Open Access Library Journal
Vol.02 No.11(2015), Article ID:68796,10 pages
10.4236/oalib.1102088
Electromicrobiology: An Emerging Reality―A Review
Clifford Nkemnaso Obi*, Grace Chigozirim Asogwa
Department of Microbiology, College of Natural Sciences, Michael Okpara University of Agriculture, Umudike, Nigeria

Copyright © 2015 by authors and OALib.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 19 October 2015; accepted 4 November 2015; published 9 November 2015

ABSTRACT
Electromicrobiology is the study of the interactions between the novel electrical properties of microorganisms and electronic devices. A diversity of microorganisms such as Geobacter and Shewanella species is capable of interacting electrically with the environment. Many recent advances in Electromicrobiology stem from studying Microbial Fuel Cells (MFCs) which are a device designed for the harvesting of electric current from organic compounds. Three types of Microbial Fuel Cells are known which are heterotrophic microbial fuel cells, photosynthetic microbial fuel cells (bio-solar cells) designed to harness the most abundant and promising energy source (solar irradiation) of earth and the hybrid microbial fuel cell. Electric microorganisms especially Sporomusa ovata can use electron derived from electrodes to reduce carbondioxide to multicarbon extracellular organic compounds in a process known as Microbial Electrosynthesis. The mechanism of electron transfer to electrodes by electric microbes is either by the use of electron shuttling molecules, redox-active proteins or via conductive pili. Conductive microorganisms and/or their nanowires have a number of potential practical applications but additional basic research will be necessary for rational applications. This review looks at the Microbial Fuel Cells, the associated mechanisms and applications.
Keywords:
Electricity, Electrons, Microbes, Microbial Fuel Cells
Subject Areas: Microbiology

1. Introduction
Electromicrobiology is a rapidly emerging field of study that investigates microbial electron exchange with electrodes, minerals, and other organisms, as well as novel properties of microorganisms that are of interest for developing electronic devices. A wide diversity of microorganisms has the ability to exchange electrons with electrodes, which contribute to a broad range of practical applications [1] . Furthermore, some microorganisms have surprising electronic characteristics. For example, biofilms of Geobacter species have conductivities that rival those of conductive polymers can function as supercapacitors or transistors [2] . The pili of these organisms is capable of long-range (>1 cm) electron transport via a metallic-like conductivity, not previously observed in a biological material [3] .
Many of the recent advances in Electromicrobiology have arisen from the study of microbial fuel cells, devices initially designed for harvesting electricity from organic matter [4] . Difficulties in scaling up microbial fuel cells for extracting energy on an industrial scale have greatly limited their short-term practical use for current production to niche applications, such as harvesting organic matter from aquatic sediments to power electronic monitoring devices [5] . Some of the most attractive practical applications of the microbial fuel cell concept are those in which there is no need to harvest current. These include monitoring rates of microbial metabolism in subsurface environments and providing electrodes as an electron acceptor to stimulate the degradation of organic contaminants in sediments [6] . Another promising application of the ability of microorganisms to transfer electrons to electrodes is the potential for balancing electron flow within microbial cells by removing excess electrons in order to promote the synthesis of desired products [7] .
In a similar manner, new potential applications for electron flow in the reverse direction, i.e. from electrodes to cells, are rapidly emerging. Feeding electrons to microorganisms living on electrode surfaces has significant potential to contribute to bioremediation of a diversity of contaminants, including radioactive and toxic metals, chlorinated compounds and nitrate [8] . Microorganisms have the potential to catalyze the production of hydrogen and methane with electrons derived from electrodes. Electrons derived from electrodes can potentially serve as the reductant for effecting microbial reduction of organic compounds to more desirable organic commodities, or for altering fermentation pathways in desired directions [9] . Furthermore, with the newly developed process of microbial electrosynthesis, it is possible to electrically power the microbial reduction of carbon dioxide to liquid transportation fuels and other useful organic commodities. When driven with electricity generated from solar technologies, microbial electrosynthesis is functionally an artificial form of photosynthesis with the potential to be much more efficient and environmentally sustainable than biomass-based strategies for fuel and chemical production.
Findings from the study of microbe-electrode exchange have also led to new insights into the functioning of anaerobic ecosystems. For over 40 years it has been considered that microorganisms in methanogenic environments exchange electrons primarily via interspecies transfer of hydrogen, with the electron-donating microorganism disposing of electrons by reducing protons to hydrogen and an electron-accepting methanogen oxidizing hydrogen with the reduction of methane [10] . However, it is feasible for different species of microorganisms to forge direct electrical connections, which are similar to those that they establish with electrodes, and direct interspecies electron transfer can be the primary mechanism for electron exchange in microbial aggregates converting wastes to methane [11] .
These developments demonstrate that a better understanding of the mechanisms by which microorganisms exchange electrons with electrodes can benefit the development from various new technologies as well as provide a better understanding of anaerobic microbial ecology. As more is learned, additional applications will probably emerge. This review summarizes current knowledge on microbe-electrode interactions and the novel electronic materials that some microorganisms can produce.
2. Aspects of Electromicrobiology
2.1. Electric Microorganisms
A wide range of microbes have been discovered that are capable of electrically interacting with their environment. Dissimilatory metal reducing bacteria is among the most studied, being capable of “breathing metals” in anaerobic environments. Derek Lovley isolated the first dissimilatory metal reducing microbe, Geobacter metallireducens, from sediment in the Potomac River in 1987 [12] . This microbe is capable of the complete oxidation of organic carbon to carbon dioxide, while utilizing an insoluble electron acceptor to support growth. Geobacter species are able to reduce a wide range of mineral and metalloids, including iron, manganese and uranium. Utilizing metal oxides in anaerobic environments for respiration; dissimilatory metal reduction plays an important role in carbon and geochemical cycles. An understanding of their function can be applied to the bioremediation of organic pollutants, heavy metals and radionuclides in anaerobic environments [5] .
An interesting aspect of the Geobacter species was the discovery, that they do not produce any electron shuttles, but directly transfer electrons extracellularly to iron oxide [13] . Subsequent studies have demonstrated electrically conductive pili and the outer membrane c-type cytochrome OmcS, as the route of extracellular electron flow between the microbes and the iron oxides. Electrically conductive pili known as microbial nanowires, are one of the most surprising discoveries in Electromicrobiology. Microbial nanowires are important components in microbe-electrode and microbe-microbe electron exchange. The nanowires of Geobacter sulfurreducens have metallic-like conductivity, similar to that observed in synthetic, organic conducting polymers [14] .
2.2. Microbial Fuel Cell (MFC)
The electricity breathing ability of these microbes can be collected from the environment as an electrical current, using a device called a Microbial Fuel Cell (MFC) [15] . Microbes utilize an electrode as an electron acceptor for anaerobic respiration. Initially designed for the harvesting of electrical current from organic compounds, difficulties in scalability above pilot scale has seen their application in power production, focused towards the powering of low power devices in aquatic environments [1] . Some of the more interesting current applications of MFC include applications where the harvesting of electrical current is not the primary focus, these include: acting as sensors for microbial metabolism in subsurface environments; stimulating bioremediation of organic environments by providing an electrode as an electron acceptor; and providing a means for balancing electron flow during the production of the microbial production of commodity chemicals [7] .
Geobacter species are extensively enriched within the electrode-associated communities of MFC, when a diverse range of environmental inoculum and organic carbon source are used. Geobacter species seem to be the most adapted in utilizing an electrode as an electron acceptor. They produce the highest current density of both pure and mixed cultures in a MFC are able to directly interact with an electrode without the use of an electron shuttle, and produce relatively thick biofilms (>60 µm) [16] . This biofilm has the unusual property of being electrically conductive, allowing all cells within the biofilm to be metabolically active and capable of transferring electrons to the electrode, across distances greater than 50 cell lengths.
Geobacter species on the electrode surface utilize a network of pilin and the outer membrane cytochromes, more specifically OmcZ, to electrically connect to the electrode. The ability to produce a conductive biofilm and act as a capacitor has seen an interest in utilizing these microbes in bioelectronics applications [17] .
2.3. Types of Microbial Fuel Cells
Microbial Fuel Cells can harvest bioelectronics from both heterotrophic and photosynthetic microorganisms or from a combination of the two in a single MFC system [18] .
2.3.1. Heterotrophic Microbial Fuel Cells
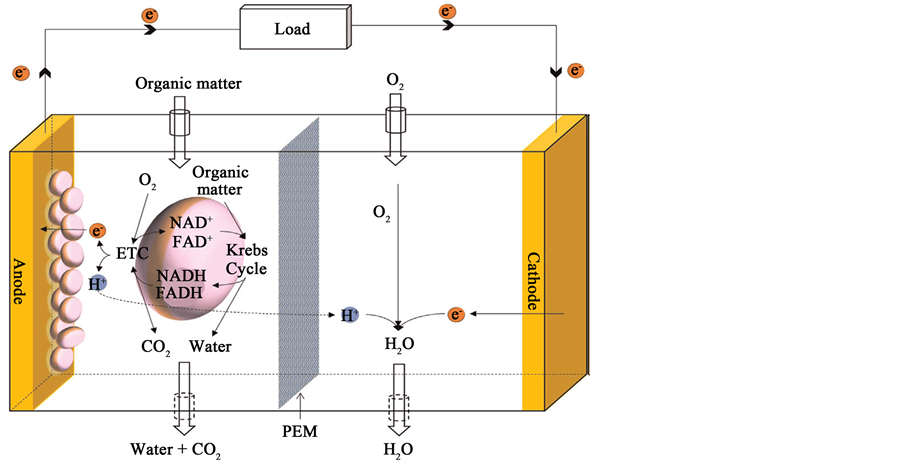
Heterotrophic MFCs can generate electrical power through the metabolic products of microbial respiration thus requiring a continual supply of organic carbon for energy. MFCs are typically comprised of anodic and cathodic chambers separated by a proton exchange membrane (PEM) allowing only H+ or other cations to pass from the anode to the cathode. A conductive load connects the two electrodes to complete the external circuit [19] . Microorganisms oxidize organic matter in the anodic chamber, completing respiration by transferring electrons to the anode. During this process, chemical energy is captured throughout the electron transport chain. Nicotinamide adenine dinucleotide (NAD+) and Nicotinamide adenine dinucleotide dehydrogenase (NADH) function as coenzymes for the reactions, repeatedly oxidizing and reducing to synthesize adenosine triphosphate (ATP), the biological energy unit of the cell (Figure 1) [20] .
MFC technology uses heterotrophic microorganisms that can transfer electrons produced via metabolism across the cell membrane to an external electrode; this process is called Extracellular Electron Transfer (EET), and it plays a key role in harvesting electrons. The primary goals for developing MFCs include identifying strategies to improve metabolic efficiency and optimize biofilm electron transfer activity to the electrode. MFC microorganisms can be planktonic cells and/or biofilms [21] . Electro active biofilms include both electrochemically active and inactive microorganisms.
While active microorganisms may support a number of functions in the microbial community (including the breakdown of complex organics through fermentation or the utilization of other electron acceptors/donors),
Figure 1. Principle of operation in a heterotrophic MFC. [18] .
electrochemically active microorganisms play an important role in enabling efficient electron transfer which maximizes both current densities and the energy efficiency [21] .
2.3.2. Photosynthetic Microbial Fuel Cells (Bio-Solar Cells)
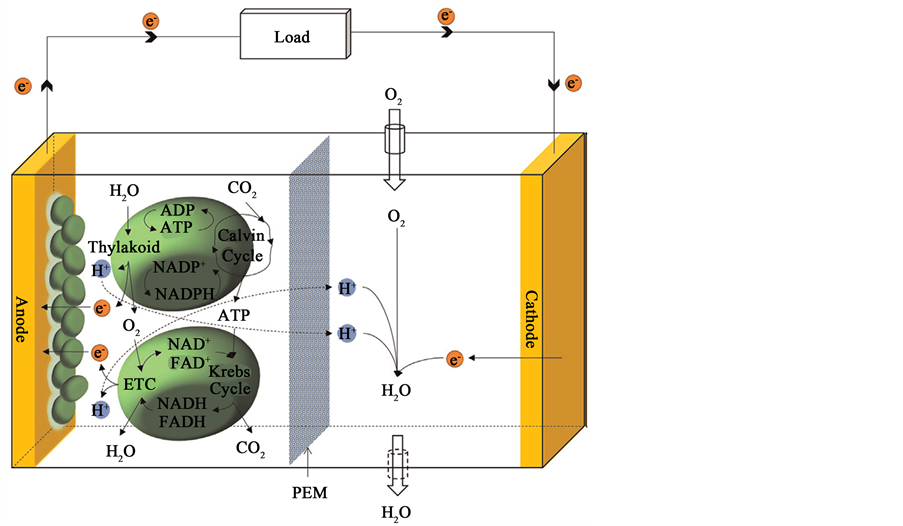
Photosynthetic MFCs (or bio-solar cells) produce bioelectric power based on the exploitation of bio-catalytic reactions of the photosynthetic microorganisms, such as cyanobacteria or algae [18] . During photosynthesis, the microorganisms capture solar energy to convert carbon dioxide and water into oxygen and carbohydrates, which will subsequently be used for their respiratory reaction, re-generating carbon dioxide and water. During those reactions, electrons are released through extracellular electron transfer pathways and flow to the cathode through the external electrical circuit (Figure 2). Simultaneously, the released protons diffuse from the anodic chamber to the cathode, where they re-combine with electrons and O2 to reform H2O. Through the aforementioned processes, photosynthetic MFCs can continuously generate electricity from solar energy without additional organic matter by increasing the electrochemical potential inside the cell to split and recreate water, producing oxygen, protons, and electrons.
Requiring only sunlight, water, and carbon dioxide to operate, photosynthetic MFCs offer advantages over potentially competing sustainable power sources such as heterotrophic MFCs or photovoltaic cells because the photosynthetic microorganisms used in photosynthetic MFCs (i) do not require an organic fuel, obviating the need for an active-feeding system, and (ii) are capable of producing power both day and night. This system resembles earth’s natural ecosystem, where living organisms work in conjunction with the nonliving components of their environment to offer self-sustainable and self-maintainable features as a system.
To date, successive efforts have focused on demonstrating the photosynthetic exoelectrogenic activities of various cyanobacteria or algae [18] . However, despite the vast potential and promise of photosynthetic MFCs, they have not yet been successfully translated into commercial applications, as they possess persistent performance limitations and scale-up bottlenecks. These challenges have relegated photosynthetic MFCs, as with ordinary MFCs, to the status of a laboratory curiosity rather than a viable alternative power source.
2.3.3. Hybrid Microbial Fuel Cell
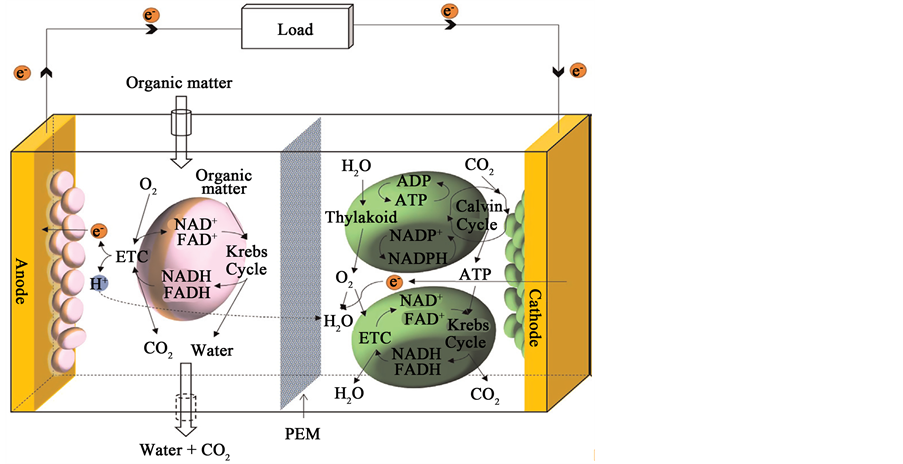
Hybrid MFCs are the collective name for a new bio-fuel cell system that integrates both heterotrophic and photosynthetic microorganisms to generate bioelectricity [21] . Within the hybrid MFC, heterotrophic microorganisms at the anode oxidize organic matter and transfer electrons to the anode while photosynthetic microorganisms provide in-situ oxygen as efficient and sustainable catalysts for the MFC cathodes.
In a dual-chamber MFC described in (Figure 3), the cathode half-cell is operated with photosynthetic micro-
Figure 2. Principle of operation in a photosynthetic MFC [18] .
Figure 3. Principle of operation in a hybrid MFC [18] .
organisms while the anode is inoculated with heterotrophic microorganisms. In the hybrid MFC with a non-cat- alyst-based cathode, oxygen can be continuously supplied for the reaction, resulting in an increase in current generation, which suggests that photosynthetic microorganisms are very efficient oxygenators.
2.4. Microbial Electrosynthesis
Microbial electrosynthesis, the process in which microorganisms use electrons derived from an electrode to reduce carbon dioxide to multi-carbon, extracellular products [22] . It is a potential strategy for converting electrical energy harvested with renewable strategies, such as solar or wind, into forms that can be stored and distributed on demand within existing infrastructure. Storage and distribution is a particular concern for solar energy, because it is a vast energy resource but harvests energy intermittently and not necessarily coincident with peak demand. The conversion of electrical energy to covalent chemical bonds may be one of the best storage and distribution options [23] . Microbial electrosynthesis powered by solar energy is an artificial form of photosynthesis with the same net overall reaction as plant-based photosynthesis: carbon dioxide and water are converted to organic compounds and oxygen [22] .
Microbial electrosynthesis depends upon electrotrophy, the ability of some microorganisms to use electrons derived from an electrode as an electron donor for the reduction of a terminal electron acceptor [24] . Although the ability of microorganisms to transfer electrons to electrodes has been studied for some time, the capacity for electron transfer in the opposite direction, from electrodes to cells, has received less attention. Geobacter species are capable of using electrons derived from graphite electrodes for the reduction of a diversity of electron acceptors, including nitrate, fumarate, and chlorinated solvents. Anaeromyxobacter dehalogenans also can reduce fumarate and reductively dehalogenate 2-chlorophenol [25] . A wide diversity of undefined microbial consortia have been inferred to contain microorganisms capable of reducing these and other electron acceptors, including oxygen, with an electrode as the sole electron donor.
Mixed cultures produced methane from carbon dioxide with neutral red, reduced at an electrode surface, as the electron donor. Methanobacterium palustre has been reported to reduce carbon dioxide to methane with electrode-derived electrons [26] , but there have been difficulties in confirming direct electron transfer in methanogens, because the low potentials required for methanogenesis also can produce significant hydrogen.
The finding that an acetogenic microorganism, Sporomusa ovata could use electrons derived from graphite electrodes for the reduction of carbon dioxide to acetate [22] provided the proof of concept that it is possible to convert carbon dioxide and water to extracellular, multicarbon products with electricity as the energy source. Biofilms of S. ovata growing on electrode surfaces produced acetate and small amounts of 2-oxobutyrate concomitantly with current consumption. Electron recovery in these products exceeded 85%, which is consistent with the reaction 2CO2 + 2H3O → CH3COOH + 2O2. The fact that carbon dioxide reduction to acetate in acetogens proceeds through acetyl-coenzyme A (CoA) and that acetyl-CoA is a central intermediate for the production of a diversity of useful organic products, including fuels. It has been suggested that microbial electrosynthesis with S. ovata can be a strategy for storing electrical energy in chemical products [22] .
2.5. Mechanisms of Electron Transfer
Many microorganisms can exchange electrons with electrodes when artificial electron shuttles are provided. Shuttles such as methylviologen, neutral red, or thionine can accept electrons from redox-active moieties within cells and transfer the electrons to electrode. Electron shuttles are often unstable and toxic, are uneconomical in large-scale processes, and cannot be employed in open environments. Microorganisms transfer electrons to an electrode without the addition of an artificial electron shuttle in three ways: (a) electron transfer via microbially produced soluble redox-active molecules; (b) short-range direct electron transfer between redox-active molecules on the outer cell surface and the electrode; and (c) long-range electron transport through conductive biofilms [27] .
2.5.1. Electron Transfer via Soluble Electron Shuttling Molecules
A diversity of both gram-negative and gram-positive microorganisms has the ability to produce electron shuttles to promote electron transfer to electrodes [28] . The concept of self-produced electron shuttles facilitating electron transfer to electrodes follows previous studies that demonstrated that some microorganisms produce shuttles that promote electron transfer between cells and insoluble Fe (III) oxides. For example, Geothrix fermentans, which can reduce Fe (III) oxide enclosed in porous alginate beads via a shuttle, also appeared to release an electron shuttle to promote electron transfer to electrodes. Shewanella species have a similar ability to reduce Fe (III) with which they are not in direct contact [22] ; this is attributed to the release of flavin in S. oneidensis cultures.
The maximum current densities produced by microorganisms that rely on electron shuttling to transfer electrons to electrodes are much lower than those for microorganisms capable of long-range electron transport through thick conductive biofilms because the slow diffusive flux of the shuttle is a major limitation. Although the shuttling mechanism may be somewhat effective in closed laboratory systems, in open environments this approach suffers from losses of the shuttle from the immediate microbe-electrode interface. For these and other reasons, it is not surprising that Shewanella species have never been found to be important constituents of anodes harvesting electricity from complex organic matter in open environments have ever been found to be important constituents of anodes harvesting electricity from complex organic matter in open environments.
2.5.2. Short Range Direct Electron Transfer via Redox-Active Proteins
The mechanisms for direct electron transfer to electrodes have been studied most extensively in G. sulfurreducens, G. sulfurreducens is closely related to the Geobacter species that typically predominate on electrodes harvesting current from organic matter, especially when oxygen intrusions are eliminated so that organic substrates are efficiently converted to current, and when the electrode potential is not artificially poised with electronics [17] . Early investigations suggested that, just as Geobacter species do not use shuttles to reduce Fe (III) oxide, shuttles are not involved in electron transfer to electrodes.
Thousands of nanowires embedded in the surface of Shewanell aoneidensis can directly transmit an electric current to inorganic minerals such as iron and manganese oxides, or the surface of electrodes. The phenomenon, known as Direct Extracellular Electron Transfer (DEET), occurs because of the way that some bacteria living in environments lacking oxygen export electrons that are generated through their respiratory cycle.
2.5.3. Long-Range Electron Transport via Conductive Pili
The possibility of a conductive biofilm was first proposed in studies on current-producing biofilms of G. sulfurreducens [29] and subsequently confirmed by direct measurements. Conductive biofilms have been invoked or inferred in other studies. However, failure to measure conductivity, as well as highly speculative and unsubstantiated models for conductivity, has led to significant debate about the mechanisms for long-range electron transport through biofilms, which can be resolved only by additional direct measurements of conductivity and rigorous experimentation [3] .
The high conductivity of current-producing biofilms of G. sulfurreducens allows cells at distances of multiple cell lengths from the anode to contribute to current production. The available evidence suggests that the conductivity of the biofilms can be attributed to a dense network of pili with metallic-like conductivity.
Long-range electron transport along pili also appears to play an important role in the recently discovered phenomenon of Direct Interspecies Electron Transfer (DIET), in which two species of microorganisms, growing syntrophically, can make electrical connections in order to directly share electrons DIET has been observed in defined syntrophic co-cultures with Geobacter metallireducens as the electron-donating organism and either Geobacter sulfurreducens, functioning as the electron-accepting partner.
3. Applications of Electromicrobiology
Research in Electromicrobiology has grown exponentially during the past decade. This has been due to the interest in new energy technologies, but it is also driven by the research for better waste treatment, water reclamation and desalination, sensors and more. Most recently, there have been new endeavours aimed at understanding how external electron transfer by microbes occurs and how it may contribute to the actions of microbes in the environment, even at relatively long distances.
These processes certainly will influence the development of the technologies noted above, but there is another area within electromicrobiology that may depend upon external electron transfer, and that is the microbial electrosynthesis of useful fuels and chemicals. Biocathodes are at the heart of electrosynthesis and require microbes to use to use the cathode as an electron donor and then catalyze the synthesis of chemicals. Waste CO2 may be used as a carbon source for the production of organic molecules, thereby avoiding the use of large arable land, and the technology can be entirely sustainable and carbon neutral if a renewable source of electricity is used.
3.1. Bioelectronics
Electrically active microorganisms have the potential to make significant contributions to the emerging field of bioelectronics. For example, the ability of microorganisms to sense a wide diversity of chemicals and environmental conditions suggests many possibilities for the development of Biosensors and Bio-computers [22] . The biosensors are used for monitoring water quality parameters such as biochemical oxygen demand (BOD) and toxicity can be related to measurement of bacterial metabolic activity.
Electronics grown or constructed from living materials have the potential benefits that they can be produced from inexpensive feed stocks with little waste generation and avoid the use of toxic compounds. If the living microorganisms and their components are part of the electronic application, they can have the capacity for self- repair and replication.
3.2. Bioremedation
The use of microorganisms in fuel cell as a catalyst for electricity generation was known 40 years ago [29] . When micro-organisms consume a substrate such as sugar in aerobic conditions they produce carbon dioxide and water, while when oxygen is not present they produce carbon dioxide, protons and electrons:
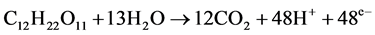 (1)
(1)
As organic material is used to “feed” the Microbial Fuel Cell (MFC), MFCs are suggested to be installed to wastewater treatment plants [17] . The bacteria would consume waste material from the water and produces supplementary power for the plant, and use inorganic mediators to tap into the electron transport chain of cells and obtain the produced electrons. This process has to be accommodated in fuel cells in which the organisms capable of producing an electric current are termed Exoelectrogens. The bioremediation process helps to harness Geobacter’s ability to oxidize organic matter as a means of removing hydrocarbon contaminants from soil and water.
3.3. Generation of Electricity from Solar Energy
As a new approach to convert solar energy into electricity is needed, and a promising one has evolved with advances in microbial fuel cell technologies. Photosynthetic MFCs are an emerging technology designed to harness earth’s most abundant and promising energy source (solar irradiation) and self-sustainably produce electrical power both day and night. The photosynthetic MFCs can continuously generate electricity from microbial photosynthetic and respiratory activities under day-night cycles. Given the fact that small-scale biological fuel cells are more energy dense than larger units, [30] the miniaturization of the photosynthetic MFCs inherently produces favorable conditions for increasing power density by reducing internal resistance and improving mass transport. Further, small-scale photosynthetic MFCs provide one possible way to scale up MFCs by connecting multiple units in a stack configuration.
3.4. Other Applications
Microbial Fuel Cells can be utilized as storage to renewable electricity similar to supercapacitors. Capacitative behaviour of bacteria on electrodes has been discovered and MFC-based supercapacitors have been proposed, (Figure 4) [31] . The MFC accumulate electrons at low current and high potentials and the stored electrons can be released as a current during discharge. MFC based supercapacitors inherently produce favourable conditions because of (i) a large surface-to-volume ratio; (ii) efficient mass transport; and (iii) short electron/proton travel distances.
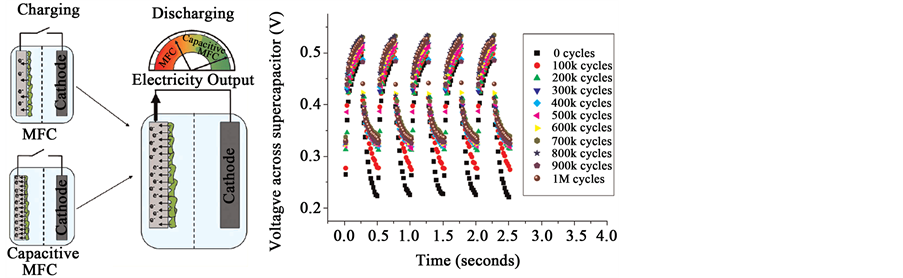
Figure 4. (a) Capacitive behavior of bacteria in MFCs; and (b) charging/discharging of the MFC-based supercapacitor up to 1,000,000 cycles [31] .
Finally, the MFCs could be a permanent power source for implantable biomedical devices, perhaps directly consuming the glucose as fuel in the blood stream.
4. Conclusions
There are many promising future research avenues in Electromicrobiology. Our understanding of how microorganisms donate electrons to electrodes is still rather superficial and even less is known about electron transfer from electrodes to cells. Furthermore, previous study on this topic has been limited to a few microbes. It is remarkable that the organisms that have been studied in detail: S. oneidensis, and G. sulfurreducens, have significantly different approaches for transferring electrons to electrodes. The intensive study of the electrophysiology of Shewanella and Geobacter species has been possible only because of substantial earlier investments that facilitate systems scale investigations. Similar in-depth investigations of other organisms are warranted. Elucidation of the mechanisms for metallic-like conductivity along pili and further investigation into the diversity of microorganisms that possess conductive filaments, the function of those filaments, and their mechanisms for conduction is needed.
Electromicrobiology has the potential to alleviate pressing societal needs. Although the justification for many of the early studies in Electromicrobiology is further optimization of Microbial Fuel Cells for energy harvesting, many more promising concepts for applications for microbe electrode interactions have recently emerged and undoubtedly more will be envisioned. For example, as noted in the introduction, the ability to favorably alter microbial fermentation with a supply of electrons from electrodes has been demonstrated, but only with the addition of soluble mediators. Developing a system for direct electron transfer may make this technology practical at a large scale. The rationale development of any of these technologies will depend on continued study of basic mechanisms of Electromicrobiology.
Cite this paper
Clifford Nkemnaso Obi,Grace Chigozirim Asogwa, (2015) Electromicrobiology: An Emerging Reality—A Review. Open Access Library Journal,02,1-10. doi: 10.4236/oalib.1102088
References
- 1. Franks, A.E. and Nevin, K.P. (2010) Microbial Fuel Cells: A Current Review. Energies, 3, 899-919.
- 2. Malvankar, N.S., Mester, T., Tuominen, M. and Lovley, D.R. (2012) Supercapacitors Based on c-Type Cytochromes Using Conductive Nanostructured Networks of Living Bacteria. A European Journal of Chemical Physics and Physical Chemistry, 13, 463-468.
http://dx.doi.org/10.1002/cphc.201100865 - 3. Malvankar, N.S., Vargas, M., Nevin, K.P., Franks, A.E. and Leang, C. (2011) Tunable Metallic-Like Conductivity in Nanostructured Biofilms Comprised of Microbial Nanowires. Nature Nanotechnology, 6, 573-579.
http://dx.doi.org/10.1038/nnano.2011.119 - 4. Lovley, D.R. (2006) Bug Juice: Harvesting Electricity with Microorganisms. Nature Reviews Microbiology, 4, 497-508.
http://dx.doi.org/10.1038/nrmicro1442 - 5. Lovley, D.R. and Nevin, K.P. (2011) A Shift in the Current: New Applications and Concepts for Microbe-Electrode Electron Exchange. Current Opinion in Biotechnology, 22, 441.
http://dx.doi.org/10.1016/j.copbio.2011.01.009 - 6. Zhang, T., Gannon, S.M., Nevin, K.P., Franks, A.E. and Lovley, D.R. (2010) Stimulating the Anaerobic Degradation of Aromatic Hydrocarbons in Contaminated Sediments by Providing an Electrode as the Electron Acceptor. Environmental Microbiology, 12, 1011-1020.
http://dx.doi.org/10.1111/j.1462-2920.2009.02145.x - 7. Bradley, R.W., Bombelli, P., Rowden, S. and Howe, C.J. (2012) Biological Photovoltaics: Intra- and Extra-Cellular Electron Transport by Cyanobacteria. Biochemical Society Transactions, 40, 1302-1307.
http://dx.doi.org/10.1042/BST20120118 - 8. Gregory, K.B., Bond, D.R. and Lovley, D.R. (2004) Graphite Electrodes as Electron Donors for Anaerobic Respiration. Environmental Microbiology, 6, 596.
http://dx.doi.org/10.1111/j.1462-2920.2004.00593.x - 9. Steinbusch, K.J.J., Hamelers, H.V.M., Schaap, J.D., Kampman, C. and Buisman, C.J.N. (2010) A Kinetic Perspective on Extracellular Electron Transfer by Anode-Respiring Bacteria. FEMS Microbiology Reviews, 34, 3-17.
http://dx.doi.org/10.1111/j.1574-6976.2009.00191.x - 10. McInerney, M.J., Sieber, J.R. and Gunsalus, R.P. (2009) Syntrophy in Anaerobic Global Carbon Cycles. Current Opinion in Biotechnology, 20, 623-634.
http://dx.doi.org/10.1016/j.copbio.2009.10.001 - 11. Morita, M., Malvankar, N.S., Franks, A.E., Summers, Z.M., Giloteaux, L., Rotaru, A.E., Rotaru, C. and Lovley, D.R. (2011) Potential for Direct Interspecies Electron Transfer in Methanogenic Wastewater Digester Aggregates. mBio, 2, e00159-11.
http://dx.doi.org/10.1128/mBio.00159-11 - 12. Lovley, D.R. and Phillips, E.J.P. (1988) Novel Mode of Microbial Energy Metabolism: Organic Carbon Oxidation Coupled to Dissimilatory Reduction of Iron or Manganese. Applied and Environmental Microbiology, 54, 1472-1480.
- 13. Nevin, K.P. and Lovley, D.R. (2000) Lack of Production of Electron-Shuttling Compounds or Solubilization of Fe(III) during Reduction of Insoluble Fe(III) Oxide by Geobacter metallireducens. Applied and Environmental Microbiology, 66, 2248-2251.
http://dx.doi.org/10.1128/AEM.66.5.2248-2251.2000 - 14. Reguera, G., Nevin, K.P., Nicoll, J.S., Covalla, S.F., Woodard, T.L. and Lovley, D.R. (2006) Biofilm and Nanowire Production Leads to Increased Current in Geobacter sulfurreducens Fuel Cells. Applied and Environmental Microbiology, 72, 7345-7348.
http://dx.doi.org/10.1128/AEM.01444-06 - 15. Bond, D.R., Holmes, D.E., Tender, L.M. and Lovley, D.R. (2002) Electrode-Reducing Microorganisms That Harvest Energy from Marine Sediments. Science, 295, 483-485.
http://dx.doi.org/10.1126/science.1066771 - 16. Nevin, K.P., Richter, H., Covalla, S.F., Johnson, J.P. and Woodard, T.L. (2008) Power Output and Columbic Efficiencies from Biofilms of Geobactersulfurreducens Comparable to Mixed Community Microbial Fuel Cells. Environmental Microbiology, 10, 2505-2514.
http://dx.doi.org/10.1111/j.1462-2920.2008.01675.x - 17. Lovley, D.R. (2012) Electromicrobiology. Annual Review of Microbiology, 66, 391-409.
http://dx.doi.org/10.1146/annurev-micro-092611-150104 - 18. Rosenbaum, M. and Angenent, L.T. (2010) Cathodes as Electron Donors for Microbial Metabolism: Which Extracellular Electron Transfer Mechanism Are Involved? Current Opinion in Biotechnology, 21, 259-264.
http://dx.doi.org/10.1016/j.copbio.2010.03.010 - 19. Logan, B.E., Hamelers, B., Rozendal, R., Schroder, U., Keller, J., Freguia, S., Aelterman, P., Verstraete, W. and Rabaey, K. (2006) Microbial Fuel Cells: Methodology and Technology. Environmental Science & Technology, 17, 5181-5192.
http://dx.doi.org/10.1021/es0605016 - 20. Rabaey, K. and Verstraete, W. (2005) Microbial Electrosynthesis: Revisiting the Electrical Route for Microbial Production. Nature Reviews Microbiology, 8, 706-716.
http://dx.doi.org/10.1038/nrmicro2422 - 21. Borole, A.P., Reguera, G., Ringeisen, B., Wang, Z., Feng, Y. and Kim, B.H. (2011) Electroactive Biofilms: Current Status and Future Research Needs. Energy & Environmental Science, 4, 4813-4834.
http://dx.doi.org/10.1039/c1ee02511b - 22. Strik, D., Timmers, R.A., Helder, M., Steinbusch, K., Hamelers, H. and Buisman, C.J.N. (2011) Microbial Solar Cells: Applying Photosynthetic and Electrochemically Active Organisms. Trends in Biotechnology, 29, 41-49.
http://dx.doi.org/10.1016/j.tibtech.2010.10.001 - 23. Nevin, K.P., Woodard, T.L., Franks, A.E., Summers, Z.M. and Lovley, D.R. (2010) Microbial Electrosynthesis: Feeding Microbes Electricity to Convert Carbon Dioxide and Water to Multicarbon Extracellular Organic Compounds. mBio, 1, e00103-10.
http://dx.doi.org/10.1128/mbio.00103-10 - 24. Lewis, N.S. and Nocera, D.G. (2006) Powering the Planet: Chemical Challenges in Solar Energy Utilization. Proceedings of the National Academy of Sciences of the United States of America, 103, 15729-15735.
http://dx.doi.org/10.1073/pnas.0603395103 - 25. Lovley, D.R. (2010) Powering Microbes with Electricity: Direct Electron Transfer from Electrodes to Microbes. Environmental Microbiology, 10, 1758-2229.
- 26. Strycharz, S.M. (2010) Reductive Dechlorination of 2-Chlorophenol by Anaeromyxobacter dehalogens with an Electrode Serving as the Electron Donor. Environmental Microbiology Reports, 2, 289-294.
http://dx.doi.org/10.1111/j.1758-2229.2009.00118.x - 27. Cheng, S., Xing, D., Call, D.F. and Logan, B.E. (2009) Direct Biological Conversion of Electrical Current into Methane by Electromethanogenesis. Environmental Science & Technology, 43, 3953-3958.
http://dx.doi.org/10.1021/es803531g - 28. Lovley, D.R. (2008) The Microbe Electric: Conversion of Organic Matter to Electricity. Current Opinion in Biotechnology, 19, 564-571.
http://dx.doi.org/10.1016/j.copbio.2008.10.005 - 29. Lovley, D.R., Ueki, T., Zhang, T., Malvankar, N.S. and Shrestha, P.M. (2011) Geobacter: The Microbe Electric’s Physiology, Ecology, and Practical Applications. Advances in Microbial Physiology, 59, 1-100.
http://dx.doi.org/10.1016/B978-0-12-387661-4.00004-5 - 30. Reguera, G., McCarthy, K.D., Mehta, T., Nicoll, J.S. and Tuominen, M.T. (2005) Extracellular Electron Transfer via Microbial Nanowires. Nature, 435, 1098-1101.
http://dx.doi.org/10.1038/nature03661 - 31. Leropoulos, I., Greenman, J. and Melhuish, C. (2010) Improved Energy Output Levels from Small-Scale Microbial Fuel Cells. Bioelectrochemistry, 78, 44-50.
http://dx.doi.org/10.1016/j.bioelechem.2009.05.009
NOTES
*Corresponding author.