Open Journal of Psychiatry
Vol.4 No.1(2014), Article ID:41667,10 pages DOI:10.4236/ojpsych.2014.41007
Disulfiram in the management of alcohol dependence: A comprehensive clinical review
1Northern Area Mental Health Services, Melbourne, Australia
2Desousa Foundation, Mumbai, India
3 Department of Psychiatry, The University of Western Ontario, Lawson Health Research Institute, London, Canada
Email: *avinashdes888@gmail.com
Copyright © 2014 Gurvinder Kalra et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accordance of the Creative Commons Attribution License all Copyrights © 2014 are reserved for SCIRP and the owner of the intellectual property Gurvinder Kalra et al. All Copyright © 2014 are guarded by law and by SCIRP as a guardian.
Received 10 November 2013; revised 2 December 2013; accepted 9 December 2013
ABSTRACT
Disulfiram remains a viable option as a treatment for alcohol dependence and has been shown in recent studies to be successful in treating patients with alcohol dependence in a manner that is superior to both naltrexone and acamprosate. It is also useful in dual diagnosis patients and those with co-morbid cocaine and alcohol dependence. Although disulfiram’s mechanism of action in alcohol dependence was long thought to be its effects as a psychological deterrent, more recent studies have uncovered potential anticraving effects as well. Recent reviews exhort to the importance of supervised disulfiram therapy in highlighting many of the potential and unique benefits of disulfiram. The present article will review the major clinical trials of disulfiram spanning nearly 60 years. It also discusses the usage of disulfiram across diverse populations along with monitoring for compliance and various adverse effects that may be encountered. The paper also reviews certain studies on long acting disulfiram therapy, recent comparative trials of disulfiram and its use in alcohol dependence. The review concludes with the role of disulfiram in the present day and long-term pharmacotherapy of alcohol dependence along with future research needs in this area.
KEYWORDS
Disulfiram; Alcohol Dependence; Alcohol; Deterrent; Pharmacotherapy; Long Acting Disulfiram
1. INTRODUCTION
Alcohol dependence is a chronic disorder that may have multiple relapses and remissions, increased mortality and low long-term abstinence rates that lead to increased psycho-social losses. Many drugs have been used in the treatment of this disorder such as the anti-craving agents, acamprosate, naltrexone and the aversive agent, disulfiram. Disulfiram has been in use since the early 1940s for the treatment of alcohol dependence and is the first FDA-approved medication for the treatment of this disorder [1,2]. Disulfiram has thus completed almost 60 years of use in alcohol use disorders and has stood the test of time. A large number of studies have been done on this molecule, ever since some proving its superiority over other drugs while others negating it.
2. MECHANISM OF ACTION OF DISULFIRAM
Ethanol undergoes metabolism in the liver initially by alcohol dehydrogenase (ADH) forming acetaldehyde; this is removed from the body primarily by oxidation into acetate by acetaldehyde dehydrogenase (ALDH) [3] (Figure 1), which finally enters the citric acid cycle.
Disulfiram acts by inhibiting the enzyme ALDH via its metabolite S-methyl N, N-diethyl-dithio-carbamate-sulphoxide [4,5], leading to accumulation of acetaldehyde in blood. This gives rise to various manifestations of disulfiram-alcohol reaction (DER) [6]. Since the inhibition of ALDH by disulfiram is irreversible, the DER will get terminated only after production of new ALDH oncedisulfiram is stopped. The new ALDH takes about a week’s time to be produced. Hence patients should be advised to take alcohol only after 2 weeks of stopping disulfiram [7]. In addition to this, disulfiram also acts on the dopaminergic system, both disulfiram and its metabolite carbon disulfide leading to inhibition of dopamine beta-hydroxylase (DBH) that leads to increase in the levels of dopa-
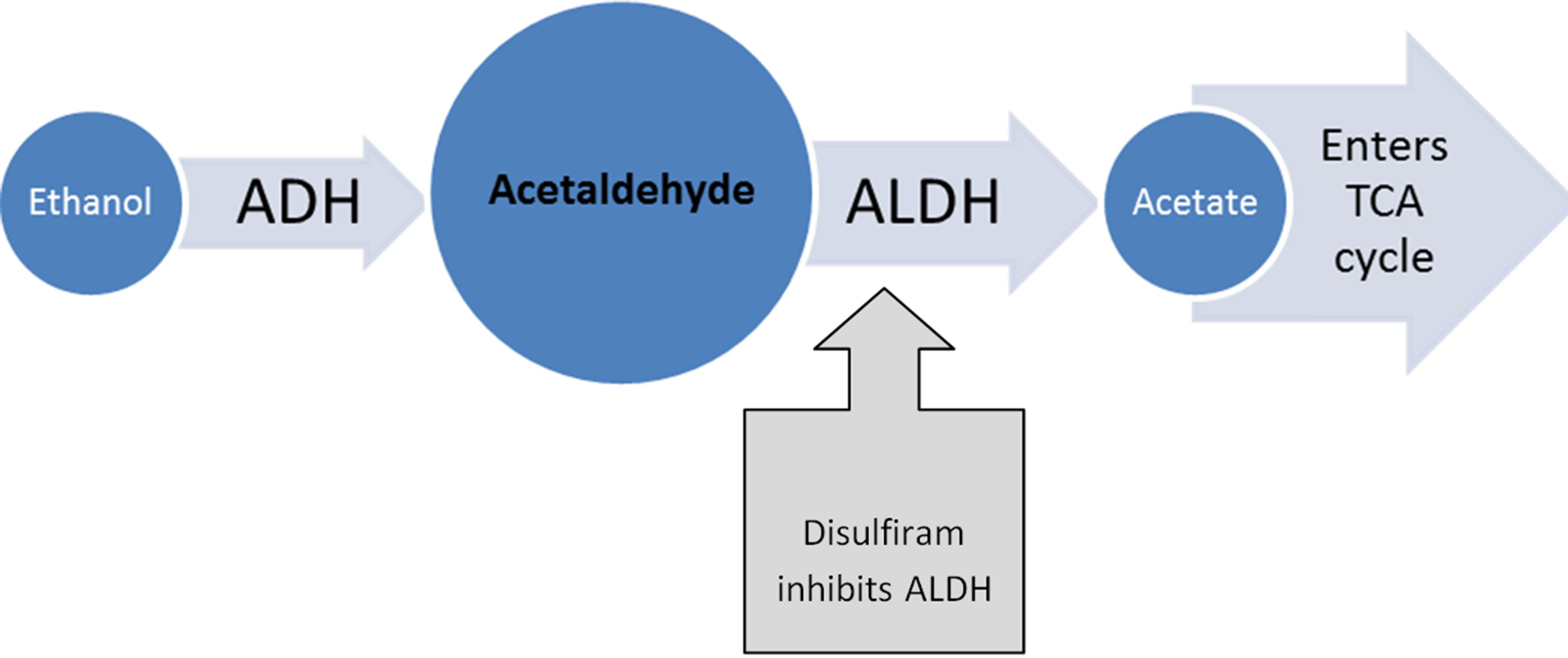
Figure 1. Mechanism of action of disulfiram. (TITLE) ADH-alcohol dehydrogenase; ALDHaldehyde dehydrogenase; TCA-Tri-Carboxylic Acid cycle.
mine. This may give rise to several neuropsychiatric manifestations such as delirium, paranoia, impairment of memory, ataxia, dysarthria and frontal lobe release signs [8]. Besides this action, disulfiram is also known to inhibit dopamine beta-hydroxylase leading to an increase in dopamine concentrations but decreased norepinephrine in the brain [9]. This may suggest an anti-craving role of disulfiram in alcohol dependence [10].
3. EFFICACY STUDIES OF DISULFIRAMWHEN USED ALONE OR COMPARED TO PLACEBO IN ADULTS
In a recent meta-analysis of 11 randomized controlled trials with a total of 1527 patients, researchers concluded that supervised disulfiram did have some effect on the short-term abstinence, days until relapse and number of drinking days when compared to placebo, no treatment or other treatments available for patients with alcohol dependence or abuse [11].
There are mixed reports and reviews on the efficacy of disulfiram in alcohol dependence. While some authors believe that it is among the most successful medications for alcohol dependence [12-14], there are others who consider it just another second-line medication with moderate efficacy [15,16]. Fears of The Disulfiram Ethanol Reaction (DER) in clinicians and its effects on their patients along with issues related to high cost and lack of easy availability of disulfiram in certain nations may have added to moderation of its use. Another factor based on clinical experience and personal communication with many psychiatrists in India by one of authors (De Sousa) is that lack of exposure to disulfiram use during psychiatry residency and training often serves as a deterrent in its use at a later stage during private clinical practice.
Ulrichsen and others have studied the effect of supervised disulfiram along with cognitive behavior therapy (CBT) in 39 patients of alcohol dependence but reported only 20% and 26% rate of abstinence in the control and disulfiram group respectively [17]. They concluded that supervised disulfiram therapy did not have any major impact on the treatment outcome in this disorder. One of the most cited disulfiram trials to date, the Veterans Affairs multisite cooperative study also showed that disulfiram and placebo treated patients had similar outcomes [18]. It is worthwhile noting that though the study by Fuller is often the most quoted for disulfiram and its efficacy, Fuller himself mentions the need for supervised disulfiram therapy. He has stated that the failure to include supervision of disulfiram therapy in his study is a serious defect. However, various authors have suggested that it is premature to reject the use of disulfiram in clinical settings based on just these studies [19]. Medication non-adherence is often cited by many authors as a strong factor that impedes the effectiveness of disulfiram since the patients themselves stop this drug. Authors studied 210 patients of alcohol dependence who had voluntarily discontinued disulfiram treatment and found that none of these patients reported adverse events as a reason for discontinuing disulfiram treatment. Around 70% of their patients gave reasons such as “a wish to drink again” or “no need for treatment anymore”. It is likely that many patients often over-estimate their self-capacity to control substance intake and in this belief, stop disulfiram but also restart alcohol [19]. Almost similar results were reported by others [20] who also reported only a small percentage (5% - 18%) of their 345 alcohol dependent inpatients having discontinued disulfiram owing to side effects. Thus it is not the side effects that lead to discontinuation of disulfiram, but the basic characteristics of “substance dependence” that lead to relapse.
Medication adherence and compliance with disulfiram can be increased easily with good psycho-education, family support and supervised disulfiram therapy when administered by a family member [12]. Clinically it is seen that patients using disulfiram reduce their alcohol consumption significantly [21] but more often than not, this may not be effective for the sole reason that many patients with alcohol dependence discontinue the drug prematurely [22]. Research suggests that the mode of action of disulfiram is a combined psychological deterrent action [14] and a physiological deterrent action. However experiencing the acetaldehyde reaction is not necessary for disulfiram’s action and does not lead to better treatment outcome in effect [23]. In one of the longest follow up studies till date (9 years from 1993-2002) authors have studied the abstinence, relapse and lapse rates in patients with alcohol dependence during an outpatient long term intensive therapy for alcoholics (OLITA) trial [24]. This is one of the few studies where sham disulfiram therapy was compared to actual disulfiram therapy. An abstinence rate of more than 50% was noted across both groups despite the long duration of the study. This indicates the role of long term deterrent (Disulfiram) therapy in the management of alcohol dependence along with a psychological component that plays a role in the mechanism of disulfiram’s action. This is because an assumption of just being on disulfiram therapy in the sham group led to similar abstinence rates compared to actual disulfiram therapy [25]. The same author has recently published a review article that emphasizes the role of disulfiram as an adjunct to psychotherapy in alcoholism treatment [7]. The article reviews disulfiram literature between 1937 and 2000 and reviews 13 clinical trials of disulfiram between the years 2000 and 2008. Disulfiram proved to be an effective therapeutic tool in all studies with superiority being documented in majority of the trials. The study mentions that initial psycho-education regarding the mechanism of action of disulfiram and its therapeutic implications are a must for efficacious use of the drug while disulfiram also may be extended to serve as a coping skill for the patient with dependence under treatment.
Disulfiram can be a useful option in the long term if given under supervision where it contributes significantly to longer periods of sobriety [26]. Even more recent papers give a thumbs-up to the long-term safety and efficacy of supervised disulfiram [27] which reports a complete abstinence in 7 out of 10 patients who were maintained on supervised disulfiram. The complete abstinence in these 7 patients was for a mean period of 70.1 ± 23.5 months; for the 3 patients that had relapse, the first relapse occurred after a mean of 34.7 ± 15.5 months. Supervision can be provided by a therapist or best by a close family member [28-30]. However a higher degree of motivation is required on the part of patients in order for them to continue disulfiram on a long term basis. Some authors have considered disulfiram therapy to be free of serious side effects [29] and beneficial only in the short term that does not improve long-term outcome in the treatment of alcoholism [31]. A few studies show the successful use of a breath analyzer to assess compliance status of patients on disulfiram [32]. But the cost of a breath analyzer is often a deterrent to its use in small routine clinical settings.
4. DISULFIRAM USE IN DUAL DIAGNOSIS PATIENTS
Use of disulfiram in the presence of psychiatric illnesses has been debated by many for two main reasons: the rate of substance use disorders is higher in patients with psychiatric illnesses especially those with psychotic spectrum disorders like schizophrenia and bipolar disorder [33] which often leads to increased psychotic symptoms [34]. Secondly, disulfiram can sometimes precipitate psychiatric symptoms such as depression, mania, psychosis and delirium [35]. However it is worth noting that reports that indicate this side-effect date back to the early 1970s when disulfiram was being used in larger doses ranging between 1000 and 3000 mg/d [36]. In a prospective open treatment study that evaluated the long-term course of drinking outcomes of 180 chronic alcoholics consecutively admitted from 1993 to 2002, subsamples are compared for 1) sham-AD versus verum-AD (disulfiram/calcium carbimide), 2) coped lapses versus finally detrimental lapses versus malignant relapses, and 3) AD use for 13 to 20 versus >20 months. In this study, the cumulative probability of not having relapsed was 0.52, and that of not having consumed any alcohol was 0.26. Disulfiram/calcium carbimide was well tolerated. Patients on sham-AD (due to contraindications to verum-AD) showed higher cumulative abstinence probability than patients on verum (S = 0.86 vs. S = 0.49, p = 0.03). The cumulative abstinence probability was S = 0.75 for patients with long-term intake compared with S = 0.50 for patients who stopped AD between months 13 and 20 (p < 0.001) [25]. An abstinence rate of >50% in this 9-year study strongly supports the concept of comprehensive, long-term outpatient treatment of alcoholics.
Mueser and others [37] conducted comprehensive chart reviews on 33 patients with alcoholism and severe mental illness that included 70% patients of schizophrenia who were maintained on disulfiram. Disulfiram use led to decreases in days hospitalized in patients and around 64% patients reported remission of alcoholism for at least one year during a 3-year follow-up. Authors [1] reported no significant changes in positive, negative or general PANSS scores in subjects with psychotic spectrum disorders who were given disulfiram. They also reported better alcohol-use outcomes for patients with a psychotic spectrum disorder who were on an active medication (disulfiram or naltrexone or combination) compared with placebo but did not report of any advantage of disulfiram or naltrexone or of the combination. In fact, Petrakis [1] supports the use of disulfiram for alcohol dependence with comorbid psychotic spectrum disorders, suggesting that these patients benefit more with such treatment methods as they may not be able to benefit fully from the forms of treatments developed for non-comorbid alcohol dependence patients. In yet another study [38], it was reported that subjects with depression reported lower craving over time with disulfiram than those on naltrexone. They concluded that disulfiram is safe for patients of alcohol use disorder and comorbid depression. Certain studies speak about reduction in craving with disulfiram therapy, while many others do not support this claim. Somewhat similar and encouraging results were also concluded by Petrakis [1] in 93 individuals who met DSMIV-TR criteria for post-traumatic stress disorder (PTSD). They reported better alcohol outcomes with active medication (naltrexone, disulfiram, or the combination) wherein overall psychiatric symptoms of PTSD improved. Authors [39] suggest that disulfiram can be safely and effectively used with patients who have comorbid diagnoses of Axis I and Axis II disorders (anti-social and borderline personality disorders); many clinicians fearing its use in personality disorders due to increased impulsivity in such cases. In their sample, a diagnosis of personality disorder did not adversely affect alcohol outcomes and they did not have a poorer response to medications than patients without diagnosis of ASPD or BPD. The results in a case series by Mutschler and others [40] do not seem very encouraging, with only 2 out of 8 patients with borderline personality disorder and alcohol dependence, remaining abstinent on supervised disulfiram therapy over a mean period of 9.25 months. However it opens up further research gates for use of disulfiram in personality disorders. Disulfiram has also been used in the treatment of alcohol dependence with pathological gambling wherein the patient reported that his desire to gamble disappeared completely [41,42].
5. DISULFIRAM USE IN ADOLESCENTS WITH ALCOHOL DEPENDENCE
In a first of its kind double-blind placebo-controlled study of disulfiram, 26 adolescents aged 16 - 19 years with chronic or episodic alcohol dependence were recruited and put on either disulfiram or placebo [43]. After 3 months of therapy, mean cumulative abstinence was reported to be significantly higher in the disulfiram group. Disulfiram was well tolerated with no major side effects. This was the first study that reported the usefulness of disulfiram as an adjunct to behavioral, psychosocial and psychotherapeutic treatments in the management of adolescent alcohol dependence. In another recent randomized study, disulfiram was compared to naltrexone in 58 adolescents with alcohol dependence [44]. Weekly psychoeducation was provided in both groups. At the end of the 6 month study, 79.3% patients in the disulfiram group remained abstinent compared to only 51.7% in the naltrexone group. This was the only study till date that has compared these drugs head on in adolescent population. However, disulfiram use in adolescents still appears a grey area of clinical practice. Further studies in larger groups and across diverse populations are needed before we reach any firm conclusions regarding its efficacy.
6. DISULFIRAM COMBINED WITH AND COMPARED TO OTHER INTERVENTIONS
Disulfiram may be either used alone or combined with other anti-craving drugs such as acamprosate or naltrexone. Some studies suggest that disulfiram may be more effective when used as an adjunct to the anti-craving medication acamprosate [45], while others show that it is superior to naltrexone, acamprosate and topiramate in relapse prevention [46-48]. On similar lines, a meta analysis of 641 randomized controlled trials and 7 longitudinal prospective studies [49] showed that supervised disulfiram administration along with psychotherapy was more effective than naltrexone plus cognitive behavior therapy or acamprosate. Similar findings were presented in earlier reviews [50]. In a controlled study on 118 patients [51], it was reported that concomitant disulfiram improved the effectiveness of acamprosate in chronic alcohol dependence.
The first published randomised controlled study comparing supervised naltrexone and supervised disulfiram in pure alcoholism was published in 2004 [46]. It showed that supervised disulfiram therapy was significantly more effective, even though—intriguingly—the naltrexone patients reported somewhat lower craving levels. The differences were not just statistically significant; they were really striking and clinically important. After 12 months, nearly twice as many patients were relapse free on disulfiram (86%) as on naltrexone (44%) (p = 0.0009). Survival time until the first relapse to heavy drinking was longer with disulfiram (119 vs. 63 days p = 0.020); and gamma-glutamyltranspeptidase (GGT) fell significantly (p = 0.038) more than in the naltrexone group.
The same authors carried out a similar study comparing disulfiram and acamprosate, except that it lasted eight months instead of twelve. It produced very similar results. At the end of the trial, 93 of 100 patients were still in contact. Relapse (the consumption of >5 drinks/40g of alcohol in one day) occurred at a mean of 123 days with disulfiram compared to 71 days with acamprosate (p = 0.0001). Eighty-eight per cent of patients on disulfiram remained abstinent compared to 46% with acamprosate (p = 0.0002). However, as with the naltrexone study, patients allocated to acamprosate had lower craving than those on disulfiram (p = 0.002) [47]. Similar results have been reported in a comparative study between disulfiram and topiramate where too, topiramate had better anti craving action than disulfiram [48].
In a study done in Finland, disulfiram, naltrexone and acamprosate in separate groups were compared when used in combination with a cognitive behavioural intervention in 243 patients with alcohol dependence [52]. All three study groups showed marked reduction in drinking from baseline to the end of the study. During the continuous medication phase, treatment with disulfiram was more effective in reducing heavy drinking days and average weekly alcohol consumption, as well as increasing time to the first drink, and the number of abstinent days. As per their findings, patients allocated to acamprosate, naltrexone and disulfiram combined with brief manual-based cognitive behavioural intervention significantly reduced their alcohol consumption and reported improved quality of life. However, supervised disulfiram appeared superior, especially during the continuous medication period when compared to naltrexone and acamprosate. In yet another first of its kind study, disulfiram alone and combination with naltrexone was found to be superior to naltrexone alone and placebo in the management of combined alcohol and cocaine dependence [53].
In another recent trial, researchers have speculated that combining disulfiram and gamma hydroxyl butyric acid may have an additive effect [54]. The authors speculated that by combining disulfiram with GHB treatment we may be able to achieve a kind of “antagonist” effect by using the “psychological threat” of disulfiram, while taking advantage of the anti-craving effect of GHB, despite the limitation of its “non-blockade” effect on alcohol. The study showed 65.4% of patients responding to this therapy. However further randomized trials are warranted to verify this hypothesis.
7. TOLERABILITY AND SAFETY STUDIES
Although considered safe at therapeutic doses with no high incidence of adverse effects [19], problems may arise in case a patient on disulfiram is exposed to alcohol [55] leading to disulfiram ethanol reaction (DER). In the absence of alcohol consumption, patients on disulfiram may complain of tiredness, sleepiness, and headache. It often leaves the patient having an unpleasant breath odor described as garlic-like or metallic taste [7]. With these side effects, patients need not stop disulfiram as most of them remit within some weeks [7].
Symptoms of DER occur mainly due to histamine-like effects of the accumulated acetaldehyde [56]. These symptoms may begin within 5 - 15 minutes after ingestion of ethanol in patients [7] who have taken disulfiram 3 - 123 hours earlier [57]. Symptoms on alcohol consumption may include flushing, sweating, nausea, vomiting, palpitations, dyspnea and hyperventilation, tremors, confusion, restlessness, drowsiness and hypotension; all these symptoms are usually self-limiting except for hypotension that can sometimes be severe and life-threatening [58,59] often this may lead to rare cases of acute myocardial infarction [60] and ischemic stroke [61]. In most clinical settings, complete recovery is the usual rule although rarely life threatening reactions may occur [61].
In this context, one has to be aware of warning patients against alcohol consumption in any form; patients also need to be warned for occupational solvent exposures while prescribing disulfiram as DER can occur with occupational exposure to products containing alcohol and other solvents [62]. One may also consider using lower doses of disulfiram at 125 mg without its deterrent effect being hampered [63]. An interesting observation has also been made by authors [64] to improve detection of any unheralded consumption of alcohol in small quantity by the patient maintained on disulfiram. Monitoring of urinary ethylglucuronide (EtG) improved detection of abstinence in such patients thereby also improving safety by preventing chronic acetaldehyde exposure which has carcinogenic, neurotoxic, and cardiotoxic properties.
Management of DER mainly consists of supportive measures such as Trendelenberg position, oxygen, intravenous fluids, and norepinephrine that is considered as the pressor agent of choice [59]. 4-methylpyrazole (4-MP) is an alcohol dehydrogenase inhibitor that inhibits the accumulation of acetaldehyde in case of disulfiram-alcohol reaction and leads to improvement in symptoms such as facial flushing and tachycardia [65]. Fomepizole, a first line antidote for methanol poisoning is also a potent inhibitor of alcohol dehydrogenase [66,67]. A single dose of fomepizole (15 mg/kg) has recently been recommended for severe DERs especially with hypotension unresponsive to fluid resuscitation or angioedema unresponsive to antihistaminics. Both these symptoms can be corrected within 1 - 1.5 hours [56].
Psychoses, including Capgras delusions are known to occur either during or after the use of disulfiram [68,69]. Though psychosis can occur in patients without any previous history of psychosis or brain disease, it is more common if there is pre-existing vulnerability in the individual such as a positive family history of psychosis or if higher doses of disulfiram are used [70]. Interestingly, psychiatric complications with disulfiram are more common in the Indian settings than in Western countries [71]. The reasons for the same are unknown though it could be cultural differences in neurobiology and genetic polymorphisms in liver metabolism pathways that need to be investigated.
Although disulfiram may also lead to hepatitis which could be fatal, the risk is very low and has been estimated to be 1 case in 30,000 treated patients per year [71]. Wright and others [72] discourage the decision to refrain from prescribing disulfiram for the fear of causing hepatotoxicity. However this adverse event can even occur at low doses and is usually seen between 16 and 120 days after the start of treatment with a peak frequency at 60 days [73]. It is therefore recommended that before a patient is started on disulfiram, baseline liver function tests be obtained, with repeat testing at two weeks, three months, and every six months thereafter [72]. However waiting for the liver function tests could also mean losing on precious time especially during the initial days when patient motivation to leave alcohol and start disulfiram may be high. In such situations one may rely on clinical signs of liver dysfunction and start the patient on disulfiram after proper patient psycho-education.
Disulfiram has also been reported to cause toxic optic neuropathy which can recover rapidly and completely after discontinuing the medicine [74]. Rarer side effects of disulfiram that are usually seen in overdoses include posterior reversible encephalopathy syndrome (PRES) [75], toxic encephalopathy with coma and convulsions [76]. Disulfiram use has also been rarely associated with development of catatonia either when given in high doses in patients with a risk of developing psychosis or even in patients without such risk [77]. Several hypotheses have been proposed in this respect: disulfiram induced dopamine hyperactivity in the meso-limbic dopaminergic pathway leads to a down-regulation of GABA-activity in thalamus and basal ganglia [78] that may in turn lead to symptoms of catatonia [79]; metabolites of disulfiram may exert direct neurotoxic effect on globus-pallidus and substantia nigra precipitating catatonic symptoms.
Polyneuropathy has also been reported in 3 out of 50 patients in a study by DeSousa and DeSousa [47]. These neuropathies may have an abrupt onset wherein patients may complain of fatigue, amnesia, and confusion; subacute that leads to ataxia, stupor, seizures and encephalopathy; and chronic that usually presents in the form of optic neuritis, ptosis and peripheral neuropathy [74].
8. ADVANTAGES OF DISULFIRAM THERAPY IN ASIAN SETTINGS
Disulfiram is the most common drug that is used for maintenance therapy for alcohol dependence in India [80]. It is cheaper than acamprosate and naltrexone in India [13] while the reverse is true for Europe and USA. In fact, supervised disulfiram therapy along with high-frequency contact with professionals is more costly compared to acamprosate in Europe [81]. One needs to evaluate the randomized trials done in India from a cost effectiveness perspective to help us determine its true cost efficacy in Indian settings [82]. It is worthwhile to note here that disulfiram is also given by many faith healers in India as “divine ash” or by traditional healers as “traditional medicine” to help an individual with alcohol dependence [83].
9. LONG ACTING DISULFIRAM THERAPY (DISULFIRAM IMPLANTS)
Disulfiram implants have been in use since the 1950s in the treatment of alcohol dependence [84]. Earlier reports of disulfiram implants reported them to be largely ineffective due to their failure to release adequate levels of disulfiram, risks of surgical complications such as infection and rejection [85]. The only assessment that was done in these studies were subject reports of abstinence and self reported DERs. There were no objective assessments included such as blood concentrations of acetaldehyde. Johnsen and Morland [86] also reported no difference between disulfiram and the placebo group, except for a higher incidence of wound complications in the disulfiram group. In this study blood tests for DSF were done (which were negative) and they also gave alcohol surreptitiously to see if patients had any true physiological reaction (which they didn’t).
10. CURRENT STATUS OF DISULFIRAM THERAPY
Long term pharmacotherapy of alcohol dependence is keenly poised today. It is important for clinicians treating alcohol dependence to note that naltrexone and acamprosate, though effective, only reduce craving and do not deter the patient from taking alcohol. He may still drink while on these drugs with no untoward effects. Disulfiram, though underused, is cheaper than the above two drugs but very effective as an alcohol deterrent as fear of a disulfiram ethanol reaction forces the patient to be off alcohol when on the drug. Combination therapy of disulfiram with any of the other drugs there by acting on different neurobiological systems may be optimal for the effective management of alcohol dependence. The use of psycho-education about disulfiram and its actions is very essential to get the best effects out of the medication. It is also important to note that since the knowledge of being on disulfiram therapy is imminent on its effective action, this impedes the conducting of double blind trials with the drug for ethical reasons while also introducing an element of bias with open studies.
11. FUTURE RESEARCH NEEDS
Although disulfiram completes its 60 years as a molecule, we still need further studies across diverse populations and subgroups of alcohol dependence with disulfiram therapy. We also need to study the effects of disulfiram when compared to other agents in patients who are alcohol dependent and have comorbid psychiatric disorders. There is a dearth of head-on comparative studies of various agents like naltrexone, acamprosate, GHB and baclofen with disulfiram in the long term management of alcohol dependence. Lack of funding for disulfiram studies with pharmaceutical industries preferring to focus on other drugs has also been an obstacle in this endeavor.
Another area worth mentioning is pharmacogenomics which deals with the influence of genetic variation on drug response in patients. Some work has already been done in this context in alcohol dependence and naltrexone and acamprosate. The functionally significant OPRM 1 Asp 40 allele predicts response to naltrexone treatment [87,88], whereas genetic variations in GATA4 genotype (GATA-binding protein 4) influences treatment response to acamprosate via modulation of atrial natriuretic polypeptide (ANP) plasma levels [89].
12. CONCLUSION
Disulfiram is being used successfully for over 6 decades now; it is the oldest molecule used in alcohol dependence pharmacotherapy, and disulfiram has stood the test of time. Since usage of disulfiram needs continued and repeated consultation with the treating psychiatrist and active decision-making by the patient, it is likely that such consultations give a chance to both of them to improve on the abstinence attempts of the patient. It has a good safety record and its pharmacokinetics have been extensively studied. The psychological aspects of supervised disulfiram therapy along with regular consultations with the health care provider contribute to a good clinical outcome with this therapy.
CONFLICT OF INTEREST
None.
REFERENCES
- Petrakis, I.L., Nich, C. and Ralevski, E. (2006) Psychotic spectrum disorders and alcohol abuse: A review of pharmacotherapeutic strategies and a report on the effectiveness of naltrexone and disulfiram. Schizophrenia Bulletin, 32, 644-654. http://dx.doi.org/10.1093/schbul/sbl010
- De Sousa, A. (2010) The pharmacotherapy of alcohol dependence: A state of the art review. Mens Sana Monographs, 8, 69-82. http://dx.doi.org/10.4103/0973-1229.58820
- Deitrich, R.A., Petersen, D. and Vasiliou, V. (2007) Removal of acetaldehyde from the body. Novartis Foundation Symposia, 285, 23-40.
- Johansson, B. (1992) A review of the pharmacokinetics and pharmacodynamics of disulfiram and its metabolites. Acta Psychiatrica Scandanavica, 362, 15-26. http://dx.doi.org/10.1111/j.1600-0447.1992.tb03310.x
- Pike, M.G., Mays, D.C., Macomber, D.W. and Lipsky, J.J. (2001) Metabolism of a disulfiram metabolite S-methyl N,N-diethyldithiocarbamate by flavinmonooxygenase in human renal microsomes. Drug Metabolism & Disposition, 29, 127-132.
- Larsen, V. (1948) The effects on experimental animals of antabuse (tetraethylthiuram disulfide) in combination with alcohol. Acta Pharmacologica Toxicology, 4, 321-332. http://dx.doi.org/10.1111/j.1600-0773.1948.tb03354.x
- Krampe, H. and Ehrenreich, H. (2010) Supervised disulfiram as adjunct to psychotherapy in alcoholism treatment. Current Pharmaceutical Design, 16, 2076-2090. http://dx.doi.org/10.2174/138161210791516431
- Fuller, R.K. and Gordis, E. (2004) Does disulfiram have a role in alcoholism treatment today? Addiction, 99, 21-24. http://dx.doi.org/10.1111/j.1360-0443.2004.00597.x
- Vaccari, A., Saba, P.L., Ruiu, S., Collu, M. and Devoto, P. (1996) Disulfiram and diethyldithiocarbamate intoxication affects the storage and release of striatal dopamine. Toxicology & Applied Pharmacology, 139, 102-108. http://dx.doi.org/10.1006/taap.1996.0147
- Muller, C.A. and Banas, R. (2011) Disulfiram: An anticraving substance? American Journal of Psychiatry, 168, 98. http://dx.doi.org/10.1176/appi.ajp.2010.10070943
- Johnson, B.A. (2010) Disulfiram. In: Stolerman, I.P., Ed., Encyclopedia of Psychopharmacology. Illustrated Edition, Heidelberg, Springer, 413.
- Brewer, C. (2005) Supervised disulfiram is more effective in alcoholics than naltrexone or acamprosate or even psychotherapy: How it works and why it matters. Adicciones, 17, 285-296.
- Barth, K.S. and Malcolm, R.J. (2010) Disulfiram: An old therapeutic with new applications. CNS Neurological Disorders & Drug Targets, 9, 5-12. http://dx.doi.org/10.2174/187152710790966678
- Krampe, H., Spies, C.D. and Ehrenreich, H. (2011) Superviseddisulfiram in the treatment of alcohol use disorder: A commentary. Alcoholism Clinical & Experimental Research, 35, 1732-1736. http://dx.doi.org/10.1111/j.1530-0277.2011.01519.x
- Garbutt, J.C. (2009) The state of pharmacotherapy for the treatment of alcohol dependence. Journal of Substance Abuse Treatment, 36, S15-S23.
- Soyka, M., Kranzler, H. and Berglund, M. (2008) World federation of societies of biological psychiatry (WFSBP) guidelines for biological treatment of substance use and related disorders, part 1: Alcoholism. World Journal of Biological Psychiatry, 9, 6-23. http://dx.doi.org/10.1080/15622970801896390
- Ulrichsen, J., Nielsen, M.K. and Ulrichsen, M. (2010) Disulfiram in severe alcoholism—An open controlled study. Nordic Journal of Psychiatry, 64, 356-362. http://dx.doi.org/10.3109/08039481003686180
- Fuller, R.K., Branchey, L., Brightwell, D.R. and The Veterans Study Group (1986) Disulfiram treatment of alcoholism: A Veterans Administration cooperative study. JAMA, 256, 1449-1455. http://dx.doi.org/10.1001/jama.1986.03380110055026
- Børup, C., Kaiser, A. and Jensen, E. (1992) Long-term Antabuse treatment: Tolerance and reasons for withdrawal. ActaPsychiatricaScandanavica, 86, 47-49.
- Liskow, B., Nickel, E.J., Tunley, N., Powell, B.J. and Penick, E.C. (1990) Alcoholics’ attitudes toward and experiences with disulfiram. American Journal of Drug and Alcohol Abuse, 16, 147-160. http://dx.doi.org/10.3109/00952999009001579
- Duckert, F. and Johnsen, J. (1987) Behavioral use of disulfiram in the treatment of problem drinking. International Journal of Addiction, 22, 445-454.
- O’Farrell, T.J. and Bayog, R.D. (1986) Antabuse contracts for married alcoholics and their spouses: A method to maintain antabuse ingestion and decrease conflict about drinking. Journal of Substance Abuse Treatment, 3, 1-8. http://dx.doi.org/10.1016/0740-5472(86)90002-4
- Mutschler, J., Dirican, G. and Funke, S. (2011) Experienced acetaldehyde reaction does not improve treatment response in outpatients treated with supervised disulfiram. Clinical Neuropharmacology, 34, 161-165. http://dx.doi.org/10.1097/WNF.0b013e3182216fd5
- Krampe, H., Stawicki, S., Wagner, T., Bartels, C., Aust, C., Ruther, E., Poser, W. and Ehrenreich, H. (2006) Follow-up of 180 alcoholic patients for up to 7 years after outpatient treatment: Impact of alcohol deterrants on outcome. Alcoholism Clinical and Experimental Research, 30, 86-95. http://dx.doi.org/10.1111/j.1530-0277.2006.00013.x
- Krampe, H., Stawicki, S., Hoehe, M.R. and Ehrenreich, H. (2007) Outpatient Long-term Intensive Therapy for Alcoholics (OLITA): A successful biopsychosocial approach to the treatment of alcoholism. Dialogues in Clinical Neuroscience, 9, 399-412.
- Kristenson, H. (1992) Long-term antabuse treatment of alcohol-dependent patients. Acta Psychiatrica Scandanavica, 369, 41-45. http://dx.doi.org/10.1111/j.1600-0447.1992.tb03314.x
- Mutschler, J., Dirican, G., Gutzeit, A. and Grosshans, M. (2011) Safety and efficacy of long-term disulfiram aftercare. Clinical Neuropharmacology, 34, 195-198. http://dx.doi.org/10.1097/WNF.0b013e3182285e2d
- Brewer, C. (1992) Controlled trials of antabuse in alcoholism: the importance of supervision and adequate dosage. Acta Psychiatrica Scandanavica, 369, 51-58. http://dx.doi.org/10.1111/j.1600-0447.1992.tb03316.x
- Kristenson, H. (1995) How to get the best out of antabuse. Alcohol & Alcoholism, 30, 775-783.
- Mueser, K.T., Noordsy, D.L., Fox. L. and Wolfe, R. (2003) Disulfiram treatment for alcoholism in severe mental illness. American Journal on Addiction, 12, 242-252.
- Milne, H.J. and Parke, T.R. (2007) Hypotension and ST depression as a result of disulfiram ethanol reaction. European Journal of Emergency Medicine, 14, 228-229. http://dx.doi.org/10.1097/MEJ.0b013e3280bef8a7
- Fletcher, K., Stone. E., Mohamad. M.W. and Chick, J. (2006) A breath test to assess compliance with disulfiram. Addiction, 101, 1705-1710. http://dx.doi.org/10.1111/j.1360-0443.2006.01602.x
- Regier, D.A., Farmer, M.E. and Rae, D.S. (1990) Comorbidity of mental disorders with alcohol and other drug abuse. Results from the Epidemiological Catchment Area (ECA) study. JAMA, 264, 2511-2518. http://dx.doi.org/10.1001/jama.1990.03450190043026
- Dixon, L. (1999) Dual diagnosis of substance abuse in schizophrenia: Prevalence and impact on outcomes. Schizophrenia Research, 35, S93-S100. http://dx.doi.org/10.1016/S0920-9964(98)00161-3
- Larson, E.W., Olincy, A., Rummans, T.A. and Morse, R.M. (1992) Disulfiram treatment of patients with both alcohol dependence and other psychiatric disorders: A review. Alcoholism Clinical and Experimental Research, 16, 125-130. http://dx.doi.org/10.1111/j.1530-0277.1992.tb00648.x
- Guild, J. and Epstein, N.B. (1951) Psychosis during the treatment of alcoholism with tetraethylthiuram disulfide. Quarterly Journal of Studies on Alcohol, 12, 360-365.
- Mueser, K.T. and Gingerich, S. (2013) Treating co-occuring psychosis and substance use disorders. Social Work & Public Health, 28, 424-439. http://dx.doi.org/10.1080/19371918.2013.774676
- Petrakis, I., Ralevski, E., Nich, C., Levinson, C., Caroll, K., Polling, J., Rounsaville, B. and VA VISN MIRECC Study Group (2007) Naltrexone and disulfiram in patients with alcohol dependence and current depression. Journal of Clinical Psychopharmacology, 27, 160-165. http://dx.doi.org/10.1097/jcp.0b13e3180337fcb
- Ralevski, E., Ball, S., Nich, C., Limoncelli, D. and Petrakis, I. (2007) The impact of personality disorders on alcohol-use outcomes in a pharmacotherapy trial for alcohol dependence and comorbid Axis I disorders. American Journal of Addictions, 16, 443-449. http://dx.doi.org/10.1080/10550490701643336
- Mutschler, J., Grosshans, M., Koopmann, A., Hermann, D., Diehl, A., Mann, K. and Kiefer, F. (2010) Supervised disulfiram in relapse prevention in alcohol-dependent patients suffering from comorbid borderline personality disorder—A case series. Alcohol and Alcoholism, 45, 146- 150. http://dx.doi.org/10.1093/alcalc/agq001
- Mutschler, J., Buhler, M., Grosshans, M., Diehl, A., Mann, K. and Kiefer, K. (2010) Disulfiram, an option for the treatment of pathological gambling? Alcohol and Alcoholism, 45, 214-216. http://dx.doi.org/10.1093/alcalc/agp093
- Mutschler, J., Dirican, G., Gutzeit, A. and Grosshans, M. (2011) Safety and efficacy of long term disulfiram after care. Clinical Neuropharmacology, 34, 195-198. http://dx.doi.org/10.1097/WNF.0b013e3182285e2d
- Niederhofer, H. and Staffen, W. (2003) Comparison of disulfiram and placebo in treatment of alcohol dependence of adolescents. Drug and Alcohol Review, 22, 295- 297. http://dx.doi.org/10.1080/0959523031000154436
- De Sousa, A. anddence. Journal of Substance Use, 13, 382-388. De Sousa, A. (2008) An open randomized trial comparing disulfiram and naltrexone in adolescents with alcohol depen http://dx.doi.org/10.1080/14659890802305861
- Verheul, R., Lehert, P., Geerlings, P.J., Koeter, M.W. and van den Brink, W. (2005) Predictors of acamprosate efficacy: Results from a pooled analysis of seven European trials including 1485 alcohol dependent patients. Psychopharmacology, 178, 167-173. http://dx.doi.org/10.1007/s00213-004-1991-7
- De Sousa, A. and De Sousa, A. (2004) A one-year pragmatic trial of naltrexone vsdisulfiram in the treatment of alcohol dependence. Alcohol& Alcoholism, 39, 528-531. http://dx.doi.org/10.1093/alcalc/agh104
- De Sousa, A. and De Sousa, A. (2005) An open randomized study comparing disulfiram and acamprosate in the treatment of alcohol dependence. Alcohol and Alcoholism, 40, 545-548. http://dx.doi.org/10.1093/alcalc/agh187
- De Sousa, A.A., De Sousa, J. and Kapoor, H. (2008) An open randomized trial comparing disulfiram and topiramate in the treatment of alcohol dependence. Journal of Substance Abuse Treatment, 34, 460-463. http://dx.doi.org/10.1016/j.jsat.2007.05.012
- Berglund, M. (2005) A better widged? Three lessons for improving addiction treatment from a meta-analytical study. Addiction, 100, 742-750. http://dx.doi.org/10.1111/j.1360-0443.2005.01106.x
- Brewer, C., Meyers, R.J. and Johnsen, J. (2000) Does disulfiram help to prevent relapse in alcohol abuse? CNS Drugs, 14, 329-341. http://dx.doi.org/10.2165/00023210-200014050-00001
- Besson, J., Aeby, F., Kasas, A., Lehert, P. and Potgeiter, A. (1998) Combined efficacy of acamprosate and disulfiram in the treatment of alcoholism: A controlled study. Alcoholism: Clinical and Experimental Research, 22, 573- 579. http://dx.doi.org/10.1111/j.1530-0277.1998.tb04295.x
- Laaksonen, E., Koski-Jannes, A., Salaspuro, M., Ahtinen, H. and Alho, H. (2008) A randomized, multicentre, openlabel, comparative trial of disulfiram, naltrexone and acamprosate in the treatment of alcohol dependence. Alcohol and Alcoholism, 43, 53-61. http://dx.doi.org/10.1093/alcalc/agm136
- Pettinati, H.M., Kampman, K.M., Lynch, K.G., Xie, H., Dackis, C., Rabinowitz, A.S. and O’Brien, C.P. (2007) A double blind, placebo-controlled trial that combines disulfiram and naltrexone for treating co-occurring cocaine and alcohol dependence. Addiction Behaviour, 33, 651-667.
- Maremmani, A.G., Pani, P.P., Rovai, L., Pacini, M., Dell Osso, L. and Maremanni, L. (2011) Long-term Ɣ-hydroxybutyric acid (GHB) and disulfiram combination therapy in GHB treatment-resistant chronic alcoholics. International Journal of Environmental Research & Public Health, 8, 2816-2827. http://dx.doi.org/10.3390/ijerph8072816
- Peachey, J.E. (1981) A review of the clinical use of disulfiram and calcium carbimide in alcoholism treatment. Journal of Clinical Psychopharmacology, 1, 368-375. http://dx.doi.org/10.1097/00004714-198111000-00004
- Sande, M., Thompson, D. and Monte, A.A. (2012) Fomepizole for severe disulfiram-ethanol reactions. American Journal of Emergency Medicine, 30, 262.e3-5.
- Heelon, M.W. and White, M. (1998) Disulfiram-cotrimoxazole reaction. Pharmacotherapy, 18, 869-870.
- Ho, M.P., Yo, C.H., Liu, C.M. Chen, C.L. and Lee, C.C. (2007) Refractive hypotension in a patient with disulfiram-ethanol reaction. American Journal of Medical Science, 333, 53-55. http://dx.doi.org/10.1097/00000441-200701000-00007
- Savas, M.C. and Gullu, J.H. (1997) The disulfiram ethanol reaction: The significance of supervision. Annals of Pharmacotherapy, 31, 374-375.
- Nucifora, G., Cassin, M., Brun, F. and Nicolosi, G.L. (2004) Anterior myocardial infarction in a chronic alcoholic man on disulfiram therapy: A case report. Italian Heart Journal, 5, 900-904.
- Prancheva, M.G., Krasteva, S.A. and Tufkova, S.G. (2010) Severe hypotension and ischemic stroke after disulfiramethanol reaction. Folia Medica, 52, 70-73. http://dx.doi.org/10.2478/v10153-010-0009-y
- Ehrlich, R.I., Woolf, D.C. and Kibel, D.A. (2012) Disulfiram reaction in an artist exposed to solvents. Occupational Medicine, 62, 64-66. http://dx.doi.org/10.1093/occmed/kqr172
- Palatty, P.L. and Saldanha, E. (2011) Status of disulfiram in present day alcoholic deaddiction therapy. Indian Journal of Psychiatry, 53, 25-29. http://dx.doi.org/10.4103/0019-5545.75557
- Mutschler, J., Grosshans, M., Koopmann, A., Mann, K., Kiefer, F. and Hermann, D. (2010) Urinary ethylglucuronide assessment in patients treated with disulfiram: A tool to improve verification of abstention and safety. Clinical Neuropharmacology, 33, 285-287. http://dx.doi.org/10.1097/WNF.0 b013e3181fc9362
- Lindros, K.O., Stowell, A., Pikkarainen, P. and Salaspuro, M. (1981) The disulfiram (Antabuse)-Alcohol reaction in male alcoholics: Its efficient management by 4-methylpyrazole. Alcoholism: Clinical & Experimental Research, 5, 528- 530. http://dx.doi.org/10.1111/j.1530-0277.1981.tb05354.x
- Megarbane, B., Borron, S.W. and Baud, F.J. (2005) Current recommendations for treatment of severe toxic alcohol poisonings. Intensive Care Medicine, 31, 189-195. http://dx.doi.org/10.1007/s00134-004-2521-0
- Bakke, S.A. and Laursen, S.B. (2011) Treatment with disulfiram can be dangerous. Ugeskrift for Laeger, 173, 3192-3193.
- Verbon, H. and de Jong, C.A. (2002) Psychosis during and after disulfiram use. Nederlands Tijdschrift Geneeskde, 146, 571-573.
- Murthy, K.K. (1997) Psychosis during disulfiram therapy for alcoholism. Journal of the Indian Medical Association, 95, 80-81.
- Quail, M. and Karelse, R.H. (1980) Disulfiram psychosis. A case report. South African Medical Journal, 57, 551- 552.
- Chick, J. (1999) Safety issues concerning the use of disulfiram in treating alcohol dependence. Drug Safety, 20, 427-435. http://dx.doi.org/10.2165/00002018-199920050-00003
- Wright IIII, C., Vafier, J.A. and Lake, C.R. (1988) Disulfiram-induced fulminating hepatitis: Guidelines for liverpanel monitoring. Journal of Clinical Psychiatry, 49, 430-434.
- Enghusen, P.H., Loft, S. and Anderson, J.R. (1992) Disulfiram therapy-adverse drug reactions and interactions. Acta Psychiatrica Scandanavica, 86, 59-66.
- Trelohan, A. and Milea, D. (2011) Reversible optic neuropathy related to disulfiram. Journal Francais Ophthalmology, 34, 382.
- Lemoyne, S., Raemaekers, J. and Daems, J. (2009) Delayed and prolonged coma after acute disulfiram overdose. Acta Neurologica Belgica, 109, 231-234.
- Liddon, S.C. and Satran, R. (1967) Disulfiram (Antabuse) psychosis. American Journal of Psychiatry, 123, 1284- 1289.
- Saddichha, S., Phutane, V. and Thirthalli, J. (2011) De novo dose-dependent catatonia due to Disulfiram therapy: A report and review. Journal of Clinical Pharmacology, 51, 285-286. http://dx.doi.org/10.1177/0091270010365556
- Stahl, S.M. (2008) Psychosis and schizophrenia. In: Stahl, S.M., Ed., Stahl’s Essential Psychopharmacology: Neuroscientific Basis and Practical Applications, 3rd Edition, Cambridge University Press, Cambridge, 294-297.
- Northoff, G. (2004) Neuroimaging and neurophysiology. In: Caroff, S.N., Mann, S.C., Francis, A. and Fricchione, G.L., Eds., Catatonia: From psychopathology to neurobiology, American Psychiatric Publishing, Washington, DC, 77-91.
- Jagadisha, T. and Prabhat, C.K. (2009) The implications of medication development in the treatment of substance use disorders in developing countries. Current Opinion in Psychiatry, 22, 274-280. http://dx.doi.org/10.1097/YCO.0b013e32832a1dc0
- Diehl, A., Ulmer, L., Mutschler, J., Herre, H., Krumm, B., Croissant, B., Mann, K. and Kiefer, F. (2010) Why is disulfiram superior to acamprosate in the routine clinical setting? A retrospective long-term study in 353 alcoholdependent patients. Alcohol & Alcoholism, 45, 271-277. http://dx.doi.org/10.1093/alcalc/agq017
- Grover, S., Basu, D. and Bhateja, G. (2007) Pharmacoprophylaxis of alcohol dependence: Review and update Part II: Efficacy. Indian Journal of Psychiatry, 49, 26-33. http://dx.doi.org/10.4103/0019-5545.31515
- Sharma, P., Murthy, P. and Bharath, M.M. (2011) Disulfiram in a “traditional” medicine sold to patients with alcohol dependence in India. Addiction, 106, 1870-1871. http://dx.doi.org/10.1111/j.1360-0443.2011.03506.x
- Johnsen, J. and Morland, J. (1991) Disulfiram implant: A double-blind placebo controlled follow-up on treatment outcome. Alcoholism: Clinical and Experimental Research, 15, 532-536. http://dx.doi.org/10.1111/j.1530-0277.1991.tb00555.x
- O’Farrell, T.J., Allen, J.P. and Litten, R.Z. (1995) Disulfiram (antabuse) contracts in treatment of alcoholism. NIDA Research Monograph, 150, 65-91.
- Johnsen, J., Stowell, A. and Stensrud, T. (1990) A doubleblind placebo controlled study of healthy volunteers given a subcutaneous disulfiram implantation. Pharmacology & Toxicology, 66, 227-230. http://dx.doi.org/10.1111/j.1600-0773.1990.tb00738.x
- Anton, R.F., Oroszi, G., O’Malley, S.,Couper, D., Swift, R., Pettinati, H. and Goldman, D. (2008) An evaluation of μ-Opioid Receptor (OPRM1) as a predictor of naltrexone response in the treatment of alcohol dependence. JAMA Psychiatry, 65, 135-144. http://dx.doi.org/10.1001/archpsyc.65.2.135
- Oslin, D.W., Berrettini, W., Kranzler, H.R., Pettinati, H., Gerlenter, J., Volpicelli, J.R. and O’Brien, C.P. (2003) A functional polymorphism of the mu-opioid receptor gene is associated with naltrexone response in alcohol-dependent patients. Neuropsychopharmacology, 28, 1546-1552. http://dx.doi.org/10.1038/sj.npp.1300219
- Kiefer, F., Witt, S.H., Frank, J., et al. (2011) Involvement of the atrial natriuretic peptide transcription factor GATA4 in alcohol dependence, relapse risk and treatment response to acamprosate. Pharmacogenomics Journal, 11, 368-374. http://dx.doi.org/10.1038/tpj.2010.51
NOTES
*Corresponding author.

