Surgical Science
Vol.4 No.2(2013), Article ID:27870,3 pages DOI:10.4236/ss.2013.42029
Pancreatic Involvement in Von Hippel-Lindau Syndrome: Report of a Total Pancreatectomy Case and Literature Review
1Department of General Surgery, Istanbul Medical Faculty, Istanbul University, Istanbul, Turkey
2Department of Radiology, Istanbul Medical Faculty, Istanbul University, Istanbul, Turkey
Email: *orhanagcaoglu@hotmail.com
Received November 25, 2012; revised December 23, 2012; accepted January 2, 2013
Keywords: Von Hippel-Lindau Syndrome; Pancreatic Cyst; Malignancy
ABSTRACT
Von Hippel-Lindau (VHL) syndrome is a rare, autosomal dominantly inherited multicancer syndrome. Disease is predisposed by hemangioblastoma, renal cell carcinoma, pheochromocytoma, pancreatic islet cell and cystic tumors or epididymis tumors under the mutation or deletion at the tumor suppressor gene. Here we report a 42-year-old woman who had been operated for cerebellar hemangioblastoma and retinal angioma, and presented with recurrent, mild abdominal pain. In radiological imaging studies, multiple pancreatic cysts without nodulation were determined. Because of the mild abdominal pain and uncertain malignant potential of the cysts, total pancreatectomy was performed. She was discharged at the fifteenth day after the surgery without any problem. The patient has been followed for 62 months without any problem, and her diabetes mellitus is being controlled by medication.
1. Introduction
Von Hippel-Lindau (VHL) syndrome is a rare disease that is inherited by the autosomal dominant pattern [1]. The disease is characterized with benign or malignant lesions that are seen primarily at the central nervous system and with multiple organ involvements such as kidney, pancreas, adrenals, liver and epididymis [2]. However, it is usually presented with neurological or ocular symptoms because the other organ involvements cause delayed symptoms [1]. History of the patient and the adjacent organ involvement findings support the diagnosis [1].
2. Case Report
Forty two-year-old woman who had been operated twice for cerebellar hemangioblastoma and retinal angioma was examined for mild abdominal pain in another hospital. The patient was referred to our clinic after the detection of gallstones and cysts at the epigastrium in ultrasound (US) examination. Liver function tests were detected higher (alkaline phosphatase(ALP): 263 U/l (30- 135), aspartate transaminase (AST): 84 U/l (5-42), alanine transaminase(ALT): 127 U/l (5-45), gamma-glutamyltranspeptidase (GGT): 96 U/l (5-85), total bilirubine: 1.42 mg/dl (0.2-1.0), direct bilirubine:0.86 mg/dl (0.0-0.3)), and total protein: 3.7 g/dl, albumine: 2.1 g/dl. All tumor markers were in normal range, Carbon-hydrate antigen (CA 19-9):21 U/ml (3-35), (CA 15-3):23 U/ml (0 -25), (CA 12-5):0.8 U/ml (1-35), carcinoembryonic antigen (CEA): 2 ng/ml (0-5). Diabetes mellitus was not detected. Her sister’s cranial tumor operation was learned after taking family history but no particular information procured.
Multiple milimetric cysts and calcifications among the pancreas, multiple milimetric stones at the gall bladder, biliary duct dilatations, and multiple and bilaterally renal anechoic cystic lesions were determined at repeated abdominal US in our institution. Computed tomography (CT) and magnetic resonance imaging (MRI) were performed. Multiple, hipoechoic cystic lesions replace the entire pancreas, and cysts at bilaterally kidneys were detected (Figure 1). Cyst walls were kept contrast. Hepatic duct dilatation (possible in the cause of choledochus compression) was detected.
Cranial CT was performed because of the cerebellar surgery history. Multiple milimetric cerebral cysts were detected; however, neither a hemangioblastoma recurrence nor a solitary (metastatic) lesion was determined. After the neurosurgery clinics consultation, the follow-up of the cerebral cystic lesions was planned. The patient
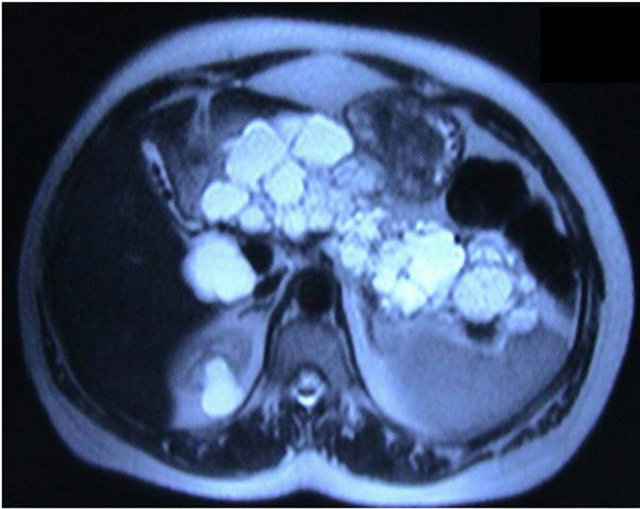
Figure 1. Magnetic resonance imaging revealed multiple hipoechoic cystic lesions replacing the entire pancreas, and cyst at right kidney.
was operated by the diagnosis of Von Hippel-Lindau syndrome with pancreatic involvement. During the surgery, multiple milimetric cysts were seen in the entire pancreas, and no pancreas parenchyma was palpated. Total pancreatectomy and splenectomy was performed. Invasive malignancy was not detected at histopathologycal examination of the pancreas. The cystic lesions with the largest one being 1 cm in diameter, were reported as microcystic adenoma (Figure 2) and at the immunophenotypic studies, the cells that were covering the cystic structures were determined CD 34 negative and cytokeratine 8/18, cytokeratine 19 positive.
The patient was discharged at the fifteenth day of the surgery without any surgical problem. Serum glucose level was controlled by insulin replacement therapy. The patient has been followed consistently for 62 months without progression at the cranial and renal cysts, and without local or systemic recurrences. Diabetes mellitus is under control by intense insulin therapy.
3. Discussion
VHL syndrome is seen 2-3/100.000 and inherited by autosomal dominant pattern [1]. It is presented with visceral organ involvements such as retinal angioma, cerebellar hemangioblastoma, renal cell carcinoma, pheochromocytoma and pancreatic cyst and neoplasms which are caused by the mutation or deletion of chromosome 3p25 [2].
Pancreatic cysts are the most frequent findings of the VHL disease and especially serous cystadenomas [3]. Pancreatic involvement in VHL ranges 16% to 29%. Neumann et al. [4] reported the frequency of pancreatic involvement 16.62% in their series and 23% in their family inherited series. Levine et al. reported the frequency
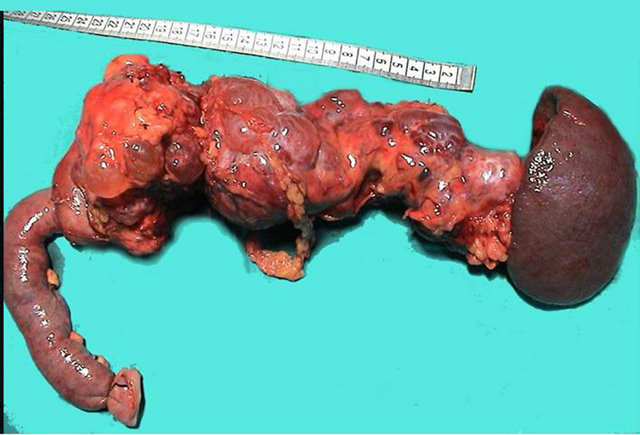
Figure 2. Serous cysts replace the entire pancreas.
of pancreatic involvement as 29% [5]. The cysts are frequently multiple and calcified [3]. The patients with pancreatic involvement are usually asymptomatic or they have non-specific complaints such as mild abdominal pain or discomfort [1]. Pancreatic insufficiency (or diabetes mellitus) is seen rarely, especially more frequent at diffuse pancreatic involvement and large cystic lesions [6-8]. The incidence of pancreatic cysts is increased by the development and improvement of the imaging techniques, and treatment chooses are still a clinical dilemma. Also surgical treatment for the pancreatic involvement of VHL syndrome has controversies. Most of the pancreatic cysts at the VHL syndrome are benign, simple cystic structures or serous cystadenomas [1,9,10]. Serous cystadenomas had been show that can undergo malignant transformation [10]. The authors argue for more aggressive approach to these tumors, most surgeons would find these procedures prudent. In our case, non-specific mild abdominal pain was presented, diffuse pancreatic involvement was seen and the cysts walls were keeping contrast, that’s why we preferred surgery. Hammel et al. declares that the lesions smaller than 1 cm diameter can be followed, but the larger lesions must be resected because of the risk of being metastatic [6]. Solid pancreatic tumors such as ductal adenocarcinoma and islet cell tumors are rarely seen with VHL and serous cysts [7,10]. Cheng et al. reported malignant islet cell tumors in 6 patients, and adenocarcinoma in 2 patients who had VHL syndrome with pancreatic involvement [7]. Adrenal pheochromocytomas, hepatic hemangiomas, hepatic adenomas, paragangliomas and polycytemia are other clinical entities that are rarely seen with VHL [2]. In case of suspicion at the diagnosis of VHL syndrome and the origins of the cystic lesion(s), surgery is the relevant treatment. Furthermore, if malignancy suspicion or mild symptoms are developed, the choice must be the surgery [9]. In our case, abdominal pain complaint was solved after surgery, and no malignancy was detected at histopathology. VHL syndrome must be kept in mind with the asymptomatic or atypically symptomatic pancreatic cysts that are presented with multiple organ involvement. Surgery should not be ignored with the symptomatic patients, and the patients with the suspicion of malignancy. It is possible to obtain cure by surgery in VHL patients with pancreatic involvement. However, the border between the conservative therapy and surgical procedure must be determined well because of the life-long replacement requirement after surgery.
REFERENCES
- B. M. Künzli, S. V. Shrikhande, M. W. Büchler, et al., “Pancreatic Lesions in Von Hippel-Lindau Syndrome: Report of a Case,” Surgery Today, Vol. 34, No. 7, 2004, pp. 626-629. doi:10.1007/s00595-004-2769-6
- C. A. Friedrich, “Von Hippel-Lindau Syndrome: A Pleamorphic Condition,” Cancer, Vol. 86, No. S11, 1999, pp. 2478-2482. doi:10.1002/(SICI)1097-0142(19991201)86:11+<2478::AID-CNCR4>3.0.CO;2-5
- B. Taouli, M. Ghouadni, J. M. Correas, et al., “Spectrum of Abdominal Imaging Findings in Von Hippel-Lindau Disease,” American Journal of Roentgenology, Vol. 181, No. 4, 2003, pp. 1049-1054.
- H. P. H. Neumann, E. Dinkel, H. J. Brambs, et al., “Pancreatic Lesions in the Von Hippel-Lindau Syndrome,” Gastroenterology, Vol. 101, No. 2, 1991, pp. 465-471.
- E. Levine, D. L. Collins, W. A. Horton, et al., “CT Screening of the Abdomen in von Hippel-Lindau Disease: An Unusual Cause of Pancreatic and Common Bile Duct Obstruction,” American Journal of Radiology, Vol. 139, 1982, pp. 505-510.
- P. R. Hammel, V. Vilgrain, B. Terris, et al., “Pancreatic Involvement in Von Hippel-Lindau Disease,” Gastroenterology, Vol. 119, No. 4, 2000, pp. 1087-1095. doi:10.1053/gast.2000.18143
- T. Y. Cheng, C. H. Su, Y. M. Shyr, et al., “Management of Pacreaticlesions in Von Hippel-Lindau Disease,” World Journal of Surgery, Vol. 21, No. 3, 1997, pp. 307-312. doi:10.1007/s002689900234
- B. Mukhopadhyay, A. Sahdev, J. P. Monson, et al., “Pancreatic Lesions in Von Hippel-Lindau Disease,” Clinical Endocrinology, Vol. 57, No. 5, 2002, pp. 603-608. doi:10.1046/j.1365-2265.2002.01637.x
- C. M. Pyke, J. A. Heerden, T. V. Colby, et al., “The Spectrum of Serous Cystadenoma of the Pancreas Clinical, Pathologic and Surgical Aspects,” Annals of Surgery, Vol. 215, No. 2, 1992, pp. 132-139. doi:10.1097/00000658-199202000-00007
- N. Agarwal, S. Kumar, J. Dass, et al., “Diffuse Pancreatic Serous Cystadenoma Associated with Neuroendocrine Carcinoma: A Case Report and Review of Literature,” Journal of Pancreas, Vol. 10, No. 1, 2009, pp. 55-58.
NOTES
*Corresponding author.

