasks containing 20 ml metal solution with concentration of 50 mg∙L−1, and agitated in an orbital shaker (Jeio Tech SI-900 R, Korea) for 120 min at 125 rpm and 25˚C. The effect of initial pH (1 - 8), time contact (15 - 120 min), initial metal ion concentration (50 - 400 mg∙L−1), temperature (15 - 40˚C), biomass dose (0.5 - 5 g∙L−1), and shaking rate (0 - 200 rpm) on the metals biosorption were studied. After the end of each experiment, the mixtures were centrifuged (for 5 min at 10000 rpm) and metal ion concentrations in the supernatant were determined. All biosorption experiments were done in triplicate and the mean values were reported. The amount of adsorbed metal ions is estimated as the amount of metal (mg) per unit of biomass dry weight (g) using the following equation:
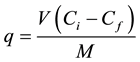 (1)
(1)
where V is the volume of metal solution (L), Ci is the initial metal concentration (mg∙L−1), Cf is the final/residual concentration (mg∙L−1) and M is the amount of biomass (g). The percent biosorption of metal ion was calculated as follows:
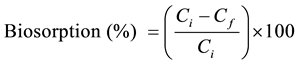 (2)
(2)
2.6. Desorption and Reuse of the Biosorbent
Following the metal biosorption experiments, metal-loaded biomasses were separated by centrifugation, washed, and contacted with 20 ml of various elutants (at solid/liquid ratio 0.1 g 20 ml−1) for 20 min on an orbital rotary shaker set at 125 rpm at 25˚C ± 1˚C. The elutants used were 0.1 M HCl, 0.1 M CaCl2, 0.1 M Na2CO3, 0.1 M EDTA, and ddH2O. The biomass was separated from elutants by filtering the reaction mixture through 0.45 μm filter paper and the filtrate was analyzed for metal concentration to investigate the desorption efficiency. The desorption efficiency was calculated from the amount of metal ions adsorbed on the biomass and the final metal ion concentration in the biosorption medium using the following equation:
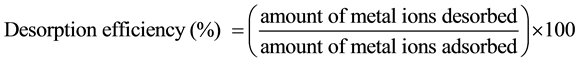 (3)
(3)
For the regeneration of biomass eluted using the most efficient elutant, a generous amount of ddH2O was used to rinse the regenerated biomass till pH in the solution reached the range of 6.8 to 7.2. Then, biomasses were dried at 60˚C for 48 h, and then re-suspended in metal containing solutions for the next biosorption cycle and this biosorption-desorption cycle was repeated five times.
2.7. Study of Mechanisms Involved in Biosorption
2.7.1. Chemical Modification of the Biomass
Chemical modifications of the fungal biomass were performed to determine which functional group/groups on the fungal biomass may be involved in binding of Ni2+ and Cd2+ ions. These modifications include:
i. Esterification of the carboxylic groups
According to Drake et al. [11] , 2 g of dried NaOH-treated biomass was added to 130 ml of methanol with 1.2 ml of HCl added to the suspension. The mixture was shaken on an orbital rotary shaker for 6 h at 125 rpm. The treatment of biomass with methanol results in esterification of carboxylic acids present on the cell wall of biomass and the reaction occurs as follows:

where R denotes the organic network of biomass molecules. Because of the esterification, metal binding capacity of carboxyl groups will be reduced. The biomass residue obtained was referred as chemical modification 1 (CM1).
ii. Methylation of amino groups
According to Loudon, [12] , 2 g of dried NaOH-treated biomass was contacted with 40 ml of formaldehyde (HCHO) and 80 ml of formic acid (HCOOH). The mixture was also shaken at 125 rpm for 6 h and the resulting reaction takes place as follows:
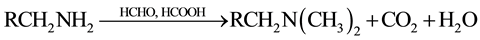
Because of the methylation of amino groups, their participation in metal biosorption is expected to be inhibited, resulting in the reduction in metal biosorption capacity on residual biomass. The obtained biomass residue was referred to as chemical modification 2 (CM2).
iii. Esterification of the phosphate groups
According to Tobin et al. [13] , 2 g of dried NaOH-treated biomass was heated under reflux and stirring conditions with 80 ml of triethylphosphite and 60 ml of nitromethane for 6 h. The obtained biomass residue was referred to as chemical modification 3 (CM3).
iv. Extraction of Lipids
According to Tobin et al. [13] , 2 g of dried NaOH-treated biomass was heated separately with 150 ml of acetone and benzene under reflux and stirring conditions. The treatment will extract the lipid fraction from the biomass. The obtained biomass residues were referred to as chemical modifications 4 (CM4) and 5 (CM5).
After chemical modification, all biomass samples were washed with ddH2O, dried at 60˚C for 48 h, and stored until use.
2.7.2. Ion Exchange Study
This experiment was carried out to evaluate the involvement of ion exchange process in Ni2+ and Cd2+ biosorption. In this experiment, 0.04 g of NaOH-treated biomass was adequately added into 20 ml of metal solutions containing 50 mg∙L−1 with desired pH. After being shaken at 125 rpm and 25˚C ± 1˚C for 2 h, the reaction mixtures were centrifuged and the supernatants were measured for metal concentrations. Release of Na+, K+, Ca2+, and Mg2+ from biomass as a result of biosorption of metal ions was studied. Appropriate control samples without metal ions added into ddH2O were set up to compare the release of monovalent and divalent ions from pretreated biomass in ddH2O. All experiments were conducted three times and the mean values were reported.
2.8. Analysis of Metal Ions
The concentrations of Ni2+, Cd2+, Ca2+, and Mg2+ ions in the supernatant of biosorption medium were determined by using an atomic absorption spectrophotometer (AAS) (Unicam 929, Philips Company, UK) with an air-acetylene flame. The instrument calibration was checked periodically by using standard metal solutions. The concentrations of Na+ and K+ ions in each sample filtrate from the experiments for ion exchange study were determined by using a flame photometer (JENWAY, UK) with reference to appropriate standard solutions.
2.9. Biosorption Isotherms
Several mathematical models have been developed to quantitatively express the relationship between the extent of adsorption and the residual solute concentration. The most widely used models are the Langmuir and Freundlich adsorption isotherm models [14] . To determine the adsorptive capacity of Eup. ludwigii for Ni2+ and Cd2+ ions, the initial metal concentration varied from 50 - 400 mg∙L−1; while the biosorbent was constant at 0.04 g 20 ml−1. A Langmuir isotherm was then obtained by plotting the values of biosorption capacity (q) versus the residual metal concentration (Cf) in solution. The classical Langmuir equation is given as follows [15] :
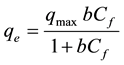 (4)
(4)
where,
qe = metal adsorbed on the biosorbent (mg∙g−1) at equilibrium;
qmax = maximum possible amount of metal adsorbed per unit weight of biosorbent;
Cf = residual concentration of metal (mg∙L−1) in the solution;
b = equilibrium constant related to the affinity of the binding sites for the metals.
Equation (4) can be linearized as follows:
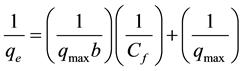 (5)
(5)
When 1/qe is plotted against 1/Cf, a straight line with slope 1/qmax b is obtained and the intercept is corresponding to 1/qmax. Also, qmax and b were determined. The classical Freundlich equation is given as follows [15] :
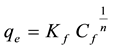 (6)
(6)
where,
qe = metal adsorbed on the biosorbent (mg∙g−1) at equilibrium;
Cf = residual concentration of metal (mg∙L−1) in the solution;
Kf = an empirical constant that provides an indication of the intensity of adsorption;
n = Freundlich adsorption constant.
This equation can be linearized by taking natural logarithm of both sides of the equation, which can be given as follows:
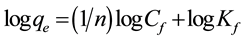 (7)
(7)
when the values of log Cf are plotted against the values of log qe, the adsorption constants (Kf and n) were obtained.
2.10. Statistical Analysis
The values presented in the study were means of three replicates and expressed as means ± standard error (SE). Statistical analysis was performed using Statistical Package for the Social Sciences (SPSS) 9.05 for Windows where it was possible to evaluate whether the effect and the interaction among the investigated factors were significant with respect to the standard error.
3. Results and Discussion
3.1. NaOH Treatment
In order to generate anionic sites without significant modification of the fungal cell wall structure, the biomass of Eup. ludwigii was treated with NaOH. Figure 1 shows the metal biosorption values obtained by untreated and NaOH-treated biomasses. As seen in Figure 1, the treatment of Eup. ludwigii with NaOH significantly enhanced the biosorption capacity from 5.24 ± 0.19 to 12.11 ± 0.28 mg∙g−1 for Ni2+ and from 13.34 ± 0.31 to 22.15 ± 0.39 mg∙g−1 for Cd2+. An enhanced biosorption capacity as a result of NaOH treatment was also observed by many researchers [9] [16] -[20] . Opposite result was reported by Li and Yuan [21] . They found that pretreatment with NaOH decreased the cadmium biosorption by Rhodotorula sp. Y11. Enhancement of biosorption capacity after NaOH treatment may be due to the exposing of active metal-binding sites embedded in the cell wall and causing availability of more anionic sites [17] . Also, alkaline treatment can activate the hydroxyl groups in fungal mycelium and therefore the epichlorohydrin will be easily introduced [22] .
Figure 1. Ni2+ and Cd2+ biosorption (mg∙g−1) by NaOH-treated and untreated biomasses of Eup. ludwigii. The data are the mean values of 3 replicates, and the bars indicate the standard error of the mean.
3.2. Effect of pH on Metal Biosorption
pH of the aqueous solution plays a vital role in the biosorption process, because it affects the chemistry of metals, and the surface charge and ionization of the functional groups on the fungal cell wall during reaction [23] . The effect of pH solution on Ni2+ and Cd2+ ions biosorption was carried out in the range of pH 1 - 8 at 50 mg∙L−1 of metal ions concentration. From the results illustrated in Figure 2, it was found that the biosorption capacities of Ni2+ and Cd2+ ions were lower at low pH and started to increase as the pH solution increased. The optimum pH values for Ni2+ and Cd2+ biosorption were 6 and 5, at which the Ni2+ and Cd2+ biosorption values were 12.50 ± 0.68 and 21.97 ± 0.64 mg∙g−1, respectively. This is in agreement with the character of metal cations biosorption. Successful biosorption of base metal cations usually takes place in the range of pH 3 - 7 and is extremely pH dependent [24] . An increase or decrease in the pH from these optimum pH values resulted in a reduction in the biosorption of these metal ions. At low pH values, protons in solution compete effectively with metals in binding to functional groups. pH effect may be further explained in relation to the competition effect between the hydronium ions (H3O+) and metal ions [25] . As initial pH increases, the active sites are being deprotonated and strengthened the charge attraction, thus leading to significant increase in Ni2+ and Cd2+ biosorption. The decrease in biosorption of Ni2+ and Cd2+ ions above pH 6 is due to precipitation of both metals as insoluble hydroxides or hydrated oxides [22] [26] [27] .
3.3. Effect of Contact Time
The effect of contact time (15 to 120 min) on the equilibrium uptake of Ni2+ or Cd2+ ions onto Eup. ludwigii for an initial metal ion concentration of 50 mg∙L−1 is shown in Figure 3. The biosorption capacity increased with increasing contact time and a large amount of metal ions was removed in the first 30 min. Equilibrium was reached in 45 and 75 min for Ni2+ and Cd2+, where the biosorption values reached 12.04 ± 0.71 and 22.23 ± 0.58 mg∙g−1, respectively. After these equilibrium periods, either in Ni2+ or Cd2+ biosorption, the amount of adsorbed metal ions was not significantly changed with contact time. This rapid initial uptake was similar to the previous reports on the biosorption of these metals by different biosorbents. Akar and Tunali [28] observed that Cd2+ biosorption by Botrytis cinerea was fast and equilibrium was reached in 60 min. A contact time of 120 min was required to reach equilibrium in the biosorption of Cd2+ by Rhizopus cohnii [29] . Pahlavanzadeh et al. [1] noted that approximately 60% of Ni2+ ions were removed by the brown algae (Cystoseria indica, Nizmuddinia zanardini, Sargassum glaucescens, and Padina australis) in the first 20 min of contact, and equilibrium was reached
Figure 2. Ni2+ and Cd2+ biosorption (mg∙g−1) by NaOH-treated Eup. ludwigii at different pH values.
in a contact time of 120 min. This observed rapid biosorption of metals is among desirable parameters for successful deployment of the biosorbents for practical application [30] .
3.4. Effect of Initial Metal Ion Concentration
The initial metal ion concentration remarkably influenced the equilibrium metal uptake and biosorption yield. The effect of initial metal (Ni2+ and Cd2+) ion concentration was investigated in the range of 50 - 400 mg∙L−1 under the determined optimum pH values and contact time. From the results presented in Figure 4, it was noted
Figure 3. Ni2+ and Cd2+ biosorption (mg∙g−1) by NaOH-treated Eup. ludwigii at different time intervals (min).
Figure 4. Ni2+ and Cd2+ biosorption (mg∙g−1) by NaOH-treated Eup. ludwigii at different metal concentrations (mg∙L−1).
that initial concentration increased the sorption of Ni2+ and Cd2+ ions, and then reached a saturation values at about 200 mg∙L−1 for Ni2+ and 300 mg∙L−1 for Cd2+. At these concentrations, the biosorbed Ni2+ and Cd2+ reached 26.23 ± 0.61 and 53.37 ± 0.84 mg∙g−1, respectively. Then the values did not significantly change with the initial metal ion concentration. This increase in uptake capacity of both metals with increasing initial metals concentration is due to higher availability of metal ions for the sorption. Moreover, higher initial concentration provides increased driving force to overcome mass transfer resistance between the biosorbent and biosorption medium [31] .
3.5. Effect of Temperature
The effects of temperature on biosorption of Ni2+ and Cd2+ onto Eup. ludwigii were carried out by varying a series of temperature from 15˚C to 40˚C. From the results presented in Figure 5, it was shown that the uptake amount of both metal ions increased with increasing temperature. So, the biosorption capacity increased from 3.67 ± 0.23 to 12.89 ± 0.21 mg∙g−1 for Ni2+ and from 9.44 ± 0.27 to 23.72 ± 0.45 mg∙g−1 for Cd2+ as the temperature increased from 15 to 40˚C. Similar results were reported on biosorption of Cd2+ onto Pycnoporus sanguineus [32] , Ni2+ onto some brown algae [1] , and Pb2+ onto Candida albicans [33] . The increase in the biosorption capacity with the increase in temperature indicates that the biosorption of Ni2+ and Cd2+ onto Eup. ludwigii was controlled by an endothermic process. This effect may be due to the fact that at higher temperatures an increase in active sites occurs due to bond rupture [1] . Opposite result was reported by Anayurt et al. [34] in the study on the biosorption of Pb2+ and Cd2+ by the fungus Lactarius scrobiculatus. They found that biosorption decreased from 98% to 90% for Pb2+ and from 95% to 88% for Cd2+ as temperature was increased from 20˚C to 50˚C.
3.6. Effect of Biomass Dose
To get the optimal biomass dose in Ni2+ and Cd2+ biosorption, different amounts (0.5 - 5 g∙L−1) of fungal biomass were used. As can be seen from Figure 6, with the dose of biomass increasing, the metal uptake of Ni2+ and Cd2+ ions per unit mass of biomass (mg∙g−1) was decreased. The biosorbed metal decreased from 38.22 ± 0.85 to 6.87 ± 0.21 mg∙g−1 for Ni2+ and from 45.62 ± 0.78 to 9.83 ± 0.35 mg∙g−1 for Cd2+ due to the increase in the fungal biomass from 0.5 to 5 g∙L−1, respectively. This is expected because as the dose of biomass increased, there was increase in the available exchangeable sites for Ni2+ and Cd2+ ions. The maximum biosorption efficiencies reached 68.66% and 98.32% for Ni2+ and Cd2+, respectively, at biomass weight 5 g∙L−1. Reduction in biomass dose in the biosorption medium at a given metal concentration enhanced the metal/biosorbent ratio and thus increased the metal uptake per unit weight of biosorbent as long as the later is not saturated [34] [35] .
Figure 5. Ni2+ and Cd2+ biosorption (mg∙g−1) by NaOH-treated Eup. ludwigii at different temperatures (˚C).
Figure 6. Ni2+ and Cd2+ biosorption (mg∙g−1) by NaOH-treated Eup. ludwigii at different biomass doses (g∙L−1).
3.7. Effect of Shaking Rate
In order to determine the optimal shaking rate, the biosorption of Ni2+ and Cd2+ ions by Eup. ludwigii was evaluated by varying the shaking rate of the biosorption media from 0 (without shaking) to 200. Figure 7 illustrates the effect of shaking rate (rpm) versus the amount of metal biosorbed (mg∙g−1) of Ni2+ and Cd2+ ions. Control units at 0 rpm (no agitation) exhibited very low Ni2+ and Cd2+ uptake. At this shaking rate, the biosorbed Ni2+ and Cd2+ reached 2.45 ± 0.12 and 7.97 ± 0.24 mg∙g−1, respectively. As shaking rate increased, the biosorption of Ni2+ and Cd2+ ions increased, and optimal values of biosorption were obtained for shaking rate of 150 and 100 rpm for Ni2+ and Cd2+ ions, respectively, and the biosorbed Ni2+ and Cd2+ were 13.34 ± 0.31 and 23.86 ± 0.64 mg∙g−1, at the same order. Nearly, similar results were observed by Tun-Guo et al. [36] in the biosorption of Cd2+ by Aspergillus niger. They found that the optimal shaking rate was 120 rpm. The optimal values of Pb2+ and Co2+ biosorption capacity by Rhizopus oryzae were obtained at shaking rate of 150 rpm [9] . Opposite result was reported by Selatnia et al. [37] for biosorption of Ni2+ by a bacterial dead Streptomyces rimosus biomass. They found that the optimum shaking rate for biosorption of this metal is 250 rpm. The lower metal uptake of both metals at higher shaking rates is attributed to non-homogeneity of the biosorption mixtures caused by vortex phenomenon [38] , making the biosorption of Ni2+ and Cd2+ ions difficult.
3.8. Desorption and Reuse of the Biosorbent
The reusability and metal recovery efficiency of the biosorbent is likely to be a key factor in accessing the potential of the biosorbent for commercial application. As they are common elutants used by many researchers, 0.1 M HCl, 0.1 M CaCl2, Na2CO3, EDTA, and ddH2O were used to identify a suitable elutant agent. Table 1 shows the percentage of Ni2+ and Cd2+ ions released from fungal biomass after treatment with different elutants. From the results, it is evident that the desorption efficiencies of Ni2+ and Cd2+ ions were more than 96% after eluting with 0.1 M HCl, and so that HCl is the most efficient elutant to be used in the biosorption-desorption cyclic studies. HCl has been found to be an effective elutant for desorption of Ni2+ from Penicillium chrysogenum [22] and baker’s yeast [39] , and Pb2+ and Cd2+ from macro-fungus Lactarius scrobiculatus [34] . On the other hand, elution of Ni2+ and Cd2+ ions by ddH2O exhibited negligible desorption capability and the desorption efficiency values do not exceed 3% for both metals, indicating a strong affinity of the fungal biomass towards Ni2+ and Cd2+ ions. After desorption using the most efficient elutant (0.1 M HCl), the biomass was washed with ddH2O, and reused for another cycle. From the results illustrated in Figure 8, it is evident that there was a gradual de-
Figure 7. Ni2+ and Cd2+ biosorption (mg∙g−1) by NaOH-treated Eup. ludwigii at different Shaking rates (rpm).
Figure 8. Ni2+ and Cd2+ biosorption (mg∙g−1) by Eup. ludwigii after desorption with 0.1 M HCl solution for 5 cycles.
crease of Ni2+ and Cd2+ biosorption on Eup. ludwigii biomass with an increase the number of desorption cycles. After a sequence of five cycles, it was observed that the biosorption capacity of Eup. ludwigii biomass has been reduced to 75.2% and 80.8% for Ni2+ and Cd2+, respectively. The results indicate that Eup. ludwigii has good potential to adsorb these metal ions repeatedly from aqueous solution.
3.9. Biosorption Isotherms
Biosorption isotherm provides a relationship between the concentration of metal in solution and the amount of metal on biosorbent when both the phases are at equilibrium. Modeling of Ni2+ and Cd2+ biosorption on the fungal biomass was realized by applying Langmuir and Freundlich adsorption isotherms. The linearized forms of the Langmuir isotherm model (Equation (5)) and Freundlich isotherm model (Equation (7)) were used to analyze and fit the data to these models. The Langmuir constants (qmax and b) with correlation coefficients (R2) were calculated from the plots in Figure 9 for biosorption of Ni2+ and Cd2+on the fungal biomass and the results are presented in Table 2. Also, Freundlich constants (Kf and n) with correlation coefficients (R2) were calculated from the plots in Figure 10, and presented in Table 2. The fit of experimental data to these models was evaluated by the correlation coefficients (R2). From the final results and based on the values of correlation coefficients (R2), Langmuir and Freundlich models best described the experimental data for biosorption of Ni2+ and Cd2+ at different temperatures, but biosorption of Cd2+ is more described by Freundlich model. In view of the Langmuir constant (qmax) values, the qmax values of Ni2+ and Cd2+ are close to the experimental qmax values at 20 and 30˚C, respectively. The favorable biosorption is indicating by higher than 1 value of Freundlich sorption constant n for fungal biomass. The values of n obtained greater than one for Ni2+ and Cd2+ indicated that physical and multilayer adsorption takes place for both metal ions. The small (Kf) values for Ni2+ indicate a lower extent biosorption, while more biosorption was observed for Cd2+ ions because of its larger (Kf) values. Generally, the higher values of Freundlich constants (Kf and n) and the lower value of Langmuir constant (b) indicating the higher affinity of the biomass [40] [41] . Similar results were also reported for biosorption of Ni2+ by Trichoderma viride [42] , Cd2+ by Rhizopus cohnii [29] , and Pb2+, Cu2+, and Cd2+ by Phanerochaete chrysosporium [43] .
Table 1. Desorption efficiencies (%) of Ni2+ and Cd2+ ions from NaOH-treated Eup. ludwigii biomass using various elutants.
Table 2. Isotherm parameters of two models for Ni2+ and Cd2+ biosorption by NaOH-treated Eup. ludwigii at different temperatures (˚C).


Figure 9. Application of Langmuir isotherm model for (a) Ni2+ and (b) Cd2+ biosorption by NaOH-treated Eup. ludwigii at different temperatures (˚C).
3.10. Mechanism Studies
3.10.1. Biosorption by Chemically Modified Biomass
The results of metal biosorption studies before and after chemical modification of functional groups of NaOH-treated Eup. ludwigii are presented in Table 3. From the results, it can be seen that when carboxyl groups were esterified (CM1), biosorption of Ni2+ and Cd2+ were significantly decreased from 12.35 ± 0.31 to 1.41 ± 0.05 mg∙g−1 and from 21.45 ± 0.29 to 3.07 ± 0.09 mg∙g−1, respectively in comparison with control (NaOH- treated biomass). The reductions were 88.6% and 85.7% for Ni2+ and Cd2+, respectively. Decreasing of biosorp-
Table 3. Metal biosorption (mg∙g−1) and biosorption efficiency (%) by NaOH-pretreated biomass (control) and chemically modified biomass residue of Eup. ludwigii.
tion was also observed for both metals by Eup. ludwigii biomass with methylated amino groups (CM2), and the biosorption values were 9.19 ± 0.25 and 17.33 ± 0.33 mg∙g−1 for Ni2+ and Cd2+, respectively. The reductions were 25.6% and 19.3% at the same order. These findings suggest that carboxyl and amine groups are important in Ni2+ and Cd2+ biosorption on Eup. ludwigii biomass. Also, these results may be sufficient to indicate the important participation of carboxyl and amine groups in the biosorption of these metal ions. Kapoor and Viraraghavan [44] have reported similar reduction in biosorption of Cd2+, Cu2+, and Pb2+ ions by A. niger subjected to esterification of its carboxyl groups. In the biosorption of Cu2+, Ni2+, Zn2+, and Cr3+ by Penicillium chrysogenum, Tan and Cheng [45] found that the main chelating sites in the mycelium are amine groups of chitosan. The interacttion between amine group and Ni2+ or Cd2+ ions is complexation. This study showed that the electrostatic attraction and complexation seem to be the most important mechanism of biosorption of Ni2+ and Cd2+ ions. On the other hand, esterification of phosphate groups (CM3) and extraction of lipids using acetone (CM4) and benzene (CM5) had no effect on both metals biosorption. Kapoor and Viraraghavan [44] observed slight decreases in biosorption of Cd2+, Cu2+, and Pb2+ ions when lipids were extracted from A. niger. They attributed the decrease to either lipid extraction or the probable structural changes that may have resulted due to the harsh conditions of the extraction process.
3.10.2. Ion Exchange Study
The contribution of ion-exchange mechanism to the biosorption of Ni2+ and Cd2+ onto Eup. ludwigii biomass was investigated by determination of the light metals Na+, K+, Ca2+, and Mg2+ in the filtrates of biosorption media. The amounts of these cations were compared with those in a control sample, which consists of Eup. ludwigii biomass and ddH2O. From the results presented in Table 4, biosorption of Ni2+ and Cd2+ ions by NaOH-treated Eup. ludwigii biomass showed that K+, Ca2+, and Mg2+ ions were released into the biosorption medium. This indicates that Ni2+ and Cd2+ possibly have been exchanged with these ions on the cell walls of Eup. ludwigii, thereby suggesting an ion exchange mechanism as one of the mechanisms of metal biosorption for Eup. ludwigii. In contrast, Na+ ion was not detected in the reaction solution. The biosorption of Sr2+, Mn2+, Zn2+, Cd2+, Cu2+, and Ti+ by S. cerevisiae, also resulted in releasing of Ca2+, Mg2+, and H+ [46] . As observed by Allaboun and Abu Al- Rub [47] , Ni2+ biosorption by palm tree leaves resulted in the release of Ca2+, Mg2+, and K+ ions from the biosorbent.
4. Conclusion
In the present study, the biosorption capacity of Eup. ludwigii for Ni2+ and Cd2+ increases after the biomass is

Figure 10. Application of Freundlich isotherm model for (a) Ni2+ and (b) Cd2+ biosorption by NaOH-treated Eup. ludwigii at different temperatures (˚C).
treated with NaOH. The biosorption process has been shown to be affected from experimental conditions, such as pH, contact time, etc. The experimental data for both metals will be described appropriately by Langmuir and Freundlich models. The mechanism of biosorption can be a combination of ion-exchange and complexation with the functional groups of the fungal biomass. The study indicates that Eup. ludwigii may be used as an inexpensive, effective, and ecofriendly biomaterial for the removal of both metals from aqueous solutions.
Table 4. Amounts of Na+, K+, Ca2+, and Mg2+ ions (mg∙L−1) released upon Ni2+ and Cd2+ biosorption by NaOH-treated Eup. ludwigii biomass.
Acknowledgements
The author would like to thank Ibb University for the financial support. The author is also grateful to the precious comments and careful corrections made by anonymous reviewers.
Cite this paper
Abdulkawi AliAl-Fakih, (2015) Biosorption of Ni2+ and Cd2+ from Aqueous Solutions Using NaOH-Treated Biomass of Eupenicillium ludwigii: Equilibrium and Mechanistic Studies. Open Journal of Applied Sciences,05,376-392. doi: 10.4236/ojapps.2015.57038
References
- 1. Pahlavanzadeh, H., Keshtkar, A.R., Safdari, J. and Abadi, Z. (2010) Biosorption of Nickel(II) from Aqueous Solution by Brown Algae: Equilibrium, Dynamic and Thermodynamic Studies. Journal of Hazardous Materials, 175, 304-310.
http://dx.doi.org/10.1016/j.jhazmat.2009.10.004 - 2. Miretzky, P., Munoz, C. and Carrillo-Chavez, A. (2010) Cd(II) Removal from Aqueous Solution by Eleocharis acicularis Biomass, Equilibrium and Kinetic Studies. Bioresource Technology, 101, 2637-2642. http://dx.doi.org/10.1016/j.biortech.2009.10.067
- 3. Akar, T., Kaynak, Z., Ulusoy, S., Yuvaci, D., Ozsari, G. and Akar, S.T. (2009) Enhanced Biosorption of Nickel(II) Ions by Silica-Gel-Immobilized Waste Biomass: Biosorption Characteristics in Batch and Dynamic Flow Mode. Journal of Hazardous Materials, 163, 1134-1141. http://dx.doi.org/10.1016/j.jhazmat.2008.07.084
- 4. Volesky, B. (2001) Detoxification of Metal-Bearing Effluents: Biosorption for the Next Century. Hydrometallurgy, 59, 203-216.
http://dx.doi.org/10.1016/S0304-386X(00)00160-2 - 5. Saeed, A., Iqbal, M. and Akhtar, M.W. (2005) Removal and Recovery of Lead(II) from Single and Multimetal (Cd, Cu, Ni, Zn) Solutions by Crop Milling Waste (Black Gram Husk). Journal of Hazardous Materials, 117, 65-73. http://dx.doi.org/10.1016/j.jhazmat.2004.09.008
- 6. Singh, H. (2006) Mycoremediation: Fungal Bioremediation. John Wiley & Sons, Hoboken. http://dx.doi.org/10.1002/0470050594
- 7. Wang, J. and Chen, C. (2006) Biosorption of Heavy Metals by Saccharomyces cerevisiae: a Review. Biotechnology Advances, 24, 427-451.
http://dx.doi.org/10.1016/j.biotechadv.2006.03.001 - 8. Sari, A. and Tuzen, M. (2009) Kinetic and Equilibrium Studies of Biosorption of Pb(II) and Cd(II) from Aqueous Solution by Macrofungus (Amanita rubescens) Biomass. Journal of Hazardous Materials, 164, 1004-1011.
http://dx.doi.org/10.1016/j.jhazmat.2008.09.002 - 9. Gharieb, M.M., Al-Fakih, A.A. and Ali, M.I. (2014) Biosorption of Pb(II) and Co(II) Ions from Aqueous Solutions Using Pretreated Rhizopus oryzae (Bread Mold). Arabian Journal for Science and Engineering, 39, 2435-2446. http://dx.doi.org/10.1007/s13369-013-0784-x
- 10. Lamelas, C., Avaltroni, F., Benedetti, M., Wilkinson, K.J. and Slaveykova, V.I. (2005) Quantifying Pb and Cd Complexation by Alginates and the Role of Metal Binding on Macromolecular Aggregation. Biomacromolecules, 6, 2756- 2764.
http://dx.doi.org/10.1021/bm050252y - 11. Drake, L.R., Lin, S., Rayson, G.D. and Jackson, P.J. (1996) Chemical Modifications and Metal Binding Studies of Datura innoxia. Environmental Science & Technology, 30, 110-114.
http://dx.doi.org/10.1021/es950131d - 12. Loudon, G.M. (1984) Organic Chemistry. Addison-Wesley Publishing Company, Boston.
- 13. Tobin, J.M., Cooper, D.G. and Neufeld, R.J. (1990) Investigation of the Mechanism of Metal Uptake by Rhizopus arrhizus Biomass. Enzyme and Microbial Technology, 12, 591-595.
http://dx.doi.org/10.1016/0141-0229(90)90132-A - 14. Rathinam, A., Maharshi, B., Janardhanan, S.K., Jonnalagadda, R.R. and Nair, B.U. (2010) Biosorption of Cadmium Metal Ion from Simulated Wastewaters Using Hypnea valentiae Biomass: A Kinetic and Thermodynamic Study. Bioresource Technology, 101, 1466-1470.
http://dx.doi.org/10.1016/j.biortech.2009.08.008 - 15. Foo, K.Y. and Hameed, B.H. (2010) Insights into Modeling of Adsorption Isotherm Systems. Chemical Engineering Journal, 156, 2-10. http://dx.doi.org/10.1016/j.cej.2009.09.013
- 16. Akar, T. and Tunali, S. (2006) Biosorption Characteristics of Aspergillus flavus Biomass for Removal of Pb(II) and Cu(II) Ions from an Aqueous Solution. Bioresource Technology, 97, 1780-1787. http://dx.doi.org/10.1016/j.biortech.2005.09.009
- 17. Amini, M., Younesi, H. and Bahramifar, N. (2009) Biosorption of Nickel(II) from Aqueous Solution by Aspergillus niger: Response Surface Methodology and Isotherm Study. Chemosphere, 75, 1483-1491. http://dx.doi.org/10.1016/j.chemosphere.2009.02.025
- 18. Baik, W.Y., Bae, J.H., Cho, K.M. and Hartmeier, W. (2002) Biosorption of Heavy Metals Using Whole Mold Mycelia and Parts Thereof. Bioresource Technology, 81, 167-170.
http://dx.doi.org/10.1016/S0960-8524(01)00148-1 - 19. Bhainsa, K.C. and D’Souza, S.F. (2008) Removal of Copper Ions by the Filamentous Fungus, Rhizopus oryzae from Aqueous Solution. Bioresource Technology, 99, 3829-3835. http://dx.doi.org/10.1016/j.biortech.2007.07.032
- 20. Kiran, I., Akar, T. and Tunali, S. (2005) Biosorption of Pb(II) and Cu(II) from Aqueous Solutions by Pretreated Biomass of Neurospora crassa. Process Biochemistry, 40, 3550-3558. http://dx.doi.org/10.1016/j.procbio.2005.03.051
- 21. Li, Z.J. and Yuan, H.L. (2006) Characterization of Cadmium Removal by Rhodotorula sp. Y11. Applied Microbiology and Biotechnology, 73, 458-463.
http://dx.doi.org/10.1007/s00253-006-0473-8 - 22. Tan, T.-W., Hu, B. and Su, H.J. (2004) Adsorption of Ni2+ on Amine-Modified Mycelium of Penicillium chrysogenum. Enzyme and Microbial Technology, 35, 508-513.
http://dx.doi.org/10.1016/j.enzmictec.2004.08.035 - 23. Tsekova, K., Todorova, D., Dencheva, V. and Ganeva, S. (2010) Biosorption of Copper(II) and Cadmium(II) from Aqueous Solutions by Free and Immobilized Biomass of Aspergillus niger. Bioresource Technology, 101, 1727-1731.
http://dx.doi.org/10.1016/j.biortech.2009.10.012 - 24. Mack, C., Wilhelmi, B., Duncan, J.R. and Burgess, J.E. (2007) Biosorption of Precious Metals: A Review. Biotechnology Advances, 25, 264-271.
http://dx.doi.org/10.1016/j.biotechadv.2007.01.003 - 25. Padmavathy, V., Vasudevan, P. and Dhingra, S.C. (2003) Biosorption of Nickel(II) Ions on Baker’s Yeast. Process Biochemistry, 38, 1389-1395.
http://dx.doi.org/10.1016/S0032-9592(02)00168-1 - 26. Congeevaram, S., Dhanarani, S., Park, J., Dexinilin, M. and Thamaraiselvi, K. (2007) Biosorption of Chromium and Nickel by Heavy Metal Resistant Fungal and Bacterial Isolates. Journal of Hazardous Materials, 146, 270-277.
http://dx.doi.org/10.1016/j.jhazmat.2006.12.017 - 27. Fan, T., Liu, Y.G., Feng, B.Y., Zeng, G.M., Yang, C.P., Zhou, M., Zhou, H.Z., Tan, Z.F. and Wang, X. (2008) Biosorption of Cadmium(II), Zinc(II) and Lead(II) by Penicillium simplicissimum: Isotherms, Kinetics and Thermodynamics. Journal of Hazardous Materials, 160, 655-661. http://dx.doi.org/10.1016/j.jhazmat.2008.03.038
- 28. Akar, T. and Tunali, S. (2005) Biosorption Performance of Botrytis cinerea Fungal By-Products for Removal of Cd(II) and Cu(II) Ions from Aqueous Solutions. Minerals Engineering, 18, 1099-1109. http://dx.doi.org/10.1016/j.mineng.2005.03.002
- 29. Lou, J.-M., Xiao, X. and Luo, S.-L. (2010) Biosorption of Cadmium(II) from Aqueous Solutions by Industrial Fungus Rhizopus cohnii. Transactions of Nonferrous Metals Society of China, 20, 1104-1111. http://dx.doi.org/10.1016/S1003-6326(09)60264-8
- 30. Tunali, S., Çabuk, A. and Akar, T. (2006) Removal of Lead and Copper Ions from Soil. Chemical Engineering Journal, 115, 203-211. http://dx.doi.org/10.1016/j.cej.2005.09.023
- 31. Dursun, A.Y. (2006) A Comparative Study on Determination of the Equilibrium, Kinetic and Thermodynamic Parameters of Biosorption of Copper(II) and Lead(II) Ions onto Pretreated Aspergillus niger. Biochemical Engineering Journal, 28, 187-195.
http://dx.doi.org/10.1016/j.bej.2005.11.003 - 32. Mashitah, M.D., Azila, Y.Y. and Bhatia, S. (2008) Biosorption of Cadmium(II) Ions by Immobilized Cells of Pycnoporus sanguineus from Aqueous Solution. Bioresource Technology, 99, 4742-4748. http://dx.doi.org/10.1016/j.biortech.2007.09.062
- 33. Baysal, Z., Çinar, E., Bulut, Y., Alkan, H. and Dogru, M. (2009) Equilibrium and Thermodynamic Studies on Biosorption of Pb(II) onto Candida albicans Biomass. Journal of Hazardous Materials, 161, 62-67. http://dx.doi.org/10.1016/j.jhazmat.2008.02.122
- 34. Anayurt, R.A., Sari, A. and Tuzen, M. (2009) Equilibrium, Thermodynamic and Kinetic Studies on Biosorption of Pb(II) and Cd(II) from Aqueous Solution by Macrofungus (Lactarius scrobiculatus) Biomass. Chemical Engineering Journal, 151, 255-261.
http://dx.doi.org/10.1016/j.cej.2009.03.002 - 35. Romera, E., González, F., Ballester, A., Blazquez, M.L. and Muñoz, J.A. (2007) Comparative Study of Biosorption of Heavy Metals Using Different Types of Algae. Bioresource Technology, 98, 3344-3353. http://dx.doi.org/10.1016/j.biortech.2006.09.026
- 36. Liu, Y.G., Fan, T., Zeng, G.-M., Li, X., Tong, Q., Ye, F., Zhou, M., Xu, W.H. and Huang, Y. (2006) Removal of Cadmium and Zinc Ions from Aqueous Solution by Living Aspergillus niger. Transactions of Nonferrous Metals Society of China, 16, 681-686.
http://dx.doi.org/10.1016/S1003-6326(06)60121-0 - 37. Selatnia, A., Madani, A., Bakhti, M.Z., Kertous, L., Mansouri, Y. and Yous, R. (2004) Biosorption of Ni2+ from Aqueous Solution by a NaOH-Treated Bacterial Dead Streptomyces rimosus Biomass. Minerals Engineering, 17, 903-911.
http://dx.doi.org/10.1016/j.mineng.2004.04.002 - 38. Vilar, V.J.P., Sebesta, F., Botelho, C.M.S. and Boaventura, R.A.R. (2005) Equilibrium and Kinetic Modeling of Pb2+ Biosorption by Granulated Agar Extraction Algal Waste. Process Biochemistry, 40, 3276-3284. http://dx.doi.org/10.1016/j.procbio.2005.03.021
- 39. Padmavathy, V. (2008) Biosorption of Nickel(II) Ions by Baker’s Yeast: Kinetic, Thermodynamic and Desorption Studies. Bioresource Technology, 99, 3100-3109.
http://dx.doi.org/10.1016/j.biortech.2007.05.070 - 40. Aksu, Z., Kutsal, T., Gun, S., Haciosmanoglu, N. and Gholminejad, M. (1991) Investigation of Biosorption of Cu(II), Ni(II) and Cr(VI) Ions to Activated Sludge Bacteria. Environmental Technology, 12, 915-921. http://dx.doi.org/10.1080/09593339109385086
- 41. Jalali, R., Ghafourian, H., Asef, Y., Davarpanah, S.J. and Sepehr, S. (2002) Removal and Recovery of Lead Using Non Living Biomass of Marine Algae. Journal of Hazardous Materials, 92, 253-262. http://dx.doi.org/10.1016/S0304-3894(02)00021-3
- 42. Sujatha, P., Kalarani, V. and Kumar, B.N. (2013) Effective Biosorption of Nickel(II) from Aqueous Solutions Using Trichoderma viride. Journal of Chemistry, 2013, Article ID: 716098.
http://dx.doi.org/10.1155/2013/716098 - 43. Gopal, M., Pakshirajan, K. and Swaminathan, T. (2002) Heavy Metal Removal by Biosorption Using Phanerochaete chrysosporium. Applied Biochemistry and Biotechnology, 102, 227-237. http://dx.doi.org/10.1385/ABAB:102-103:1-6:227
- 44. Kapoor, A. and Viraraghavan, T. (1997) Fungi as Biosorbents. In: Wase, J. and Forster, C., Eds., Biosorbents for Metal Ions, Taylor & Francis, London, 67-85.
- 45. Tan, T.W. and Cheng, P. (2003) Biosorption of Metal Ions with Penicillium chrysogenum. Applied Biochemistry and Biotechnology, 104, 119-128.
http://dx.doi.org/10.1385/ABAB:104:2:119 - 46. Vasudevan, P., Padmavathy, V. and Dhingra, S.C. (2002) Biosorption of Monovalent and Divalent Ions on Baker’s Yeast. Bioresource Technology, 82, 285-289.
http://dx.doi.org/10.1016/S0960-8524(01)00181-X - 47. Allaboun, H. and Abu Al-Rub, F.A. (2008) Dynamics, Mechanistic and Equilibrium Studies for the Biosorption of Nickel on Palm Tree Leaves. Jordan Journal of Civil Engineering, 2, 125-138.










