Journal of Biophysical Chemistry
Vol.1 No.1(2010), Article ID:1882,6 pages DOI:10.4236/jbpc.2010.11006
Correlations between delayed fluorescence of chlorophyll, metabolism and yield of plants. II. Influence of moisture of leaf and temperature condition on delayed fluorescence of leaves
Research and Industry Center of Photosynthesizing Organisms, Fееd Additives and Physiologically Active Compounds, Yerevan, Armenia; armin.av@hotmail.com
Received 22 April 2010; revised 28 April 2010; accepted 8 May 2010.
Keywords: delayed fluorescence of chlorophyll; temperature; moisture; frost; weakly associated polypeptides
ABSTRACT
During various temperatures of incubation the dehydration of leaves up to 3.2-3.8% mainly induced increase maximum amplitude of delayed fluorescence of chlorophyll. It was shown that moisture loss with this range could be determined for the most part by the growth of the electrochemical potential of thylakoid membranes. The further incubation of detached leaves at 36℃ temperature, with more notable moisture loss, resulted in specific its decline as opposed to cases of 22 and 6℃ of thermal incubation. It was confirmed that the increased temperatures and moisture loss damage of the cells of plants occurred together induce a greater influence on plants than in case of occurring apart. The results allow to suppose that this can be mostly caused weakly associated polypeptides fallen out from the chloroplast membrane, which may be stipulated by high temperature combined with change ionic and osmotic stresses due moisture loss. Simultaneously, the results showed that the exposure of the critical lowered air temperature led to considerable typical changes of leaves delayed fluorescence parameters of field plants. Therefore, their use can constitute new approaches to elucidate the molecular basis of plant freezing tolerance in a timely manner, based on concentration-related changes and the efficiency of coupling between light and dark processes of plants.
1. INTRODUCTION
The optimum temperature for various reactions of photosynthesis of plants generally varies from 20 to 35℃. In case of short treatment time the increased temperature induced energy metabolism activation of chloroplasts [1,2]. It is well known that water deficit in plants can induce change of osmotic potential [3]. During dehydration of leaves the intensity of photosynthesis increases noticeably at first (up to 4% of moisture loss, named effect of Brilliant) and at second stage it inhibits in case of more moisture loss. The electro-microscopic research showed that at the first period only small changes of packaging the lamellae grains and stroma were obtained, and since the moisture loss of 4% already significant swellings in the lamellae system and infringements of membrane structures were observed [1]. It is noteworthy that in case of interaction between the increased temperatures and the drought the structural organization and regular processes in the cells of plants were broken in more degree [3,4]. Simultaneously, in condition of the water loss of wheat leaves, the roots of which were submerged into polyethylene glycol solutions, an increase in electrolyte leakage, decrease in contents of chlorophylls a and b, major PSII proteins, including the D1 and D2 proteins in the PSII and the LHC2 and I, whole inhibition of PSII activity were obtained [5]. Northern hybridization showed that progressive water stress remarkably reduced contents of the chloroplast gene psbA and psbD and nuclear gene cab transcripts [5] and 62 genes differentially expressed under osmotic stress [6]. On the other hand, it was assumed that water stress had no effects on the primary photochemistry of PSII, but modified the PSII photochemistry in the light-adapted leaves and such modifications may be a mechanism to down-regulate the photosynthetic electron transport to match a decreased CO2 assimilation [7]. It was also assumed that PSII is to be highly droughtresistant: even a drastic desiccation in the air of detached leaf samples only marginally affected the quantum yield for photochemistry in PSII, and water stress markedly modified the responses of PSII to superimposed constraints [9]. The stability of PSII to heat was observed in leaves exposed to water stress conditions: heat treatments (e.g. 42℃ in the dark), which caused a complete and irreversible inhibition of PSII in well-watered leaves, resulted in a small and fully reversible reduction of the photochemical efficiency of PSII in drought-stressed leaves. These results allow to suppose that water stress enhanced resistance of PSII to constraints (heat, strong light at high temperature) that are usually associated with drought in the field [8].
PSII is the main producer of DF (the intensity of the PSI delayed fluorescence is several times lower [9], which possesses raised sensitivity to the action of plant metabolism changes and various environmental factors [10]. DF transients have been shown to be driven by many mechanisms, including electrical and pH gradients [11], acceptor availability, donor availability [12], and redox state of the oxygen-evolving system [13,14]. The delayed fluorescence induction curve, registered in a several minutes time period, encompasses two major phases-a fast one, taking place in the first second of induction, and a slow one, that can last for minutes (Figure 1). Each phase is shaped by several processes running at the same time [13,14]. Those initial phases of the induction which many authors attribute to the changes of the electrical potential, depending on the size of ECP on a membrane on the state of the PSII reaction center, since the light induced potential difference and the proton gradient across the thylakoid membrane are thought to reduce the amount of activation energy necessary for the back reaction between QA– and Z+ to occur and on the other hand, the I level follows the extent of the variable component of fluorescence and has been suggested to be proportional to the amount of oxidized QA at the beginning of illumination [11,13,14], as well as the concentration of active centers of the PSII [11]. It was previously assumed that the time course of DF after the maximum (Figure 1) is possibly influenced by a number of dark photosynthetic processes (directly or indirectly modifying the characteristics of PSII) [13,14]. The decay of induction curve was generally explained by a decrease in the membrane energization as a result of secondary cation efflux from thylakoids [13,14]. Our results showed that half time of I decay-τ describe the level of interaction LR with dark processes of plants [10]. These are a biological base triggering the use of these DF parameters in physiological research.
Thus, our goal was to study the correlations between values of DF parameters and both temperature and moisture influences on leaves.
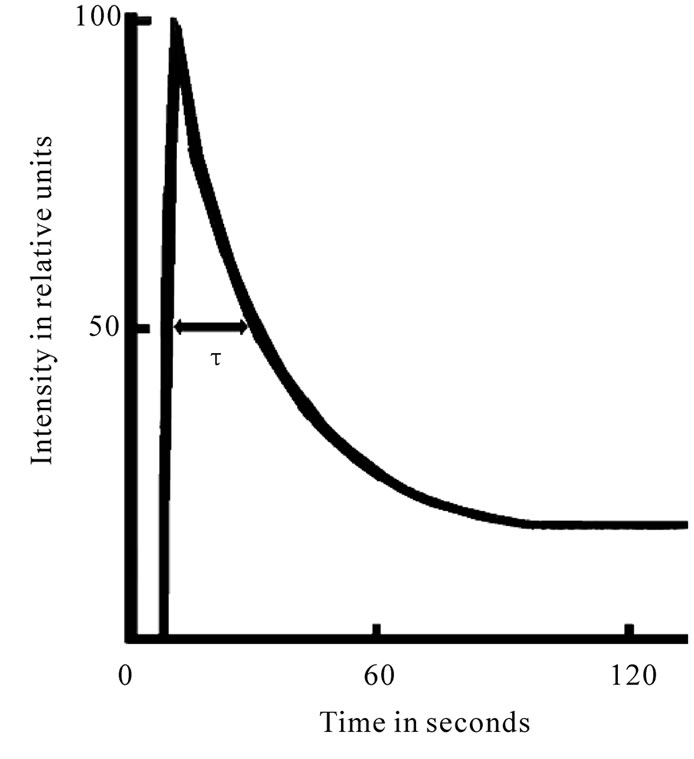
Figure 1. Induction curve of delayed fluorescence of chlorophyll.
2. MATERIALS AND METHODS
2.1. Plant Material and Experimental Design
The potato (Solanum tuberosum) variety Priekulsky and the pea (Pisum sativum) variety Pobeditel were grown on the clay loam soils in the Armenian Ararat Valley, with the soil moisture of 67-71% of field water capacity. The potato was planted on August 24, and the pea was sown on July 30.
2.2. Measurement and Sample Protocol
The moisture of leaves was identified by their drying at 80℃ in oven. The thermal influence was measured on leaves with the initial average moisture of 86.01%, which were detached from field plants in the early hours at air temperature of 22-24℃. In each replicates of thermal treatment of variants the measurement was done on 40 unpaired lobes from the same quantity of plants, with 4 replicates. Each detached from-leave lobe was incubated and measured at various standing temperature of variant.
The influence of frosts was measured on leaves of the field potato (in a plot of 19 plants) and pea plants (in a plot of 33 plants), which had not a visual trace of damage. Each detached from-leave lobe was incubated about 1.5 hour in the dark between paper sheets at temperature of 22℃.
The parameters of the leaves DF induction curve were measured from the center of lobes by using a single-disc phosphoroscope (time period between excitation and recording of DF was 7 ms), Exciting light was obtained from a 200 W glow lamp (KGM-200, provided maximum intensity of modulated visual light at the sample surface of 2000 μmol photons m2/s) and photodiode at temperature of 22℃.
2.3. Statistical Analyses
The data were processed from a purely mathematical standpoint, using the Student’s and Fisher's exact test, with 5% significance level, and by the use of the standard methods of correlation analyses.
3. RESULTS
In case of incubation at 22 and 6℃ temperature affects the dehydration of detached leaves with range up to 3.2-3.8% induced the I significant increase (Tables 1-3, rIM(T) at incubation of 130 and 220 min). In the event of elimination of moisture loss affect the temperature increase induced significant rise of I in all variants (Tables 1 and 3, rIT(M)). The incubation of detached leaves at 36℃ with further more notable moisture loss resulted in I decline as opposed to variants with other thermal treatment (Tables 1-3, rIM(T) at incubation of 400 min). I depends on the size of ECP on a membrane [11,13,14] donor availability [11-13], as well as possible concentration of active PS II [10]. Our results have shown that half time of I decay-τ describe the level of interaction LR with dark processes of plants [10]. In cases of eliminating the τ or M influences on I a significant correspondence was obtained between I changes with the moisture decrease, as well as with τ of variants (Tables 1 and 2).
The moisture loss induced reliable τ increase in cases of elimination of I influence (Tables 2 and 3, rτM(I), rτM(T)). On the other hand, the induced I increase by moisture loss with range up to 3.2-3.8% stipulated τ reliable decrease (Table 2, rIτ(M) in case of 22 and 6℃ temperature effects, Table 3, rτM(T)). Only in case the incubation of detached leaves at 36℃ temperature effect with more notable moisture loss, I declines accompanied by τ significant increase (Table 2, rIτ(M), Table 3 rIτ(T)). The incubation temperature increase induced I growth, as well as stipulated τ decrease of variants (Table 3).
After the preliminary incubation in extreme temperatures in 90 minutes, their affects on DF parameters were excluded through leaves subsequent adaptation to 22℃ (Table 1). In this case, after the subsequent 1.5-hour incubation at 22℃ no significant distinctions for the DF parameters of leaves up to 4% moisture loss were obtained. Consequently, moisture loss in such leaves did not induce significant influence on the concentration of the PSII and the level of interaction between LR and DR. Therefore, this result showed that in this range of moisture loss, I growth was caused by the increased ECP on thylakoid’s membranes due to water loss. These results are consistent also with previous electronic-microscopic finding that the changes obtained in this range of moisture are analogous to those induced by plasmolytic agent’s effect on structural organization of thylakoid membranes [1]. Previously also was shown that the short-term influence of high temperatures and the small dehydration of leaves did not cause a substantial change in the activity of the DR key enzymes [15]. Henceforth it was possible to conclude that the enhancement of the LR and DR interaction efficiency in the case of 1.7-2.5% moisture loss could for the most part be determined by the growth of the thylakoid’s membranes ECP. Therefore, it is apparent that the effect named Brilliant was induced due ECP growth in case of small moisture loss.
After the preliminary incubation of leaves at 375 minutes in extreme temperatures, the thermal effects on leaves’ DF parameters were excluded through leaves subsequent adaptation to 22℃ (Table 1). In this case, after the 1.5-hour incubation of leaves at 22℃ the leaves, which earlier were processed at 36℃, showed a significant reduction in I, compared with other both variants (Tables 1). On the other hand, it was previously obtained that leaves incubated at 22℃ showed I reduction only after loosing over 35-40% moisture [16]. Therefore,
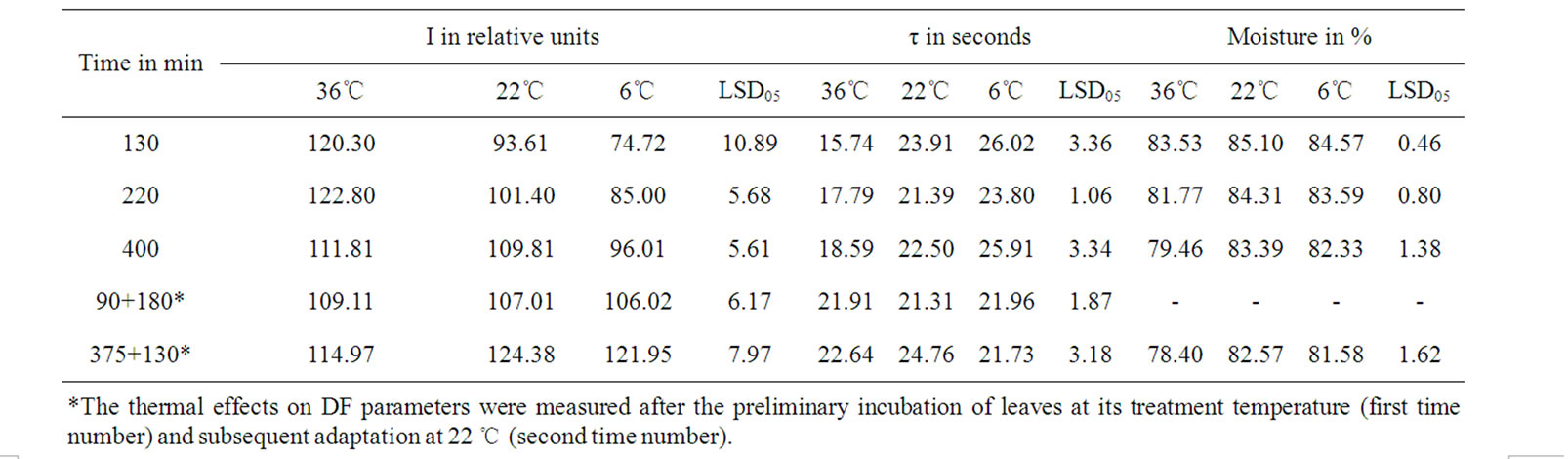
Table 1. Temporary variation of DF parameters and leaf moisture during incubation at different temperatures.
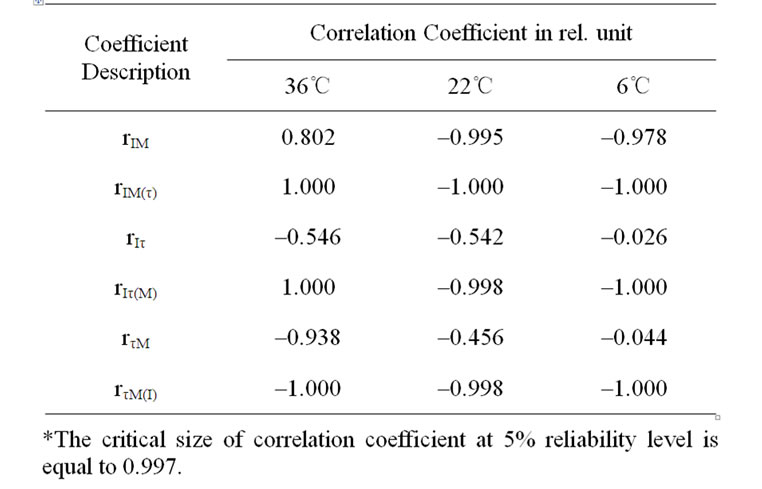
Table 2. Correlative correspondence between DF parameters and leaf moisture in variants of different temperature treatments*.
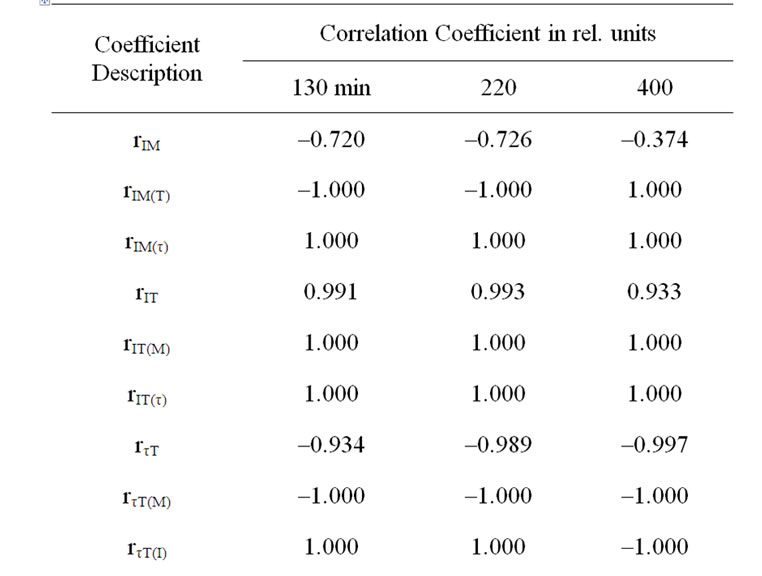
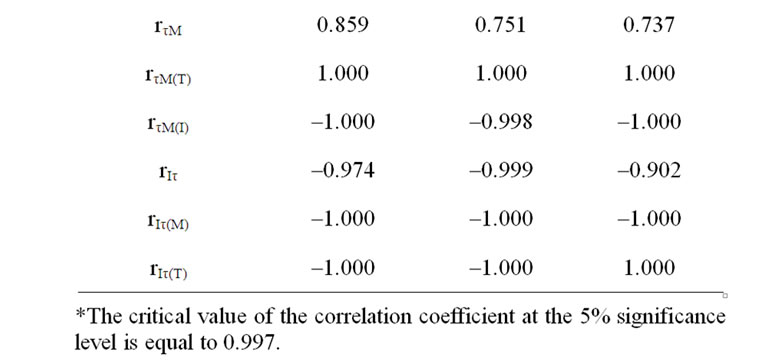
Table 3. Correlative correspondence of DF parameters and temperature and leaf moisture between variants during Incubation at different temperatures.
taking into account the well-known higher DR key centers resistance to increased temperature affect [15], our results showed that obtained specific correlations between parameters of I decrease at 36℃ with more notable moisture loss, caused by both temperature and moisture loss specific influence on PSII quantity reduction
 (a) (b)
(a) (b)
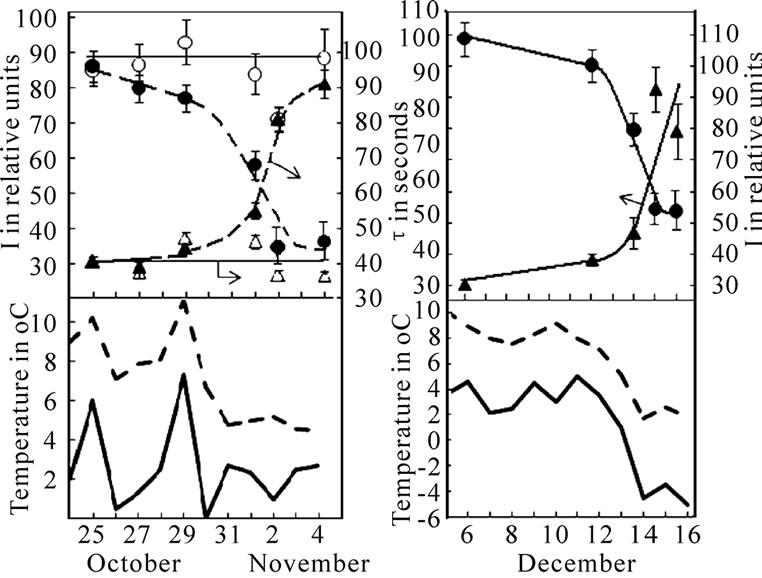
Figure 2.Temporary variation DF parameters (A. potato (●, ▲) and pea (○, ∆), B. pea) and minimal and mean air temperatures. Air temperature is presented by the data of State Hydrometeorological service.
per unit of area.
The critical range of resistance to low temperature is 0 ℃ for potato and from –4 up to –6℃ for pea plants [17]. During the vegetation, the first fall to 0℃ of the minimum daily temperature was observed on October 30. After the frost a gradual I decrease and τ increase were observed in potato leaves, while no significant changes in DF parameters were obtained for the pea leaves (Figure 2(a)). After October 30, the average daily air temperature was about 5℃ that allowed to exclude the influence of the reactivation process on the changes in the potato leaves’ parameters, since the depression of its vital activity occurs already up to an air temperature of 7-8℃ [17]. The lack of significant changes in the pea leaves’ DF parameters was observed (Figure 2(a)) because it’s vital activity reduction occurs at 2℃ of air temperature. However, after the exposures to frost with intensity ranging from –4 to –5℃ a subsequent decrease of I and growth of τ were observed in pea leaves (Figure 2(b)). After frost affects the average daily air temperature was at a lower level, which was necessary for reactivation processes of plant (Figure 2(b)).
4. DISCUSSION
The effect of high temperatures on the photosynthetic apparatus of the leaves and isolated chloroplasts were also performed through their 5-min thermal treatment [16,18]. After subsequent adaptation leaves to the temperature of 22℃ intensity of chlorophyll fluorescence of isolated chloroplasts, Hill’s reaction indicated the damage in the area of PSII at processing above 40℃. The amplitude of electro paramagnetic resonance Signal II (g = 2.0026, ∆Hmax = 7-8 Gs), caused by the functioning of the water-splitting system of PSII, increased above 40℃ thermal affect on chloroplast. In the meantime, the microcalorimetric observation of isolated chloroplasts warmedup at 45℃ has not shown significant damages in the structural organization of basic membranes. Other studies reported that in case of thermal treatment using the water-splitting system was damaged in the first place [19,20]. Our investigation by using fluorescent probe method showed that at the temperature of above and more than 40℃ the weakly associated polypeptides had fallen out from the chloroplast membrane [21-23]. After the separation of chloroplasts from incubation medium content of the obtained weakly associated with membrane polypeptides was 5-6% of the total protein amount in non separated suspension (or 17-20% of membrane whole weakly associated polypeptides). There was also polypeptide of 12-34 kdalton in the separated solution and among them later it was identified 17.2 kdalton thermolabile weakly associated polypeptide of the watersplitting system [24].
Previously it was described that the mineral salts’ induced influences on I mostly depends on the effects of salt concentration on concentration of active PSII, compared to the changes of ECP of membranes [10]. Under high salt conditions, plant growth is severely inhibited due to both osmotic and ionic stresses [6]. In case of salt stress, osmotic stress is triggered by an excess of salt in the soil, and ionic stress is caused by the over-accumulation of salt in the cells and recovery is greater under osmotic stress than in case of salt stress [6]. It was shown that the weakly associated polypeptides had fallen out of the chloroplast membrane also in case of increase in amount of mineral salts in suspension of chloroplast [25]. Last findings showed that in the leaf thermograms curve of DF, stationary level decline was observed starting from temperature at 35℃, which was interpreted as a result of PSII oxygen-evolving system inhibition [26]. These results allow to suppose that the specific effect of both increased temperature and moisture loss could be stipulated by the PSII activity in consequence of disturbance in interaction between weakly associated polypeptides with chloroplast membrane.
Experiments were shown that initial moisture loss could be determined for the most part by the growth of the thylakoid membranes ECP. Therefore, it is apparent that the effect named Brilliant was mainly resulted from the ECP growth. Simultaneously our results confirmed that the combined effects of drought and high temperature were much more severe than those of each separate treatment cells of plants [3,4] may be case of deactivation of water-splitting reaction center. Obtained data allow to suppose that both the increased temperature and moisture loss specific effect could be stipulated by the PSII deactivation through the weakly associated polypeptides fallen out from the chloroplast membrane.
It was shown that frost induced the water crystal formation and as a result increased the cell hydrolytic processes, observed in the content of lipid falls and registered deactivation of PSII and PSI [27-29]. The obtained data conform to the results of both photosynthesis intensity inhibition and PSII activity loss in case of frost. Therefore, our results allow to conclude that the exposure of the critical low air temperature led to considerably typical changes of DF parameters of leaves, which may be then associated with the deactivation of PSII and whole damage of photosynthetic apparatus. The obtained results confirmed our previous assumption that changes in active PSII concentrations and interaction between LR centers and dark processes of plant affect on parameters of DF and moisture loss to stimulate a deactivation of PSII in leaves [10]. Therefore, the use of DF parameters can constitute new approaches to elucidate the molecular basis of plant freezing tolerance in a timely manner, based on concentration-related changes and the efficiency of coupling between LR and dark processes of plants. At the same time, our experiments showed the targets of additional applications of DF parameters aimed at the thermal and moisture influences research. The interest of using the parameters of this luminescence for the diagnosis of plants’ state is identified also by the possibility of their fast measurements on intact objects by the use of comparatively simple and accessible equipment.
REFERENCES
- Niculina, G.N. and Semichatova, O.A. (1983) Influence of a heat on the contents of adenylate in leaves of peas. The influence of a high temperature on the contents of the adenylates in pea leaves. Russian Plant Physiology, 30, 998-1005.
- Tezara, W., Mitchell, V., Driscoll, S.P. and Lawlor, D.W. (2002) Effects of water deficit and its interaction with CO2 supply on the biochemistry and physiology of photosynthesis in sunflower. Journal of Experimental Botany, 53, 1781-1791.
- Polimbetova, F.A., Mamonov, L.K. and Kim, G.G. (1974) Estimation of cereal crops to drought and heat resistance in field conditions. Russian Agricultural Biology, 9, 235- 237.
- Nicolas, M.E., Gleadow, R.M. and Dalling, M.J. (1984) Effects of drought and high temperature on grain growth in wheat. Australian Journal of Plant Physiology, 11, 553-566.
- Liu, W., Yuan, S., Zhang, N., Lei, T., Duan, H., Liang, H. and Lin, H. (2006) Effect of water stress on Photosystem 2 in two wheat cultivars. Biologia Plantarum, 50, 597- 602.
- Ueda, A., Kathiresan, A., Inada, M., Narita, Y., Nakamura, T., Shi, W., Takabe, T. and Bennett, J. (2004) Osmotic stress in barley regulates expression of a different set of genes than salt stress does. Journal of Experimental Botany, 55, 2213-2218.
- Lu, C. and Zhang, J. (1999) Effects of water stress on Photosystem II photochemistry and its thermostability in wheat plants. Journal of Experimental Botany, 50, 1199- 1206.
- Havaux, M. (1992) Stress tolerance of Photosystem II in vivo. Antagonistic effects of water, heat, and photoinhibition stresses. Plant Physiology, 100, 424-443.
- Mar, T., Brehlner, J. and Roy, G. (1975) Induction kinetics of delayed light emission in spinach chloroplasts. Biochimica et Biophysica Acta, 376, 345-353.
- Avagyan, A.B. (2010) Correlations between Delayed Fluorescence of Chlorophyll, Metabolism and Yield of Plants. I. Influence of Fertilizers on Correlations. Journal of Biophysical Chemistry, 1. (in press)
- Wraight, C.A. and Crofts, A.R. (1971) Delayed fluorescence and the high-energy state of chloroplast. European Journal of Biochemistry, 19, 386-397.
- Mar, T., Brehlner, J. and Roy, G. (1975) Induction kinetics of delayed light emission in spinach chloroplasts. Biochimica et Biophysica Acta, 376, 345-353.
- Goltsev, V., Zaharieva, I., Lambrev, P., Yordanov, I. and Strasser, R. (2003) Simultaneous analysis of prompt and delayed chlorophyll a fluorescence in leaves during the induction period of dark to light adaptation. Journal of Theoretical Biology, 225, 171-183.
- Goltsev, V., Chernev, P., Zaharieva, I., Lambrev, P. and Strasser, R.J. (2005) Kinetics of delayed chlorophyll a fluorescence registered in milliseconds time range. Photosynthesis Research, 84, 209-215.
- Avagyan, A.B. (1986) Effect of air temperature and soil moisture content on delayed fluorescence in Pisum sativum leaves in field conditions. Russian Fiziologiya Rastenii, 33, 23-28. http://cat.inist.fr/?aModele = afficheN& cpsidt = 8764680
- Berry, J.A. and Downton, W.J.S. (1987) Photosynthesis dependence from influence of environment factors. Photosynthesis, 2, 276-354.
- Korovin, A.I. (1984) Plants and extreme temperatures. Gidrometizdat, Sankt Petersburg.
- Avagyan, A.B., Venedictov, P.S., Dobrecov, G.E. and Rubin, A.B. (1982) Influence heat damage of pea chloroplasts on their interaction with fluorescent probe ANS. Biological Sciences, 11, 30-35.
- Muller, A. and Witt, H.T. (1961) Trapped primary product of photosynthesis in green plants. Nature, 189, 944-945.
- Kafalieva, D.N. and Bushuev, M.C. (1979) Influence of temperature on photoinduced proton gradient in isolated chloroplasts. Russian Biophysics, 24, 676-680.
- Avagyan, A.B., Venedictov, P.S. and Rubin, A.B. (1982) Interaction between fluorescent probe ANS and chloroplasts. Russian Biophysics, 27, 415-419.
- Avagyan, A.B., Venedictov, P.S., Rubin, A.B. (1984) Application of rodamin 6g as a fluorescent probe for study of chloroplast membranes. Russian Biophysics, 29, 980-983.
- Avagyan, A.B. (1987) The application of the method of fluorescent probes in research of chloroplast membrane properties. Biological Journal of Armenia, 40, 443-448.
- Becker, D.W., Callahan, F.E. and Chiniae, G.M. (1985) Photoactivation of NH2OH-treated leaves: reassembly of realized extrinsic PSII polypeptides and relegation of Mn catalyst of water oxidation. FEBS Letters, 192, 209-214.
- Avagyan, A.B., Venedictov, P.S., Dobrecov, G.E. and Rubin, A.B. (1983) Influence uniand bivalence cations on interaction fluorescent probe ANS and rodamin 6g with chloroplasts. Biological Sciences, 5, 33-36.
- Zaharieva, I. and Goltsev, V. (2003) Advances on Photosystem II investigation by measurement of delayed chlorophyll fluorescence by a phosphoroscopic method. Photochemistry and Photobiology, 77, 292-298.
- Neuner, G. and Pramsohler, M. (2006) Freezing and high temperature thresholds of Photosystem II compared to ice nucleation, frost and heat damage in evergreen subalpine plants. Physiologia Plantarum, 126, 196-204.
- Yi, X.F. and Zhang, Z.B. (2008) Influence of insectinfested cotyledons on early seedling growth of Mongolian oak, Quercus mongolica. Photosynthetica, 46, 139- 142.
- Ehlert, B. and Hincha, D.K. (2008) Chlorophyll fluorescence imaging accurately quantifies freezing damage and cold acclimation responses in Arabidopsis leaves. Plant Methods. http://www.plantmethods.com/content/4/1/12
Abbreviations:
PSII-photosystem II
LHC-light harvesting complex
ECP-electrochemical potential
DF-delayed fluorescence of chlorophyll
I-maximum amplitude of DF curve
τ-time of I half decay
LR-light reactions of photosynthesis
DR-dark reactions
M-leaf moisture
T-temperature
r-correlation coefficient

