Paper Menu >>
Journal Menu >>
 Open Journal of Gastroenterology, 2011, 1, 1-6 OJGas doi: 10.4236/ojgas.2011.11001 Published Online August 2011 (http://www.SciRP.org/journal/ojgas/). Published Online August 2011 in SciRes. http://www.scirp.org/journal/OJGas Emergency gastrectomy for gastric necrosis 5 years after laparoscopic adjustable gastric banding (LAGB) Rani Kanthan1, Jenna-Lynn Senger1, Selliah Chandra Kanthan2 1Department of Pathology and Laboratory Medicine, University of Saskatchewan, Saskatoon, Canada; 2Department of Surger y, University of Saskatchewan, Saskatoon, Canada. Email: rani.kanthan@saskatoonhealthregion.ca Received 27 June 2011; revised 3 August 2011; accepted 10 August 2011. ABSTRACT Laparoscopic adjustable gastric banding (LAGB) is currently one of the most common bariatric surgical procedures performed worldwide for the treatment of morbid obesity. Among bariatric surgeries, the percentage of LAGB has increased from 24.4% to 42.3%. In North America alone, the number of LAGB procedures has increased exponentially by 944.2% (from 9,270 to 96, 800 cases). Though early and late complications following LAGB are well-understood, data regarding long term complications remains in- definite due to the limited follow-up periods. We re- port a case of acute gastric necrosis associated with band slippage presenting as a surgical emergency five years after LAGB. Gastric necrosis represents a rare, but life threatening complication of gastric banding that has been reported in the early post-operative pe- riod. To the best of our knowledge, this case report is the longest time-interval reported between LAGB and the diagnosis of this very rare complication. Accurate diagnosis is often delayed due to its rarity and non- specific clinical presentation. Increased awareness of this delayed complication should facilitate early recog- nition as it often requires urgent surgical interven- tion to prevent fatal outcomes. Keywords: LAGB; Gastric Necrosis; Band Slippage 1. INTRODUCTION Since its introduction in 1993, worldwide, more than 100,000 laparoscopic adjustable gastric banding (LAGB) procedures have been performed as the surgical treat- ment of choice for morbid obesity [1]. Features includ- ing the a) ease of the procedure, b) minimally invasive nature, c) the potential reversibility, and d) the ability to calibrate the stoma contribute to the continued growing popularity of this technique [2]. Early or late complica- tions related to LAGB include band slippage, pouch dilatation, band erosion, gastric perforation, injection- port malfunctio n and rarely gastric ne crosis [3]. Though, specific late complications associated with this proce- dure are recognized in 10% - 20% of cases [1] the data remains incomplete due to limited follow-up periods. Gastric necrosis is a very rare complication that is po- tentially life threatening and constitutes an absolute sur- gical emergency with dire consequences if unrecognized. Including this report, a total of eighteen cases have been reported in the literature (Table 1) [1-16]. We report the case of a patient who, five years after LAGB, presented with vague abdominal symptoms to the Emergency Room (ER) on two occasions prior to being accurately diagnosed as gastric pouch necrosis which necessitated urgent surgical intervention. 2. CASE REPORT A 46-year-old woman presented to our emergency de- partment with progressive abdominal pain, abdominal distension, nausea and vomiting. Abdominal examina- tion showed mild tenderness in the epigastrium on pal- pation with no other positive signs. She had been to the ER on two previous occasions in the past few weeks for similar complaints. Relevant past history included a laparoscopic adjustable gastric band placement in Mex- ico five years ago for weight reduction (225 lbs). Due to the repeated visits and the persistence of symptoms over 24 hours a surgical consult with radiological investiga- tions were undertaken. Plain films of the abdomen confirmed the presence of a LAGB attached to its external port as seen in Figure 1. A contrast enhanced abdominal CT scan revealed mark- edly dilated esophagus (Figure 2a) and stomach with a distal migration of the LAG band (Figure 2b) at the py- lorus (Figure 2c). An abnormal enhancement pattern with early ‘target sign’ of alternating layers of high and low attenuation suggestive of submucosal edema and/or hemorrhage in keeping with mucosal ischemia is de- 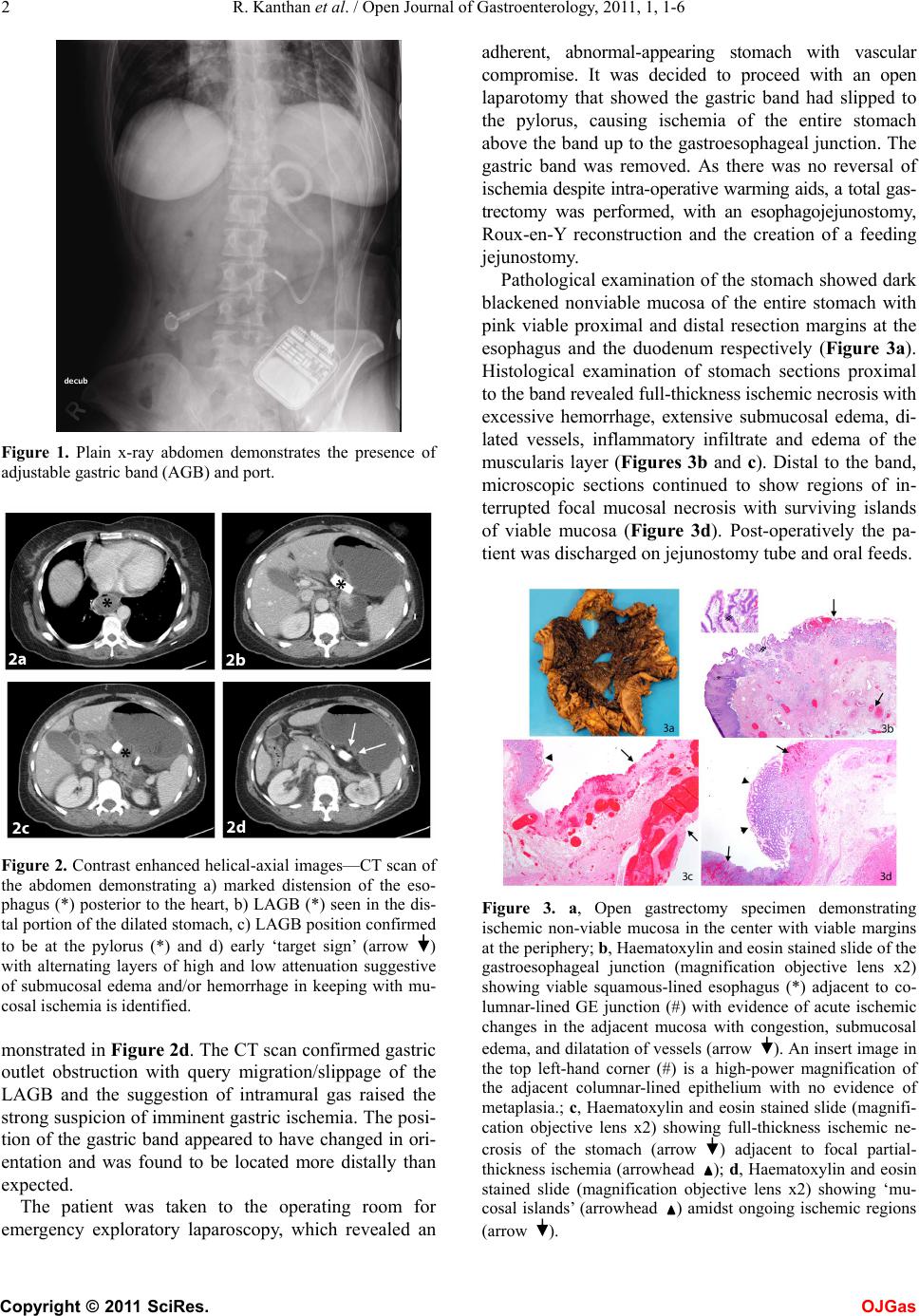 R. Kanthan et al. / Open Journal of Gastroenterology, 2011, 1, 1-6 Copyright © 2011 SciRes. OJGas 2 Figure 1. Plain x-ray abdomen demonstrates the presence of adjustable gastric band (AGB) and port. Figure 2. Contrast enhanced helical-axial images—CT scan of the abdomen demonstrating a) marked distension of the eso- phagus (*) posterior to the heart, b) LAGB (*) seen in the dis- tal portion of the dilated stomach, c) LAGB position confirmed to be at the pylorus (*) and d) early ‘target sign’ (arrow ) with alternating layers of high and low attenuation suggestive of submucosal edema and/or hemorrhage in keeping with mu- cosal ischemia is identified. monstrated in Figure 2d. The CT scan confirmed gastric outlet obstruction with query migration/slippage of the LAGB and the suggestion of intramural gas raised the strong suspicion of imminent gastric ischemia. The posi- tion of the gastric band appeared to have changed in ori- entation and was found to be located more distally than expected. The patient was taken to the operating room for emergency exploratory laparoscopy, which revealed an adherent, abnormal-appearing stomach with vascular compromise. It was decided to proceed with an open laparotomy that showed the gastric band had slipped to the pylorus, causing ischemia of the entire stomach above the band up to the gastroesophageal junction. The gastric band was removed. As there was no reversal of is chemia despite intra-operative warming aids, a total gas- trectomy was performed, with an esophagojejunostomy, Roux-en-Y reconstruction and the creation of a feeding jejunostomy. Pathological examination of the stomach showed dark blackened nonviable mucosa of the entire stomach with pink viable proximal and distal resection margins at the esophagus and the duodenum respectively (Figure 3a). Histological examination of stomach sections proximal to the band revealed full-thickness ische mic necrosis with excessive hemorrhage, extensive submucosal edema, di- lated vessels, inflammatory infiltrate and edema of the muscularis layer (Figures 3b and c). Distal to the band, microscopic sections continued to show regions of in- terrupted focal mucosal necrosis with surviving islands of viable mucosa (Figure 3d). Post-operatively the pa- tient was discharged on jejunostomy tube an d oral feed s. Figure 3. a, Open gastrectomy specimen demonstrating ischemic non-viable mucosa in the center with viable margins at the periphery; b, Haematoxylin and eosin stained slide of the gastroesophageal junction (magnification objective lens x2) showing viable squamous-lined esophagus (*) adjacent to co- lumnar-lined GE junction (#) with evidence of acute ischemic changes in the adjacent mucosa with congestion, submucosal edema, and dilatation of vessels (arrow ). An insert image in the top left-hand corner (#) is a high-power magnification of the adjacent columnar-lined epithelium with no evidence of metaplasia.; c, Haematoxylin and eosin stained slide (magnifi- cation objective lens x2) showing full-thickness ischemic ne- crosis of the stomach (arrow ) adjacent to focal partial- thickness ischemia (arrowhead ); d, Haematoxylin and eosin stained slide (magnification objective lens x2) showing ‘mu- cosal islands’ (arrowhead ) amidst ongoing ischemic regions (arrow ).  R. Kanthan et al. / Open Journal of Gastroenterology, 2011, 1, 1-6 Copyright © 2011 SciRes. OJGas 3 One week later she returned to the ER complaining of abdominal pain with cramping, depression and a general sense of being overwhelmed. She was admitted with the suspicion of a partial small bowel obstruction; however, abdominal CT scan, HIDA scan and gastroscopy all failed to demonstrate the same. She was started on TPN to give bowel rest, and upon re-starting oral feeds did well. She was followed up in the clinic 6 weeks post- operatively and is progressing well. Her current weight is 121 lbs. 3. REVIEW AND DISCUSSION Using PubMed, a literature search was performed using the text words ‘laparoscopic gastric band*’ and ‘gastric necrosis’. Overall, 27 results including case reports and case series were retrieved. All papers relevant to this study were reviewed and of these, 22 papers have been included in this study. The findings of the cases with gastric necrosis following LAGB includ ing details o f : a) reference #, b) author, c) study type, d) number of cases/patients reported, e) age and sex, f) time interval between LAGB and gastric necrosis, g) presenting symptoms, h) management, i) outcome and j) cause of the gastric necrosis are summarized in Table 1 [1-16]. Since its introduction in 1993 and approval by the Foods and Drug Administration in 2001, Laparoscopic Adjustable Gastric Banding (LAGB) has gained popu- larity for the treatment of morbid obesity, making it one of the most common surgeries in Europe, Australia, Latin America, and the USA [4,5]. The ease of this sur- gical technique coupled with the minimal invasiveness and its reversibility has contributed to its increasing popularity over the past five years [2]. Worldwide, while the percentage of LAGB carried out among bariatric surgeries has increased from 24.4% in 2003 to 42.3%; in North America alone in 2008 the number of LAGB pro- cedures has increased exponentially by 944.2% (from 9,270 to 96, 800 cases) [17]. It is estimated that band related complications arise in 12.2% of LAGB cases [5,18]. These can occur as early or late postoperative events. Band related complications include band migration/erosion, band dislocation/ leak- age, band slippage/prolapse and port related leaks/rup- ture [19]. The most common complication is band slip- page or gastric prolapse occurring in 4.5% - 5% of pa- tients [5,18]. Additional complications may include bleeding, gastric perforation, pouch or esophageal dila- tation, stoma obstruction, band erosion and gastric ne- crosis [5,8]. Rarely, small bowel obstruction secondary to intragastric erosion and migration of a gastric band has also been reported [20]. Clinical presenting symp- toms of these late complications usually include gradual onset of reflux, dysphagia, and vomiting [2]; however, acute retrosternal chest pain has also been reported as the primary presenting complaint [6]. In many cases, band deflation relieves symptoms [1]. When this is in- sufficient, laparoscopic treatment, including band repo- sitioning,replacement, or removal is an adequate remedy for many of these complications. Though specific late complications associated with this procedure are recog- nized in 10% - 20% of cases [1] the data remains indefi- nite due to limited follow-up period s. Gastric necrosis is a very rare complication that is potentially life threaten- ing and constitu tes an absolute surgical emergency. As seen in Ta b l e 1 , several mechanisms as the cause of gastric necrosis were identified by individual authors in their respective reports. In agreement with the litera- ture, gastric prolapse was the most commonly reported cause of necrosis [21]. Additional mechanisms reported as the cause of necrosis include band slippage [22], fun- dal herniation [1], pouch dilatation [16], late prolapse [10], Type II paraesophageal hernia with strangulation [6] paragastric Richter’s hernia [22] and organo-axial vol- vulus [11]. Though many complications arising from LAGB occur during the immediate or early post-opera- tive phase of healing, gastric necrosis appears to be a late delayed complication, with the majority of cases occurring 1-3 years after LAGB (Ta ble 1). If the necro- sis is detected early, conservative treatment with com- plete deflation of the adjustable band with close fol- low-up [7], or limited gastrectomy procedures such as sleeve gastrectomy [8] have been reported as effective treatment. In the event of complete gastric necrosis, a total gastrectomy is often required. Therefore, early de- tection of gastric necrosis is crucial to satisfactory pa- tient management. The reported data of follow-up peri- ods following LAGB are generally less than five years [4]. As gastric necrosis can occur as a late delayed com- plication (after five years) as seen in this reported case, a more vigilant long term follow-up of LAGB cases is recommended. The stomach is a highly vascular organ with an exten- sive arcade of arterial anastomotic network which there- fore rarely succumbs to ischemic necrosis [3]. In this context, for ischemia to occur, several predisposing risk factors have been identified to contribute to the onset of gastric devitalization. These i nclude: A) Gastric prolapse occurs where a part of the stom- ach slips through the band. This can decrease the blood flow within the gastric pouch. This band slippage may occur immediately after surgery or many mon ths later, in either an anterior or posterior direction which causes an obstruction between the upper and lower stomach [21]. B) Pouch dilatation increases the pressure on the gastric wall, impairing the vasculature [8]. This can be caused by patients who chronically overeat, and will  R. Kanthan et al. / Open Journal of Gastroenterology, 2011, 1, 1-6 Copyright © 2011 SciRes. OJGas 4 Table 1. Composite literature review of gastric necrosis following LAGB (pubmed listed 2001-2011) in descending chronological order. a) Ref # b) Author c) Study Type d) #Cases #Pa- tients e) Age (Sex) f) Time interval since LAGB g) Presenting symptoms of this complication h) Management i) Outcome j) Cause of Necrosis Kanthan 2011 Case Report 46 (F) 5 yearsAbd pain, Distension, Nausea, Vomiting Emergency total gastrectomy Recovery Band slippage 4 Polat, 2010 Case Series 1/232 NR 1 year NR Laparoscopic band removal NR NR 5 Fragkouli , 2010 Short Report 37 (M) 8 days Vomiting, Abd pain, Fever, Hypotension, Tachycardia, Leukocytosis Surgical explora- tion Death NR 6 Bernante, 2008 Case Report 42 (F) 16 mo Chest pain, Vomiting, Tachycardia, Tachypnea, Leukocytosis Laparoscopic band removal, sleeve gastrec- tomy Recovery Paraesophageal hernia 2 Iannelli, 2005 Case Report 45 (F) 2 yearsAbd pain, Vomiting, Fever, Tachycardia, LeukocytosisUpper polar gastrectomy Recovery Gastric prolapse 7 Foletto, 2005 Case Report 59 (F) 2 yearsVomiting, Dysphasgia, Adb pain, Leuko cytosis Laparoscopic band removal and drainage Recovery Gastric prolapse 8 Lunca, 2005 Case Report 41 (F) 3 yearsAbd p ain, Vomiting, Dysphagia Sleeve gastrectomy Recovery Gastric prolapse 45 (F) 3 yearsChest pain, Food intolerance Laparoscopic band deflation Recovery Pouch dilata- tion, gastric strangulation 1 Landen, 2005 2 Case Reports 55 (F) 4 months Food intolerance, Abd painTotal gastrectomy and splenectomy Recovery Herniation of fundus, cardiac ulcer 3 Yitzhak, 2005 Case Report 43 (F) 3 yearsAbd pain, Food intolerance, Tachycardia Endpouch retriever with fundectomy Recovery Band slippage, fundus stran- gulation 9 Irtan, 2004 Obser- vation 45 (F) 2 yearsAbd p ain, Vomiting, Leukocytosis, Hyperthermia, Aphagia Total gastrectomy Recovery Band slippage 10 Yoffe, 2004 Case Report 20 (F) 3 yearsVomiting, Abd pain, FeverLaparoscopic band removal and drainage Recovery Gastric prolapsed, pouch dilatation 11 Bortul, 2004 Case Report 19 (F) 17 months Abd pain, Vomiting, Leukocytosis Total gastrectomy & splenectomy Recovery Organo-axial volvulus 12 Dargent, 2004 Case Series 1/1 180 NR NR NR NR Death NR 13 Zinzin- dohoué, 2003 Case Series 1/500 NR NR NR Gastrectomy NR NR NR 20 months NR Proximal gastrectomy Death 14 Angri- sani, 2003 Case Series 2/1 863 NR NR NR Gastrectomy and esophagojeju- nostomy Recovery NR 15 Cheval- lier, 2002 Case Series 1/400 45 (F) 13 months Abd tenderness, PeritonitisTotal gastrectomy Recovery NR 16 Kir- chmayr, 2001 Case Report 24 (F) 20 months Dysphagia, Vomiting, Abd Pain, Vomiting, Leukocytosis Resection of greater curve Recovery Pouch dilata- tion, gastric perforation, herniation often present with vomiting caused by stoma obstruction and gastroesophageal junction motility disorders [16,21]. C) Band slippage/migration/erosion may result in the exertion of pressure against the fundus, limiting blood flow to the gastric wall [6,19]. The risk of a slipped band increases dramatically in patients who self-vomit after meals and in those who ingest large amounts of food [7]. D) Despite the rich arterial anastomoses in the stom- ach that can prevent necrosis, pre-existing co-morbid- ities found among obese people may fu rther increase the risk of gastric necrosis [5].  R. Kanthan et al. / Open Journal of Gastroenterology, 2011, 1, 1-6 Copyright © 2011 SciRes. OJGas 5 E) Finally, over-inflation of the gastric band can cause the same increased pressure, limiting blood flow. No more than 0.5cc should be injected per visit and checked monthly [10]. Due to its rarity, the recognition and accurate diagno- sis of gastric n ecrosis is often delayed; therefore, its pres- entation often constitutes a medical emergency necessi- tating urgent surgical intervention. As illustrated by our index case, gastric necrosis can present as a delayed complication five years after LAGB. Though delayed gastric necrosis has been reported earlier as listed in Ta- ble 1, this case has the longest post procedural time in- terval (5 years). Failure to recognize this urgent clinical situation can have dire consequences which, if untreated, will result in death of the patient. [5]. While conserva- tive treatment has been described in some cases of focal limited early necrosis, most cases require surgical inter- vention with procedures ranging from minimally inva- sive resection [3,16] including emergency sleeve gas- trectomy [6] to emergency total gastrectomy [1,9,14, 15]. In conclusion, as obesity rates rise to epidemic pro- portions, it is likely that LAGB will continu e to escalate in popularity as the preferred method of surgical treat- ment. Awareness and recognition of complications that may arise immediately or years following LAGB is im- portant data to be collected for continued best practice care of these patients. Delayed occurrence of gastric necrosis represents a rare but life threatening complica- tion of gastric banding. Accurate diagnosis is often de- layed due to its rarity and nonspecific clinical signs at presentation. Increased awareness of this delayed com- plication should facilitate early recognition as it will often require urgent surgical intervention to avert dire consequences. REFERENCES [1] Landen, S., Majerus, B. and Delugeau, V. (2005) Com- plications of gastric banding presenting to the ED. American Journal of Emergency Medicine, 23, 368-370. doi:10.1016/j.ajem.2005.02.016 [2] Iannelli, A., Facchiano, E., Sejor, E., et al. (2005) Gastric necrosis: A rare complication of gastric banding. Obesity Surgery, 15, 1211-1214. doi:10.1381/0960892055002194 [3] Yitzhak, A., Avinoach, E. and Mizrahi, S. (2005) A minimally invasive solution for necrotic fundus follow- ing slipped adjustable gastric band. Obesity Surgery, 15, 439-441. doi:10.1381/0960892053576631 [4] Polat, F., Poyck, P.P.C., Dickhoff, C., et al. (2010) Out- come of 232 morbidly obese patients treated with lapa- roscopic adjustable gastric banding between 1995-2003. Digestive Surgery, 27, 397-402. doi:10.1159/000318778 [5] Fragkouli, K., Mitselou, A. and Vougiouklakis, T. (2010) Death-related gastric necrosis after laparoscopic adjust- able gastric banding in the early post-operative period. Diagnostic Pathology, 5, 68. doi:10.1186/1746-1596-5-68 [6] Bernante, P., Breda, C., Zangrandi, F., et al. (2008) Emer- gency sleeve gastrectomy as a rescue treatment for acute gastric necrosis due to type II paraesophageal hernia in an obese woman with gastric banding. Obesity Surgery, 18, 737-741. doi:10.1007/s11695-007-9374-x [7] Foletto, M., De Marchi, F., Bernante, P., et al. (2005) Late gastric pouch necrosis after Lap-Band treated by an individualized conservative approach. Obesity Surgery, 15, 1487-1490. doi:10.1381/096089205774859272 [8] Lunca, S., Vix, M., Rikkers, A., et al. (2005) Late gastric prolapse with pouch necrosis after laparoscopic adjust- able gastric banding. Obesity Surgery, 15, 571-575. doi:10.1381/0960892053723420 [9] Irtan, S., Forestier, D., Scesa, J.L., et al. (2004) Nécrose gastrique après gastroplastie. La Presse Médicale, 33, 718-720. doi:10.1016/S0755-4982(04)98728-3 [10] Yoffe, B., Sapojnikov, S. and Goldblum, C. (2004) Gas- tric wall necrosis following late prolapse after laparo- scopic banding. Obesity Surgery, 14, 142-144. doi:10.1381/096089204772787473 [11] Bortul, M., Scaramucci, M., Tonello, C., et al. (2004) Gastric wall necrosis from organo-axial volvulus as a late complication of laparoscopic gastric banding. Obesity Surgery, 14, 285-287. doi:10.1381/096089204322857726 [12] Dargent, J. (2004) Surgical treatment of morbid obesity by adjustable gastric band: the case for a conservative strategy in the case of failure—a 9-year series. Obesity Surgery, 14, 986-990. doi:10.1381/0960892041719545 [13] Zinzindohoué, F., Chevallier, J.M., Douard, R., et al. (2003) Laparoscopic gastric banding: a minimally inva- sive surgical treatment for morbid obesity: Prospective study of 500 consecutive patients. Annals of Surgery, 237, 1-9. doi:10.1097/00000658-200301000-00001 [14] Angrisani, L., Furbetta, F., Doldi, S.B., et al. (2003) Lap Band adjustable gastric banding system: The Italian ex- perience with 1863 patients operated on 6 years. Surgical Endoscopy, 17, 409-412. [15] Chevallier, J.M., Zinzindohoué, F., Elian, N., et al. (2002) Adjustable gastric banding in a public university hospital: prospective analysis of 400 patients. Obesity Surgery, 12, 93-99. doi:10.1381/096089202321144658 [16] Kirchmayr, W., Ammann, K., Aigner, F., et al. (2001) Pouch dilatation after gastric banding causing gastric ne- crosis. Obesity Surgery, 11, 770-772. doi:10.1381/09608920160558768 [17] Buchwald, H. and Oien, D.M. (2009) Metabolic/bariatric surgery worldwide 2008. Obesity Surgery, 19, 1605- 1611. doi:10.1007/s11695-009-0014-5 [18] Carelli, A.M., Youn, H.A., Kurian, M.S. et al. (2010) Safety of the laparoscopic adjustable gastric band: 7-year data from a U.S. center of excellence. Surgical Endo- scopy, 24, 1819-1823. doi:10.1007/s00464-009-0858-8 [19] Forsell, P., Hallerbäck, B., Glise, H. et al. (1999) Com- plications following Swedish Adjustable Gastric Banding: a long-term follow-up. Obesity Surgery, 9, 11-16. doi:10.1381/096089299765553647 [20] Egbeare, D.M., Myers, A.F. and Lawrance, R.J. (2008) Small bowel obstruction secondary to intragastric erosion and migration of a gastric band. Journal of Gastrointes- tinal Surgery, 12, 983-984.  R. Kanthan et al. / Open Journal of Gastroenterology, 2011, 1, 1-6 Copyright © 2011 SciRes. OJGas 6 doi:10.1007/s11605-007-0304-y [21] Spivak H and Favretti F. (2002) Avoiding postoperative complications with the LAP-BAND system. The Ameri- can Journal of Surgery, 184, 31S-37S. doi:10.1016/S0002-9610(02)01177-7 [22] Srikanth, M.S., Oh, K.H., Keskey, T., et al. (2005) Criti- cal extreme anterior slippage (paragastric Richter’s her- nia) of the stomach after laparoscopic adjustable gastric banding: Early recognition and prevention of gastric strangulation. Obesity Surgery, 15, 207-215. doi:10.1381/0960892053268471 |

