Paper Menu >>
Journal Menu >>
 J. Biomedical Science and Engineering, 2008, 1, 116-120 Published Online August 2008 in SciRes. http://www.srpublishing.org/journal/jbise JBiSE An image analysis method for quantification of he- patic perfusion based on contrast-enhanced ultra- sound imaging Yi-xin Li, Fang Yang & Ning Gu* State Key Laboratory of Bioelectronics, Jiangsu Laboratory for Biomaterials and Devices, School of Biological Science and Medical Engineering, Southeast University, Nanjing 210096, PR China. *Correspondence should be addressed to N. Gu (guning@seu.edu.cn). ABSTRACT Information about hepatic perfusion is used in clinical liver disease diagnosis. An image analy- sis system can help physicians make efficient and accurate diagnosis. The objective of this study is to propose an image analysis method for the quantification of the hepatic perfusion based on contrast-enhanced ultrasound imaging (CEUI). The proposed method contains frame selection, image registration, digital subtraction and grey-scale calculation. Then, by processing an image sequence, a time-intensity curve (TIC) for hepatic perfusion is derived. The kernel of this image analysis technology is digital subtrac- tion and its accuracy is improved by frame selec- tion and image registration. The advantage of this method is that it can obtain the perfusion information of the whole liver w hich is rarely ob- tained by traditional image analysis technology; therefore, it is a supplement of the traditional image analysis method. This method is applied on the quantification of a rabbit’s hepatic perfu- sion and the result shows the efficiency of it. Keywords: Hepatic perfusion quantification, Image analysis, Image registration, Digital sub- traction 1. INTRODUCTION Blood perfusion describes the amount of blood delivered to the capillary beds of a block of tissue in a certain pe- riod of time and its quantity determines whether the en- ergy status of the tissue is likely to become compromised [1]. Thus, when blood perfusion of the tissue is abnormal, many diseases will be caused and on the contrary, when diseases occur, abnormal blood perfusion will be present. Various liver diseases lead to significant alterations of hepatic perfusion and quantification of hepatic perfusion has the potential to improve the assessment and manage- ment of liver diseases [2]. Consequently, much effort has been done to quantify hepatic perfusion with the help of different imaging modalities, such as MRI, CT and so on [1-3]. As a versatile, non-invasive, low risk, low cost and portable real-time imaging technique, ultrasound is the most frequently used imaging modality [4]. CEUI is a new ultrasonic imaging modality developed with the in- troduction of ultrasonic contrast agent (UCA) and the evolution of corresponding imaging technologies [5]. Since CEUI can image the blood flow, it has been exten- sively used in clinical diagnosis [4,6,7]. One of the most important possible applications in the clinic is that it is more and more used to assess and quantify the blood flow and blood perfusion of various tissues and organs [8--12]. Efficient image analysis technology is needed during the quantification of hepatic perfusion using CEUI. The conventional analysis method is to measure mean signal intensity (mean grey-scale) of the pixels in a manual placed, equally sized and shaped region of interest (ROI) in each of the image frame, and then the TIC of the ROI is obtained [1,3,11]. Some important perfusion parame- ters, for example the mean transit time (MTT) and blood volume fraction (BVF), can be obtained easily using this analysis method. Despite its efficiency, there are still is- sues to be addressed. Since the manual placed ROI is usu- ally local, the perfusion information of the whole liver such as the three vascular phases of the liver perfusion, as an important perfusion parameter, can hardly be derived directly from the TIC of the local ROI. As a supplement of the traditional image analysis method, to reflect the three vascular phases of the liver perfusion directly, the proposed image analysis method in this paper focuses on the derivation of the TIC of all the automatically segmented contrast-enhanced areas rather than that of the local ROI. The segmentation is imple- mented by subtracting the baseline image (background) from the contrast-enhanced image. After the subtraction, all the contrast-enhanced areas (all the pixels with a grey- scale above zero in the subtraction image) are obtained. To verify the method, a rabbit’s hepatic perfusion video is acquired using CEUI and is analyzed using the proposed method. The result of the application shows the efficiency SciRes Copyright © 2008  Y. X. Li et al. / J. Biomedical Science and Engineering 1 (2008) 116-120 117 SciRes Copyright © 2008 JBiSE of the proposed method. The layout of the paper is as follows: Section 2 pre- sents the theoretical framework of the image analysis method. The experiment is described in section 3 along with results and discussion in section 4. The paper con- cludes in section 5. 2. METHOD The main processes of this method can be described as follows: 1) a frame before the injection of the UCA is selected as the baseline image, and then one appropriate frame for each second after the injection is selected as the contrast-enhanced image, and all the contrast-enhanced images construct the image sequence; 2) for each of the contrast-enhanced image, the baseline image is registered and subtracted to acquire all the contrast-enhanced areas in the image; 3) the mean signal density of all the con- trast-enhanced areas is calculated for every contrast- enhanced image, and then the TIC can be derived at last. 2.1. Selection of appropriate frames Tissue motion induced by respiration makes the frames at different time point have lots of motion artefacts which will heavily decrease the efficiency of digital subtraction. Therefore, after the first frame is selected, all the other selected frames should have the least motion with the first frame. The purpose of this step is to reduce the global motion between the frames. In this method, the energy of the histogram of differences (EHD), proved to be compu- tationally cheap and yield accurate results [13], is applied as the similarity measurement criterion and the frame with highest EHD has the greatest similarity with the first frame. The theory of EHD can be briefly described as follows: 2 () () G EHD gG dHg =− Μ= ∑ (1) Here MEHD (d ) is the energy of the histogram of differ- ences, g∈[-G, G] ⊆ [-255,255] ⊂ Z. H(g) is defined as: 1 ()( (,),) d W H gIxyg KL δ =∑ (2) Here K, L are the width and height of the block to be processed (the image frame in this study); Id (x, y) = I (x, y) − I0 (x+∆x, y+∆y) is the subtracted image, where I (x, y) is the contrast-enhanced image and I0 (x+∆x, y+∆y) is the aligned baseline image. δ (x, y) is the Kronecker delta function. 1, (, )0, x y xy x y δ = ⎧ ⎨ ≠ ⎩ (3) 2.2. Image registration and digital subtraction The global artefacts are reduced by selecting the appro- priate frame; however, the local motion artefacts still exist between the contrast-enhanced image and baseline image. Figure 1. The schematic diagram for the selections of control points. To remove the local motion artefacts, the baseline image is registered by finding the local motion vectors of the pixels in the baseline image and then aligning them. The most ideal method for registration is to align every pixel; however the computational cost makes this hard to be implemented. Given to the continuous motion of neigh- bouring area, an alternative approach is to align the se- lected control points first and then all the other pixels are aligned by interpolation. To acquire the control points, both the baseline image and the contrast-enhanced image are divided into an array of rectangular tiles as shown in Figure 1 and each vertex of the grid is selected as control point. Based on the selection of control points, the whole process of the registration and digital subtraction can be described as follows: Process 1: Control points of both the images are selected as the centres and R×R pixels around each centre are set to be the ROIs. The ROI of baseline image is denoted as Ab(i, j), and the corresponding area of the contrast-enhanced im- age is denoted as Ac(i, j) (i, j = 1 - R). Process 2: Ab(i, j) is shifted according to the rules of three-step search algorithm [14] and the position is located where Ab(i+∆x, j+∆y) and Ac(i, j) had the largest MEHD (d). The centre of Ab(i+∆x, j+∆y) is recorded and pixel unit (-∆x, - ∆y) is defined as the local motion vector of the control point, where ∆x and ∆y are the number of pixels that have been shifted in x (horizontal) and y (vertical) directions. All the control points are processed by repeating this process. Process 3: Figure 2. The bilinear interpolation. 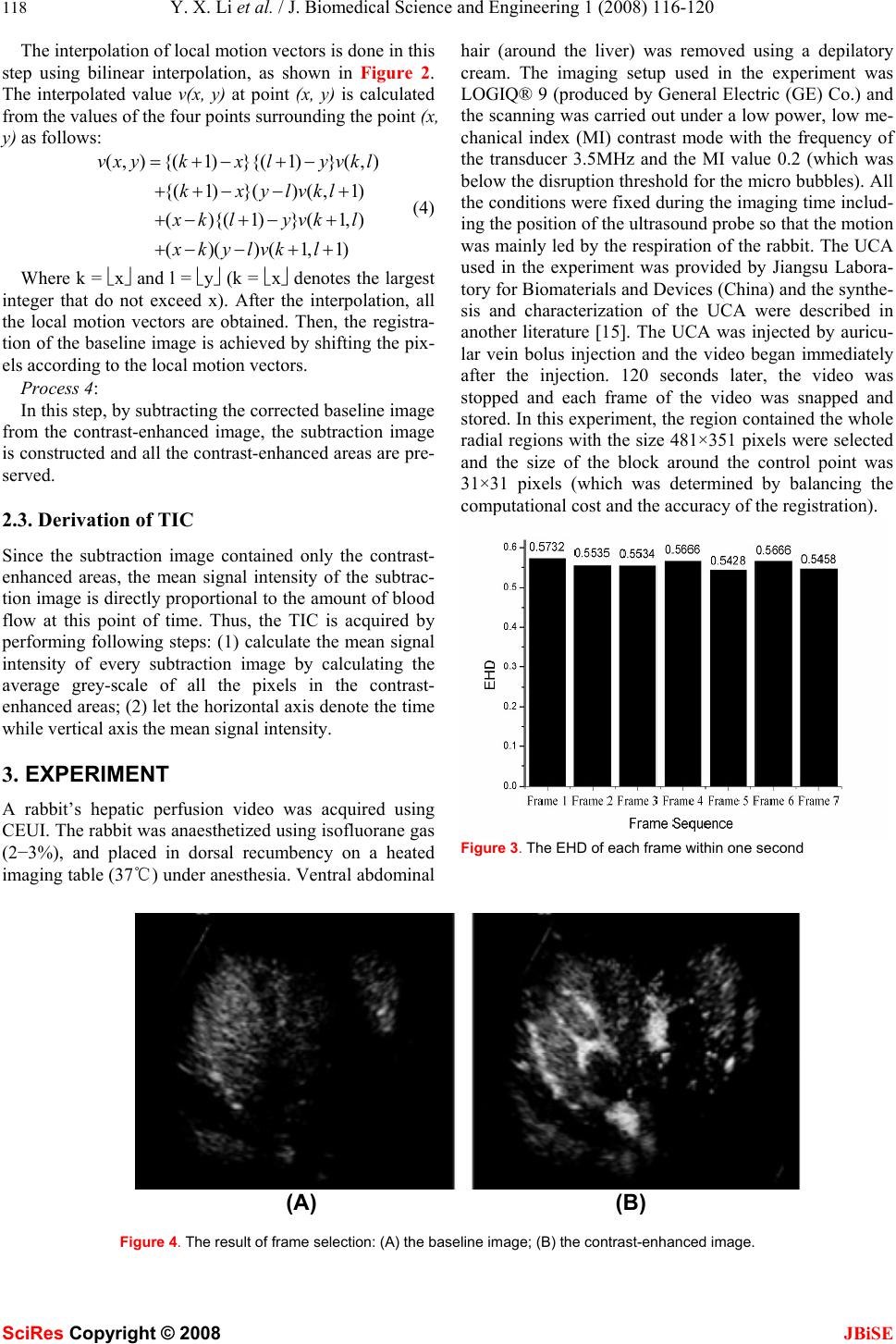 118 Y. X. Li et al. / J. Biomedical Science and Engineering 1 (2008) 116-120 SciRes Copyright © 2008 JBiSE The interpolation of local motion vectors is done in this step using bilinear interpolation, as shown in Figure 2. The interpolated value v(x, y) at point (x, y) is calculated from the values of the four points surrounding the point (x, y) as follows: (,){(1)}{(1)}(,) {( 1) }()(,1) (){(1)}(1,) ()()(1,1) vxykx lyvkl kxylvkl x klyvkl xkylvk l =+−+− ++− −+ +− +−+ +−−+ + (4) Where k = ⎣x⎦ and l = ⎣y⎦ (k = ⎣x⎦ denotes the largest integer that do not exceed x). After the interpolation, all the local motion vectors are obtained. Then, the registra- tion of the baseline image is achieved by shifting the pix- els according to the local motion vectors. Process 4: In this step, by subtracting the corrected baseline image from the contrast-enhanced image, the subtraction image is constructed and all the contrast-enhanced areas are pre- served. 2.3. Derivation of TIC Since the subtraction image contained only the contrast- enhanced areas, the mean signal intensity of the subtrac- tion image is directly proportional to the amount of blood flow at this point of time. Thus, the TIC is acquired by performing following steps: (1) calculate the mean signal intensity of every subtraction image by calculating the average grey-scale of all the pixels in the contrast- enhanced areas; (2) let the horizontal axis denote the time while vertical axis the mean signal intensity. 3. EXPERIMENT A rabbit’s hepatic perfusion video was acquired using CEUI. The rabbit was anaesthetized using isofluorane gas (2−3%), and placed in dorsal recumbency on a heated imaging table (37℃) under anesthesia. Ventral abdominal hair (around the liver) was removed using a depilatory cream. The imaging setup used in the experiment was LOGIQ® 9 (produced by General Electric (GE) Co.) and the scanning was carried out under a low power, low me- chanical index (MI) contrast mode with the frequency of the transducer 3.5MHz and the MI value 0.2 (which was below the disruption threshold for the micro bubbles). All the conditions were fixed during the imaging time includ- ing the position of the ultrasound probe so that the motion was mainly led by the respiration of the rabbit. The UCA used in the experiment was provided by Jiangsu Labora- tory for Biomaterials and Devices (China) and the synthe- sis and characterization of the UCA were described in another literature [15]. The UCA was injected by auricu- lar vein bolus injection and the video began immediately after the injection. 120 seconds later, the video was stopped and each frame of the video was snapped and stored. In this experiment, the region contained the whole radial regions with the size 481×351 pixels were selected and the size of the block around the control point was 31×31 pixels (which was determined by balancing the computational cost and the accuracy of the registration). Figure 3. The EHD of each frame within one second (A) (B) Figure 4. The result of frame selection: (A) the baseline image; (B) the contrast-enhanced image. 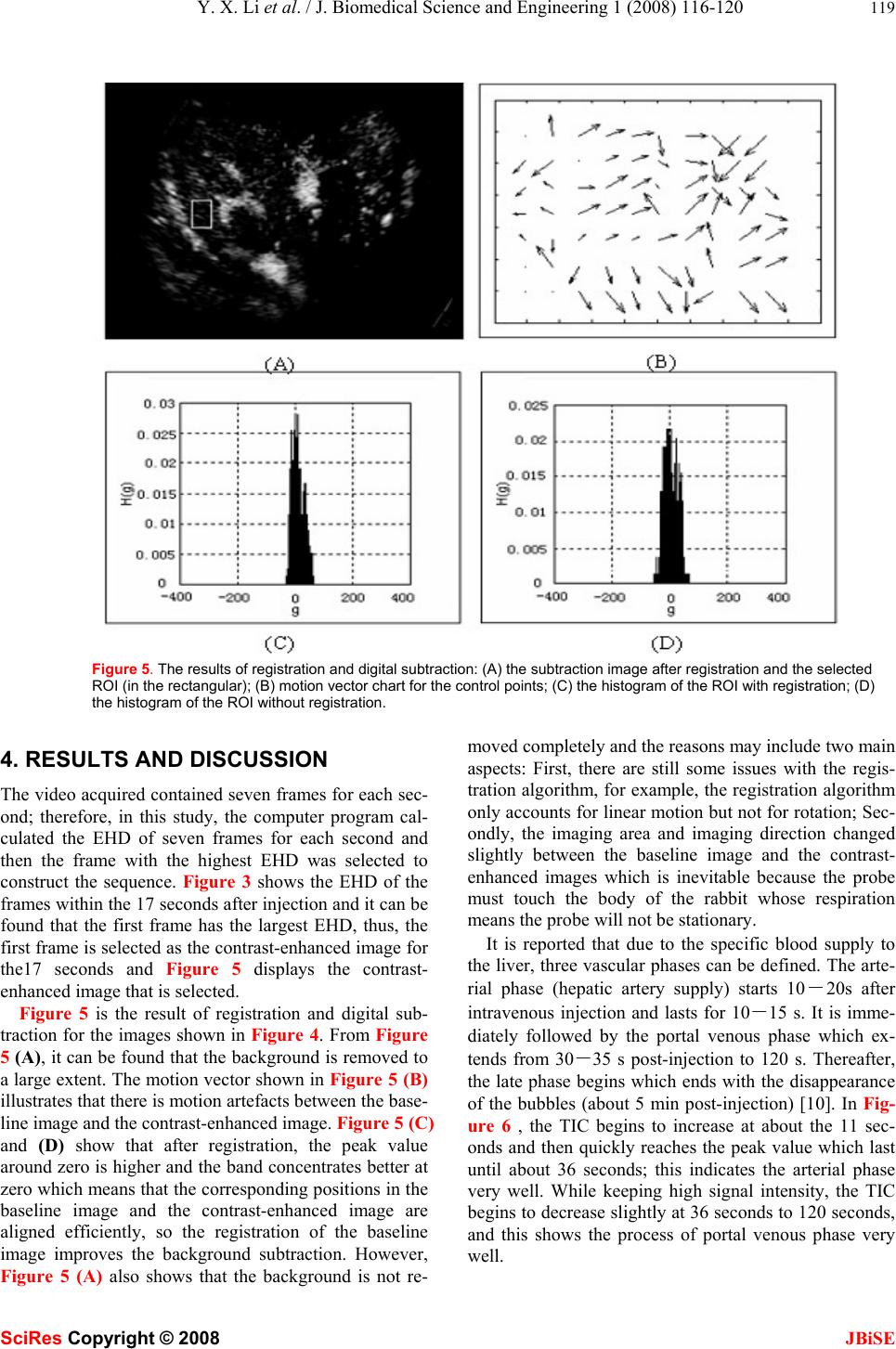 Y. X. Li et al. / J. Biomedical Science and Engineering 1 (2008) 116-120 119 SciRes Copyright © 2008 JBiSE Figure 5. The results of registration and digital subtraction: (A) the subtraction image after registration and the selected ROI (in the rectangular); (B) motion vector chart for the control points; (C) the histogram of the ROI with registration; (D) the histogram of the ROI without registration. 4. RESULTS AND DISCUSSION The video acquired contained seven frames for each sec- ond; therefore, in this study, the computer program cal- culated the EHD of seven frames for each second and then the frame with the highest EHD was selected to construct the sequence. Figure 3 shows the EHD of the frames within the 17 seconds after injection and it can be found that the first frame has the largest EHD, thus, the first frame is selected as the contrast-enhanced image for the17 seconds and Figure 5 displays the contrast- enhanced image that is selected. Figure 5 is the result of registration and digital sub- traction for the images shown in Figure 4. From Figure 5 (A), it can be found that the background is removed to a large extent. The motion vector shown in Figure 5 (B) illustrates that there is motion artefacts between the base- line image and the contrast-enhanced image. Figure 5 (C) and (D) show that after registration, the peak value around zero is higher and the band concentrates better at zero which means that the corresponding positions in the baseline image and the contrast-enhanced image are aligned efficiently, so the registration of the baseline image improves the background subtraction. However, Figure 5 (A) also shows that the background is not re- moved completely and the reasons may include two main aspects: First, there are still some issues with the regis- tration algorithm, for example, the registration algorithm only accounts for linear motion but not for rotation; Sec- ondly, the imaging area and imaging direction changed slightly between the baseline image and the contrast- enhanced images which is inevitable because the probe must touch the body of the rabbit whose respiration means the probe will not be stationary. It is reported that due to the specific blood supply to the liver, three vascular phases can be defined. The arte- rial phase (hepatic artery supply) starts 10-20s after intravenous injection and lasts for 10-15 s. It is imme- diately followed by the portal venous phase which ex- tends from 30-35 s post-injection to 120 s. Thereafter, the late phase begins which ends with the disappearance of the bubbles (about 5 min post-injection) [10]. In Fig- ure 6 , the TIC begins to increase at about the 11 sec- onds and then quickly reaches the peak value which last until about 36 seconds; this indicates the arterial phase very well. While keeping high signal intensity, the TIC begins to decrease slightly at 36 seconds to 120 seconds, and this shows the process of portal venous phase very well.  120 Y. X. Li et al. / J. Biomedical Science and Engineering 1 (2008) 116-120 SciRes Copyright © 2008 JBiSE Figure 6. The TIC of the rabbit’s hepatic perfusion ob- tained using the proposed method 5. CONCLUSION In this study, as a supplement of the traditional image analysis method, an image analysis method used to quan- tify the total hepatic perfusion using CEUI is proposed. The main contributions included in this method are frame selection, image registration, digital subtraction and grey-scale calculation. In the steps of frame selection and registration, EHD is selected as the criterion of simi- larity measure. The usefulness of the method is proved by applying it on the quantification of a rabbit’s hepatic perfusion. Since ultrasound is the most widely used im- aging modality, and as the development of the technol- ogy of CEUI, this method has great potential clinical value. In addition, it can also be used to quantify the perfusion of other organs such as the kidney. Although this method shows great potential advantages, there are still some issues to be addressed. First, the method is not accurate enough to make the background removed com- pletely which affects the efficiency of quantification; secondly, the program for this method available now is off-line, which hampers the real-time clinical application. Further works are needed to address these issues so that the method can be better used for clinical diagnosis. ACKNOWLEDGMENT The authors gratefully acknowledge the financial support given to this research from National Important Science Research Pro- gram of China (Nos. 2006CB933206) REFERENCES [1] D.L. Thomas, M.F. Lythgoe, G.S. Pell, et al. (2000) The meas- urement of diffusion and perfusion in biological systems using magnetic resonance imaging, Phys Med Biol, 45, 97–138. [2] R. Materne, B.E. Van Beers, A.M. Smith, et al. (2000) Non- invasive quantification of liver perfusion with dynamic computed tomography and a dual-input one-compartmental model. Clinical Science, 99, 517–525. [3] B.E. Van Beers, I. Leconte, R. Materne, et al. (2001) Hepatic perfusion parameters in chronic liver disease: dynamic CT meas- urements correlated with disease severity. AJR, 176, 667–673. [4] R. Thomas (2007) Focal liver lesions: Role of contrast-enhanced ultrasound. European Journal of Radiology, 64, 173–182. [5] E.G. Schutt, D.H. Klein, R.M. Mattrey, et al. (2003) Injectable microbubbles as contrast agents for diagnostic ultrasound imaging: the key role of perfluorochemicals. Angew Chem Int Ed, 42, 3218–3235. [6] C. Serra, M. Guido, M. Antonio, et al. (2007) Ultrasound assess- ment of vascularization of the thickened terminal ileum wall in Crohn’s disease patients using a low-mechanical index real-time scanning technique with a second generation ultrasound contrast agent. European Journal of Radiology, 62, 114–121. [7] T.D. Zordo, S.P. Mlekusch, G.M. Feuchtner, et al. (2007) Value of contrast-enhanced ultrasound in rheumatoid arthritis, European Journal of Radiology, 64, 222–230. [8] S.J. Rim, H. Leong-Poi, J.R. Lindner, et al. (2001) Quantification of cerebral perfusion with "real-time" contrast-enhanced ultra- sound. Circulation, 104, 2582-2587. [9] S. Malm, S. Frigstad, F. Helland, et al. (2005) Quantification of resting myocardial blood flow velocity in normal humans using real-time contrast echocardiography: A feasibility study. Cardio- vascular Ultrasound, 3, 16. [10] K. Wei, E. Le, J.P. Bin, et al. (2001) Quantification of renal blood flow with contrast-enhanced ultrasound, J Am Coll Cardiol, 37, 1135-1140. [11] R. Metoki, F. Moriyasu, N Kamiyama, et al. (2006) Quantifica- tion of hepatic parenchymal blood flow by contrast ultrasonogra- phy with flash-replenishment imaging. Ultrasound in Medicine & Biology, 32, 1459-1466. [12] T. Jansson, H.W. Persson, K. Lindström (1999) Estimation of blood perfusion using ultrasound, Journal of Engineering in Medicine, 213, 91-106. [13] E.H.W. Meijering, W.J. Niessen, M.A. (1999) Viergever Retro- spective Motion Correction in Digital Subtraction Angiography: A Review, IEEE Transactions on Medical Imaging, 18(1), 2-21. [14] T. Koga, K. Iinuma, A. Hirano, et al. (1981) Paper presented at the Motion compensated interframe coding for video conferenc- ing, New Orleans, LA, Nov-Dec. [15] F. Yang, A.Y. Gu, Z.P. Chen, et al. (2008) Multiple emulsion microbubbles for ultrasound imaging, Materials Letters, 62, 121- 124. |

