Chinese Medicine
Vol.4 No.3(2013), Article ID:37184,23 pages DOI:10.4236/cm.2013.43015
Houttuynia cordata Thunb: A Review of Phytochemistry and Pharmacology and Quality Control
College of Chemistry and Chemical Engineering, Central South University, Changsha, China
Email: *hongmeilu@csu.edu.cn
Copyright © 2013 Jiangang Fu et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received July 1, 2013; revised August 6, 2013; accepted August 21, 2013
Keywords: Houttuynia cordata Thunb; Phytochemical constituents; Pharmacological activity; Quality control
ABSTRACT
Houttuynia cordata Thunb is an important medicinal plant widely distributed in East Asia. The collected information is an attempt to cover recent developments in the pharmacology, phytochemistry and quality control of this species. During the past several decades, the medicinally important phyto-constituents have been identified including essential oil, flavonoids and other polyphenols, fatty acids and alkaloids. A survey of the literatures shows H. cordata possesses a variety of pharmacological activities including antiviral, antitumor, antimicrobial, anti-inflammatory, and antioxidative effects. Little attempt has been done to review the techniques used for its quality control. Future efforts should concentrate more on in vitro, in vivo studies and clinical trials in order to confirm traditional wisdom in the light of a rational phytotherapy. The information summarized here is intended to serve as a reference tool to practitioners in the fields of ethnopharmacology and natural products chemistry.
1. Introduction
There is a long history of herbal medicine in far Eastern countries; in particular, Chinese people have utilized herbs and plants to treat various diseases for more than 8000 years [1]. With the advance of modern medicine and drug research, chemical synthesis has replaced plants as the primary source of medicinal agents in industrialized countries. However, in 1985, the World Health Organization estimated that about 80% of the world’s population relied on traditional medicines including herb medicines for their primary health care needs [2]. Several aspects: 1) the high cost of chemical synthesis drug discovery, 2) the efficiency of herb medicine on complex illnesses such as cancer and cardiovascular disease, 3) the unique activities of herbal medicine aiming at the system level via interactions with a multitude of targets in the human body, make people return to the herbal medicine, a potential reservoir for new drugs [3].
Houttuynia cordata Thunb, the sole species in the genus Houttuynia that belongs to the Saururaceae family, is a flowering and perennial herb native to China, Japan, Korea and Southeast Asia. In China, this plant is distributed widely, eastwards to Taiwan, southwest to Yunnan and Tibet, and north to Shaanxi and Gansu. It grows optimally on moist, shady hillside, wayside and ridge of field with an altitude of 300 - 2600 m.
H. cordata is a well known traditionally used medicinal material in the indigenous medicine systems of Southeast Asia. It is commonly called Yu-Xing-Cao, Ji-Cai, historically called Cen-Cao (Wuyue Chunqiu), Ji (Mingyi Bielu), Zi-Bei-Yu-Xing-Cao (Lü Chan Yan Ben Cao), Zi-Ji (Jiuji Yifang), Zu-Zi (Bencao Gangmu), Zu-Cao (Xinxiu Bencao), Chou-Zhu-Cao (Yilin zuanyao··yaoxing), Ce-Er-Gen (Zunyifu Zhi), Zhu-Bi-Kong (Tianbao Bencao), Jiu-Jie-Lian (Lingnan Caiyao Lu), Zhe-Er-Gen or Fei-Xing-Cao (Guizhou Mingjian Fangyao Ji), and Chou-Xing-Cao (Quanzhou Bencao) in China; dokudame in Japan; E-Sung-Cho in Korea; Khaotong or Plu-khao in Thailand; giấp cá or diếp cá in Vietnam. It has the functions of relieving fever, resolving toxin, reducing swelling, draining pus and promoting urination [4]. During the period of the Severe Acute Respiratory Syndrome (SARS) outbreak, it was one of the ingredients in the SARS prevention formulas recognized by the Health Ministry of China. Recently, several studies also provided scientific data to support and unveil its anti-SARS [5], anti-inflammatory [6,7], anti-allergic [8,9], virucidal [10,11], antileukemic [12], anti-oxidative [13,14] and anti-cancer [15] activities. It was reported that H. cordata contains groups of such chemical components as flavones, essential oil and alkaloids [16].
Over the last few years, there has been a rapid increase in the information available on the structures and pharmacological activities of H. cordata (Figure 1). In this review, we present recent H. cordata plant research in three sections: phytochemistry, pharmacological activities and quality control. The information was mainly collected from databases (Scifinder, ISIWeb of Knowledge) and several books.
2. Morphological Description
H. cordata is herbaceous perennial plant growing to 20 - 80 cm, the flowers are greenish-yellow, borne on a terminal spike 2 - 3 cm long with 4 - 6 large white basal bracts; spike terminal yellowish-brown. The odour is fishy on rubbing and the taste is slightly adstringent (Figure 2).
3. Traditional Uses of H. cordata
The uses differ from one country to another. In China, H. cordata has been used to treat anisolobis sores. The documented folk uses and indications in China were listed in detail in Table 1. In Korea, it has been used for the treatment of cough, pneumonia, bronchitis, dysentery, dropsy, leukorrhea, uteritis, eczema, herpes simplex, acne, chronic sinusitis and nasal polyps [11,17]. In Thailand, it has been used for immune stimulization and as anticancer agent [18]. In Japan, it has been mainly used as diuretics [19] and also used for the treatment of stomach ulcers [20], the control of the infection [21], and as antimicrobial [22,23], antitumor [24], promoting agents for the production of an antibiotic substance by a strain of gram-positive, spore-bearing bacilli [25]. In India, the shoot has been used for the freshness, good sleep, heart disorders by Apatani who have traditionally settled in seven villages in the Ziro valley of Lower Subansiri district of Arunachal Pradesh in the Eastern Himalayan region of India [26]. Besides general medicinal uses, H. cordata is employed as food (Table 2) and cosmetic formulations. In Korea and Japan, H. cordata is frequently used in combination with other herbal medicines as cosmetic. Its extraction are used as cosmetic composition for preventing or treating wrinkle [27], preventing chapped skin [28], antiaging [29,30], improving the skin conditions [31], removing freckles and skin-whitening [30]. The fermented extract with other herbal medicine are used for alleviating atopic dermatitis [32] and other skin troubles [33], owing to the anti-inflammatory and skincalming effect, pruritus-alleviating effect, and humidifying effect of this composition. The extraction is also used for protecting or nourishing hair and preventing dandruff [34,35]. In addition, H. cordata is used to prepare the massage pack which is able to treat acne, chloasma, atopy, and freckle without leaving any scar [36].
4. Phytochemistry
To date, the majority of phytochemical studies on H. cordata have focused on three types, namely: essential oil, flavonoid and alkaloids components [16]. Recent pharmacological studies indicated the essential oil components in H. cordata possess anti-inflammatory, antibacterial and antiviral activities [6,10]. The flavonoid components revealed antineoplastic, antioxidant, antimutagenic and free radical scavenging capacity [14, 37,38]. Similarly, the alkaloid components demonstrated significant potent antiplatelet and cytotoxic activities [15].
4.1. Essential Oil
Most of previous studies were mainly focused on the chemistry of essential oil. The volatile oils of H. cordata were extracted by various methods, supercritical CO2 extraction [39,40], steam distillation [41-43], petroleum ether extraction [39,42], solid-phase microextraction [43], flash evaporation and simultaneous distillation-extraction [44,45], isolated by preparative HPLC [45,46], and analyzed qualitatively and quantitatively by GC , GCMS, GC-MS with on-column derivatization procedure [41] TLC [47,48], flash GC [49], combined gas-liquid chromatography and mass spectroscopy [48]. The considerable differences may depend on the extraction procedure, the season, the part, the dry process, the stage of development and the distinct habitat in which the plant has been collected [47,50-54]. A total of 346 volatile components were reported. They were composed mainly of terpenoids (27.0%), hydrocarbons (16.8%), esters (11.9%), alcohols (11.6%), ketones (7.2%), aldehydes (4.9%), acids (3.8%), phenols (1.7%), aethers (0.9%) and mixed compounds (14.2%). Among these, which were found with higher frequency are: methyl n-nonyl ketone, b-myrcene, houttuynin, decanal, trans-caryophyllene, decanoic acid, camphene, β-pinene, lauraldehyde, bornyl acetate, α-pinene, limonene, 4-terpineol, caryophyllene oxide, nonanol and linalool so on.
4.2. Flavonoids and Other Polyphenols
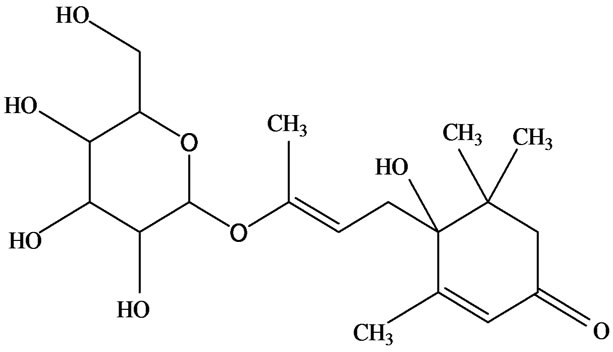
As would be expected in biologically active plants, a number of flavonoids and other polyphenols have been isolated and identified from H. cordata (Table 3). The quercitrin is the first flavonoids extracted from the leaves and stems of H. cordata [55]. Quercetin-3-O-β-D-galactoside-7-O-β-D-glucoside, kaempferol 3-O-[α-L-rhamnopyranosyl-(1→6)-β-D-glucopyranoside], quercetin
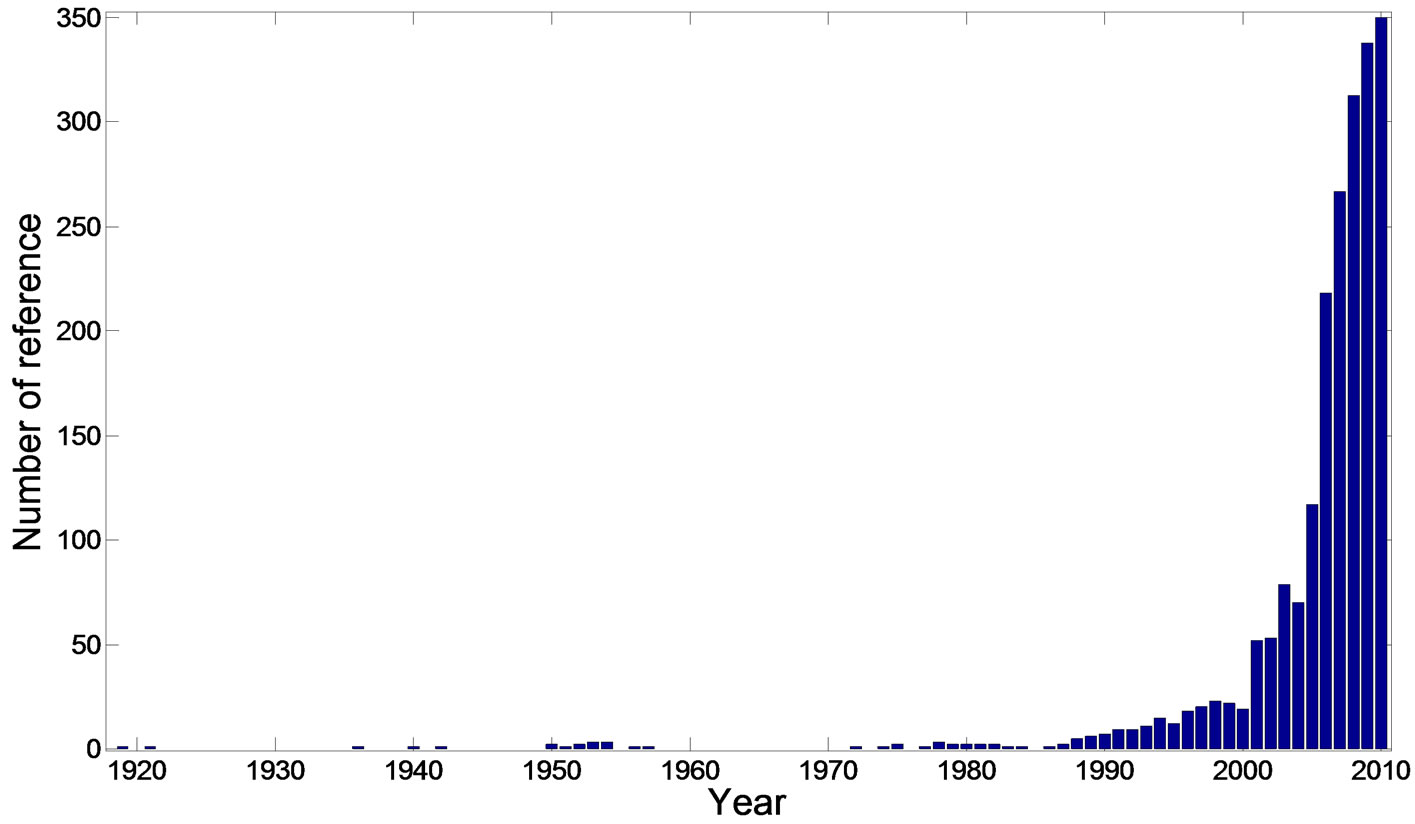
Figure 1. The increasing trend of the research work about H. cordata (The data was collected from Scifinder database).

Table 1. Ethnomedical uses of H. cordata in China.
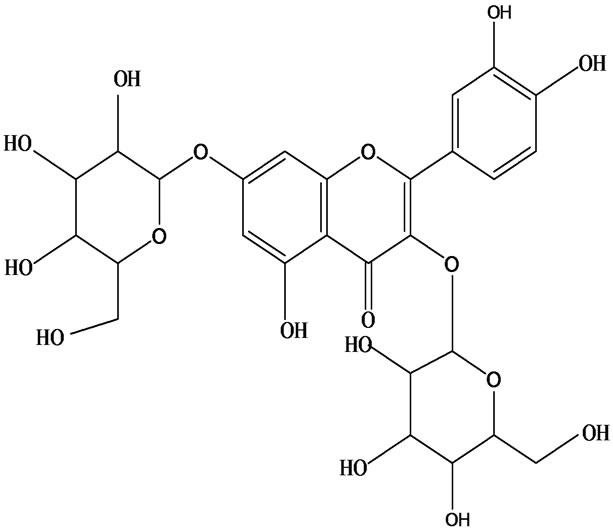
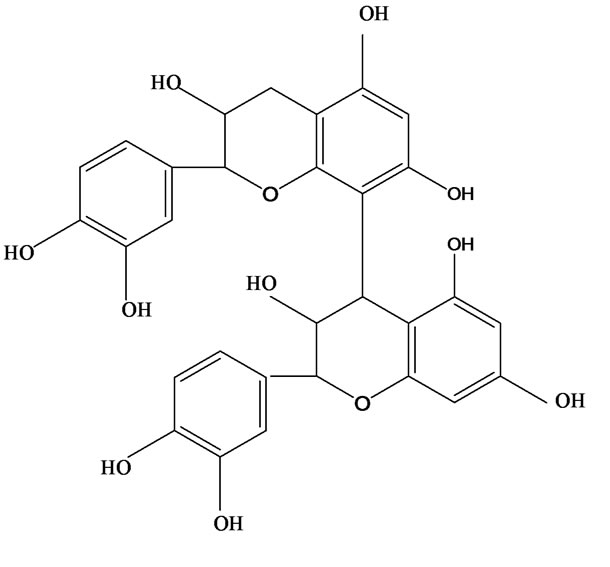
3-O-α-L-rhamnopyranosyl-7-O-β-D-glucopyranoside three flavonoid glycosides [56], chlorogenic acid methyl ester, 4-[(2E)-3-(b-D-glucopyranosyloxy)-2-buten-1-yl]-4-hydroxy-3,5,5-trimethyl-2-cyclohexen-1-one, 2-(4-hydroxyphenyl)ethyl-β-D-glucopyranoside, 2-(3,4-dihydroxyphenyl) ethyl-b-D-glucopyranoside, 4-(b-D-glucopyrano-syloxy)-3-hydroxybenzoic acid five polyphenols [57], catechin, procyanidin B, houttuynamide A, houuttuynoside A [18,58] (Figure 3), were isolated from H. cordata. The structures of new compounds are listed in
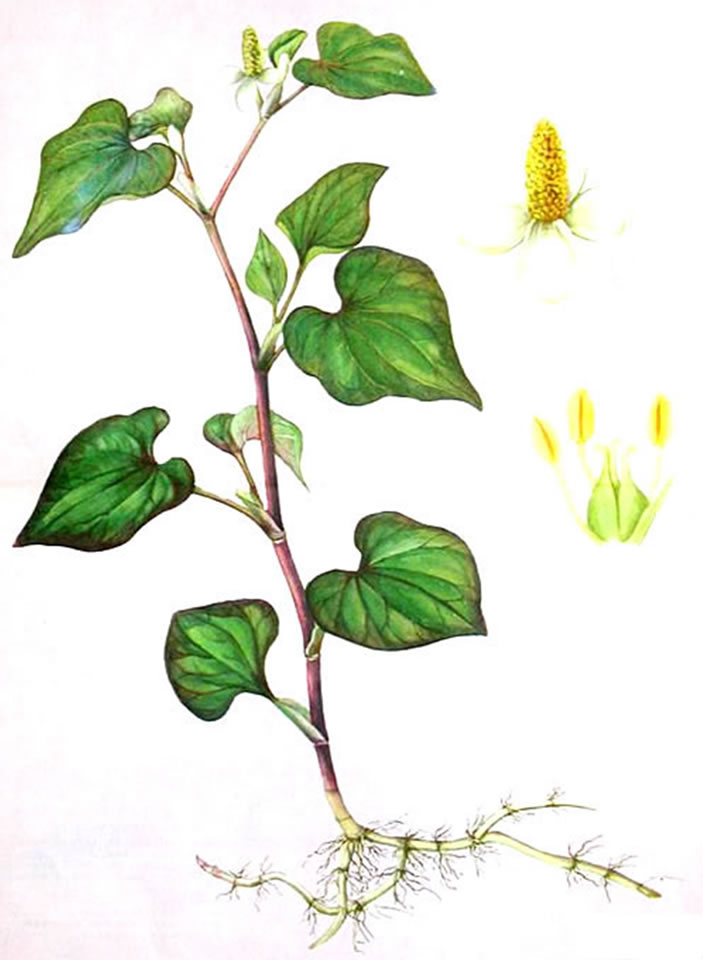
Figure 2. Houttuynia cordata Thunb.
The flavonoids and other polyphenols of H. cordata were extracted by various methods, hot soaking extraction [59,60], EtOAc extraction [61], ethanol/methanol refluent extraction [56], Soxhlet extraction [59], ultrasonic extraction [62], microwave-assisted extraction [63, 64] and pressurized liquid extraction [60], isolated and purified by macroreticular resin [65], various column chromatography [57] and bioactivity-guided fractionation and isolation [66], and identified and analyzed by physiochemical properties analysis [67], capillary electrophoresis with wall-jet amperometric detection [68], TLC [67,69], GC [69], HPLC [69-72], HPLC-MS [73], HPLC-DAD-ESI-MS [66,74] and spectral analysis including UV-VIS [59,67], NMR and MS [56,75]. Among the above-mentioned extraction methods, microwave-assisted extraction and pressurized liquid extraction are favorable to the other methods. The microwave-assisted extraction not only has higher extraction efficiency but also the advantages of being fast and energy saving.
The amounts of five flavonoid glycosides contained in H. cordata, quercitrin, hyperin, rutin, isoquercitrin and afzelin, were determined by HPLC [70,76]. The order of the flavonoids content in the different parts was as: flower > leaf > fruit > stem. No flavonoid was found in rhizome.
A correlation between flavonoid glycoside contents of H. cordata and light intensity has been reported. The flavonoid glycoside content was the highest when the plant was cultivated without shade and decreased as the shading rate increased [77].
The contents of flavonoids in freeze-dried H. cordata from different habitats were measured by HPLC-MS. It was found that flavonoids in Hongkong were higher than those in Sichuan and Guangdong Province. The relationship between the content of flavonoids in H. cordata and their biological characteristics such as morphologic and growth traits or a geographic origin were analyzed. The result revealed that the levels of three major flavonoids, hyperin, quercitrin, and quercetin, varied remarkably in the plants from different provinces, and variation in quercitrin was significantly correlated to the biological characteristics of the plant but not correlated to the geographic region where the plant grows [72].
4.3. Alkaloids
During the past 20 years, many kinds of alkaloid have been isolated from H. cordata, including aporphine, pyridine and the others. The structures of these compounds are shown in Table 4. cis-N-(4-Hydroxystyryl) benzamide and trans-N-(4-Hydroxystyryl) benzamide were isolated from the CHCl3 extraction of this herb by a combination of HPLC and other techniques [78]. Probstle and co-workers isolated aristolactam A, aristolactam B, piperolactam A and norcepharacdione B from H. cordata. Jong and Wang isolated 7-chloro-6-demethyl-cepharadione, long chain substituted pyridine alkaloids 3,5-didecanoyl-pyridine,2-nonyl-5-decanoylpyridine which are rare in nature and N-methyl-5-methoxy-pyr-rolidin-2- one from this plant [79,80], respectively.
4.4. Organic Acid and Fatty Acid
Palmitic acid, stearic acid, heptanoic acid, nonanoic acid, undecanoic acid, octanoic acid, hexanoic acid, lauric acid, capric acid, heptadecanoic acid, tetradecanoic acid, tridecanoic acid, pentadecanoic acid, octadecenoic acid, hexadecenoic acid, octadecadienoic acid, aspartic acid, glutamic acid capric acid, lauric acid and palmitic acid in this plant were identified using gas chromatograph [81]. In addition, chlorogenic acid, crypto-chlorogenic acid, neo-chlorogenic acid, quinic acid and caffeic acid were identified using mass spectra and fragmentation patterns [18]. Takagi and coworkers extracted chlorogenic acid, detected palmitic acid, linoleic acid, oleic acid, and stearic acid in the benzene fraction [82]. Wu and co-workers seperated and purified the chemical components by solvent extraction, thin-layer chromatograph and silica gel column chromatograph, and identified the structures by IR, EI-MS, 1H-NMR and 13C-NMR [83]. Bauer and colleagues identified linolenic, linoleic, oleic, palmitic and stearic acid by phytochemical examination [16]. Wang and colleagues isolated and purified succinic acid from dried rhizome of this plant by solvent extraction, silica gel and Sephadex LH-20 column chromatographs [80].

Table 2. Food applications of H. cordata.
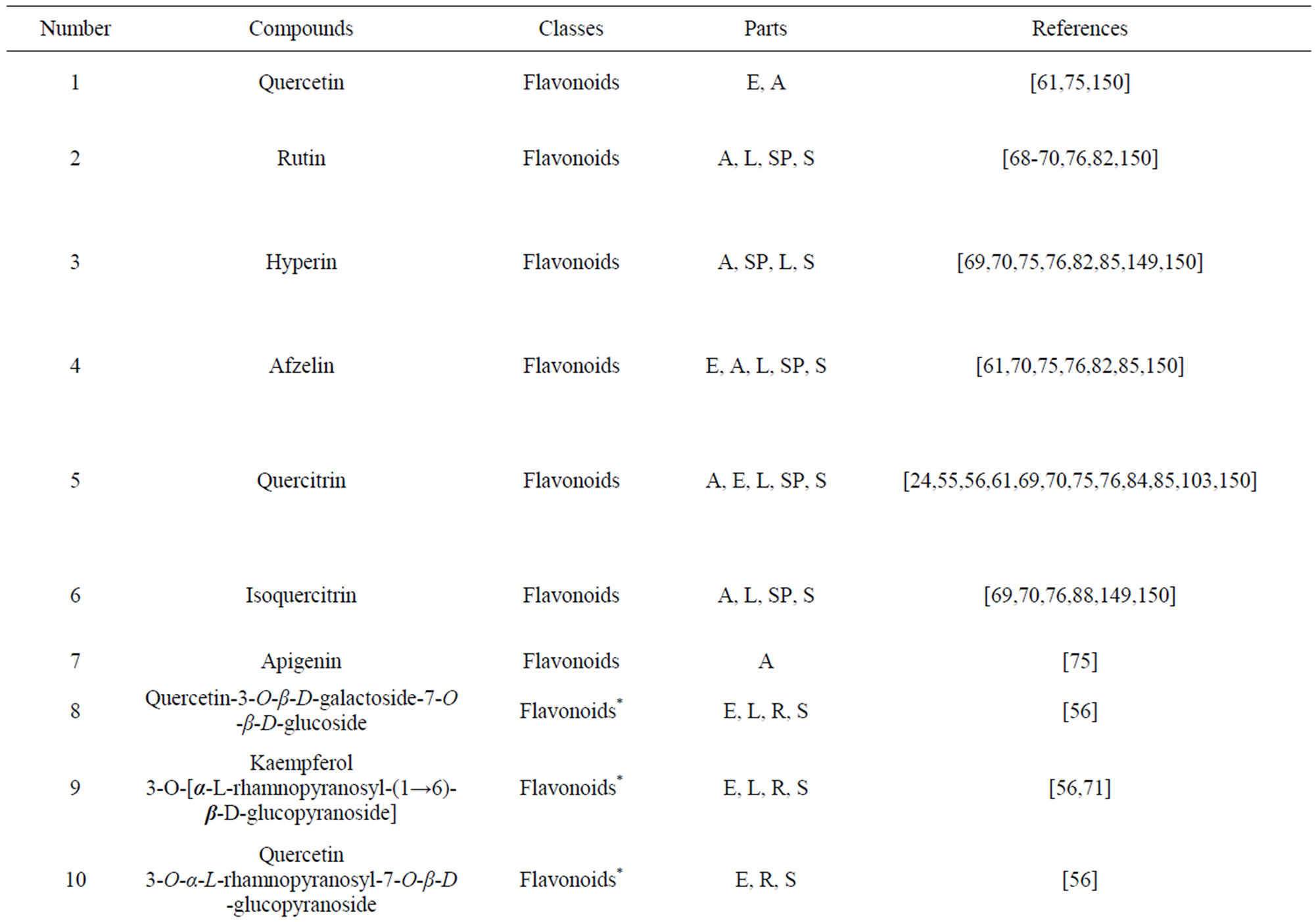


Table 3. Flavonoids and other polyphenols from H. cordata.
4.5. Sterols
A number of common sterols have been isolated from H. cordata. Stigmast-4-en-3-one, 3b-hydroxystigmast-5-en- 7-one, 5α-stigmastane-3,6-dione and stigmast-4-ene-3,6- dione were isolated from this plant [84]. Stigmast-3,6- dione, sitoindoside I and daucosterol were isolated and purified from dried rhizome of H. cordata by solvent extraction, silica gel and Sephadex LH-20 column chromatographs [80]. b-Sitosterol [4,82,85], b-sitosterol glucoside [85], brassicasterol [4], stigmasterol [4], spinasterol [4], and stigmast-4-ene-3,6-dione [16] were also found in this plant.
4.6. Amino Acid and Microelements
H. cordata contains more than 20 amino acids, including alanine, valine, glutamic acid, aspartic acid, isoleucine, proline, leucine, glycine, serine, lysine, cystine, tyrosine, methionine, phenylalanine, histidine, threonine, tryptophane, arginine, hydroxyproline and citrulline [81,86]. The major components were alanine, valine, glutamic acid, aspartic acid, isoleucine, proline leucine [81]. Among all amino acids, the glutamic acid content was the highest followed by leucine and aspartic acid [86]. Many microelements including iron, magnesium, manganese, potassium, copper, zinc and calcium, etc., can be in H. cordata [86,87]. The involucre of H. cordata containes high levels of zinc, copper, and Zn/Cu ratios at the stages of fructification [86].
4.7. Other Compounds
N-phenethyl-benzamide, glyceryl linoleate and n-butyl- α-D-fructopyranoside were isolated and purified from dried rhizome of H. cordata by solvent extraction, silica gel and Sephadex LH-20 column chromatographs [80]. Vomifoliol, sesamin and 1,3,5-tridecanoylbenzene were isolated from the aerial parts of H. cordata. 1,3,5-tridecanoylbenzene was also isolated [84]. Chou characterized N-(1-hydroxymethyl-2-phenylethyl) benzamide, N- (4-hydroxyphenylthyl) benzamide, 4-hydroxybenzamide, 4-hydroxy-3-methoxybenzamide, 6,7-dimethyl-1-ribitol- 1-yl-1,4-dihydroquinoxaline-2,3-dione, (1H)-quinolinone, indole-3-carboxylic acid, dihydrovomifoliol, reseoside, 7-(3,5,6-trihydroxy-2,6,6-trimethylcyclohexyl)but-3-en-2-one, 6-(9-hydroxy-but-7-enyl)-1,1,5-trimethylcyclhexane- 3,5,6-triol, 4-hydroxybenzoic acid, methylpara-ben, phydroxybenzaldehyde, benzyl-b-D-glucopyranoside and cycloart-25-ene-3b,24-diol from H. cordata using 1D and 2D NMR and mass spectra [85]. The carotenoids were observed in H. cordata. The content of b-carotene and violaxanthin are relatively high in the cotyledonous stage and decreased in fructification, while the content of lutein is low in the cotyledonous stage and progressively increased with the growth [86].
5. Pharmacological Activities
5.1. Diuretic Effects
The components quercitrin extracted from the leaves and
 (a)
(a) 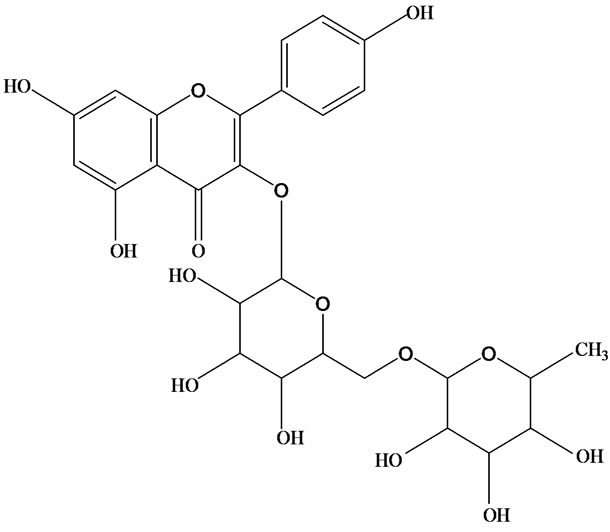 (b)
(b) 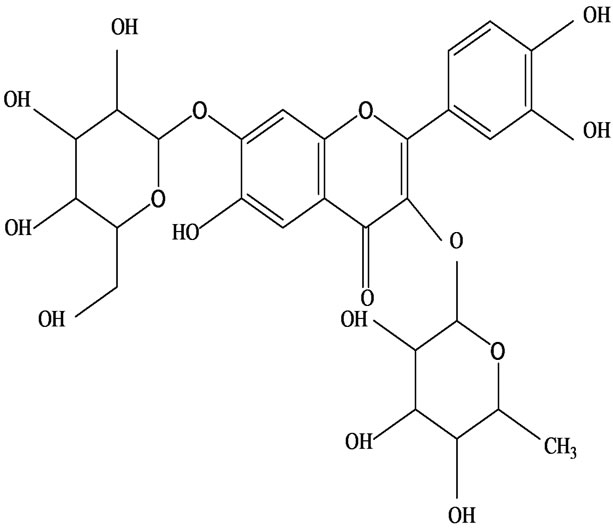 (c)
(c) 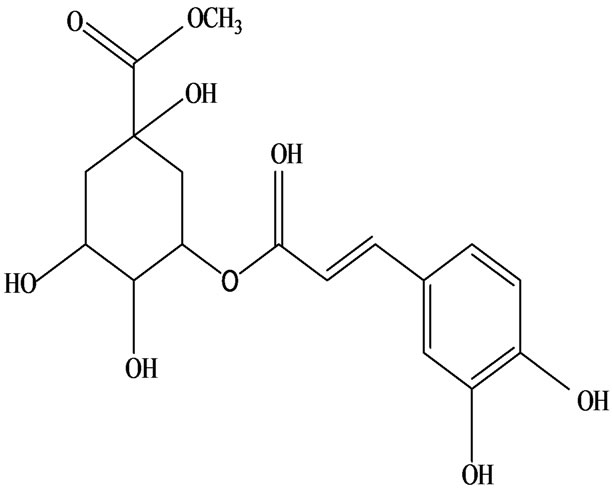 (d)
(d)
 (e)
(e) 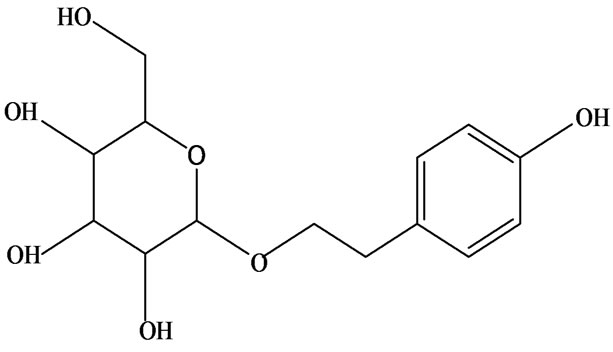 (f)
(f) 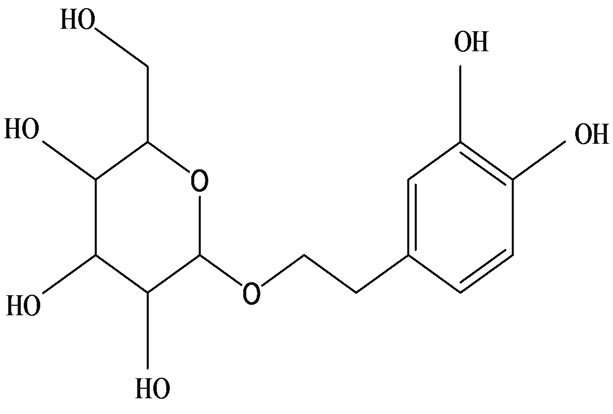 (g)
(g) 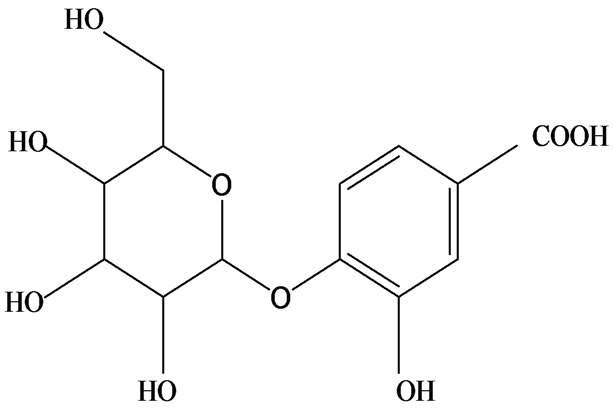 (h)
(h)
 (i)
(i) 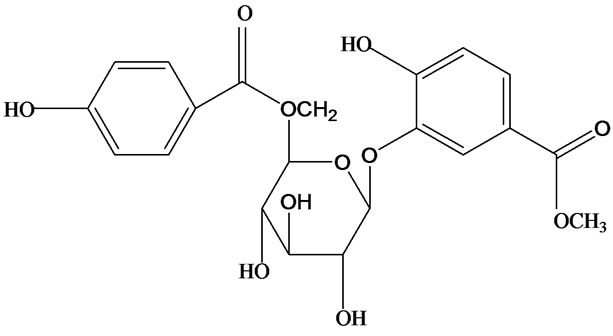 (j)
(j) 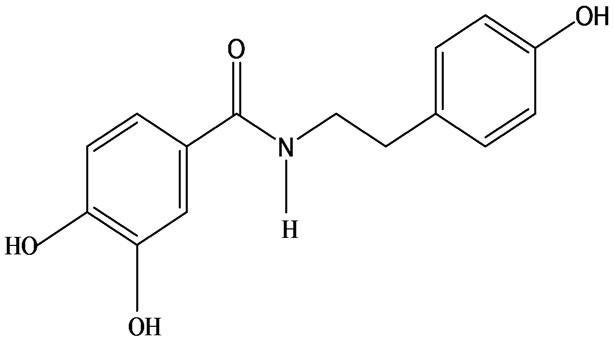 (k)
(k) 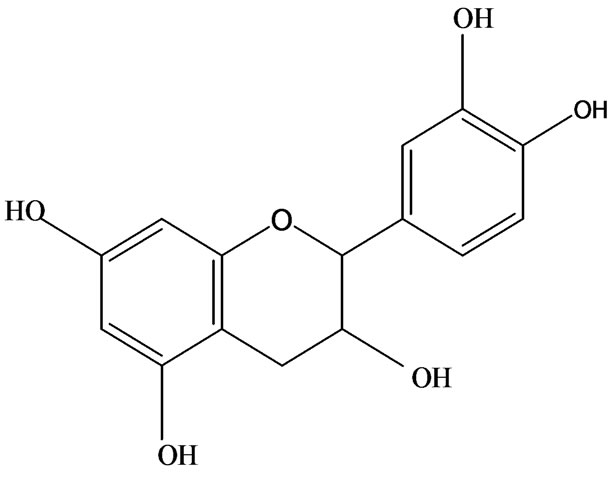 (l)
(l)
Figure 3. Flavonoids and other polyphenols were isolated or detected from H. cordata for the first time. (a) Quercetin 3-O-α-L-rhamnopyranosyl-7-O-β-D-glucopyranoside; (b) Kaempferol 3-O-[α-L-rhamnopyranosyl-(1→6)-β-D-glucopyranoside]; (c) Quercetin 3-O-α-L-rhamnopyranosyl-7-O-β-D-glucopyranoside; (d) Chlorogenic acid methyl ester; (e) 4-[(2E)- 3-(b-D-glucopyranosyloxy)-2-buten-1-yl]-4-hydroxy-3,5,5-trimethyl-2-cyclohexen-1-one; (f) 2-(4-Hydroxyphenyl)ethyl-β-Dglucopyranoside; (g) 2-(3,4-Dihydroxyphenyl)ethyl-b-D-glucopyranoside; (h) 4-(b-D-glucopyranosyloxy)-3-hydroxybenzoicacid; (i) Procyanidin B; (j) Catechin; (k) Houttuynamide A; (l) Houttuynoside A.
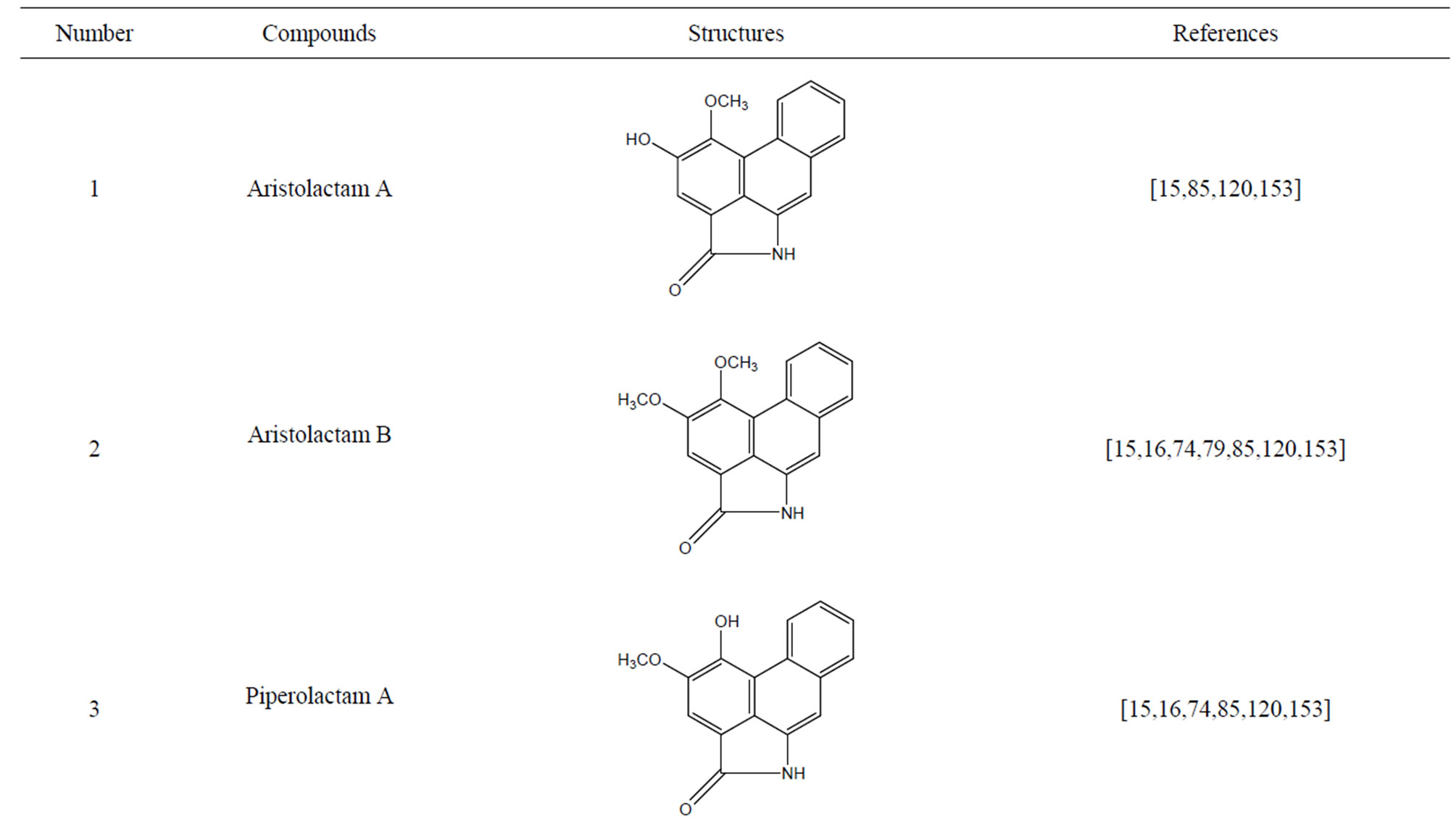
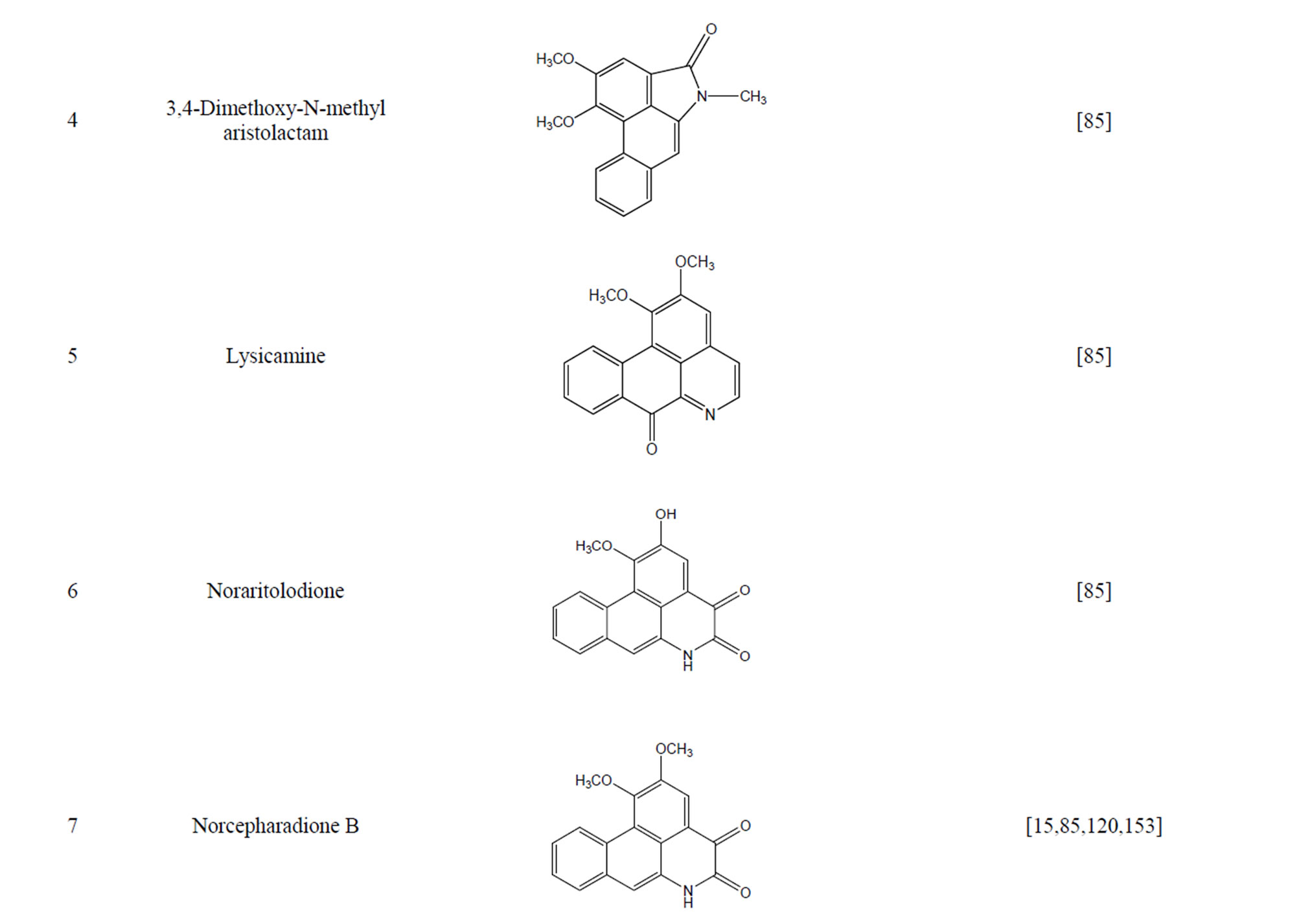
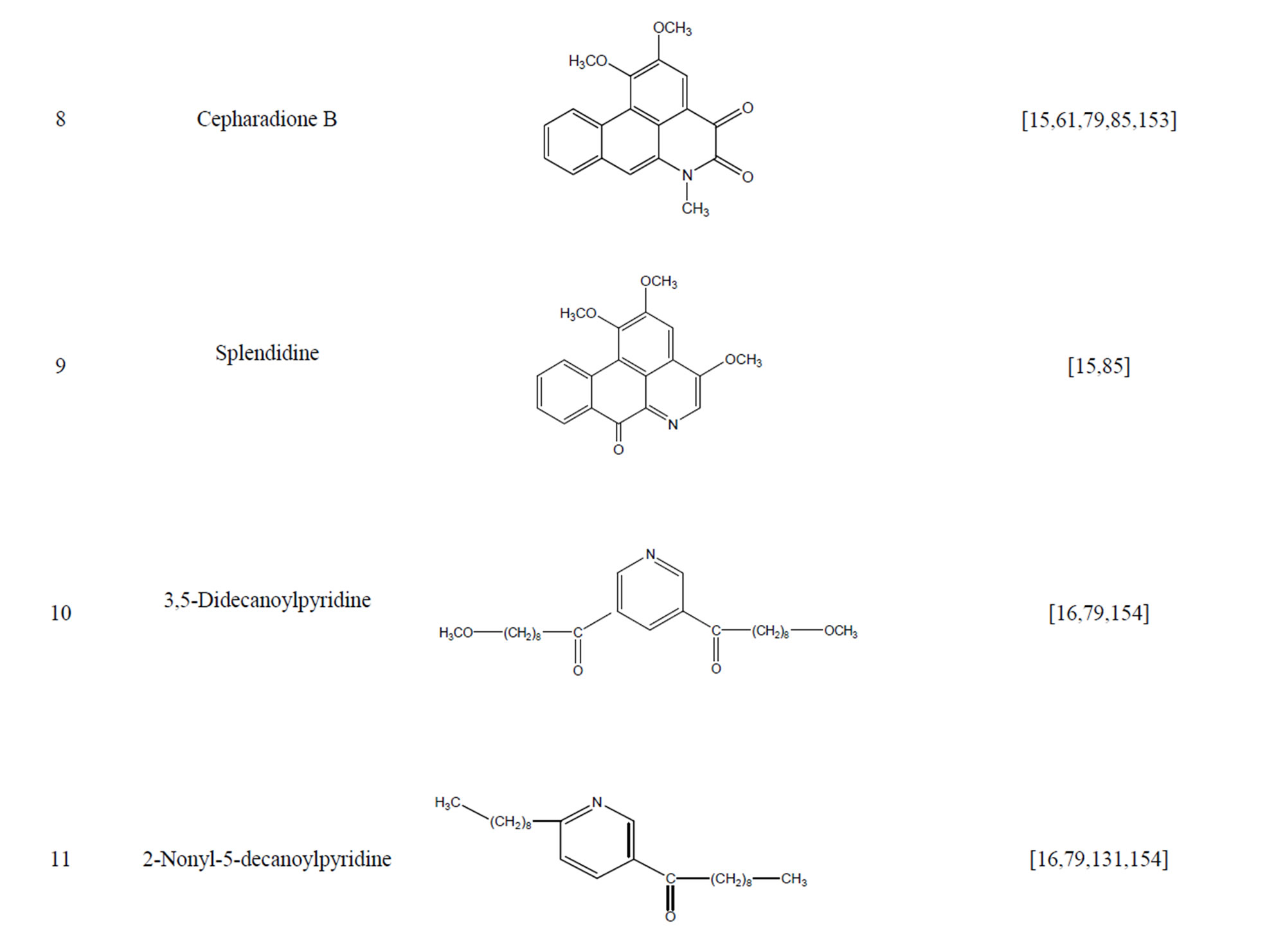
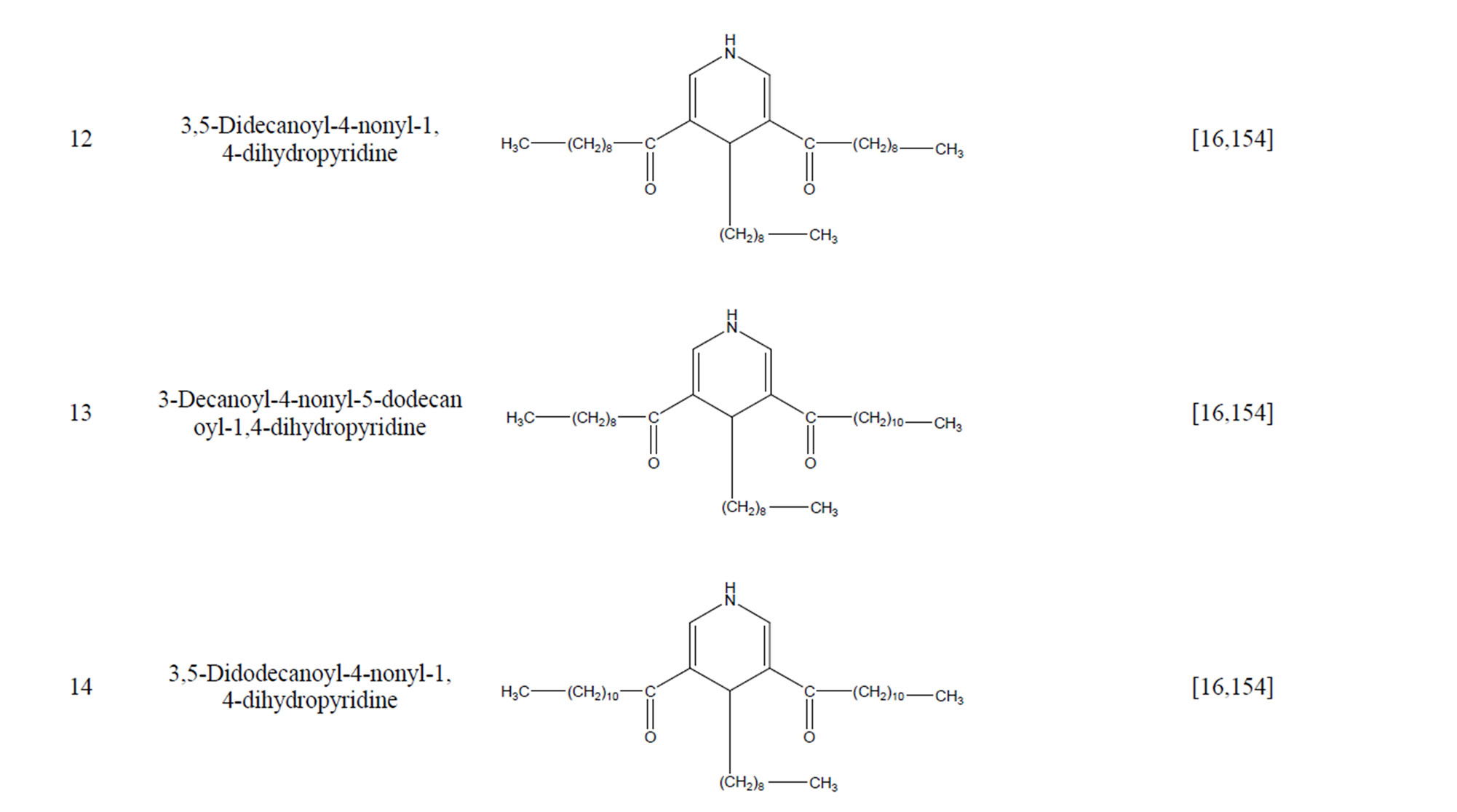

Table 4. Alkaloids from H. cordata.
stems, isoquercitrin from the floral spikes and fruit spikes of this herb show diuretic action [55,88]. The diuretic action is attributed to quercitrin, KCl and K2SO4 [89]. The extract of H. cordata showed similar diuretic action of acetylcholine, lactic acid and aspartic acid [19].
5.2. Antimicrobial Effects
Zhang [90] found that oil extract had inhibitory effect on β-Hemolytic streptococcus, Streptococcus aureus, Pseudomonas aeruginosa and Escherichia coli. Kwon [44] noticed that the volatile flavor components showed strong antibacterial activities against Bacillus cereus, Bacillus subtilis, Vibrio cholerae and Vibrio parahaemolyticus. Meng [91] found that water and ethanol extracts of fresh and dry H. cordata showed antimicrobial activity against Staphylococcus aureus and Escherichia coli. The minimal inhibitory concentrations (MICs) against Staphylococcus aureus of water extracts of fresh and dry H. cordata were 12.5 and 100 mg/mL, and those of ethanol extracts were 25 and 100 mg/mL respectively. The fresh extract had better pharma-cological activity than dry one, and water extract had better pharmacological activity than ethanol extract. Lu et al reported that oils obtained by hydrodistillation from the above and the below ground parts shows antimicrobial activity with MIC values of 0.0625 × 10−3 to 4.0 × 10−3 mL/mL against Staphylococcus aureus and Sarcina ureae [53]. Zhang et al. found that the MICs of the essential oil against Staphylococcus aureus and Sarcina increased with the storage time [92]. Moderate antibacterial activities are observed against Escherichia coli and Staphyloccocus aureus by [93,94], but not by [95].
The volatile essential oil isolated from H. cordata was seperated into 11 fractions by preparative HPLC. In a test of the antibacterial activity of 11 fractions, the growth of nine Gram-negative bacteria was inhibited when treated with Fraction 6 including methyl n-nonyl ketone, β- myrcene, β-ocimene, 1-decanol and houttuynin, and fraction 5 including decanal, bornyl acetate, fenchene and decanoic acid, respectively [46]. Houttuynin isolated from the rhizome of H. cordata suppressed the growth of yeasts and molds [22] and 3-nonylpyrazole inhibited the growth of Staphylococcus aureus, Bacillus subtilis, Trichophytons, Zygosaccharomyces salsus, and Aspergillus niger [96]. Kim et al. observeed that the antibacterial activity of H. cordata water extract (HCWE) against Salmonella typhimurium increased in a dose-dependent manner at concentrations from 25 to 100 mg/ml during 8-h incubation in vitro, without showing cytotoxicity in RAW 264.7 cells. Furthermore, HCWE showed virulence reduction effects in S. typhimurium-infected BALB/c mice in vivo [97].
5.3. Antiviral Effects
Over the past decade, substantial progress has been made in research on the natural products for the treatment of AIDS. Several plants and their products including H. cordata have shown anti-HIV activity [98]. The distillate and three major compounds from fresh plants of H. cordata showed dose-dependent virucidal acitivity against HIV-1 without showing cytotoxicity in vitro. At 2-fold dilution, approximately 20% and 40% of HIV-1 were inactivated by the pretreatment with the distillate for 2 h and 6 h, respectively. While no significant activity was observed at the concentrations less than 0.0017% (w/v), each of three components exerted virucial activity against HIV-1 at 0.0083% (w/v). Lauryl aldehyde was found to be the most potent constituent of the three components [10]. Vpr, an accessory gene product of HIV-1 induced abnormality in cell cycle leading to the increased HIV replication, was supposed to be a possible target for antiAIDS drugs. Quercetin, a compound from this crude drug, efficiently inhibited Vpr function without affecting its expression. Furthermore, the data suggested that Vprinduced transcription from HIV-LTR was considerably abrogated by quercetin. These in vitro data indicated that quercetin, a flavonoid previously reported inhibited HIV replication, also targeted Vpr [99].
An in vitro study evaluated the anti-HSV (herpes simplex virus) activity of H. cordata, using XTT-based colorimetric assay. BCC-1/KMC cells were infected with HSV and were cultured in HCWE. The results showed that HCWE significantly inhibited the replication of HSV at a concentration of 250 µg/mL, 10.2% for HSV type 1 (HSV-1) (p < 0.05) and 32.9% for HSV type 2 (HSV-2) (p < 0.005). The ED50 of HSV-1 and HSV-2 were 822.4µg/mL and 362.5 µg/mL respectively. H. cordata had better effect against HSV-2 than HSV-1, and had a low ED50 against HSV-2 [11]. The distillates extracted from fresh plants of H. cordata and three components, methyl n-nonyl ketone, lauryl aldehyde, and capryl aldehyde are also assayed for anti-HSV activity. The distillates, lauryl aldehyde and capryl aldehyde exhibited moderate antiviral activity against HSV-1 (ED50 = 0.0013%, ED50 = 0.0008%, ED50 = 0.00038%, w/v). Methyl n-nonyl ketone was not so effective against it (ED50 = 0.0091%, w/v) [10]. In another in vitro study, norcepharadione B isolated from the whole plant of H. cordata significantly suppresses HSV-1 replication by 46.38% at the concentration of 100 µM [58].
Severe acute respiratory syndrome (SARS) is a lifethreatening form of pneumonia caused by SARS coronavirus (SARS-CoV). From late 2002 to mid 2003, it infected more than 8000 people worldwide, of which over 7000 cases were found in China (http://www.who. int/en/). Owing to the high infectious rate and the absence of definitive therapeutic Western medicines, State Administration of Traditional Chinese Medicine of the People’s Republic of China proposed six traditional Chinese medicine formulae to general public as preventive measures on 24 April 2003 (http://www.satcm.gov.cn/ zhuanti/jbfz/20060901/100052.shtml). H. cordata was one of the component herbs in a heatremoving and detoxifying formula. Recently, immunomodulatory effect H. cordata was investigated in mouse splenic lymphocytes, inhibitory activity on SARS coronaviral 3C-like protease (3CLpro) and RNA-dependent RNA polymerase (RdRp). The results showed that HCWE stimulated the proliferation of mouse splenic lymphocytes significantly and dose-dependently. By flow cytometry, it revealed that H. cordata increased the proportion of CD4(+) and CD8(+) T cells. H. cordata exhibited significant dose-dependent inhibitory effects on SARS-CoV 3C-like protease (3CL (pro)) and RdRp activity. At concentration of 200 µg/ml or above, significantly inhibited the activity of 3CLpro (p < 0.05). At the highest testing dose (1000 µg/ml), the 3CLpro activity was decreased by 50%. The essential oil could inhibit the growth of influenza virus in cultures with ED50 = 41% (v/v) [100], and a complete inhibition at 250 mg/mL [90]. The antiviral assays demonstrated that quercetin 3-rhamnoside (Q3R) possessed strong antiviral activity of about 86% against influenza A/WS/33 virus at concentration of 100 µg/ml and antiviral activity of about 66% at the same virus at concentration of 10 µg/ml. The research on antivirus activities was carried out using amantadine, ribavirin and H. cordata injection (HCI) in vitro and in vivo. The abilities of treating pneumonia of mice when three kinds of drugs were used cooperatively were far higher than those alone in BALB/c mice [101]. Therefore, these findings provided important information for the utilization of H. cordata for influenza treatment.
Another one flavonoid compound, quercetin 7-rhamnoside (Q7R), showed antiviral activity against rotavirus and rhinovirus [102]. Q7R was used to inhibit porcine epidemic diarrhea virus (PEDV), was the predominant cause of severe entero-pathogenic diarrhea in swine. It inhibited PEDV replication by 50% using 0.014 µg/mL. Several structural analogues of Q7R, quercetin, apigenin, luteolin and catechin, also showed moderate anti-PEDV activity. Q7R did not directly interact with or inactivate PEDV particles and affect the initial stage of PEDV infection by interfering of PEDV replication. The effectiveness of Q7R against transmissible gastroenteritis virus (TGEV) and porcine respiratory coronavirus (PRCV), was lower compared to PEDV. Q7R could be considered as a lead compound for development of anti-PEDV drugs [103].
The inhibitory effect of H. cordata on epidemic hemorrhagic fever virus (EHFV) infection was also reported [104]. H. cordata extract (HCE) could neutralize EV71- induced cytopathic effects in Vero cells. The IC50 of HCE for EV71 was 125.92 ± 27.84 µg/mL. Antiviral screening of herb extracts was also conducted on 3 genotypes of EV71, coxsackievirus A16 and echovirus 9. HCE had the highest activity against genotype A of EV71. A plaque reduction assay showed that HCE significantly reduced plaque formation. Viral protein expression, viral RNA synthesis and virus-induced caspase 3 activation were inhibited in the presence of HCE, suggesting that it affected apoptotic processes in EV71-infected Vero cells by inhibiting viral replication. The antiviral activity of HCE was greater in cells pretreated with extract than those treated after infection. Therefore, it was concluded that HCE had antiviral activity, and it offered a potential to develop a new anti-EV71 agent [105].
5.4. Anticancer/Antitumour Effects
Some extracts and compounds from H. cordata and some traditional Chinese medicine formulae containing H. cordata were reported to have anticancer effect. A traditional Chinese medicine formulae containing H. cordata was used for treating lung cancer and improving human immunity. It had the advantages of good curative effect, no obvious toxic or side effect, simple manufacturing process, and low cost [106,107].
HCE treatment caused lowering of cell viability in various human cancer cell lines [108], and administered on the acupuncture point prevented the increase of mass weight of melanoma BBL16 tumor cells inoculated into mice [109]. The cellular effects of HCE and the signal pathways of HCE-induced apoptosis in HL-60 human promyelocytic leukemia cell line were investigated. HCE treatment caused apoptosis of cells as evidenced by discontinuous fragmentation of DNA, the loss of mitochondrial membrane potential, release of mitochondrial cytochrome c into the cytosol, activation of procaspase-9 and caspase-3, and proteolytic cleavage of poly(ADP-ribose) polymerase (PARP). Pretreatment of Ac-DEVD-CHO, caspase-3 specific inhibitor, or cyclosporin A, a mitochondrial permeability transition inhibitor, completely abolished HCE-induced DNA fragmentation. Together, these results suggested that HCE possibly caused mitochondrial damage leading to cytochrome c release into cytosol and activation of caspases resulting in PARP cleavage and execution of apoptotic cell death in HL-60 cells [110]. The 100 µg/mL of methanolic extract of H. cordata root showed significant protective effects (p < 0.01) against hydrogen peroxide-induced DNA damage in HepG2 cells and increased cell viability against hydrogen peroxide. The study indicated that H. cordata root methanol extract acted as a potential antioxidant, and exhibited potential anticancer properties [111]. The investigation examined the anticancer activity of the methanol extract from H. cordata on ICR mouse with induced abdominal cancer and L1210 cancer cells. To get an insight into the reaction mechanism undelying the anticancer activity, 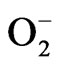 ion quantity and antioxidant enzyme activities such as superoxide dismiutase (SOD) and glutathione peroxidase (GPx) of L1210 cells in the presence of HCE were measured. The increased values of SOD and GPx enzyme activities in addition to the augmented generation of
ion quantity and antioxidant enzyme activities such as superoxide dismiutase (SOD) and glutathione peroxidase (GPx) of L1210 cells in the presence of HCE were measured. The increased values of SOD and GPx enzyme activities in addition to the augmented generation of 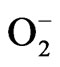 ion in L1210 cells implied that the reactive oxygen species including
ion in L1210 cells implied that the reactive oxygen species including 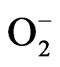 ion which were presumably induced by HCE might have participated in the process of L1210 cells cytotoxicity [112,113]. In another study, HCWE inhibited five leukemic cell lines, namely L1210, U937, K562, Raji and P3HR1, with IC50s between 478 µg/mL and 662 µg/mL. It was proved to be a potential medicinal plant for treating leukemia [12].
ion which were presumably induced by HCE might have participated in the process of L1210 cells cytotoxicity [112,113]. In another study, HCWE inhibited five leukemic cell lines, namely L1210, U937, K562, Raji and P3HR1, with IC50s between 478 µg/mL and 662 µg/mL. It was proved to be a potential medicinal plant for treating leukemia [12].
In a study, forty-eight Sprague-Dawley rats were fed with a diet containing 0%, 2% or 5% H. cordata powder and 15% fresh soybean oil or 24-h oxidized frying oil (OFO) for 28 days. The level of microsomal protein, total cytochrome 450 content (CYP450) and enzyme activities including NADPH reductase, ethoxyresorufin O-deethhylase (EROD), pentoxyresorufin O-dealkylase (PROD), aniline hydroxylase (ANH), aminopyrine demethylase (AMD), and quinone reductase (QR) were determined. The oxidized frying oil feeding produced a significant increase in the content of CYP450, microsomal protein, activities of NADPH reductase, EROD, PROD, ANH, AMD and QR in rats (P < 0.05). In addition, the activeties of EROD, ANH and AMD decreased and QR increased after feeding with H. cordata in OFO-fed group (P < 0.05). The feeding with 2% H. cordata diet showed the most significant effect. The findings suggested that polyphenol in H. cordata could be an important and necessary factor in the defense against CYP450-mediated cancers and other chronic diseases [114].
The anticancer activity of flavonoid extracts from H. cordata was studied on Sarcoma-180. They exhibited cytotoxic activity on S-180. The total flavonoid extract of H. cordata gave highest rate of death and showed good inhibitory effect on the growth of ascites tumor by S-180 in mice [37]. The inhibiting rate of flavonoids was detected by MTT assay. The apoptosis of HL60 and B16- BL6 was detected by FCM. Flavonoid from H. cordata also inhibited HL60 and B16BL6 and induced cell apoptosis. The IC50 in HL60 and B16BL6 was 0.410 and 0.122 g/L respectively [115].
Furthermore, six alkaloids exhibited cytotoxicity against five human tumor cell lines (A-549, SK-OV-3, SKMEL-2, XF-498 and HCT-15) in vitro, including aristolactam B, piperolactam A, aristolactam A, norcepharadione B, cepharadione B and splendidine isolated by bioactivity-guided fractionation of a methanolic extract. Among them, splendidine exhibited the strongest cytotoxicity and aristolactam B selectively suppressed XF- 498 (ED50 = 0.84 μg/ml) [15].
5.5. Anti-Inflammatory Effects
Many kinds of extracts from H. cordata showed antiinflammatory activity. The inflammation induced by xylene in the mice ear edema model was adopted to study the anti-inflammatory activity of chloroform extract, water extract, ethanol extract and n-butanol extract. All forms showed good anti-inflammatory activity, and the water extract had better pharmacological activity than ethanol extract, while the extract of fresh H. cordata had better pharmacological activity than that of dry one [91,116].
Shuang-Qing-Cao (SQC), a folk Chinese medicinal formula composed of Lonicera japonica Thunb (Caprifoliaceae), Isatis indigotica Fort (Cruciferae) and H. cordata, was used for treating diseases related with inflamemation in the folk of China. The results indicated that SQC extract in β-cyclodextrin was greatly effective on an experimental model of acute lung inflammation induced by the intratracheal instillation of lipopolysaccharide (LPS) in vivo [117]. 40 - 400 mg·kg−1 of fresh HCE reduced the increase of LPS-induced leucocytes in brochoalveolar lavage fluid (BALF) in ICR mice, and alleviated the infiltration of inflammatory cells in pathological lung tissue, which showed that fresh HCE inhibited the LPS-induced lung inflammation [118].
A study investigated the effect of HCE on the migration of the human mast cell line, HMC-1, in response to stem cell factor (SCF). Treatment with HCE at a concentration of 10 µg/mL for 24 h showed no significant decrease in the survival rate of the HMC-1 cells. SCF showed the typical bell-shape curve for the HMC-1 cell chemoattraction with the peak of the curve at the SCF concentration of 100 ng/mL. HC-1, which was the whole plant extracted with 80% EtOH, and HC-3, which was the residue successively partitioned with EtOAc, both had inhibitory effects on HMC-1 cell movement. After the treatment with 10 µg/mL HC-1 extract for 6 and 24 h, the chemotactic index (CI) of HMC-1 cells decreased to 74% and 63%, respectively. HC-3 extract treatment for 6 and 24 h lowered the CI to 72% and 44%, respectively. The HC-1 and HC-3 extracts had no inhibitory effect on the mRNA and surface protein expressions of c-kit, SCF receptor. SCF mediated the chemotaxis signaling via nuclear factor kB (NF-kB) activation, and both extracts inhibited the activation. Therefore, the results indicated that HC-1 and HC-3 extracts decreased the chemotactic ability of HMC-1 cells in response to SCF by inhibiting NF-kB activation, and these substances may be useful for treating mast cell-induced inflammatory diseases [8]. Ethanol fraction was obtained from dried and powdered whole plants of H. cordata. The residue was diluted with water and was successively partitioned with n-hexane, EtOAc and BuOH. H. cordata fractions (HcFs) inhibited the expression of IL-4 and IL-5 in response to phorbol 12-myristate 13-acetate (PMA) and calcium ionophore (CaI) in Jurkat T cells and the human mast cell line, HMC-1. IL-4 and tumor necrosis factor-alpha (TNF-α)- induced thymus and activationregulated chemokine (TARC) production was blocked by HcFs in skin fibroblast CCD-986sk cells, particularly by the ethanol extract of Hc. Stimulants included in PMA, phytohemagglutinin (PHA) and CaI, increase the mRNA level of CC chemokine receptor 4 (CCR4), a receptor of TARC, in Jurkat T cells, and the ethanol extract of HcF weakly blocks the increased mRNA level. The ethanol extract inhibited TARC-induced migration, as well as basal migration of Jurkat T cells. Recent studies showed the usefulness of HcFs in the ethnopharmacological treatment of Th2-mediated or allergic inflammation, through the down-regulation of the production of Th2 cytokines, TARCand cell migration [119].
The effects of aqueous extract of H. cordata on the production of the pro-inflammatory mediators, nitric oxide (NO) and TNF-α were examined in an activated macrophage-cell line; RAW 264.7. The aqueous extract from H. cordata inhibited NO production in a dose-dependent manner, but minimally (~30%) TNF-α secretion at 0.0625 and 0.125 mg/mL [7]. Another study investigated the effects of aqueous extract on passive cutaneous anaphylaxis (PCA) in mice and on IgE-mediated allergic response in rat mast RBL-2H3 cells. Oral administration of aqueous extract inhibited IgE-mediated systemic PCA in mice. It also reduced antigen (DNP-BSA)-induced release of β-hexosaminidase, histamine, and reactive oxygen species in IgE-sensitized RBL-2H3 cells. In addition, it inhibited antigen-induced IL-4 and TNF-α production and expression in IgE-sensitized RBL-2H3 cells. It inhibited antigen-induced activation of NF-kB and degradation of IKB-α, and suppressed antigen-induced phosphorylation of Syk, Lyn, LAT, Gab2, and PLCγ2 and phosphorylation of Akt and MAP kinases (ERK1/2 and JNK1/2 but not p38 MAP kinase).
HCE showed remarkable COX inhibitory activity. Phytochemical investigation of HCE has led to the isolation of three aristolactams, two 4,5-dioxoaporphine derivatives, and several pyridine and dihydropyridine derived alkaloids. Finally it was found that unsaturaed fatty acids, like oleic and linoleic acid, were responsible for the high acitivity of the extract (IC50: 13, 4 and 0, 25 µM respectively; indomethacin: 1, 15 µM) [120]. The antiinflammatory activities of quercitrin isolated from H. cordata were evaluated in mice, rats, and guinea pigs. Quercitrin (50, 100, and 200 mg/kg orally) inhibited the rat hind paw edema induced by carrageenin, dextran, histamine, serotonin, and bradykinin in a dose-dependent manner; at 200 mg/kg this compound also inhibited the scald edema induced by 54˚C hot water. Quercitrin did not show any inhibition of the UV light-induced erythema in guinea-pigs and of the increase of vascular permeability induced by acetic acid in mice. Quercitrin did not affect the granuloma formation in a cotton pellet implant and the development of adjuvant arthritis in rats. Quercitrin had an inhibitory effect on acute inflammation [121].
HCI, an aqueous solution of the steam distillate from plants of H. cordata, was used to treat ulcerative colitis (UC). Forty-two first episode type UC patients were randomly divided into HCI treatment group (n = 21) and Sulfasalazine group (n = 21). Clinical effects were observed in the 2 groups while ultrastructure of colonic mucosa, ICAM-1 and the pressure of distant colon were studied in HCI group. The clinical effect of HCI group (complete remission in 20, 95.2%; improvement in 1, 4.8%) was better than that of Sulfasalazine group (complete remission in 15, 72.4%, improvement in 5, 23.8%; inefficiency in 1, 3.8%, P < 0.01). The time of stool frequency recovering to normal (5.6 ± 3.3 d), and blood stool disappearance (6.7 ± 3.8 d) and abdominal pain disappearance (6.1 ± 3.5 d) in HCI group compared with Sulfasalazine group (9.5 ± 4.9 d, 11.7 ± 6.1 d, 10.6 ± 5.3 d, P < 0.01). HCI inhibited the epithelial cell apoptosis of colonic mucous membrane and the expression of ICAM- 1 (45.8 ± 5.7% vs 30.7 ± 4.1%, P < 0.05). Compared with normal, the mean promotive speed of contraction wave steped up (4.6 ± 1.6 cm/min vs 3.2 ± 1.8 cm/min, P < 0.05) and the mean amplitude of the wave decreased (14.2 ± 9.3 kPa vs 18.4 ± 8.0 kPa, P < 0.05) in active UC patients. After treatment with HCI, these 2 indexes improved significantly (17.3 ± 8.3 kPa, 3.7 ± 1.7 cm/min, P < 0.05). In normal persons, the postprandial pressure of sigmoid (2.9 ± 0.9 kPa) was higher than that of descending colon (2.0 ± 0.7 kPa) and splenic flexure (1.7 ± 0.6 kPa), while the colonic pressure (1.5 ± 0.5 kPa, 1.4 ± 0.6 kPa, 1.3 ± 0.6 kPa) decreased significantly (P<0.05) in active UC patients. The pain threshold of distant colon (67.3 ± 18.9 mL) in active UC patients decreased significantly compared with normal persons (216.2 ± 40.8 mL, P < 0.05) and recovered to normal after treatment with HCI (187.4 ± 27.2 mL, P < 0.05) [122]. Injection of carrageenan into the pleural cavity elicited an acute inflammatory response characterized by protein rich fluid accumulation and leukocyte infiltration in the pleural cavity. The inflammatory responses including fluid volume, protein concentration, C-reactive protein and cell infiltration reached peak after 24 h. The results showed that these parameters were attenuated by HCI and touched bottom at dose of 0.54 mL/100g. The results clearly indicated that HCI had anti-inflammatory activity [6]. The subsequent GC-MS analysis result indicated that main effect compounds in HCIs were methyl n-nonyl ketone, houttuynin, lauryl aldehyde, capryl aldehyde, bpinene, b-linalool, 1-nonanol, 4-terpineol, α-terpineol, bornyl acetate, n-decanoic acid and acetic acid, geraniol ester etc [123,124].
5.6. Antioxidative Effects
Chen [14] showed that both aqueous and methanolic extracts of H. cordata had antioxidant properties under OFO feeding-induced oxidative stress on Sprague-Dawley rats. The rats were fed diets containing 0%, 2%, or 5% H. cordata and 15% fresh or OFO for 28 days. The levels of polyphenols in feces, blood plasma, and liver were determined. The low-density lipoprotein (LDL) oxidation lag time, plasma total antioxidant status (TAS), and levels of thiobarbituric acid-reactive substances (TBARS) were used as antioxidant indexes and the protein carbonyl groups were used as oxidative index. The polyphenol levels decreased in blood plasma and increased in feces when feeding OFO; the apparent absorption of polyphenols also decreased. The polyphenol levels in plasma increased when feeding H. cordata. The OFO-fed rats had higher plasma TBARS and hepatic protein carbonyl group concentrations and shorter LDL lag times than controls. The total TAS was elevated and the LDL lag time was prolonged with H. cordata feeding.
Ng [13] examined the antioxidant properties of H. cordata and its protective effect on bleomycin-induced pulmonary fibrosis in rats. Results showed that aqueous extract of H. cordata exhibited a different magnitude of antioxidant activities in all model systems tested. Although H. cordata showed weaker free radical scavenging and xanthine oxidase inhibitory activity than vitamin E, its anti-lipid peroxidation activity in rat liver homogenate was close to that of vitamin E. In animal studies, H. cordata significantly decreased the levels of superoxide dismutase, malondialdehyde, hydroxyproline, interferon-γ, and TNF-α. However, an increase in the concentration of catalase was noted in the bronchoalveolar lavage fluid. H. cordata also remarkably improved the morphological appearance of the lung of bleomycin-treated rats. This protective effect was more pronounced than that of vitamin E. In Korea, the reactive oxygen radical species (ROS) scavenging effects of 50 kinds of Korean medicinal plant leaves were examined. H. cordata exhibited over a 95% scavenging effect of superoxide anion in 1 ppm concentration of test solution. The correlation coefficient of total phenolic content with superoxide anion, hydrogen peroxide, hydroxyl radical and DPPH radical scavenging effects were 0.8111, 0.8057, 0.8809 and 0.9810, respectively [125]. Furthermore, the polyphenols isolated from H. cordata, quercetin, quercetin-3-O-bD-galactoside, quercetin-3-O-α-L-rhamnoside and kaempferol-3-O-α-L-rhamnoside, exerted strong DPPH radicalscavenging activity [126]. In addition, the antioxidative activities of those medicinal plants and the compounds were investigated using the thiocyanate method to evaluate inhibitory effects on lipid peroxidation in the linoleic acid system. The peroxide levels gradually increased during incubation in the presence of linoleic acid over 3 days, and most of the plants inhibited lipid peroxidation. The aerial part of H. cordata reduced lipid peroxidation more effectively as lipid peroxidation progressed, resulting in inhibition of about 80% relative to the control value by the 3rd day of incubation. The polyphenols isolated from H. cordata, quercetin, quercetin-3-O-b-D-galactoside and quercetin-3-O-α-L-rhamnoside, also showed marked and dose-dependent inhibitory effects on lipid peroxidation. Moreover, quercetin glycosides showed stronger activity than quercetin, suggesting that glycosylation increased the antioxidative activity of quercetin [127]. The antioxidative and anti-lipid peroxidative efficacies of the fractions (H2O, 20% MeOH, 40% MeOH, 60% MeOH, 100% MeOH) from H. cordata were measured by DPPH method and TBARS assay on rat liver homogenate. It was revealed that 60% MeOH fractions had potential antioxidative activity and inhibited lipid peroxidation significantly. The DNA damage was analyzed by tail moment (TM) and tail length (TL), which used markers of DNA strand breaks in SCGE. The 100 µg/mL of methanolic extract of H. cordata root showed significant protective effects (P < 0.01) against H2O2-induced DNA damage in HepG2 cells and increased cell viability against H2O2. The results of this study indicated that H. cordata root methanol extract acted as a potential antioxidant, and exhibited potential anticancer properties, which might provide a clue to find applications in new pharmaceuticals for oxidative stability. The free radical scavenging capacities and antioxidant activities of H. cordata were evaluated using commonly accepted assays, including xanthine-xanthine oxidase assay, linoleic acid peroxidation and DPPH spectrophotometric assays, rapid screening of antioxidant by dot-blot and DPPH staining, TLC analysis with DPPH staining and DNA strand scission by hydroxyl radicals. H. cordata was extracted with dichloromethane, methanol or ethanol, respectively and selected for the best antioxidant results. Each sample under assay condition showed a dose-dependent free radical scavenging effect of DPPH and a dose-dependent inhibitory effect of xanthine oxidase and lipid peroxidation. They also showed a protective effect on DNA damage caused by hydroxyl radicals generated from UV-induced photolysis of hydrogen peroxide. A rapid evaluation for antioxidants using TLC screening and DPPH staining methods demonstrated each extract having various free radical scavenging capacity. Stained silica layer revealed a purple background with yellow spots at the location of drops, which showed radical scavenging capacity [138].
5.7. Antidiabetic Effects
The essential oils from H. cordata has been shown to have an effect on improving fat metabolism, the urinary albumin and insulin resistance of diabetes mellitus rats [128,129]. The rat model of diabetes mellitus was induced with streptozotocin (STZ) and high glucose-lipoids animal feeds, treated with H. cordata for 8 weeks. After treatment, fasting insulin level was lower in H. cordata group than in control, rosiglitazone and losartan group (P < 0.05). Insulin sensitivity index increased in H. cordata group and rosiglitazone group (P < 0.05 compared with the losartan group). The urinary albumin and 24 h urine volume were the lowest in H. cordata group (P < 0.05). The concentration of triglyceride in serum was lower in H. cordata and rosiglitazone group than that in control group (P < 0.05). H. cordata had a protective effect on the renal tissues in diabetic rats, which was probably correlated with the decrease of the expression of transforming growth factor-b1 and collagen I and with the increase of the expression of bone morphogenetic protein-7 in the renal tissues [130,131].
5.8. Anti-Allergic Effects
Human basophilic KU812F cells express a high-affinity immunoglobulin (Ig) E receptor, FcεRI, which plays an important role in IgE-mediated allergic reactions. Flow cytometric analysis showed that the FcεRI expression and the IgE binding activity were suppressed when the cells were cultured with HCE. Reverse transcriptionpolymerase chain reaction analysis showed that levels of the mRNAs for FcεRIα- and γ-chains were decreased by the treatment of HCE. Addition of HCE to culture medium also resulted in a reduction in the release of histamine from the cells. These results suggested that HCE may exert its anti-allergic activity through down-regulation of FcεRI expression and a subsequent decrease in histamine release [17]. Oral administration of HCWE inhibited compound 48/80-induced systemic anaphylaxis in mice. HCWE also inhibited the local allergic reaction, PCA, activated by anti-dinitrophenyl (DNP) IgE antibody in rats. HCWE reduced the compound 48/80-induced mast cell degranulation and colchicine-induced deformation of rat peritoneal mast cells (RPMC). Moreover, HCWE dose-dependently inhibited histamine release and calcium uptake of RPMC induced by compound 48/80 or anti-DNP IgE. Another study investigated the effects of HCWE on PCA in mice and on IgEmediated allergic response in rat mast RBL-2H3 cells. Oral administration of HCWE inhibited IgE-mediated systemic PCA in mice, and reduced antigen (DNP-BSA)- induced release of b-hexosaminidase, histamine, and reactive oxygen species in IgE-sensitized RBL-2H3 cells. In addition, HCWE inhibited antigen-induced IL-4 and TNF-α production and expression in IgE-sensitized RBL- 2H3 cells. HCWE inhibited antigen-induced activation of NF-kB and degradation of IkB-α. To investigate the inhibitory mechanism of HCWE on degranulation and cytokine production, the activation of intracellular FcεRI signaling molecules were examined. HCWE suppressed antigen-induced phosphorylation of Syk, Lyn, LAT, Gab2, and PLC γ2, further downstream, inhibited antigen-induced phosphorylation of Akt and MAP kinases (ERK1/2 and JNK1/2 but not p38 MAP kinase).
5.9. Antimutagenic Effects
The antimutagenic effects of both aqueous and methanolic extracts of H. cordata were examined using the Ames test. They had dose-dependent antimutagenic effects on benzo(a)pyrene, aflatoxin B1, and OFO, and the antimutagenic ability of aqueous extracts was higher than of methanolic extracts [14]. The inhibitory actions of chloroform extracts from H. cardata on the mutagenicity of Trp-P-2 and 1-NP was examined [132]. Inhibitory action was clarified on the following three points: 1) extract had the repressive effect on the two kinds of mutagenic substances which were composed of the indirect mutagen (Trp-P-2) requiring metabolic activation with S-9mix and of the direct mutagen (1-NP), 2) extract had the repressive effect on Trp-P-2 (NHOH), an activated form of Trp-P-2, and 3) extract had the represssive effect on metabolic process of Trp-P-2 with S-9mix. These results suggested that the antimutagens in chloroform extracts had wide mechanism of repression.
5.10. Others
Two compounds, cisand trans-N-(4-hydroxystyryl) benzamide, were isolated from the CHCl3 extract of H. cordata, and proved to be potent inhibitors of platelet aggregation [78]. The injection and aqueous extract of H. cordata were illustrated to enhance immune function by modulating ex vivo pro-inflammatory cytokine and NO production as well as the expression of iNOS and COX-2 [133,134]. Effects of H. cordata extracts on the level of lipid peroxide and the enzyme activities of the liver were investigated in bromobenzene-induced rats. Lipid peroxide content in liver was increased by bromobenzene. It was decreased when the methanol extract of H. cordata was given to the rat. The methanol extract reduced the activities of aminopyrine N-demethylase and aniline hydroxylase that increased by bromobenzene, however did not affect glutathione S-transferase activity. The methanol extract recovered the activity of epoxide hydrolase activity that decreased significantly by bromobenzene. It was suggest that the extract might play an important play in the prevention of hepatotoxicity by reduction of aminopyrine N-demethylase and aniline hydroxylase activities as well as enhancement of epoxide hydrolase activity [75].
6. Quality Control
During our market surveillance, H. cordata has been sold in medicinal materials, tablet, injection, formula, grain, capsule and oral liquid, either presenting as single herb or as collections of herbs. However, like most of traditional herbal medicines and their preparations, despite its existence and continued use over many centuries, and its popularity and extensive use during the last decade, the quantity and quality of the safety and efficacy data are far from sufficient to meet the criteria needed to support its use world-wide. During the past several decades, with the extensive herbal treatment, adverse drug reactions accompany. HCI, the extract of sterile volatile oils from distillation of fresh H. cordata with a milliliter equivalent to 2 g of herbal materials, was reported a substantial incidence of life threatening anaphylaxis. The Chinese National Adverse Reaction Monitoring Center reports over 5000 adverse reactions to HCI from January 1988 to April 2006 including 222 serious cases [135]. The State Food and Drug Administration of China (SFDA) halted the use of HCI in hospitals in June 2006. Later, Lei and co-worker found that polysorbate-80, one of supplementary material in the injection caused serious adverse reactions in dogs with intravenous drip of HCI, when the concentration of polysorbate-80 was greater than 0.01 mg/ml. The distillate without polysorbate-80 showed no adverse reaction. This revealed that the adverse reactions of this injection may cause by supplementary material, not by the raw material herb [136,137]. Then, after reexamining HCI manufacturing conditions and quality control, the SFDA gave permission to few manufacturers to recommence HCI production. An allergen-warning label was requested to be added to the product.
As metioned above, H. cordata herb contains dozens of components. However, only one or two markers or pharmacologically active components is currently employed for evaluating the quality and authenticity, identifying the single herb or herb medicine preparations. The quality of HCI is assured based on the content of methyl n-nonyl ketone, whose content is set to the least 1.0 µg/ml injection. The quality of “fufang yuxingcao” tablet is assured based on the content of baicalin, whose content is set to the least 2.7 mg per tablet [138]. This standard is neither sufficient to determine the identity of an extracted plant material nor to ensure the quality of products. The chemical constituents in H. cordata vary depending on harvest seasons, plant origins, drying processes and other factors. Thus, it is necessary to develop accepted guideline for evaluating traditional medicine [139] and for determining most of the phytochemical constituents of herbal products in order to ensure the reliability and repeatability of pharmacological and clinical research, to understand their bioactivities and possible side effects of active compounds and to enhance product quality control [140].
The concept of phytoequivalence is developed in Germany in order to ensure consistency of herbal products [141]. According to this concept, a chemical profile concept, such as a fingerprint, is developed and proposed for standardizing medicinal products manufactured from herb medicines and their raw materials. In 2004, the Chinese State Food and Drug Administration (SFDA) regulated the compositions of liquid injection with herb medicine ingredients, using stringent quality procedures such as chemical assay and standardization. Fingerprints of herb medicine liquid injections are compulsorily carried out for this purpose. Comparing with conventional method focusing mainly on the determination of a certain active compound, fingerprinting can offer integral characterization of a complex system with a quantitative degree of reliability. The method for authentication and quality assessment of herb medicine has recently been accepted by the World Health Organization (WHO) as a strategy for the assessment of herbal medicines [142].
Under such circumstances, China Pharmacopoeia Committee organizes several scientific research institutions including our group, Research center for modernization of Chinese herbal medicine, college of chemistry and chemical engineering, Central south university, to establish the fingerprint of 70 traditional Chinese medicine injections and the technology platforms.
7. Conclusions
Throughout our literature review, many modern chemical, physical and biological methods are applied for the investigation of H. cordata. Despite the great progress, there are still some key tasks to do:
1) Find actual bioactive components. As mentioned above, the chemical composition of this herb is complex and diverse. Essential oil, alkaloids, flavonoids and other polyphenols are studied frequently in this species. The remaining chemical compounds are not well studied. Most of the mentioned studies focus on crude preparations of H. cordata, and the chemical profiles are not well detailed or standardized. There is a scarcity of detailed isolation studies works published. We believe that the isolation of new active principles for drug discovery from individual perspective, and establishment of detail chemical profiles for standardized extracts from holistic perspective would be of great scientific merit.
2) Understand the mechanism. Despite the extensive past and present traditional uses, and recent progress in pharmacological studies, biological data to correlate the ethnobotany to the chemistry are still lacking. In addition to recognized antibacterial ingredients such as decanoyl acetaldehyde and undecanone, some non-volatile components such as flavonoids, alkaloids and water-soluble polysaccharide have been found to have strong pharmacological activities gradually. Many studies on the pharmacological effects of H. cordata are reported, but the anti-inflammatory effects and immune mechanism are not clear, which are more important. The quantification of individual phytoconstituents and pharmacological profile of extracts based on in vitro, in vivo studies and on clinical trials are urgently needed in order to confirm traditional wisdom in the light of rational phytotherapy.
3) Find new ways. It is very difficult to find how the mixtures of ingredients act in concert, partly due to the complexity of H. cordata but also due to the lack of appropriate approaches within research of complex herbal mixtures. Recent arisen technologies, such as proteomics and metabolomics, offer additional complementary approaches and provide a deeper and holistic insight in systems biology. They are addressed as promising tools to investigate herb medicines [143].
4) Establish effective quality control methods. Allergic compounds in the product HCI are not entirely clear. And the current Chinese Pharmacopoeia only provides TLC distinction of undecanone for this herb [144]. The comprehensive and effective quality control methods for complex active ingredients of this herb are lack.
Although HCI has been halted, there is no doubt about the efficacy of this herb. This herb, particularly its oral formulations, has high value and broad application prospects. Therefore, in order to better development and utilization of this herb, it is urgent to use modern technology to clarify the material basis of its efficacy and mechanism, improve the quality control standards, and expand the clinical use for traditional efficacy and pharmacological effect. The collected information reviewed here provides a resource for future ethnopharmacological and phytochemical studies of the genus. The scientific validation for the popular use of H. cordata deserves more investigations.
8. Acknowledgements
The authors would like to thank National Natural Science Foundation of China for support of the projects (No. 20975115, 21175157 and 21375151), China Hunan Provincial science and technology department for support of the project (No. 2012FJ4139), Central South University for special support of the basic scientific research project (No. 2010QZZD007), China Postdoctoral Science Foundation for support of the project (No. 201104511).
REFERENCES
- P. Drasar and J. Moravcova, “Recent Advances in Analysis of Chinese Medical Plants and Traditional Medicines,” Journal of Chromatography B, Vol. 812, No. 1-2, 2004, pp. 3-21.
- N. R. Farnsworth, O. Akerele, A. S. Bingel, D. D. Soejarto and Z. Guo, “Medicinal Plants in Therapy,” Bulletin of the World Health Organization, Vol. 63, No. 6, 1985, pp. 965-981.
- S. Frantz, “Drug Discovery: Playing Dirty,” Nature, Vol. 437, No. 7061, 2005, pp. 942-943. doi:10.1038/437942a
- H. Zheng, Z. Dong and J. She, “Modern Study of Traditional Chinese Medicine,” Xue Yuan Press, Beijing, 1998.
- K.-M. Lau, K.-M. Lee, C.-M. Koon, C. S.-F. Cheung, C.-P. Lau, H.-M. Ho, M. Y.-H. Lee, S. W.-N. Au, C. H.-K. Cheng, C. B.-S Lau, et al., “Immunomodulatory and Anti-SARS Activities of Houttuynia Cordata,” Journal of Ethnopharmacology, Vol. 118, No. 1, 2008, pp. 79-85. doi:10.1016/j.jep.2008.03.018
- H. Lu, Y. Liang, L. Yi and X. Wu, “Anti-Inflammatory Effect of Houttuynia cordata Injection,” Journal of Ethnopharmacology, Vol. 104, No. 1-2, 2006, pp. 245-249. doi:10.1016/j.jep.2005.09.012
- E. Park, S. Kum, C. Wang, S. Y. Park, B. S. Kim and G. Schuller-Levis, “Anti-Inflammatory Activity of Herbal Medicines: Inhibition of Nitric Oxide Production and Tumor Necrosis Factor-Alpha Secretion in an Activated Macrophage-Like Cell Line,” American Journal of Chinese Medicine, Vol. 33, No. 3, 2005, pp. 415-424. doi:10.1142/S0192415X05003028
- I. S. Kim, J.-H. Kim, J. S. Kim, C.-Y. Yun, D.-H. Kim and J.-S. Lee, “The Inhibitory Effect of Houttuynia cordata Extract on Stem Cell Factor-Induced HMC-1 Cell Migration,” Journal of Ethnopharmacology, Vol. 112, No. 1, 2007, pp. 90-95. doi:10.1016/j.jep.2007.02.010
- G. Z. Li, O. H. Chai, M. S. Lee, E.-H. Han, H. T. Kim and C. H. Song, “Inhibitory Effects of Houttuynia cordata Water Extracts on Anaphylactic Reaction and Mast Cell Activation,” Biological & Pharmaceutical Bulletin, Vol. 28, No. 10, 2005, pp. 1864-1868. doi:10.1248/bpb.28.1864
- K. Hayashi, M. Kamiya and T. Hayashi, “Virucidal Effects of the Steam Distillate from Houttuynia cordata and Its Components on HSV-1, Influenza Virus, and HIV,” Planta Medica, Vol. 61, No. 3, 1995, pp. 237-241. doi:10.1055/s-2006-958063
- L.-C. Chiang, J.-S. Chang, C.-C. Chen, L.-T. Ng and C.-C. Lin, “Anti-Herpes Simplex Virus Activity of Bidens pilosa and Houttuynia cordata,” American Journal of Chinese Medicine, Vol. 31, No. 3, 2003, pp. 355-362. doi:10.1142/S0192415X03001090
- J.-S. Chang, L.-C. Chiang, C.-C. Chen, L.-T. Liu, K.-C. Wang and C.-C. Lin, “Antileukemic Activity of Bidens pilosa L. var. minor (Blume) Sherff and Houttuynia cordata Thunb,” American Journal of Chinese Medicine, Vol. 29, No. 2, 2001, pp. 303-312. doi:10.1142/S0192415X01000320
- L.-T. Ng, F.-L. Yen, C.-W. Liao and C.-C. Lin, “Protective Effect of Houttuynia cordata Extract on BleomycinInduced Pulmonary Fibrosis in Rats,” American Journal of Chinese Medicine, Vol. 35, No. 3, 2007, pp. 465-475. doi:10.1142/S0192415X07004989
- Y.-Y. Chen, J.-F. Liu, C.-M. Chen, P.-Y. Chao and T.-J. Chang, “A Study of the Antioxidative and Antimutagenic Effects of Houttuynia cordata Thunb. Using an Oxidized Frying Oil-Fed Model,” Journal of Nutritional Science and Vitaminology, Vol. 49, No. 5, 2003, pp. 327-333. doi:10.3177/jnsv.49.327
- S.-K. Kim, S. Y. Ryu, J. No, S. U. Choi and Y. S. Kim, Cytotoxic Alkaloids from Houttuynia cordata,” Archives of Pharmacal Research, Vol. 24, No. 6, 2001, pp. 518- 521. doi:10.1007/BF02975156
- R. Bauer, A. Proebstle and H. Lotter, “Cyclooxygenase Inhibitory Constituents from Houttuynia cordata,” Phytomedicine, Vol. 2, No. 4, 1996, pp. 305-308. doi:10.1016/S0944-7113(96)80073-0
- S.-Y. Shim, Y.-K. Seo and J.-R. Park, “Down-Regulation of Fc.vepsiln.RI Expression by Houttuynia cordata Thunb Extract in Human Basophilic KU812F Cells,” Journal of Medicinal Food, Vol. 12, No. 2, 2009, pp. 383-388. doi:10.1089/jmf.2007.0684
- N. Nuengchamnong, K. Krittasilp and K. Ingkaninan, “Rapid Screening and Identification of Antioxidants in Aqueous Extracts of Houttuynia cordata Using LC-ESIMS Coupled with DPPH Assay,” Food Chemistry, Vol. 117, No. 4, 2009, pp. 750-756. doi:10.1016/j.foodchem.2009.04.071
- H. Masuzawa, “The Diuretic Action of the Extract of Phytolacca Root (Phytolacca esculenta) and of Extracts of Some Plants and Drugs. II. The Diuretic Action of Some Plants and Drugs,” Journal of Okayama Medical Association, Vol. 52, No. 1, 1940, pp. 1813-1821.
- S. Oyama, “Extraction of an Active Principle from Houttuynia cordata,” JP Patent No. 25000880, 1950.
- G. Inagak, “Houttuynia cordata Extracts for Treatment of Athlete’s Foot,” JP Patent No. 53050313, 1978.
- Y. Isogai, “An Antimicrobial Substance Isolated from the Rhizome of Houttuynia cordata,” Scientific Papers of the College of General Education, University of Tokyo, Tokyo, 1952.
- H. Chikane, S. Yuka and Y. Takao, “Antibacterial Activity of Extracts from Houttuynia cordata and It’s Components,” Bulletin of Saitama Medical School Junior College, Vol. 14, No. 2, 2003, pp. 1-6.
- C. Lee and S.-J. Lin, “Constituents of Houttuynia cordata,” Beiyi Xuebao, Vol. 6, No. 1, 1974, pp. 75-78.
- K.-I. Hoshishima and H. Okabe, “Promoting Agents Obtained from Houttuynia cordata for the Production of an Antibiotic Substance by a Strain of Gram-Positive, SporeBearing Bacilli,” Tohoku Journal of Experimental Medicine, Vol. 52, No. 3, 1950, pp. 265-271.
- C. P. Kala, “Ethnomedicinal Botany of the Apatani in the Eastern Himalayan Region of India,” Journal of Ethnobiology and Ethnomedicine, Vol. 1, No. 11, 2005, pp. 11- 18. doi:10.1186/1746-4269-1-11
- J. W. Kim, H. J. Ji, J. H. Ahn and M. S. Kim, “Cosmetic Composition Containing Herbal Medicine Extracts for Preventing Wrinkle,” KR Patent No. 2009002678, 2009.
- M. Ida, “Cosmetics,” JP Patent No. 54145226, 1979.
- T. Tehara, “Antiaging Cosmetics Containing Natural Products,” JP Patent No. 2006241036, 2006.
- Y. S. You, “Houttuynia cordata Face Lotion,” KR Patent No. 2001016594, 2001.
- M. Araki, “Additives for Cosmetics to Improve the Skin Conditions,” JP Patent No. 2007246516, 2007.
- S. H. So and Y. O. Kim, “Cosmetic Composition for Alleviating Skin Inflammation,” KR Patent No. 2009- 049401, 2009.
- H. M. Kim, H. J. Kim, S. G. Park, J. J. Kim and J. H. Choi, “Cosmetic Composition for Alleviating Skin Trouble,” KR Patent No. 2009002678, 2009.
- N. Takagi, M. Kamiya and K. Yoshida, “Cosmetics Containing Rhinacanthus nasuta Extracts, Ganoderma lucidum Extracts and/or Houttuynia cordana Extracts for Skin Aging Control and Hair Protection,” JP Patent No. 09143025, 1997.
- J. S. Choi, “Hair Care Product for Nourishing Hair and Preventing Dandruff and Method for Manufacturing the Same,” KR Patent No. 2004037057, 2004.
- Y. S. You, “Method of Preparing Functional Massage Pack Useful for Treating Acne from Rice Powder, Houttuynia Cordata Thunb. And Green Tea Leaves,” KR Patent No. 2006059930, 2006.
- T. H. Hoang, V. B. Ha, Q. H. Tran and V. H, Ha, “Antineoplastic Activity of Flavonoid Components Extracted from Leaves of Houttuynia cordata Thunb. in Vietnam,” Tap Chi Duoc Hoc, Vol. 51, No. 10, 2003, pp. 9-10.
- C. W. Choi, S. C. Kim, S. S. Hwang, B. K. Choi, H. J. Ahn, M. Y. Lee, S. H. Park and S. K. Kim, “Antioxidant Activity and Free Radical Scavenging Capacity between Korean Medicinal Plants and Flavonoids by AssayGuided Comparison,” Plant Science, Vol. 163, No. 6, 2002, pp. 1161-1168. doi:10.1016/S0168-9452(02)00332-1
- H. Zeng, L. Jiang and Y. Zhang, “Chemical Constituents of Volatile Oil from Houttuynia cordata Thunb,” Journal of Plant Resources and Environment, Vol. 12, No. 3, 2003, pp. 50-52.
- J. Meng, X. Dong, Y. Zhou, Z. Jiang, S. Liang and Z. Zhao, “Orthogonal Experiment Using SFE-CO2 in Extraction of Essential Oil from Fresh Houttuynia cordata and Analysis of Essential Oil by GC-MS,” China Journal of Chinese Materia Medica, Vol. 32, No. 3, 2007, 215- 217.
- M. I. Ch, Y. F. Wen and Y. Cheng, “Gas Chromatographic/Mass Spectrometric Analysis of the Essential Oil of Houttuynia cordata Thunb by Using On-Column Methylation with Tetramethylammonium Acetate,” Journal of AOAC International, Vol. 90, No. 1, 2007, pp. 60-67.
- X. Hao, L. Li, Z. Ding and Y. Yi, “Analysis of Essential Oil from Houttuynia cordata in Guizhou,” Acta Botanica Yunnanica, Vol. 17, No. 3, 1995, pp. 350-352.
- Z. Zeng, Y. Liang and C. Xu, “Comparing Chemical Fingerprints of Herbal Medicines Using Modified Window Target-Testing Factor Analysis,” Analyical And Bioanalytical Chemistry, Vol. 381, No. 4, 2005, pp. 913-924
- H.-D. Kwon, I.-H. Cha, W.-K. Lee, J.-H. Song and I.-H. Park, “Antibacterial Activity of Volatile Flavor Components from Houttuynia cordata Thunb,” Journal of Food Science and Nutrition, Vol. 1, No. 2, 1996, pp. 208-213.
- J.-M. Kang, I.-H. Cha, Y.-K. Lee and H.-S. Ryu, “Identification of Volatile Essential Oil, and Flavor Characterization and Antibacterial Effect of Fractions from Houttuynia cordata Thunb. I. Identification of Volatile Essential Oil Compounds from Houttuynia cordata Thunb,” Han’guk Sikp’um Yongyang Kwahak Hoechi, Vol. 26, No. 2, 1997, pp. 209-213.
- J.-M. Kang, I.-H. Cha, Y.-K. Lee and H.-S. Ryu, “Identification of Volatile Essential Oil, and Flavor Characterization and Antibacterial Effect of Fractions from Houttuynia cordata Thunb. II. Flavor Characterization and Antibacterial Effect of Fraction from Houttuynia cordata Thunb Obtained by Preparative HPLC,” Han’guk Sikp’um Yongyang Kwahak Hoechi, Vol. 26, No. 2, 1997, pp. 214-221.
- L. Chen, W. Wu and Y. Zheng, “TLC Analysis on Essential Oil in Houttuynia cordata with Different Chromosome Numbers,” Chinese Traditional and Herbal Drugs, Vol. 35, No. 12, 2004, pp. 1399-1402.
- Y.-L. Liu and Z.-F. Deng, “Study on the Chemical Constituents of the Essential Oil from Houttuynia cordata Thunb,” Chinese Bulletin of Botany, Vol. 21, No. 3, 1979, pp. 244-249.
- M. Qi, X. Ge, M. Liang and R. Fu, “Flash Gas Chromatography for Analysis of Volatile Compounds from Houttuynia cordata Thunb,” Analytica Chimica Acta, Vol. 527, No. 1, 2004, pp. 69-72. doi:10.1016/j.aca.2004.08.073
- H. Lu, “Fingerprint and Fingerprint-Efficacy Study of Houttuynia cordata Thunb. and Injection, a Traditional Chinese Medicine (TCM),” Central South University, Chemistry & Chemical Engineering College, Changsha, 2006.
- C. Huang, W. Wu and Y. Zheng, “Analysis on the Chemical Constituents of Essential Oil from Different Parts of Houttuynia cordata Thunb,” Chinese Journal of Pharmaceutical Analysis, Vol. 27, No. 1, 2007, pp. 40- 44.
- W.-F. Yang, Y. Chen and Y.-Y. Cheng, “Essential Oils from Various Parts of Houttuynia cordata Thunb,” Chinese Traditional and Herbal Drugs, Vol. 37, No. 8, 2006, pp. 1149-1151.
- H. Lu, X. Wu, Y. Liang and J. Zhang, “Variation in Chemical Composition and Antibacterial Activities of Essential Oils from Two Species of Houttuynia cordata Thunb,” Chemical & Pharmaceutical Bulletin, Vol. 54, No. 7, 2006, pp. 936-940. doi:10.1248/cpb.54.936
- H. Lu, Y. Liang, X. Wu, L. Yi and S. Chen, “Comparative Study of Fingerprints of Houttuynia cordata Injection Made of Fresh and Dry Raw Material,” Chinese Journal of Analytical Chemistry, Vol. 34, No. 6, 2006, pp. 813- 816.
- H. Nakamura, T. Ota and G. Fukuchi, “The Constituents of Diuretic Drugs. II. The Flavonol Glucoside of Houttuynia cordata Thunb,” Journal of the Pharmaceutical Society of Japan, Vol. 56, No. 3, 1936, p. 68.
- J. Meng, X. Dong, Z. Jiang, S. Leung and Z. Zhao, “Study on Chemical Constituents of Flavonoids in Fresh Herb of Houttuynia cordata,” China Journal of Chinese Materia Medica, Vol. 31, No. 16, 2006, pp. 1335-1337.
- J. Meng, X. P. Dong, Y. S. Zhou, Z. H. Jiang, S.-Y. K. Leung and Z. Z. Zhen, “Studies on Chemical Constituents of Phenols in Fresh Houttuynia cordata,” China Journal of Chinese Materia Medica, Vol. 32, No. 10, 2007, pp. 929-931.
- S.-C. Chou, C.-R. Su, Y.-C. Ku and T.-S. Wu, “The Constituents and Their Bioactivities of Houttuynia cordata,” Chemical & pharmaceutical Bulletin, Vol. 57, No. 11, 2009, pp. 1227-1230. doi:10.1248/cpb.57.1227
- S. H. Li and X. J. Wu, “Studies on Flavonoids Extracting Methods in Different Parts of Houttunia cordata Thunb,” Journal of Jishou University, Vol. 28, No. 3, 2007, pp. 117-118.
- Y. Zhang, S. Li and X. Wu, “Pressurized Liquid Extraction of Flavonoids from Houttuynia cordata Thunb,” Separation and Purification Technology, Vol. 58, No. 3, 2008, pp. 305-310. doi:10.1016/j.seppur.2007.04.010
- D. S. Jang, J. M. Kim, Y. M. Lee, J. L. Yoo, Y. S. Kim, J.-H. Kim and J. S. Kim, “Flavonols from Houttuynia cordata with Protein Glycation and Aldose Reductase Inhibitory Activity,” Natural Product Sciences, Vol. 12, No. 4, 2006, pp. 210-213.
- S. Huang, C. Wan, Y. Zou and R. Li, “Extraction and Identification of Flavones from Houttuynia cordata Thunb. by Ultrasonic Wave,” Li Shizhen Medicine And Materia Medica Research, Vol. 17, No. 11, 2006, pp. 2261-2262.
- C. Ye, J. Kan, S. Tan, J. Fan and G. Yang, “Microwave Assisted Extraction of Total Flavonoids from Houttuynia cordata Thunb. Leaves,” China Brewing, No. 1, 2009, pp. 134-137.
- C. Ye, J. Kan, S. Tan, Q. Zhu, L. Xu and G. Yang, “Extraction and Separation of Flavonoids from Houttuynia cordata Thunb. Leaves,” Transactions of the Chinese Society of Agricultural Engineering, Vol. 24, No. 10, 2008, pp. 227-232.
- S. Y. Xu, J. X. Hong, G. M. Yan, Y. F. Lin and J. Y. Sun, “Study on Purification Process of Extract of the Fresh Houttuynia cordata,” Research and Practice on Chinese Medicines, Vol. 22, No. 2, 2008, pp. 55-58.
- T. Zhang and D. Chen, “Anticomplementary Principles of a Chinese Multiherb Remedy for the Treatment and Prevention of SARS,” Journal of Ethnopharmacology, Vol. 117, No. 2, 2008, pp. 351-361. doi:10.1016/j.jep.2008.02.012
- Y. Guo and J. Xu, “Extraction and Purification of Houttuyniae cordata Flavonoids and Identification of Flavonoids Type,” Food Science, Vol. 28, No. 9, 2007, pp. 287- 291.
- X. Xu, H. Ye, W. Wang, L. Yu and G. Chen, “Determination of Flavonoids in Houttuynia cordata Thunb. and Saururus chinensis (Lour.) Bail. by Capillary Electrophoresis with Electrochemical Detection,” Talanta, Vol. 68, No. 3, 2006, pp. 759-764. doi:10.1016/j.talanta.2005.05.027
- K. H. Choe, S. J. Kwon and D. S. Jung, “A Study on Chemical Composition of Saururaceae Growing in Korea. 4. On Flavonoid Constituents of Houttuynia cordata,” Analytical Science & Technology, Vol. 4, No. 3, 1991, pp. 285-288.
- J.-I. Fuse, H. Kanamori, I. Sakamoto and S. Yahara, “Flavonol Glycosides in Houttuynia cordata,” Natural Medicines, Vol. 48, No. 4, 1994, pp. 307-311.
- Q. C. Eng, Z. N. Yang, J. W. Hu, W. S. Cheng and C. X. Li, “Determination of Contents of Seven Flavonoids in Houttuynia cordata Thunb by HPLC,” Journal of Jiangxi Normal University, Vol. 32, No. 6, 2008, pp. 645-648, 661.
- L.-S. Wu, J.-P. Si and X.-Q. Yuan, “Quantitive Variation of Flavonoids in Houttuynia cordata from Different Geographic Origins in China,” Chinese Journal of Natural Medicines, Vol. 7, No. 1, 2009, pp. 40-45.
- J. Meng, H. Liao, X. Dong and Z. Zhao, “Quantitative Analysis of Flavonoids in Freeze-Dried Houttuynia cordata from Different Habitats by HPLC-MS,” Journal of Guangdong College of Pharmacy, Vol. 24, No. 2, 2008, pp. 114-117.
- J. Meng, K. S.-Y. Leung, Z. Jiang, X. Dong, Z. Zhao and L.-J. Xu, “Establishment of HPLC-DAD-MS Fingerprint of Fresh Houttuynia cordata,” Chemical & Pharmaceutical Bulletin, Vol. 53, No. 12, 2005, pp. 1604-1609. doi:10.1248/cpb.53.1604
- J.-C. Park, J.-M. Hur, J.-G. Park, S.-J. Park, J.-H. Lee, N.-J. Sung, M.-R. Choi, S.-H. Song, M.-S. Kim and J.-W. Choi, “The Effects of Houttuynia cordata on the Hepatic Bromobenzene Metabolizing Enzyme System in Rats and Isolation of Phenolic Compounds,” Korean Journal of Pharmacognosy, Vol. 31, No. 2, 2000, pp. 228-234.
- T. Kawamura, Y. Hisata, K. Okuda, Y. Noro, T. Tanaka, M. Yoshida and E. Sakai, “Pharmacognostical Studies of Houttuyniae Herba. (1) Flavonoid Glycosides Contents of Houttuynia cordata Thunb,” Natural Medicines, Vol. 48, No. 3, 1994, pp. 208-212.
- E. Sakai, T. Shibata, T. Kawamura, Y. Hisata, Y. Noro, M. Yoshida and T. Tanaka, “Pharmacognostical Studies of Houttuyniae Herba: 2. Growth and Flavonoid Glycoside Contents of Houttuynia cordata Thunb. Cultivated under Shade Condition,” Natural Medicines, Vol. 50, No. 1, 1996, pp. 45-48.
- H. Nishiya, K. Ishiwata, K. Komatsu, O. Nakata, K. Kitamura and S. Fujii, “Platelet Aggregation Inhibitors from Jyu-Yaku (Houttuyniae Herb),” Chemical & Pharmaceutical Bulletin, Vol. 36, No. 5, 1988, pp. 1902-1904. doi:10.1248/cpb.36.1902
- T. T. Jong and M. Y. Jean, “Alkaloids from Houttuynia cordata,” Journal of the Chinese Chemical Society, Vol. 40, No. 3, 1993, pp. 301-303.
- L. Wang, Y. Zhao, L. Zhou and J. Zhou, “Chemical Constituents of Houttuynia cordata,” Chinese Traditional and Herbal Drugs, Vol. 38, No. 12, 2007, pp. 1788-1790.
- K. H. Choe, S. J. Kwon and K. C. Lee, “Chemical Composition of Saururaceae Growing in Korea. (3). On Fatty Acids and Amino Acids of Houttuynia cordata and Saururus chinensis,” Punsok Kwahak, Vol. 2, No. 2, 1989, pp. 285-292.
- S. Takagi, M. Yamaki, K. Masuda and M. Kubota, “On the Constituents of the Terrestrial Part of Houttuynia cordata Thunb,” Shoyakugaku Zasshi, Vol. 32, No. 2, 1978, pp. 123-125.
- X. Wu, S. Li, A. Li and J. Zhang, “Chemical Components from Herba of Houttuynia cordata Thunb,” Journal of Chinese Medicinal Materials, Vol. 31, No. 8, 2008, pp. 1168-1170.
- T. T. Jong and M. Y. Jean, “Constituents of Houttuyniae cordata and the Crystal Structure of Vomifoliol,” Journal of the Chinese Chemical Society, Vol. 40, No. 4, 1993, pp. 399-402.
- S.-C. Chou, “The Constituents of the Houttuynia cordata Thumb,” National Cheng Kung University, Tainan, 2005.
- M. Mori, K. Suzuki, Y. Tsukahara and M. Nada, “Changes in the Components of Houttuynia cordata during Growth,” Sagami Joshi Daigaku Kiyo, Shizenkei, Vol. 59B, No. 3, 1995, pp. 89-95.
- X. Gong and M. Cheng, “Analysis and Appraisal of Nutrients of Houttuynia cordata Thunb,” Food and Fermentation Industries, Vol. 30, No. 11, 2004, pp. 102-105.
- Y. Kimura and Y. Nishikawa, “Standardization of Crude Drugs. III. 2. Component of Houttuynia cordata,” Journal of the Pharmaceutical Society of Japan, Vol. 73, No. 47, 1953, pp. 196-198.
- T. Ohta, “The Diuretic Components of Houttuynia cordata,” Journal of the Pharmaceutical Society of Japan, Vol. 62, No. 2, 1942, pp. 105-106.
- W. Zhang, F. Lu, S. Pan and S. Li, “Extraction of Volatile Oil from Houttuynia cordata and Its Anti-Biotic and Anti-Virus Activities,” Practical Preventive Medicine, Vol. 15, No. 2, 2008, pp. 312-316.
- J. Meng, X. Zong and X. Dong, “Study on Pharmacological Effects of Fresh and Dry Houttuynia cordata Thunb,” Li Shizhen Medicine and Materia Medica Research, Vol. 19, No. 6, 2008, pp. 1315-1316.
- J. Zhang, X. Wu, Z. Luo and X. Zhong, “Determination of Chemical Components and Antibacterial Activities of Houttuynia cordata Thunb. during Desiccation and Storage,” Food Science, Vol. 28, No. 11, 2007, pp. 565-569.
- M. Fu, X. Wu, D. Liu, J. Zhang, L. Wei and X. Jiang, “Research on Antibacterial Activity in Vitro of the Houttuynia cordata Thunb Injection,” Journal of Anhui Agricultural Science, Vol. 35, No. 8, 2007, pp. 2264-2265.
- P. Qian, Y. Liang and H. Lu, “The Study of Antibacterial Activity in Quality Control of Houttuynia cordata Injection,” Journal of Xiangtan Normal University, Vol. 29, No. 3, 2007, pp. 12-14.
- Y. Hou and X. Zhang, “Antiphlogistic Action of Houttuynia cordata Injection in Vitro and in Mice,” China Journal of Chinese Materia Medica, Vol. 15, No. 4, 1990, pp. 221-222, 255.
- T. Kosuge and H. Okeda, “Nonylpyrazole, a New Antimicrobial Substance,” Journal of Biochemistry, Vol. 41, No. 2, 1954, pp. 183-186.
- G. Kim, D. Kim and J. Lim, “Biological and Antibacterial Activities of the Natural Herb Houttuynia cordata Water Extract against the Intracellular Bacterial Pathogen Salmonella within the RAW 264.7 Macrophage,” Biological & Pharmaceutical Bulletin, Vol. 31, No. 11, 2008, pp. 2012-2017. doi:10.1248/bpb.31.2012
- S. B. Bharate, “Medicinal Plants with Anti-HIV Potential,” Journal of Medicinal and Aromatic Plant Sciences, Vol. 25, No. 2, 2003, pp. 427-440.
- M. Shimura, Y. Zhou, Y. Asada, T. Yoshikawa, K. Hatake, F. Takaku and Y. Ishizaka, “Inhibition of VprInduced Cell Cycle Abnormality by Quercetin: A Novel Strategy for Searching Compounds Targeting Vpr,” Biochemical and Biophysical Research Communications, Vol. 261, No. 2, 1999, pp. 308-316. doi:10.1006/bbrc.1999.0994
- N. Morita, K. Hayashi, A. Fujita and H. Matsui, “Extraction of Antiviral Substances from Houttuynia cordata Thunb,” JP Patent No. 07118160, 1995.
- Y. Yan, X. Chen, X. Yang, S. Wu, D. Fang and C. Dong, “Cooperative Anti-Influenza Virus Activities of Amantadine, Ribavirin and Herb Houttuynia,” Virologica Sinica, Vol. 17, No. 2, 2002, pp. 192-194.
- D. H. Kwon, W. J. Choi, C. H. Lee, J. H. Kim and M. B. Kim, “Flavonoid Compound Having an Antiviral Activity,” WO Patent No. 2007069823, 2007.
- H.-J. Choi, J.-H. Kim, C.-H. Lee, Y.-J. Ahn, J.-H. Song, S.-H. Baek and D.-H. Kwon, “Antiviral Activity of Quercetin 7-Rhamnoside against Porcine Epidemic Diarrhea Virus,” Antiviral Research, Vol. 81, No. 1, 2009, pp. 77-81.
- X. Zheng, X. Tang and X. Su, “Experimental Study of Inhibitory Effect of the Four Traditional Chinese Herb Medicines on Epidemic Hemorrhagic Fever Virus,” Bulletin of Hunan Medical University, Vol. 18, No. 2, 1993, pp. 165-167.
- T.-Y. Lin, Y.-C. Liu, J.-R. Jheng, H.-P. Tsai, J.-T. Jan, W.-R. Wong and J.-T. Horng, “Anti-Enterovirus 71 Activity Screening of Chinese Herbs with Anti-Infection and Inflammation Activities,” American Journal of Chinese Medicine, Vol. 37, No. 1, 2009, pp. 143-158.
- X. Kou, “Traditional Chinese Medicine Composition Containing Taraxacum and Glycyrrhiza and Others for Treating Lung Cancer,” CN Patent No. 101406657, 2009.
- Q. Li, “Chinese Medical Honeyed Pill for Treating Lung Carcinoma,” CN Patent No. 101391036, 2009.
- H. Jung, J. Choi and C. Jin, “Effects of Flos Lonicerae and Herba Houttuyniae on Human Cancer Cell-Lines,” Korean Journal of Oriental Physiology & Pathology, Vol. 10, No. 3, 1996, pp. 126-132.
- W. Bae, H. Go and C. Kim, “Experimental Study on the Effect of Houttuyniae Herbal Accupuncture on the Growth of Melanoma B16 in Mice,” The Korean Acupuncture and Moxibustion Journal, Vol. 18, No. 2, 2001, pp. 186-201.
- K.-B. Kwon, E.-K. Kim, B.-C. Shin, E.-A. Seo, J.-Y. Yang and D.-G. Ryu, “Herba Houttuyniae Extract Induces Apoptotic Death of Human Promyelocytic Leukemia Cells via Caspase Activation Accompanied by Dissipation of Mitochondrial Membrane Potential and Cytochrome c Release,” Experimental and Molecular Medicine, Vol. 35, No. 2, 2003, pp. 91-97.
- D. S. Hah, C. H. Kim, J.-D. Ryu, E. K. Kim and J. S. Kim, “Evaluation of Protective Effects of Houttuynia cordata on H2O2-Induced Oxidative DNA Damage Using an Alkaline Comet Assay in Human HepG2 Cells,” Journal of Toxicology and Public Health, Vol. 23, No. 1, 2007, pp. 25-31.
- H. Ha, D. Y. Jung and S. W. Park, “Anticancer Effect of Houttuynia cordata Extract on Cancered ICR Mouse and L1210 Cells with Changes of SOD and GPx Activities,” Yakhak Hoechi, Vol. 48, No. 4, 2004, pp. 219-225.
- A. Murakami, S. Jiwajinda and K. Koshimizu, “Screening for in Vitro Anti-Tumor Promoting Activities of Edible Plants from Thailand,” Cancer Letters, Vol. 95, No. 1-2, 1995, pp. 139-146.
- Y.-Y. Chen, C.-M. Chen, P.-Y. Chao, T.-J. Chang and J.-F. Liu, “Effects of Frying Oil and Houttuynia cordata Thunb on Xenobiotic-Metabolizing Enzyme System of Rodents,” World Journal of Gastroenterology, Vol. 11, No. 3, 2005, pp. 389-392.
- H. Fan, W. Qu, Y. Li and M. Sun, “Experimental Investigation for Anti-Tumor Activity of Flavonoid from the Houttuynia cordata Thunb. in Vitro,” Chinese Journal of Hospital Pharmacy, Vol. 28, No. 7, 2008, pp. 528-531.
- J. Meng, D. He, Y. Zhou and X. Dong, “Comparison on the Pharmacological Effects of Different Extract Parts from Houttuynia cordata Thunb,” Li Shizhen Medicine And Materia Medica Research, Vol. 19, No. 5, 2008, pp. 1050-1051.
- Q. Feng, Y. Ren, Y. Wang, H. Ma, J. Xu, C. Zhou, Z. Yin and L. Luo, “Anti-Inflammatory Effect of SQC-Beta-CD on Lipopolysaccharide-Induced Acute Lung Injury,” Journal of Ethnopharmacology, Vol. 118, No. 1, 2008, pp. 51-58.
- J. Hong, B. Wang, Y. Gao, B. Feng, J. Mo, Y. Wang, H. Yao, X. Xu, W. Zhao and F. Tang, “Effect of Fresh Herba Houttuyniae Extract on Lipopolysaccharide-Induced Inflammation in Mice,” The Chinese Journal of Modern Applied Pharmacy, Vol. 25, No. 5, 2008, 376-378.
- J. Lee, I. Kim, J. Kim, J. Kim, D. Kim and C. Yun, “Suppressive Effects of Houttuynia cordata Thunb (Saururaceae) Extract on Th2 Immune Response,” Journal of Ethnopharmacology, Vol. 117, No. 1, 2008, pp. 34-40.
- A. Probstle and R. Bauer, “Aristolactams and a 4,5-Dioxoaporphine Derivative from Houttuynia cordata,” Planta Medica, Vol. 58, No. 6, 1992, pp. 568-569.
- K. Taguchi, Y. Hagiwara, K. Kajiyama and Y. Suzuki, “Pharmcological Studies of Houttuyniae Herba: The Antiinflammatory Effect of Quercitrin,” Journal of the Pharmaceutical Society of Japan, Vol. 113, No. 4, 1993, pp. 327-333.
- X.-L. Jiang and H.-F. Cui, “Different Therapy for Different Types of Ulcerative Colitis in China,” World Journal of Gastroenterology, Vol. 10, No. 10, 2004, pp. 1513- 1520.
- H.-M. Lu, Y.-Z. Liang and P. Qian, “Profile-Effect on Quality Control of Houttuynia cordata Injection,” Acta pharmaceutica Sinica, Vol. 40, No. 12, 2005, pp. 1147- 1150.
- H.-M. Lu, Y.-Z. Liang, X.-J. Wu and P. Qiu, “Tentative Fingerprint-Efficacy Study of Houttuynia cordata Injection in Quality Control of Traditional Chinese Medicine,” Chemical & Pharmaceutical Bulletin, Vol. 54, No. 5, 2006, pp. 725-730.
- Y.-C. Kim and S.-K. Chung, “Reactive Oxygen Radical Species Scavenging Effects of Korean Medicinal Plant Leaves,” Food Science and Biotechnology, Vol. 11, No. 4, 2002, pp. 407-411.
- E. Cho, T. Yokozawa, D. Rhyu, S. Kim, N. Shibahara and J. Park, “Study on the Inhibitory Effects of Korean Medicinal Plants and Their Main Compounds on the 1,1-diphenyl-2-picrylhydrazyl Radical,” Phytomedicine, Vol. 10, No. 6-7, 2003, pp. 544-551.
- E. J. Cho, T. Yokozawa, D. Y. Rhyu, H. Y. Kim, N. Shibahara and J. C. Park, “The Inhibitory Effects of 12 Medicinal Plants and Their Component Compounds on Lipid Peroxidation,” American Journal of Chinese Medicine, Vol. 31, No. 6, 2003, pp. 907-917.
- H. Wang and Y. Xiu, “Effect of Houttuynia cordata on Urinary Albumin and Insulin Resistance of Diabetes Mellitus Rats,” Traditional Chinese Drug & Clinical Pharmacology, Vol. 19, No. 1, 2008, pp. 12-14.
- H. Wang, Y. Xiu and K. Sun, “Effect of Houttuynia cordata on Fat Metabolism in Plasma and Insulin Resistance of Diabetes Mellitus Rats,” Chinese Journal of Basic Medicine in Traditional Chinese Medicine, Vol. 15, No. 1, 2009, pp. 72-73, 78.
- F. Wang, F. Lu, G. Chen and L. Xu, “Influence of Houttuynia cordata Thumb on Expression of TGF-β1 and HGF in Kidney Tissue of Streptozotocin Induced Diabetic Rats,” The 3rd World Integrative Medicine Congress Abstracts, Guangzhou, 21-24 September 2007, p. 722.
- F. Wang, F. Lu and L. Xu, “Effects of Houttuynia cordata Thumb on Expression of BMP-7 and TGF-β1 in the Renal Tissues of Diabetic Rats,” Journal of Traditional Chinese Medicine, Vol. 27, No. 3, 2007, pp. 220-225.
- Y. Kimata, Y. Eto, M. Asanoma, Y. Sakabe and H. Horitsu, “Inhibitory Actions of Chloroform Extracts from Natural Medicines on the Mutagenicity of Trp-P-2 and 1-NP,” Chukyo Joshi Daigaku Kenkyu Kiyo, Vol. 31, No. 2, 1997, pp. 123-130.
- Z. Song, C. Wang, J. Cheng, F. Li, Z. Zhu, Y. Ning and M. Zhang, “Effect of Injection of Houttuynia cordata Thunb, Hyperiaum japonlcum Thunb and Erycibe obtusifoIia Benth on Immune Function of Rats,” Chinese Traditional and Herbal Drugs, Vol. 24, No. 12, 1993, pp. 643-644, 438.
- J. Kim, C.-S. Park, Y. Lim and H.-S. Kim, “Paeonia japonica, Houttuynia cordata, and Aster scaber Water Extracts Induce Nitric Oxide and Cytokine Production by Lipopolysaccharide-Activated Macrophages,” Journal of Medicinal Food, Vol. 12, No. 2, 2009, pp. 365-373.
- K.-M. Ji, M. Li, J.-J. Chen, Z.-K. Zhan and Z.-G. Liu, “Anaphylactic Shock and Lethal Anaphylaxis Caused by Houttuynia cordata Injection, a Herbal Treatment in China,” Allergy, Vol. 64, No. 5, 2009, pp. 816-817.
- L. Lei, X. Pu, K. Yang, H. Wang, Y. Liu, D. Zhang and L. Zhang, “Adverse Response Caused by Intravenous Drip of Houttuynia cordata Injection I,” Pharmacology and Clinics of Chinese Materia Medica, Vol. 23, No. 5, 2007, pp. 131-133.
- Y. Liu, X. Pu and L. Lei, “Adverse Response Caused by Intravenous Drip of Houttuynia cordata Injection Ⅱ,” Pharmacology and Clinics of Chinese Materia Medica, Vol. 24, No. 2, 2008, pp. 61-63.
- China TPCo (Ed.), “The Chinese Pharmacopoeia,” China Medical Science Press, Beijing, 2010.
- WHO “General Guidelines for Methodologies on Research and Evaluation of Traditional Medicines,” 2000, p. 1.
- Y.-Z. Liang, P. Xie and K. Chan, “Quality Control of Herbal Medicines,” Journal of Chromatography B, Vol. 812, No. 1-2, 2004, pp. 53-70.
- V. E. Tyler, “Phytomedicines: Back to the Future,” Journal of Natural Products, Vol. 62, No. 11, 1999, pp. 1589- 1592.
- WHO “Guidelines for the Assessment of Herbal Medicines,” 1991.
- J. Wang, R. van der Heijden, S. Spruit, T. Hankermeier, K. Chan, J. van der Greef, G. Xu and M. Wang, “Quality and Safety of Chinese Herbal Medicines Guided by a Systems Biology Perspective,” Journal of Ethnopharmacology, Vol. 126, No. 1, 2009, pp. 31-41.
- C. P. Commission (Ed.), “The Pharmacopoeia of the People’s Republic of China,” China Medical Science Press, Beijing, 2010.
- China Mohotpsro, “Farther Standardization of Raw Materials Administrators for Health Foods,” 2002.
- http://en.wikipedia.org/wiki/Houttuynia
- Y. Wata, “Therapeutic Drinks from Houttuynia cordata,” JP Patent No.56097234, 1981.
- K. Mori, “Deodorizing Fish Oil,” JP Patent No. 35240, 1919.
- J. S. Choi, “Knife-Cut Noodles Using Houttuyniae Herba and Method for Preparing the Same,” KR Patent No. 2004063884, 2004.
- S. H. Kwon and M. S. Kim, “Method for Manufacturing Soy Sauce by Using Houttuynia Cordata and Saururus Chinensis,” KR Patent No. 2009076250, 2009.
- Y. S. You, “Method for Manufacturing Carbonic Drink of Houttuynia Cordata,” KR Patent No. 2002028976, 2002.
- T. H. Hoang, Q. H. Tran, V. B. Ha and D. T. Nguyen, “Study on the Flavonoid Component Extracted from Leaves of Houttuynia cordata Thunb in Vietnam,” Tap Chi Duoc Hoc, No. 9, 2002, pp. 13-15.
- H. Wagner, R. Bauer, P. Xiao, J. Chen and A. Probstle, “Herba Houttuyniae cordatae,” Chinese Drug Monographs and Analysis, Vol. 1, No. 6, 1997, pp. 1-11.
- A. Proebstle, A. Neszmelyi, G. Jerkovich, H. Wagner and R. Bauer, “Novel Pyridine and 1,4-Dihydropyridine Alkaloids from Houttuynia cordata,” Natural Product Letters, Vol. 4, No. 3, 1994, pp. 235-240.
NOTES
*Corresponding author.

