Paper Menu >>
Journal Menu >>
 Materials Sciences and Application, 2011, 2, 977-980 doi:10.4236/msa.2011.28131 Published Online August 2011 (http://www.SciRP.org/journal/msa) Copyright © 2011 SciRes. MSA 977 Preparation of Copper (Cu) and Copper Oxide (Cu2O) Nanoparticles under Supercritical Conditions M. Asharf Shah*, M. S. Al-Ghamdi Department of Physics, Faculty of Sciences, King Abdul Aziz University, Kingdom of Saudi Arabia, Saudi. Email: *shahkau@hotmail.com Received May 19th, 2011; revised May 20th, 2011; accepted May 31st, 2011. ABSTRACT Temperature and pressure, both of which can affect the supersaturation and nucleation are responsible for solvents properties. In this study, we use water as solvent under supercritical conditions and report copper (Cu) and (Cu2O) nanoparticles of size ranging from 9 nm to 60 nms . This synthetic technique has the following advantages: Firstly, it is one step synthesis approach, making it easy to control the growth kinetics. Secondly, the synthesis needs no sophisti- cated equipments. Thirdly, the approach is non-toxic without producing hazardous waste as water is being used as sol- vent as well as source of oxygen. Forth, it is a surfactant free synthesis and has bright prospects. Keywords: Supercritical Water, Copper Powder, Synthesis, Characterization 1. Introduction In chemical sciences, synthesis of transition metal and metal oxide nanoparticles is a growing research field. As the metal particles are reduced in size, bulk properties of the particles disappear to be substituted to that of quan- tum dot following quantum mechanical rules. It can thus be easily understood that metal nanoparticles chemistry differs from that of the bulk materials. Since with size reduction the high surface area to volume ratio lead to enhanced catalytic activity [1]. Among various metal nanoparticles, copper (Cu) and copper oxide (Cu2O) nanoparticles have attracted con- siderable attention because copper is one of the most important in modern technologies and is readily available [2]. Considerable interest has been focused on copper nanoparticles due to their optical, catalytic, mechanical and electrical properties [3,4]. It has been used as het- erogeneous catalysts in many important chemical proc- esses, such as degradation of nitrous oxide with ammonia and oxidation of carbon monoxide, hydrocarbon and phenol in supercritical water [5]. Cu and Cu2O nanoparticles have been synthesized through different methods such as thermal decomposition, metal salt reduction, microemulsion, laser ablation, DC arc discharge, solvothermal and sonochemical reactions [6-10]. Among various techniques for synthesis, the hydrothermal method of producing metal oxide nanomaterials is unique and economical. Recently, we have reported the synthesis of Cu2O nanoparticles of almost uniform size at 180˚C without any surfactants and additives [11]. In the continuation of the development of simple syn- thesis of nanoparticles, we herein report the synthesis of copper and copper oxide nanoparticles from a simple, green, low cost and reproducible process. The average size of the nanoparticles is ~25 nms. Detailed and system- atic studies would be necessary to optimize the conditions for obtaining nanoparticles of desired dimensions. The aim of the study is to provide the feasibility of the simple route for the preparation of copper oxide nanostructures without additives and organics. The reported method besides being organics free is economical, fast and free of pollution, which will make it suitable for large scale production. 2. Experimental In a typical preparation process, 3 mg of copper powder was added to 30 ml of distilled water in a glass vial and was well sonicated for about 25 minutes in a sonicator. The reaction mixture was transferred into a stainless steel teflon lined metallic bomb of 100 ml capacity and sealed under ordinary conditions. The autoclave was then placed inside a preheated furnace and the mixture was heated to 140˚C and maintained at this temperature for 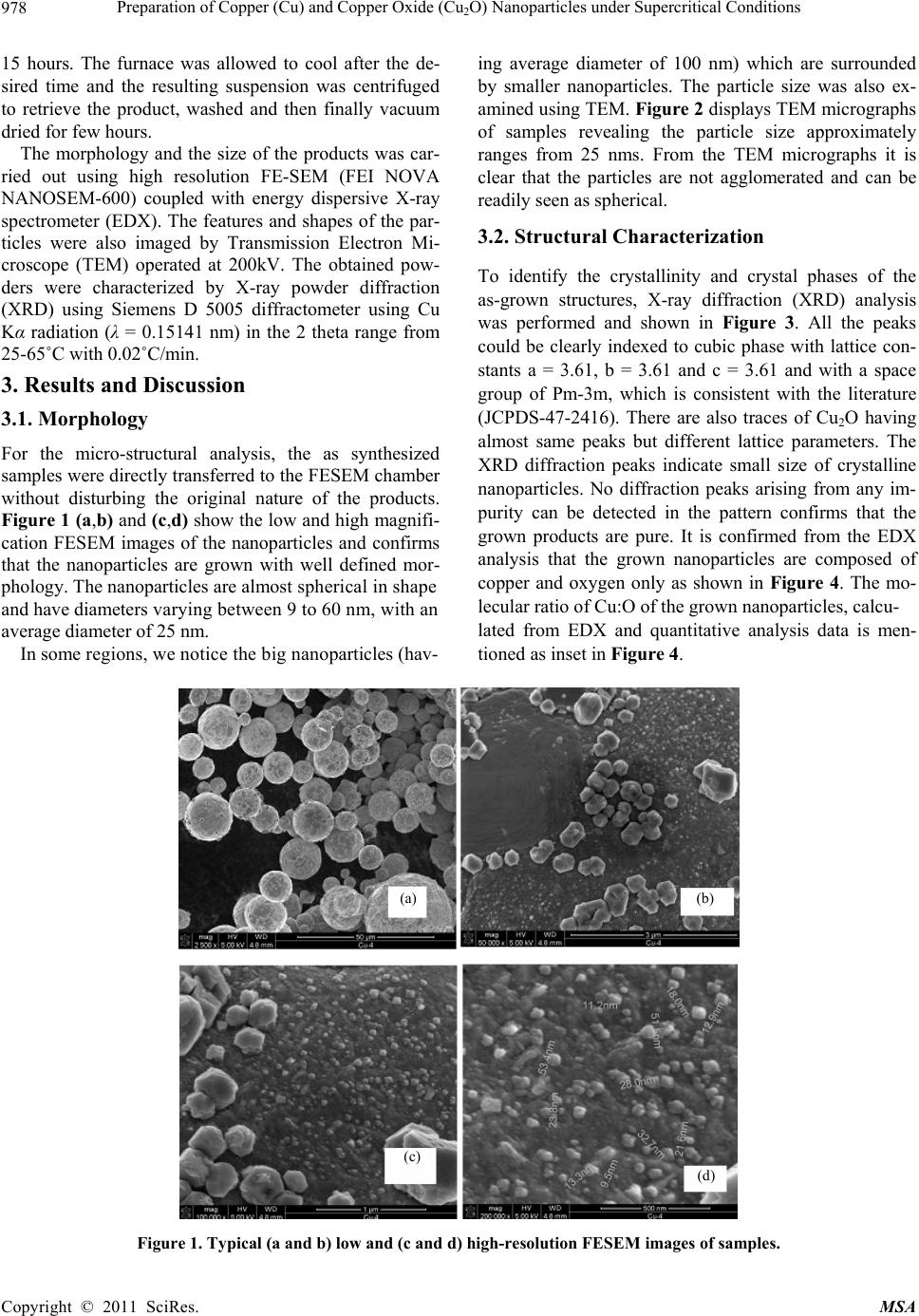 Preparation of Copper (Cu) and Copper Oxide (Cu2O) Nanoparticles under Supercritical Conditions Copyright © 2011 SciRes. MSA 978 15 hours. The furnace was allowed to cool after the de- sired time and the resulting suspension was centrifuged to retrieve the product, washed and then finally vacuum dried for few hours. The morphology and the size of the products was car- ried out using high resolution FE-SEM (FEI NOVA NANOSEM-600) coupled with energy dispersive X-ray spectrometer (EDX). The features and shapes of the par- ticles were also imaged by Transmission Electron Mi- croscope (TEM) operated at 200kV. The obtained pow- ders were characterized by X-ray powder diffraction (XRD) using Siemens D 5005 diffractometer using Cu Kα radiation (λ = 0.15141 nm) in the 2 theta range from 25-65˚C with 0.02˚C/min. 3. Results and Discussion 3.1. Morphology For the micro-structural analysis, the as synthesized samples were directly transferred to the FESEM chamber without disturbing the original nature of the products. Figure 1 (a,b) and (c,d) show the low and high magnifi- cation FESEM images of the nanoparticles and confirms that the nanoparticles are grown with well defined mor- phology. The nanoparticles are almost spherical in shape and have diameters varying between 9 to 60 nm, with an average diameter of 25 nm. In some regions, we notice the big nanoparticles (hav- ing average diameter of 100 nm) which are surrounded by smaller nanoparticles. The particle size was also ex- amined using TEM. Figure 2 displays TEM micrographs of samples revealing the particle size approximately ranges from 25 nms. From the TEM micrographs it is clear that the particles are not agglomerated and can be readily seen as spherical. 3.2. Structural Characterization To identify the crystallinity and crystal phases of the as-grown structures, X-ray diffraction (XRD) analysis was performed and shown in Figure 3. All the peaks could be clearly indexed to cubic phase with lattice con- stants a = 3.61, b = 3.61 and c = 3.61 and with a space group of Pm-3m, which is consistent with the literature (JCPDS-47-2416). There are also traces of Cu2O having almost same peaks but different lattice parameters. The XRD diffraction peaks indicate small size of crystalline nanoparticles. No diffraction peaks arising from any im- purity can be detected in the pattern confirms that the grown products are pure. It is confirmed from the EDX analysis that the grown nanoparticles are composed of copper and oxygen only as shown in Figure 4. The mo- lecular ratio of Cu:O of the grown nanoparticles, calcu- lated from EDX and quantitative analysis data is men- tioned as inset in Figure 4. Figure 1. Typical (a and b) low and (c and d) high-resolution FESEM image s of sample s. (a) (b) (c) (d) 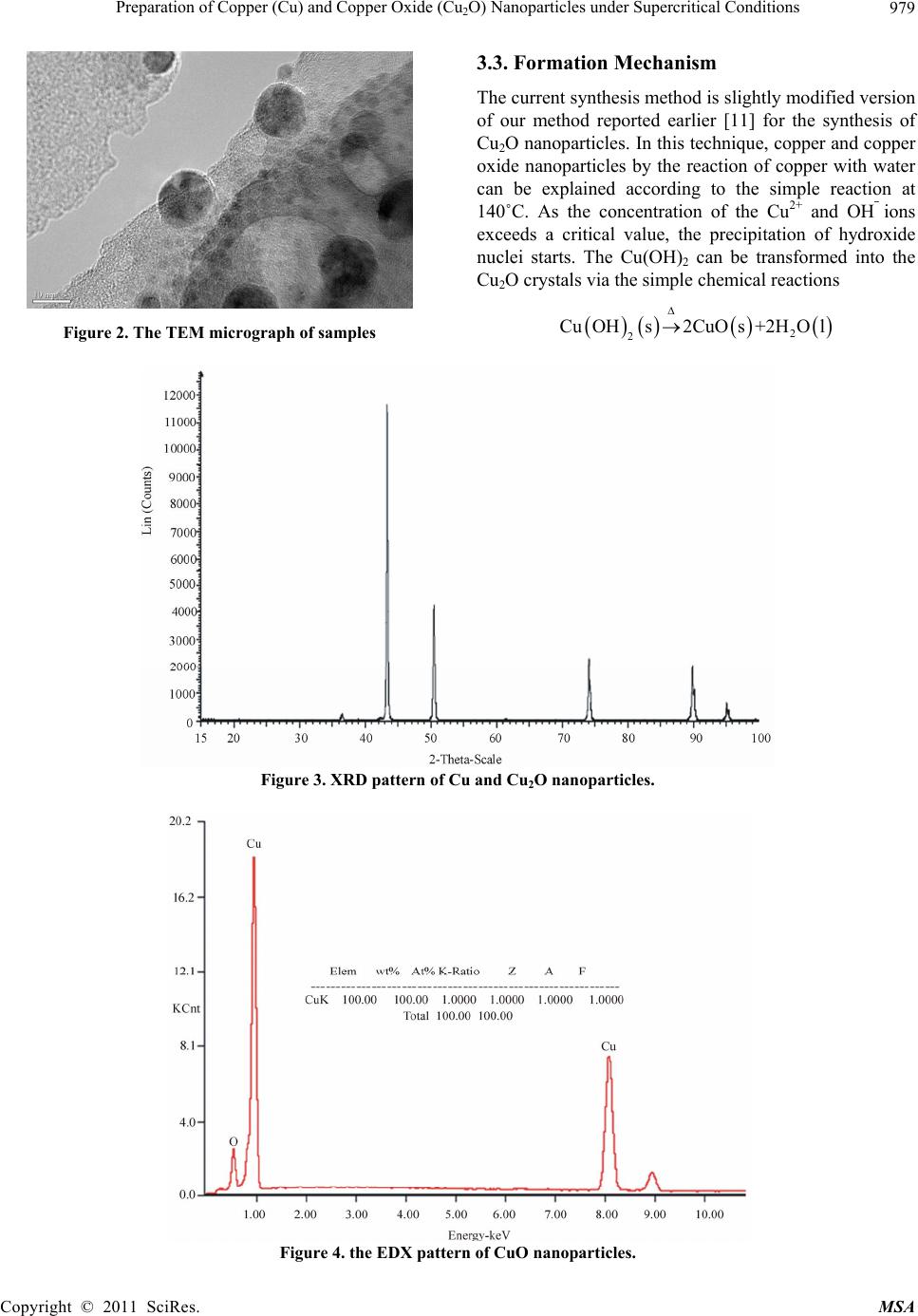 Preparation of Copper (Cu) and Copper Oxide (Cu2O) Nanoparticles under Supercritical Conditions Copyright © 2011 SciRes. MSA 979 Figure 2. The TEM micrograph of samples 3.3. Formation Mechanism The current synthesis method is slightly modified version of our method reported earlier [11] for the synthesis of Cu2O nanoparticles. In this technique, copper and copper oxide nanoparticles by the reaction of copper with water can be explained according to the simple reaction at 140˚C. As the concentration of the Cu2+ and OH⎯ ions exceeds a critical value, the precipitation of hydroxide nuclei starts. The Cu(OH)2 can be transformed into the Cu2O crystals via the simple chemical reactions 2 2 CuOHs2CuOs+2H Ol Figure 3. XRD pattern of Cu and Cu2O nanoparticles. Figure 4. the EDX pattern of CuO nanoparticles.  Preparation of Copper (Cu) and Copper Oxide (Cu2O) Nanoparticles under Supercritical Conditions Copyright © 2011 SciRes. MSA 980 The Cu metal on reaction with water slowly gives out hydrogen (g) and the liberated oxygen reacts with metal to give oxides as shown in the above reaction. The similar study has been reported earlier, where evolution of hydrogen has been documented [12-14]. The Cu reacts with oxygen and forms nuclei, which further serve as seed for Cu2O nanoparticles growth. The growth of nanopart- icles could be occurring at the small oxide nuclei that may be present on the metal surfaces. 4. Conclusions We have presented a very versatile, non toxic and envi- ronmental friendly approach for the synthesis of Cu and Cu2O nanoparticles at 140˚C without using any organics. This facile, reproducible and low cost approach should promise us a future large scale synthesis of nanostruc- tures for many applications in nanotechnology. The tech- nique could be extended and expanded to provide a con- venient strategy for the synthesis of oxide nanostructures. 5. Acknowledgements One of the authors (Shah M.A) is pleased to acknowledge the KACST, Riyadh and Bei Zhang for characterization of samples. REFERENCES [1] M. S. Niasari, F. Davar and N. Mir, “Synthesis and Characterization of Metallic Copper Nanoparticles via Thermal Decomposition,” Polyhedron, Vol. 27, No. 17, 2008, pp. 3514-3518. HHHdoi:10.1016/j.poly.2008.08.020 [2] M. J. G. Pacheo, J. E Morales-Sanchz, J. Gernandez and F. Ruiz, “Synthesis of Copper Nanoparticles Using Soy- beans as a Chelant Agent,” Material Letters, Vol. 64, No. 12, 2010, pp. 1361-1364. HHHdoi:10.1016/j.matlet.2010.03.029 [3] Y. Xi, C. Hu, P. Gao, R. Yang, X. E., X. Wang and B Wan, “Morphology and Pase Selective Synthesis of CuxO (x = 1, 2) Nanostructures and Their Catalytic Deg- radation Activity,” Material Science and Engineering B, Vol. 166, No. 1, 2010, 113-117. HHHdoi:10.1016/j.mseb.2009.10.008 [4] Y. He, “A Novel Solid Stabilized Emulsion Approach to Cuo Nanostructures Microspheres,” Materical Research Bull, Vol. 42, No. 1, 2007, pp. 190-195. HHHdoi:10.1016/j.materresbull.2006.05.020 [5] R. Motogoshi, T. Oku, A. Suzuki, K. Kkikchi, S. kihuchi, B. Jeyadwan and J. Cuya, “Fabrication and Characteriza- tion of Cupprious Oxide Fullerene Solar Cells,” Synthetic metals, Vol. 160, No. 11-12, 2010, pp. 1219-1222. doi:10.1016/j synthemet. 2010.03.012 [6] Q. Liu, X. Ling, H. Liu, J. Hang and Z. Hu “Solution Phases Synthesis of CuO Nanorods,” Material Chemistry and Physical, Vol. 98, No. 2-3, 2006, pp. 519-522. HHHdoi:10.1016/j.matchemphys.2005.09.073 [7] H. Wang, J. Zhang, J. J. Zhu and H. Y. Chen, “Prepara- tion of Copper Oxide Nanoparticles by Microwave Irra- diation,” Journal Crystal Growth, Vol. 244, No. 1, 2002, pp. 88-99. HHHdoi:10.1016/S0022-0248(02)01571-3 [8] Z. Meng, X. Xiaodang and Z. Millin, “Hydrothermal Synthesis of Sheaf Like CuO via Ionic Solids,” Materials Letters, Vol. 62, No. 3, 2008, pp. 385-388. HHHdoi:10.1016/j.matlet.2007.05.046 [9] F. Xu, W. Ji, Z. Shen, S. H. Tang, X. R. Ye and Z. A. Jia, “Preparation and Characterization of CuO Nanocrystals,” Journal Solid State Chemistry, Vol. 147, No. 2, 1999, pp. 516-519. HHHdoi:10.1006/jssc.1999.8409 [10] M. A. Al-Mamun, Y. Kusumoto and M. Muruganandham, “Simple New Synthesis of Cu Nanoparticles in Water/Ac- etoninitrile Mixed Solvents and Their Characterization,” Materials Letters, Vol. 63, No. 23, 2004, pp. 2007-2009. HHHdoi:10.1016/j.matlet.2009.06.037 [11] M. A. Shah and F. Al-Nowasir, “A Green and an Envi- ronmentally Benign Approach to Prepare Cu2O Nanocry- stals and Their Potential Applications”, International Journal of Nanoparticles, Vol. 4, No. 1, 2011. [12] M. A. Shah, A. Quarshi, “Novel Surfactant Free Synthesis of MgO Nanoflakes,” Journal Alloys and Compdounds, Vol. 482, No. 1-2, 2009, pp. 548-552. HHHdoi:10.1016/j.jallcom.2009.04.129 [13] W. Jisen, Y. Jinkai, S. Jinjuan and B. Ying, “Synthesis of Copper Oxide Nanocrystals and the Growth Mechanism of Copper Oxide Nanorods,” Materials and Design, Vol. 25, No. 7, 2004, pp. 625-629. HHHdoi:10.1016/j.matdes.2004.03.004 [14] J. S. Wang, J. K. Yang, J. J. Sun and Y. Bio, “Synthesis of Copper Oxide Nanocrystals and the Growth Mecha- nism of Copper Oxide Nanorods,” Materials and Design, Vol. 25, No. 7, 2004, pp. 625-629. |

