Open Access Library Journal
Vol.02 No.02(2015), Article ID:68052,6 pages
10.4236/oalib.1100777
The Epidemiology of Adenovirus Associated Lower Respiratory Tract Infection in Hospitalized Children in Hangzhou City, China
Qi Zheng1, Meiping Lu1,2*, Wei Li3
1Department of Respiratory, Children’s Hospital, Zhejiang University School of Medicine, Hangzhou, China
2Department of Rheumatology Immunology and Allergy, Children’s Hospital, Zhejiang University School of Medicine, Hangzhou, China
3Department of Pediatrics, Jiaxing Maternity and Child Health Care Hospital, Jiaxing, China
Email: *lu_meiping@126.com
Copyright © 2015 by authors and OALib.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 22 January 2015; accepted 7 February 2015; published 11 February 2015
ABSTRACT
This study was performed to investigate the epidemiology of adenovirus related acute lower respiratory tract infection in children through a large sample collection. Totally 76,518 patients admitted to Children’s Hospital Affiliated to Zhejiang University School of Medicine were investigated from July 2006 to June 2012. Nasopharyngeal aspirates were collected and examined by immunofluorescence assay to confirm viral etiology. There were 479 patients positive for adenovirus (6.3‰) and pneumonia accounted for most of these patients. The median age of adenovirus infected was 16 months and peaked at the second six-month of life. Winter, spring and summer had the similar positive rate, while autumn had the lowest occurrence rate. The detection rates of adenovirus on each spring from 2007 to 2012 were 18.01‰, 5.73‰, 11.85‰, 6.35‰, 11.77‰ and 2.66‰, respectively, which indicated a prevalence appearing once every two years.
Keywords:
Adenovirus, Respiratory Infection, Epidemiology
Subject Areas: Epidemiology, Pediatrics

1. Introduction
Adenovirus was first described in the 1950s and is associated with a wide range of diseases, such as keratoconjunctivitis, febrile respiratory illness and gastrointestinal diseases [1] - [3] . Severe cases can occur in infants or immunodeficient patients. Serious infections are often associated with fetal short-term complications including respiratory distress, cardiac failure and hepatic dysfunction or with intractable long-term complications, such as obliterative bronchiolitis and bronchiectasis [4] . Due to the severity of adenovirus infection, it has attracted extensive attention from clinicians worldwide [5] [6] .
Significant discrepances exist in the incidences of adenovirus infection in different regions as the geographic disparity and different test methods. In some tropical countries such as Cambodia, adenovirus was detected in 3% of acute lower respiratory tract infection in children less than five years old [7] . While in Oman, the detection of adenovirus was much higher, which accounted for about 7.7% [8] . Existing of different detection rate is also due to the lack of large sample research. So understanding of the epidemiology through large sample date is crucial to prioritize the preventive strategies like immunization. As a result, we conducted this research through a six-year continuous monitoring of hospitalized children and summarized the investigations of adenovirus infected cases by laboratory detection and the epidemiological information.
2. Material and Methods
2.1. Patients
The study was performed from July 2006 to June 2012 at the Children’s Hospital Zhejiang University School of Medicine. Children who were hospitalized for acute lower respiratory infection were enrolled. Records of adenovirus positive patients were collected for further study. The experimental protocol was approved by the ethics committee from Zhejiang University School of Medicine.
2.2. Clinical Manifestations of Acute Lower Respiratory Infection
Recognition of pediatric acute lower respiratory infection was based on the physician diagnosis of bronchitis, bronchiolitis and pneumonia. Acute respiratory infection was defined as respiratory symptoms lasting <3 weeks. Patients with chronic lung or heart diseases, and primary or secondary immunodeficiency were excluded. Physicians categorized the patients into lower and upper respiratory tract infections according to the clinical manifestations described in the Nelson Textbook of Pediatrics [9] .
2.3. Specimens
Nasopharyngeal samples were collected on admission by inserting a catheter tube into the nasopharynx and aspirating with a vacuum constriction device. The aspirates were then made into slides, each of which should have at least 20 exfoliative cells. The aspirates were placed in a universal transport medium and stored at 4˚C until sent to the central laboratory of children’s hospital for analysis.
2.4. Virus Determination
Immunofluorescence assay was conducted for the qualitative identification of adenovirus, influenza A, influenza B, parainfluenza-type 1, 2, 3 and respiratory syncytial virus. The slides were examined under magnification of 200 times with a fluorescent microscopy. Positive staining was represented by the presence of at least two intact cells with a type of fluorescence for certain virus.
2.5. Statistical Analysis
All statistical analyses were performed using SPSS software (version 19). Data are presented as the percentage or mean ± SD. The Chi-square test was used to compare the difference of proportion between different seasons and years. P-value < 0.05 was considered significant.
3. Results
The detection rate of Adenovirus in acute lower respiratory infection was 6.25‰ (479/76,518). Medical records of adenovirus identified cases were analyzed as follows.
3.1. Demographics
The age distribution was from one month to 172 months (median age, 16 months). There were 55.32% children younger than 24 months of age, and 8.14% younger than 6 months. Children over than 5 years old had the lowest morbidity. The highest occurrence of adenovirus in acute lower respiratory infection was in the first year of life and peaked at the second six-month. Boys accounted for over two thirds of all patients (331/479). About half of the patients had direct-contact history of respiratory diseases before getting illness.
3.2. Clinical Information for ADV Positive Children (Table 1)
Pneumonia and bronchiolitis are the most common admitting diagnoses of all patients. The mean length of hospitalization was 10.3 days (10.3 ± 7.0). All of the ADV positive patients had cough and a quarter of them had wheezing. Over 90% patients had fever, some of them even got continuous high fever (body temperature > 39˚C) persisting for about 8 days (8.67 ± 4.31). The main positive signs of ADV infected children were moist rales in the lung. Respiratory failure and liver damage were the most common complications of ADV infection.
3.3. Seasonal Occurrence of Adenovirus Infection
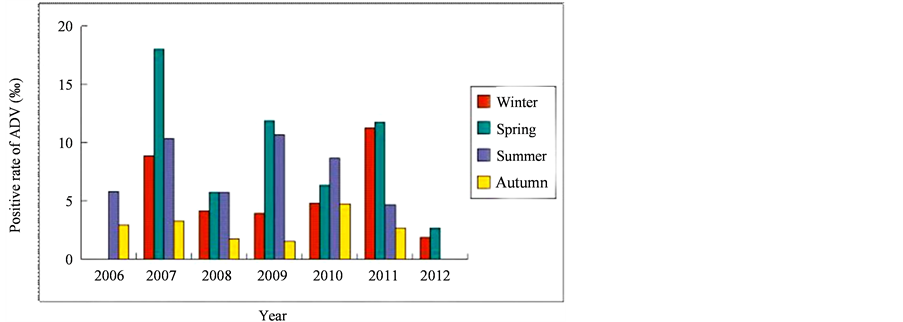
Adenovirus was isolated sporadically with the detection rate highest in the spring (April-June, 36.7%) and lowest in the autumn (October-December, 10.6%). There was significant difference of the detection rate between autumn and the other seasons (P < 0.05). The detection of adenovirus in different seasons was revealed in Figure 1.
3.4. Prevalent Trends of Adenovirus Associated Acute Lower Respiratory Infection
Spring has the highest detection rate in all seasons. In this season, positive rate of adenovirus-acute lower respiratory infection from 2006 to 2012 were 18.01‰, 5.73‰, 11.85‰, 6.35‰, 11.77‰ and 2.66‰, respectively. The occurrence of adenovirus-acute lower respiratory infection seemed to have prevalence at spring once every two years. The trends of adenovirus infection in each season of the six years were shown in Figure 2.
Table 1. Clinical information for ADV positive patients.
Figure 1. The positive detection rate of adenovirus in different seasons.
Figure 2. The trends of adenovirus infection in each season.
4. Discussion
The occurrence of adenovirus-acute lower respiratory infection is often sporadic with different age and season distribution. In this study, we noticed that patients less than two years of age occupied over half of all cases. Children are much more likely to get trapped into serious adenovirus infection as the specific antibody to adenovirus is generated and gradually increased since two years of age [10] . It is generally thought that infants less than six months are under the protection of antibodies that obtained from their mothers. But we found the occurrence of adenovirus-acute lower respiratory infection in little babies less than six months was higher than our expected, which accounted for about 8.14% of all cases. Fatal pneumonia in neonate was observed in previous study, who was ultimately developed into acute respiratory distress and died of multi-visceral failure [11] . It is predicted that once adenovirus infection occur in the neonatal period, it is more likely to get into serious situation even though the child has none underlying diseases [11] [12] . While, in view of this relatively high occurrence as well as the serious outcome, it is necessary to monitor the epidemic of adenoviral infection in little babies so as to give clinical intervention at early stage.
A time series study shows that some climatic conditions such as temperature and humidity were correlated with the incidence of respiratory virus infection. Here we found that adenovirus appeared to be active all year round with a little prominent of incidence peaking at spring and had the lowest detection rate at autumn which was consistent with the previous report [13] . Furthermore, the occurrence of adenovirus infection seemed to have a two-year rhythm like some other common respiratory viruses, such as respiratory syncytial virus, human metapneumovirus and parainfluenza virus [14] [15] . Direct contact and inhalation is the main way of virus transmission which is often influenced by geographic or climatic factors. Lower temperatures as well as higher humidity may prolong the survival time of respiratory viruses in vitro [16] . Some other climatic factors such as ultraviolet B radiance and wind velocity were also linked with the overall number of respiratory diseases hospitalizations [17] [18] . In
5. Summary
This study showed that the occurrence of adenovirus-acute lower respiratory infection seemed to have a two- year rhythm and the incidence of adenovirus-acute lower respiratory infection in spring was higher than other seasons. Infants less than six months are common infection population of adenovirus-acute respiratory infection. Therefore, those patients should be intensively monitored.
Acknowledgements
Thanks to all of the members of Respiratory Department of Children’s Hospital, Zhejiang University School of Medicine in helping collecting the clinical specimens.
Funding
This work was supported by the Chinese National Natural Science Foundation (No. 81070012) and Qianjiang talents project of technology in Zhejiang (No. 2011R10028).
Ethical Approval
The experimental protocol was approved by the ethics committee from the medical school of Zhejiang University.
Competing Interests
The authors declare that they have no competing interests.
Contributors
All authors participated in drafting and revising the manuscript and approved the submitted version.
Cite this paper
Qi Zheng,Meiping Lu,Wei Li, (2015) The Epidemiology of Adenovirus Associated Lower Respiratory Tract Infection in Hospitalized Children in Hangzhou City, China. Open Access Library Journal,02,1-6. doi: 10.4236/oalib.1100777
References
- 1. Larranaga, C., Kajon, A. and Avendano, L.F. (2000) Hospital-Acquired Adenovirus 7 h Infantile Respiratory Infection in Chile. Pediatric Infectious Disease Journal, 19, 527-531.
http://dx.doi.org/10.1097/00006454-200006000-00007 - 2. Edwards, K.M., Thompson, J., Paolini, J. and Wright, P.F. (1985) Adenovirus Infections in Young Children. Pediatrics, 76, 420-424.
- 3. Uhnoo, I., Svensson, L. and Wadell, G. (1990) Enteric Adenoviruses. Baillière’s Clinical Gastroenterology, 4, 627-642.
http://dx.doi.org/10.1016/0950-3528(90)90053-J - 4. Delacourt, C. (2007) Sequelae of Viral Lower Respiratory Tract Infections in Children. La Revue du Praticien, 57, 1919-1922.
- 5. Munoz, F.M., Piedra, P.A. and Demmler, G.J. (1998) Disseminated Adenovirus Disease in Immunocompromised and Immunocompetent Children. Clinical Infectious Diseases, 27, 1194-1200.
http://dx.doi.org/10.1086/514978 - 6. Louie, J.K., Kajon, A.E. and Holodniy, M. (2008) Severe Pneumonia Due to Adenovirus Serotype 14: A New Respiratory Threat. Clinical Infectious Diseases, 46, 421-425.
http://dx.doi.org/10.1086/525261 - 7. Guerrier, G., Goyet, S. and Chheng, E.T. (2012) Acute Viral Lower Respiratory Tract Infections in Cambodian Children: Clinical and Epidemiological Characteristics. Pediatric Infectious Disease Journal, 32, e8-e13.
http://dx.doi.org/10.1097/INF.0b013e31826fd40d - 8. Khamis, F.A., Al-Kobaisi, M.F. and Al-Areimi, W.S. (2012) Epidemiology of Respiratory Virus Infections among Infants and Young Children Admitted to Hospital in Oman. Journal of Medical Virology, 84, 1323-1329.
http://dx.doi.org/10.1002/jmv.23330 - 9. Kliegman, R.M., Stanton, B.F. and St. Geme, J.W. Nelson Textbook of Pediatrics. 18th Edition, Part 18, Section 2.
- 10. Endo, R., Ishiguro, N. and Kikuta, H. (2007) Seroepidemiology of Human Bocavirus in Hokkaido Prefecture, Japan. Journal of Clinical Microbiology, 45, 3218-3223.
http://dx.doi.org/10.1128/JCM.02140-06 - 11. Henquell, C., Boeuf, B. and Mirand, A. (2009) Fatal Adenovirus Infection in a Neonate and Transmission to Health-Care Workers. Journal of Clinical Virology, 45, 345-348.
http://dx.doi.org/10.1016/j.jcv.2009.04.019 - 12. Faden, H., Wilby, M. and Hainer, Z.D. (2011) Pediatric Adenovirus Infection: Relationship of Clinical Spectrum, Seasonal Distribution, and Serotype. Clinical Pediatrics, 50, 483-487.
http://dx.doi.org/10.1177/0009922810392775 - 13. Munoz, F.M., Piedra, P.A. and Demmler, G.J. (1998) Disseminated Adenovirus Disease in Immunocompromised and Immunocompetent Children. Clinical Infectious Diseases, 27, 1194-1200.
http://dx.doi.org/10.1086/514978 - 14. Weigl, J.A., Puppe, W. and Meyer, C.U. (2005) RSV-Prevention in Children Guided by a Web-Based Early Warning System. Klinische Pädiatrie, 217, 47-52.
http://dx.doi.org/10.1055/s-2004-836246 - 15. Weigl, J.A., Puppe, W. and Meyer, C.U. (2007) Ten Years’ Experience with Year-Round Active Surveillance of up to 19 Respiratory Pathogens in Children. European Journal of Pediatrics, 166, 957-966.
http://dx.doi.org/10.1007/s00431-007-0496-x - 16. Shaman, J. and Kohn, M. (2009) Absolute Humidity Modulates Influenza Survival, Transmission, and Seasonality. Proceedings of the National Academy of Sciences of the United States of America, 106, 3243-3248.
http://dx.doi.org/10.1073/pnas.0806852106 - 17. Welliver, R. (2009) The Relationship of Meteorological Conditions to the Epidemic Activity of Respiratory Syncytial Virus. Paediatric Respiratory Reviews, 10, 6-8.
http://dx.doi.org/10.1016/S1526-0542(09)70004-1 - 18. Loh, T.P., Lai, F.Y.L. and Tan, E.S. (2011) Correlations between Clinical Illness, Respiratory Virus Infections and Climate Factors in a Tropical Paediatric Population. Epidemiology Infection, 139, 1884-1894.
http://dx.doi.org/10.1017/S0950268810002955 - 19. du Prel, J.-B., Puppe, W. and Grondahl, B. (2009) Are Meteorological Parameters Associated with Acute Respiratory Tract Infections? Clinical Infectious Diseases, 49, 861-868.
http://dx.doi.org/10.1086/605435
NOTES
*Corresponding author.