Journal of Applied Mathematics and Physics
Vol.04 No.02(2016), Article ID:63503,6 pages
10.4236/jamp.2016.42022
Comparison of the Levels of Five Heavy Metals in Human Urine and Sweat after Strenuous Exercise by ICP-MS
Shengxiang Tang1, Xinzhe Yu2, Cinan Wu3*
1College of Life Science, Guizhou University, Guiyang, China
2College of Pharmaceutical Science, Guizhou University, Guiyang, China
3Zunyi Normal College, Zunyi, China



Received 6 December 2015; accepted 13 February 2016; published 17 February 2016
ABSTRACT
Inductively coupled plasma mass spectrometry (ICP-MS) was used to measure the levels of trace heavy metals in human urine and sweat, to compare the performance of ICP-MS using three sample processing methods, namely direct dilution, wet digestion, and microwave digestion. The results showed that the wet digestion ICP-MS method has the highest accuracy (relative standard deviation ≤10%) and is more useful for measuring the levels of trace heavy metals in urine and sweat. Hence, we used this method to compare the levels of the five trace heavy metals, namely chromium, copper, zinc, cadmium, and lead, in the urine and sweat of adults after strenuous exercise. The results showed that the levels of these five trace heavy metals in sweat were significantly higher than those in urine after strenuous exercise (The differences between the measurements in urine and those in sweat were significant P ≤ 0.01). The results suggested that exercise-induced sweating can effectively remove the harmful heavy metals from the human body.
Keywords:
Strenuous Exercise, Urine, Sweat, ICP-MS, Heavy Metals

1. Introduction
Physical exercise can enhance blood circulation and metabolic rate [1]. The secondary metabolites and harmful waste products produced by the body during exercise are excreted through sweat and urine [2] [3]. Urination is crucial for the regulation of the body’s fluid and electrolyte balance, as well as for the removal of metabolic wastes from the body [4]. Perspiration can dissipate body heat via evaporation and remove a large amount of harmful substances from the body in the form of inorganic salts [5] [6]. Harmful heavy metals can also be excreted from the human body through these pathways.
Heavy metals are biologically unavailable and are non-degradable elements that can destroy protein and enzyme activities after ingestion into the body, in which excessive accumulation of heavy metals can lead to chronic poisoning [7]. Chromium is widely used in various industries such as metallurgy, electroplating, and leather tanning. Trivalent chromium (Cr3+) and hexavalent chromium (Cr6+) are the most common forms of Cr to which humans are exposed, with hexavalent Cr having the highest level of toxicity. Additionally, Cr and its compounds can enter the human body via direct contact and ingestion and can cause great harm to the respiratory and digestive systems [8] [9]. Copper is an important non-ferrous metal applied in construction, manufacturing, electrical, and military industries. Excessive intake of Cu in the body can easily lead to Menke’s and Wilson’s disease [10] [11]. Zinc is not only an important metal in chemical, electroplating, and alloy industries but also an essential element for human health, and both excessive Zn intake and Zn deficiency can damage human health [12]. Cadmium is widely applied in the semiconductor and nuclear industries. However, Cd is also widely recognized as a heavy metal environmental pollutant and a highly carcinogenic substance, which can damage and disease the respiratory, reproductive, immune, and blood circulatory systems of the human body [13] [14]. Lead is one of the earliest metals used by humans and is currently widely applied in batteries, paints, and radiation protection. However, Pb is also recognized as a highly carcinogenic heavy metal that causes great damage to the heart, kidney, blood, nervous system, and digestive system [15] [16]. Some of these five heavy metals are essential trace elements found in the human body (e.g., Cr3+, Cu, and Zn), whereas others are non-essential elements (e.g., Cr6+, Cd, and Pb). However, excessive accumulation of these heavy metals in the body can adversely affect human health. Therefore, excessive amounts of these five heavy metals in the human body need to be removed through normal mechanisms of metabolism to maintain the balance of trace elements.
In this study, we investigated the effects of perspiration and urination on the balance of heavy metals in the human body by comparing the levels of the five trace heavy metals, namely Cr, Cu, Zn, Cd, and Pb, in the sweat and urine excreted from the body after physical exercise.
2. Materials and Methods
2.1. Selection of Test Participants
The test subjects were healthy, adult males selected from among sophomore undergraduates enrolled in Guizhou University. All test participants volunteered for the study after being clearly informed of the experimental content and purposes. Before the experiments, the test participants were determined to be enrolled students at the North Campus of Guizhou University, who lived on campus, had similar diets, and were not consumers of alcohol or tobacco, nor of vitamin or mineral supplements. The study was performed in full accordance with the ethical and safety standards for human physiological experiments [17].
2.2. Sample Collection
The experiment was conducted in a climate-controlled indoor stadium. Prior to initiating physical exercise, the participants were directed to urinate and to cleanse their upper bodies with ultrapure water (Thermo Barnstead GenPure xCAD Ultrapure Water System). Then, the participants engaged in a strenuous badminton session, with their upper bodies exposed to the elements for 2 hours, rehydrating with demineralized pure water during the exercise session. The polyethylene sample bottles were treated by immersion in 30% (v/v) HNO3 for more than 48 hours and repeatedly washed with ultrapure water six times, followed by natural air-drying prior to use. These bottles were used to collect more than 20 mL of sweat from each participant by scraping. After the exercise session, urine samples of more than 20 mL each were collected from each participant. All the samples collected were allowed to stand for 30 minutes and then filtered using a 9-mm filter paper. Then, 10 mL of each sample was transferred into a test tube. The samples were stored at 4˚C until testing.
2.3. Sample Preparation and Elemental Analysis
Both sweat and urine are biological fluids containing urea, creatinine, uric acid, and other organic substances, as well as inorganic salts such as sodium, potassium, and chloride, in addition to water [2]. Hence, sample pro- cessing is needed to reduce interference by impurities during the elemental analysis. Currently, three types of sample processing methods are used for biological samples, namely direct dilution [18], wet digestion [19], and microwave digestion [20]. In direct dilution, 1 mL of sweat sample was diluted with 5% HNO3 to 50 mL. In wet digestion, 3 mL of HNO3 and 1 mL of HClO4 were added to 5 mL of sweat sample, followed by slow heating to 200˚C using a LabTech DB
The elemental analysis was performed using inductively coupled plasma mass spectrometry (ICP-MS) [18]- [21]. The mixed standard solution consisted of five heavy metals (Cr, Cu, Zn, Cd, and Pb) provided by the National Center of Analysis and Testing for Nonferrous Metals and Electronic Materials. The solution was diluted with dilute nitric acid solution (5% HNO3) into four concentration gradients of 0.00, 1.00, 10.00, and 100.00 μg/L. The solutions were then subjected to analysis using the Thermo X Series 2 ICP-MS with the following instrument parameters: carrier gas flow rate,
3. Results
3.1. Accuracy
The urine and sweat samples processed using the different methods were analyzed with ICP-MS in parallel to calculate the accuracy of each sample processing method while using the ultrapure water as a blank.
Table 2 shows the accuracy of each sample processing method for the sweat and urine samples, excluding invalid data with ≥90% errors. A higher relative standard deviation value indicates a lower sample concentration with weaker line intensity. In addition, the instrument performance and analytic conditions can also affect the actual accuracy of the measurement [22]. In the direct dilution method, Cr was detected in sweat with an accuracy of ≤10%, while the other measurements had poor results (≥10%). Pb was detected in urine with an accuracy of ≥10%, while other measurements had better results (≤10%). In the microwave digestion method, only a few elements were detected with a higher accuracy (Pb in sweat; Zn and Pb in urine), while the results of other measurements were not satisfactory (≥10%). Wet digestion is an ideal sample processing method with an accuracy of ≤10% for all measurement results.
Table 1. Parameters of microwave digestion system.
Table 2. Results of test for precision (n = 9).
Note: The precision is the relative standard deviation (RSD), RSD = S/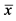 ×100%, S is the standard deviation,
×100%, S is the standard deviation, 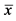 is the average.
is the average.
3.2. Comparison of the Levels of the Five Heavy Metals in Sweat and Urine
The wet digestion method with an accuracy of ≤10% was selected to compare the levels of these five heavy metals in sweat and urine excreted by the human body after physical exercise (Table 2). The SPSS 19 software was used for the statistical analysis.
After a 2-hour strenuous badminton session, all five trace heavy metals (Cr, Cu, Zn, Cd, and Pb) were detected in the urine and sweat samples from the healthy male adults using ICP-MS, after being processed by the wet digestion method (Table 3). The results showed that the differences in the levels of these five heavy metals in human urine and sweat were statistically significant. After physical exercise, the levels of Cr, Cu, Zn, Cd, and Pb in sweat were significantly higher than those in urine (P ≤ 0.01).
4. Discussion
Knowledge about the accumulation and excretion mechanisms of toxic elements in the human body is still limited. The conventional method of toxic element analysis for the human body is primarily analysis via detection of toxic elements by sampling blood and urine. Recent studies have shown that although human blood and urine do not completely reflect the cumulative capacity and ability of the body to remove the toxic elements, sweat may be a better test sample for monitoring the accumulation and removal of toxic elements in the body [2]. However, the number of studies that have analyzed the trace heavy metals in the body through sweat analysis is still limited. Moreover, a widely accepted sample collection and detection method for sweat, which impedes the promotion of sweat as a test sample in the monitoring of human health, is lacking. In this study, we selected the most commonly applied ICP-MS method after referring to recent studies on detection methods for trace elements in other biological samples (such as blood and urine) [18] [20] [23]. ICP-MS has several advantages such as small degree of interference, high accuracy, high detection speed, and good detection limits and linear dynamic range. In addition, ICP-MS can simultaneously perform multi-element analysis, detection, and quantitative measurement of isotopes [18] [20]. The comparison between these three sample processing methods (Table 2) indicates that the wet digestion ICP-MS method is more suitable for measuring trace elements in human sweat.
In this study, the detection results of trace heavy metals in human urine and sweat are similar to those from previous studies [3] [19]. The levels of these five trace heavy metals (Cr, Cu, Zn, Cd, and Pb) excreted in sweat were significantly higher than those excreted in urine after the strenuous exercise (Table 3). These trace heavy metals accumulated in the human body can be eliminated through perspiration and urination to maintain the balance of trace elements in the body and prevent excessive accumulation of heavy metals, which are hazardous to human health. Meanwhile, the accumulated heavy metals cannot be utilized or degraded by the human body and can only be excreted via limited mechanisms. Hence, the loss of balance between intake and removal will cause the accumulation of heavy metals in the body, which destroys homeostasis and results in the dysfunction of bodily systems [7]. The human body has two major sources of heavy metal intake. The first source is the mother, during infancy, and the second source is daily exposure. Daily exposure is the main source of heavy metals in the body for most people. The participants in this study were not living in areas of heavy industrial production but in areas rich in mineral resources (e.g., coal resources). In recent years, improper mining has caused environmental damage, in which Cd, Pb, Cu, Zn, and Cr are the representatives of heavy metals polluting the environment [24] [25]. Therefore, heavy metals entering the human body via contact and ingestion have to be eliminated via the body’s own mechanisms to maintain good health.
Table 3. Determination results of human sweat and urine.
Note: Date are measured value ± standard deviation, **P ≤ 0.01.
Current studies have indicated that physical exercise can enhance the removal of trace elements from the human body; however, these studies mainly involved the essential trace elements, and the studies on heavy metals are rare and majorly focused on Cu and Zn (heavy metals that are beneficial to the human body) [26] [27]. In this study, we drew the conclusions from previous studies to further investigate the ability of sweat and urine to excrete heavy metals from the human body during physical exercise. The test results (Table 3) indicated that sweat has a higher capacity than urine in removing these five heavy metals (Cr, Cu, Zn, Cd, and Pb) from the human body after strenuous exercise. The results not only showed that physical exercise has a significant effect on the balance of trace elements but also indicated that perspiration during physical exercise can effectively remove the toxic heavy metals from the body and reduce the accumulation of trace heavy metals in the body. According to the conclusion drawn from this study, we suggest that people living in these areas may have ingested the trace heavy metals from their polluted environment; thus, they should actively participate in physical exercise to enhance the detoxification capability of their bodies and reduce the damages caused to their bodies by heavy metals. However, our study has certain shortcomings due to the small sampling size and geospatial limitations. Hence, a further in-depth study is needed to elaborate the principle and efficacy of physical exercise in removing the harmful heavy metals from the human body.
Acknowledgements
The study was supported by the Graduate Innovation Fund of Guizhou University (Agricultural Research 2015017). We would like to thank the Physical Education College of Guizhou University for providing the human and material resources. In addition, we would like to express our gratitude to the Geological Laboratory of the
Conflict of Interest
The authors declare that they have no conflict of interest.
Cite this paper
Shengxiang Tang,Xinzhe Yu,Cinan Wu, (2016) Comparison of the Levels of Five Heavy Metals in Human Urine and Sweat after Strenuous Exercise by ICP-MS. Journal of Applied Mathematics and Physics,04,183-188. doi: 10.4236/jamp.2016.42022
References
- 1. Opondo, M.A., Sarma, S. and Levine, B.D. (2015) The Cardiovascular Physiology of Sports and Exercise. Clinics in Sports Medicine, 34, 391-404. http://dx.doi.org/10.1016/j.csm.2015.03.004
- 2. Genuis, S.J., Beesoon, S., Birkholz, D. and Lobo, R.A. (2012) Human Excretion of Bisphenol a: Blood, Urine, and Sweat (BUS) Study. Journal of Environ-mental & Public Health, 2012, Article ID: 185731. http://dx.doi.org/10.1155/2012/185731
- 3. Genuis, S. J., Birkholz, D., Rodushkin, I. and Beesoon, S. (2011) Blood, Urine, and Sweat (BUS) Study: Monitoring and Elimination of Bioaccumulated Toxic Elements. Archives of Environmental Contamination & Toxicology, 61, 344- 357. http://dx.doi.org/10.1007/s00244-010-9611-5
- 4. Bouatra, S., Aziat, F., Mandal, R., Guo, A.C., Wilson, M.R. and Knox, C., et al. (2013) The Human Urine Metabolome. Plos One, 8, e73076. http://dx.doi.org/10.1371/journal.pone.0073076
- 5. Shin-Ya, H., Yoshida, T., Tsuneoka, H., Nakai, S. and Ito, T. (2004) Effect of Sports Wear on Thermoregulatory Response During Exercise in a Hot Environment. Japanese Journal of Physical Fitness & Sports Medicine, 53, 347-355. (In Japanese)
- 6. Pluim, B.M., Racinais, S. and Périard, J.D. (2015) Blood, Sweat and Tears: Training and Competing in the Heat. British Journal of Sports Medicine, 49, 1161. http://dx.doi.org/10.1136/bjsports-2015-095113
- 7. Mishra, S., Dwivedi, S.P. and Singh, R.B. (2010) A Review on Epigenetic Effect of Heavy Metal Carcinogens on Human Health. Open Nutraceuticals Journal, 3, 188-193. http://dx.doi.org/10.2174/18763960010030100188
- 8. Shadreck, M. and Mugadza, T. (2013) Chromium, an Essential Nutrient and Pollutant: A Review. African Journal of Pure & Applied Chemistry, 7, 310-317.
- 9. Ye, X., Cao, Z.J., Wang, Q., Qu, Y.L. and Cai, J.Y. (2015) Chromium Exposure-Related Biomarkers and Its Application in Assessment on Health Effects. Journal of Environment and Health, 32, 336-369. (In Chinese)
- 10. Stern, B. R., Solioz, M., Krewski, D., Aggett, P., Aw, T.C., Baker, S., et al. (2007) Copper and Human Health: Biochemistry, Genetics, and Strategies for Modeling Dose-Response Relationships. Journal of Toxicology & Environmental Health Part B, 10, 157-222. http://dx.doi.org/10.1080/10937400600755911
- 11. Schlief, M.L., Tim, W., Ann Marie, C., Holtzman, D.M. and Gitlin, J.D. (2006) Role of The Menkes Copper-Transporting Atpase in Nmda Receptor-Mediated Neuronal Toxicity. Proceedings of the National Academy of Sciences of the United States of America, 103, 14919-14924. http://dx.doi.org/10.1073/pnas.0605390103
- 12. Chasapis, C.T., Loutsidou, A.C., Spiliopoulou, C.A. and Stefanidou, M.E. (2012) Zinc and Human Health: An Update. Archives of Toxicology, 86, 521-534. http://dx.doi.org/10.1007/s00204-011-0775-1
- 13. Yong, F., Sun, X., Yang, W., Ning, M., Xin, Z., Jin, F., et al. (2014) Concentrations and Health Risks of Lead, Cadmium, Arsenic, and Mercury in Rice and Edible Mushrooms in China. Food Chemistry, 147, 147-151.
- 14. Zhang, W.L., Yu, D., Zhai, M.M. and Qi, S. (2014) Cadmium Exposure and Its Health Effects: A 19-Year Follow-Up Study of a Polluted Area in China. Science of the Total Environment, 470-471, 224-228. http://dx.doi.org/10.1016/j.scitotenv.2013.09.070
- 15. Zhang, Y., Geng, C. and Cai, C. (2013) Review on Models for Lead Exposure on Human Health Risk Assessment. Environmental Chemistry, 6, 943-951. (In Chinese)
- 16. Mason, L.H., Harp, J.P. and Han, D.Y. (2014) Pb Neurotoxicity: Neuropsychological Effects of Lead Toxicity. Biomed Research International, 2014, 1-8. http://dx.doi.org/10.1155/2014/840547
- 17. Westra, A.E. and Beaufort, I.D. (2013) Improving the Helsinki Declaration’s Guidance on Research in Incompetent Subjects. Journal of Medical Ethics, 41, 278-280. http://dx.doi.org/10.1136/medethics-2013-101496
- 18. Ying, L., Kippler, M., Harari, F., Grandér, M., Palm, B., Nordqvist, H., et al. (2014) Alkali Dilution of Blood Samples for High Throughput ICP-MS Analysis—Comparison with Acid Digestion. Clinical Biochemistry, 48, 140-147.
- 19. Hou, K., Ji, H.B., Li, H.T., Wang, L.Z., Wang, W.Y., Liu, Q.B., et al. (2009) Determination of Life Elements in Hair and Urine with Brick-Tea Fluorosis in Inner Mongolia by ICP—OES. Spectroscopy and Spectral Analysis, 29, 1100- 1103. (In Chinese)
- 20. Cruz, S.M., Schmidt, L., Nora, F.M.D., Pedrotti, M.F., Bizzi, C.A., Barin, J.S., et al. (2015) Microwave-Induced Combustion Method for the Determination of Trace and Ultratrace Element Impurities in Graphite Samples by ICP- OES and ICP-MS. Microchemical Journal, 123, 28-32. http://dx.doi.org/10.1016/j.microc.2015.05.008
- 21. Zhang, X.W., Li, Y.H., Yang, L.S., Li, H.R. and Wang, W.Y. (2010). Determination of Five Trace Elements (Pb, Cd, Se, As and Hg) in Human Whole Blood by Temperature-Controllable Wet Digestion and ICP-MS Technique (in Chinese). Spectroscopy and Spectral Analysis, 7, 1972-1974.
- 22. Xin, R.X. (2011) Plasma Emission Spectrum Analysis. 3rd Edition, Chemical Industry Press, Beijing, 123-124, 156-157. (In Chinese)
- 23. Lech, T. (2014) ICP OES and CV AAS in Determination of Mercury in an Unusual Fatal Case of Long-Term Exposure to Elemental Mercury in a Teenager. Forensic Science International, 237, e1-e5. http://dx.doi.org/10.1016/j.forsciint.2014.02.015
- 24. Wu, Y., Liu, C. and Tu, C. (2008). Distribution and Sequential Extraction of Some Heavy Metals in Urban Soils of Guiyang City, China. Chinese Journal of Geochemistry, 4, 401-406. http://dx.doi.org/10.1007/s11631-008-0401-y
- 25. Xiaoyan, L.I., Cao, Y. and Le, Q.I. (2012) The Distribution Characteristics of Heavy Metals in Guiyang Urban Soils. Chinese Journal of Geochemistry, 31, 174-180. http://dx.doi.org/10.1007/s11631-012-0564-4
- 26. Soria, M., González-Haro, C., Ansón, M. and Escanero, J.F. (2015) Plasma Levels of Trace Elements and Exercise Enduced Stress Hormones in Well-Trained Athletes. Journal of Trace Elements in Medicine & Biology, 31, 113-119. http://dx.doi.org/10.1016/j.jtemb.2015.04.004
- 27. Akio, H., Hiromi, W., Momoko, C., Yutaka, I., Matatoshi, K., Naoto, K., et al. (2002) Seasonal Variation of Trace Element Loss to Sweat during Exercise in Males. Environmental Health & Preventive Medicine, 7, 60-63. http://dx.doi.org/10.1007/BF02897331


