World Journal of Vaccines
Vol.2 No.3(2012), Article ID:21527,6 pages DOI:10.4236/wjv.2012.23020
Purification of Dengue Virus Particles by One-Step Ceramic Hydroxyapatite Chromatography
![]()
1R&D Department, PENTAX New Ceramics Division, HOYA Corporation, Tokyo, Japan; 2Protein Technos Institute, Atsugi-shi, Japan.
Email: yae.kurosawa@pc.hoya.co.jp
Received April 4th, 2012; revised May 5th, 2012; accepted June 7th, 2012
Keywords: Ceramic Hydroxyapatite; Virus Purification; Dengue Virus; Virus Particle; Virion
ABSTRACT
Dengue virus type 2 ThNH7/93 retained infectious activity after purification by ceramic hydroxyapatite chromatography. Dengue virus type 2 culture fluid was loaded onto the ceramic hydroxyapatite column and eluted with a linear gradient of sodium phosphate buffer. Culture fluid and protein contaminants derived from host cells were eluted initially, followed by elutions of dsDNA, and then dengue virus type 2. The recoveries of dengue virus type 2 were 64 ± 14% (n = 11) in the hemagglutination (HA) test and 60% (n = 2) determined by focus assay for viral infectivity. This protocol was highly reproducible, simple, rapid, and appears applicable to other virus species such as influenza virus, Japanese encephalitis virus and adenovirus.
1. Introduction
Purification of viruses is very important for vaccine development and gene therapy. The upstream process for the development of vaccines, has recently undergone dramatic development, but the downstream process has not changed significantly, despite its importance. Chromatography has been used for virus purification [1].
Previously, we reported the adsorption of Japanese encephalitis virus (JEV) particles on ceramic hydroxyapatite by use of scanning electron microscopy [2]. We further reported on the purification of JEV particles from mouse brain homogenate by use of ceramic hydroxyapatite chromatography [1,3]. Infectious activity in these virus particles was maintained. This report describes a reliable chromatographic purification of dengue virus type 2 from culture fluid; viral infectivity was retained.
2. Materials and Methods
2.1. Ceramic Hydroxyapatite and Column Preparation
CHT Ceramic Hydroxyapatite, Type II, 40 μm particle size was purchased from Bio-Rad Laboratories, Inc. (Hercules, CA, USA). This material is a standardized ceramic type, which is sintered material; use of in-house hydroxyapatite could lead to varying results. Ceramic hydroxyapatite (0.358 g) was packed into an empty stainless steel column (i.d. 4.6 mm × 35 mm; Sugiyama Shoji Co., Ltd., Kanagawa, Japan) by the dry method.
2.2. Culture of Dengue Virus
Dengue virus type 2 ThNH7/93 strain [4] and C6/36 cells [5] were received from Professor K. Morita of Department of Virology, Institute of Tropical Medicine, Nagasaki University, Japan. Flasks (225 cm2, Sumitomo Bakelite Co., Ltd., Tokyo, Japan) were precoated with poly-L-lysine (Sigma-Aldrich Co., St. Louis, MO, USA) at 100 g/mL in phosphate buffered saline (PBS), stored at room temperature and rinsed with PBS, before use. The C6/36 cells were cultured in Minimum Essential Medium Eagle (modified) (modified EMEM, MP Biomedicals, Irvine, CA, USA) containing 10% fetal bovine serum (FBS; Invitrogen Corporation, Carlsbad, CA, USA) in poly-L-lysine coated flasks at 28˚C for 1 week. After the cells reached confluence, dengue virus type 2 was inoculated onto the cell monolayer in 75 mL of modified EMEM (MP Biomedicals) containing 0.5% FBS and MEM Vitamin Solution (Invitrogen Corporation), and cultured at 28˚C. Medium was changed at Day 3 and culture fluid (about 75 mL) was collected at Day 7 (Scheme 1). Culture fluid was filtered through a 0.22 μm pore size filter to remove cells and large cell debris.
2.3. Standard Chromatographic Procedure
The chromatography was performed on a BioLogic DuoFlow system (Bio-Rad Laboratories, Inc.). The column

Scheme 1. Preparation of samples. Sample 1, culture fluid of dengue virus-infected C6/36 cells; Sample 2, culture fluid of C6/36 cells without virus infection; and Sample 3, virus culture medium.
was set on a BioLogic DuoFlow system and rinsed with 600 mM sodium phosphate buffer (NaPB) pH 7.2. The column was then equilibrated with 10 mM NaPB pH 7.2. Ten milliliters of culture fluid from dengue virus type 2 were loaded onto the column with 10 mM NaPB pH 7.2, and then eluted with a linear gradient up to 600 mM NaPB pH 7.2 for 15 min at a flow rate of 1.0 mL/min. Chromatography was monitored with UV absorbance at 280 and 260 nm, and conductivity. Eluate was collected by 2 mL fractions, for the first 20 min (10 fractions), and then by 1 mL fractions (the remaining 20 fractions). The collected fractions were used for the evaluation. As a control, the culture medium and the cell culture fluid without virus were also loaded onto the column. After the procedure, the column was washed with distilled water, sterilized with 0.5 M NaOH for 5 column volumes, and that was replaced with 10 mM NaPB. The system was further washed with distilled water.
2.4. Hemagglutination (HA) Test
Preserved goose blood was purchased from Nippon Biotest Laboratories Inc., Tokyo, Japan. Goose red blood cells (GRBC) were washed 3 times with saline. GRBC was suspended in saline at a final concentration of 8% as a stock suspension. This was further diluted 1:24 to prepare 0.33% GRBC working suspension in virus adjusting diluent (VAD) containing 150 mM NaCl and 200 mM sodium phosphate at pH 6.2. Each fraction was diluted 10-fold, and 50 μL of diluted sample was further diluted serially, 2 fold with 0.4% BSA in BS9 (120 mM NaCl, 50 mM boric acid and 24 mM sodium hydrate) in Ubottom 96-well plates (Becton, Dickinson and Company, Franklin Lakes, NJ, USA). And then, 50 μL of 0.33% GRBC suspension was added into each well, the plates were mixed gently, and then incubated at 37˚C for 30 min. The HA titer was the highest dilution of virus showing the agglutination pattern on the bottom of the well.
2.5. Estimating of Double-Stranded DNA (dsDNA)
The concentrations of dsDNA in fractions were determined using Quant-iT PicoGreen dsDNA Assay Kit (Invitrogen Corporation), according to the manufacturer's instructions. A standard curve was prepared with lambda DNA (kit component) at a concentration between 1.56 and 100 ng/mL. Total dsDNA quantities in fractions were corrected by determined concentrations and fraction volumes.
2.6. Focus Assay for Dengue Viral Infectivity
Focus assay for dengue virus infectivity was performed with a modification of the method described by Okuno et al. [6,7]. Virus titer was detected in a confluent monolayer of Vero cells(American Type Culture Collection, Manassas, VA, USA) in 96-well microplates. To prepare the confluent monolayer, 100 μL of Vero cells (2 × 105 cells/mL) were cultured in Eagle’s minimum essential medium (EMEM) (Sigma-aldrich Co.) containing 10% FBS at 37˚C in 5% CO2 for 1 day. Each fraction underwent 10-fold serial dilution with EMEM containing 10% FBS. After the culture fluid was removed from each well, 25 μL of diluted virus samples were inoculated and cultured for 1 hour by tilting gently every 15 min for the adsorption of viruses to cells. And 75 μL of EMEM with 10% FBS was then added to each well, and the plates were further incubated at 37˚C for 76 hours. The cells were washed with 100 μL of PBS once and fixed with 100 μL of ethanol (Wako Pure Chemical Industries, Ltd., Osaka, Japan) for 5 min at room temperature, twice. After the removal of ethanol, the plates were dried and stored at –30˚C until staining. The foci were visualized with peroxidase-anti-peroxidase (PAP) staining. The plates stored at –30˚C were reconstituted by adding 100 μL of PBS containing 0.1% polyoxyethylene(20) sorbitan monolaurate (commercially also known as tween 20; Wako Pure Chemical Industries, Ltd.). The cells were treated with 100 μL (30 min at 37˚C) of the supernatant of hybridoma JE114, which was established in-house and produced IgG1 to react to flaviviruses, as the primary antibody. After washing the plates with 100 μL of PBS, 100 μL of 1:500-diluted goat anti-mouse IgG (H + L) (Rockland Immunochemicals Inc., Gilbertsville, PA, USA) in PBS was added into each well and incubated for 30 min at 37˚C. In the same way, the plates were washed and incubated with 100 μL of 1:250-diluted mouse peroxidase-anti-peroxidase immune complex (Nordic Immunological Laboratories, AX Eindhoven, The Netherlands) in PBS. Finally, the peroxidase reaction was developed for 0.25 mg/mL of 3,3’-diaminobenzidine tetrahydrochloride (Sigma-aldrich Co.) and 0.01% hydrogen peroxide (Wako Pure Chemical Industries, Ltd.) in PBS at room temperature for 20 min. The plates were washed with tap water and the foci were counted.
2.7. Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis and Immunoblotting
Fractions were concentrated 20-fold by ultrafiltration with centrifugal filter devices (Microcon Ultracel YM-10, Millipore Corporation, Billerica, MA, USA). Concentrated fractions were analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) [8] and immunoblotting using 15% polyacrylamide gel (cPAGEL; ATTO Corporation, Tokyo, Japan). Each protein band was defined after silver staining in SDS-PAGE analysis. For immunoblotting, the gel was transferred to a nitrocellulose membrane (Hybond-ECL; GE Healthcare UK Ltd., Buckinghamshire, England) at 70 V (constant voltage) for 2 hours after electrophoresis. The membrane was blocked with 1% bovine serum albumin (BSA; Wako Pure Chemical Industries, Ltd.) in PBS at room temperature for 30 min. Next, it was incubated with 1:500 anti-dengue virus type 2 mouse antiserum in 1% BSA/PBS at room temperature for 30 min. After washing with PBS containing 0.1% tween 20, it was incubated with 1:1000 peroxidase-conjugated rabbit anti-mouse IgG + A + M (H + L) (Zymed Laboratories Inc., South San Francisco, CA, USA) in 1% BSA/PBS at room temperature for 30 min. The membrane was then washed and dipped into the substrate solution (Vector VIP Substrate Kit for Peroxidase; Vector laboratories, Inc., Burlingame, CA, USA) to visualize the specific bands.
3. Results
3.1. Chromatograms of Culture Fluid of Dengue Virus, Cell Culture Fluid without Virus and the Culture Medium
The chromatogram of the culture fluid of dengue virus type 2-infected C6/36 cells (Sample 1) from the ceramic hydroxyapatite column (Figure 1(a)) was compared with

Figure 1. Chromatograms of ceramic hydroxyapatite column applied with culture fluids and medium. (a) Culture fluid of dengue virus-infected C6/36 cells (Sample 1); (b) Culture fluid of C6/36 cells without virus infection (Sample 2); and (c) Culture medium containing 0.5% FBS without virus (Sample 3). Black line, UV absorbance at 260 nm; gray line, UV absorbance at 280 nm; and broken line, conductivity of elution buffer.
those of the culture fluid of C6/36 cells without virus infection (Sample 2; Figure 1(b)) and the culture medium itself (Sample 3; Figure 1(c)). After 30 min of retention time, the culture medium showed the same UV absorbance values at 280 and 260 nm (Figure 1(c)). Although the absorbance at 260 nm was a little greater than at 280 nm in the culture fluid of cells without virus (Figure 1(b)); furthermore, the chromatogram of virus culture fluid showed a small UV absorbance peak at 260 nm (Figure 1(a)). This suggests that the peak at 260 nm was caused by the virus culture.
3.2. Determination of Fractions of Hydroxyapatite Chromatography
3.2.1. Virus Activity and Protein Contaminants
To determine the dengue virus activity, a hemagglutination (HA) test and a focus assay for infectious activity were performed in fractions eluted from ceramic hydroxyapatite chromatography (Figure 2). The HA test can detect E protein on the envelope of dengue virus, regardless of an intact virion. The presence of infectious activity means that the virus maintains the ability to infect
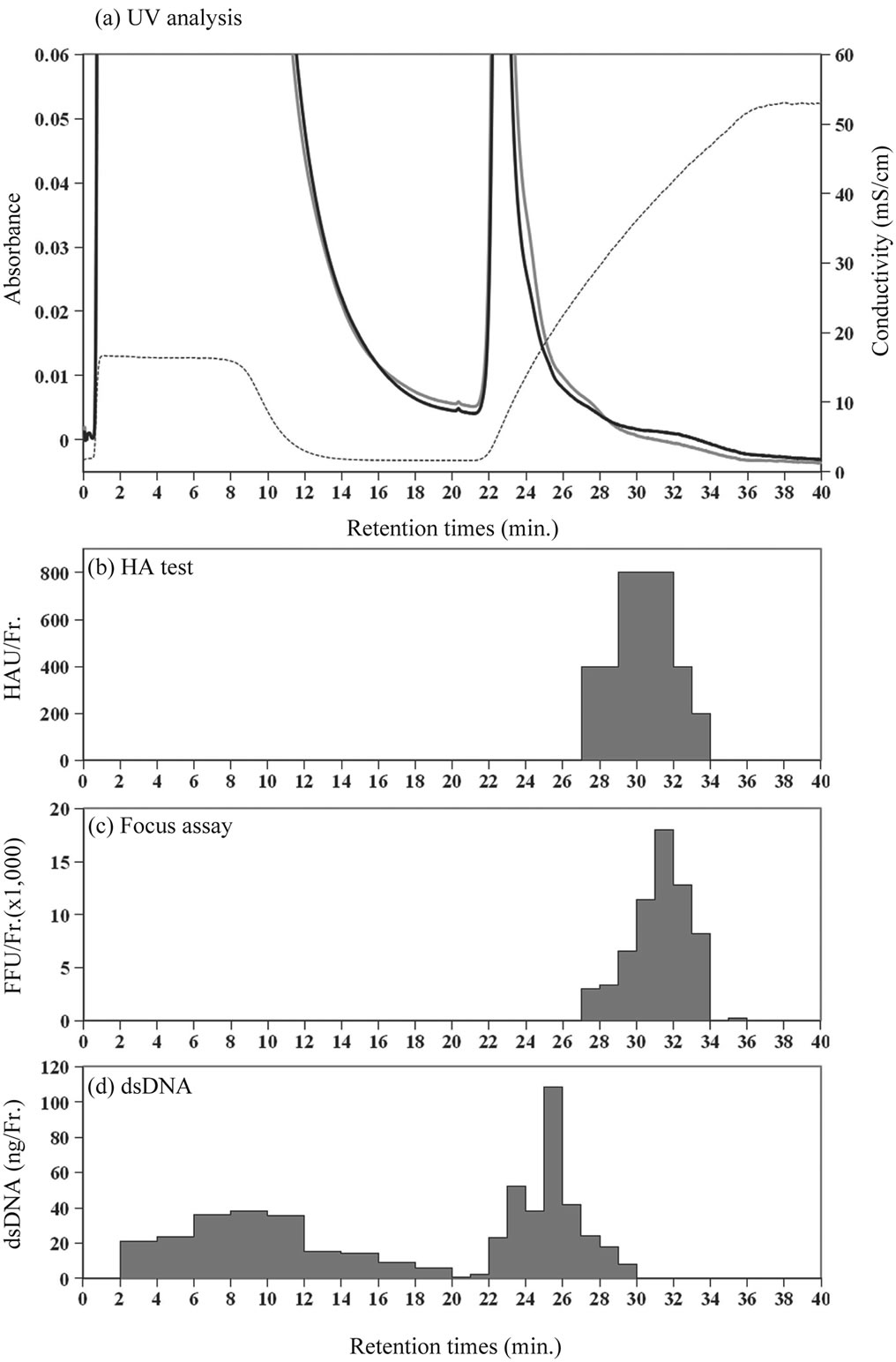
Figure 2. Eluate analysis in fractions from ceramic hydroxyapatite column. (a) UV analysis; bold black line, UV absorbance at 260 nm; gray line, absorbance at 280 nm; broken line, conductivity of elution buffer; (b) HA test; hemagglutination units in each fraction (HAU/Fr.); (c) Infectious activity in focus assay; focus forming units in each fraction (FFU/Fr.); and (d) Contents of dsDNA in each fraction.
to cells and should represent a whole particle. Neither of the virus activities was detected in flow-through fractions (before about 22 min of retention time). As shown in Figures 2(b) and (c), peaks for both virus activities were recorded at similar retention times, which were consistent with the small UV absorbance peak noted at 260 nm (Figure 2(a)); they were separated from the large UV absorbance peak recorded at about around 22 - 26 min of retention time, for contaminants. This result indicated that dengue virus type 2 was clearly separated from culture fluid in a one-step purification by ceramic hydroxyapatite chromatography.
Figure 3(a) shows that the viral active fractions (retention time 29 - 33 min) had fewer contaminants than did the fractions with a large UV peak (retention time 22- 27 min). Viral structure proteins were detected by immunoblotting in the viral active fractions (Figure 3(b)); immunologically, therefore, it confirmed that the viral active fractions contained dengue viruses.
3.2.2. Contamination of dsDNA
Contaminants included proteins and dsDNA from the culture medium and from host cells. The concentration of dsDNA derived from host cells in fractions was determined by use of a PicoGreen dsDNA Quantitation Kit. As showed in Figures 2(a) and (d), the dsDNA peak was eluted between the UV absorbance peaks for proteins and infectious activity. This suggests that the peak for dengue virus could be distinguished from those representing contaminants of culture fluid.

Figure 3. SDS-PAGE and immunoblotting analysis of fractions. Only fraction samples were concentrated 20-fold by ultrafiltration; Protein bands on gel were visualized by silver staining, and dengue virus-specific proteins were detected with anti-dengue virus type 2 mouse antiserum; (a) SDS-PAGE; and (b) Immunoblotting; D2 control, dengue virus type 2 purified by ultracentrifugation method; and CF, culture fluid (not concentrated).
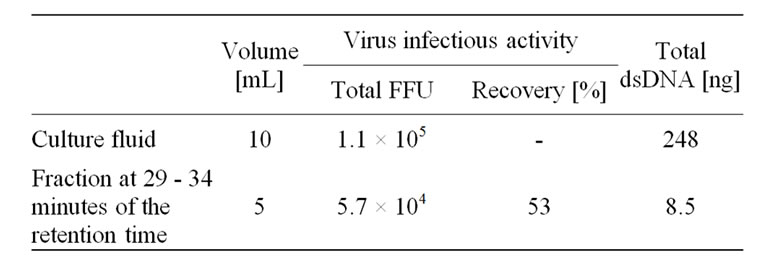
Table 1. Comparison of the infectious activity and dsDNA contaminants between culture fluid and purified fractions of dengue virus.

Table 2. Recovery of dengue virus from ceramic hydroxyapatite chromatography.
3.3. Recovery of Dengue Virus from Hydroxyapatite Chromatography
Dengue virus type 2 was eluted from ceramic hydroxyapatite chromatography, and the infectious activities of the chromatographic fractions were determined. Typical results are summarized in Table 1. The recovery at the virus peak (the retention time 29 - 34 min) was 53%. The initial amount of dsDNA in culture fluid was 248 ng, and the majority of dsDNA was decreased to 8.5 ng by hydroxyapatite chromatography purification.
This chromatographic result was highly reproducible. The retention times for virus peaks were always detected around 28 min to 35 min, in the repeated chromatographic elutions. The recovery in HA test was confirmed in 11 independent experiments (Table 2). The same level of recovery for infectious activity was detected. These results indicate that the ceramic hydroxyapatite chromatography described in this report is a reproducible method.
4. Discussion and Conclusions
Ceramic hydroxyapatite is a mild packing media for biological material and can be applied in the neutral region. The neutral condition leads to a good separation of biological materials, while maintaining biological activity. The results of this study confirm that dengue virus type 2 was purified by ceramic hydroxyapatite chromatography but its viral infectivity.
The present study showed that a one-step purification method for dengue virus type 2 by ceramic hydroxyapatite chromatography is both reproducible and robust. This method may be applicable to the other virus species, and it may be subsequently found at various sites in the future.
5. Acknowledgements
C6/36 cells and dengue virus type 2 were kindly supplied by Professor Koichi Morita of Department of Virology, Institute of Tropical Medicine, Nagasaki University, Japan. We thank Dr. Chang-Kweng Lim and Dr. Tomohiko Takasaki (Department of Virology I, National Institute of Infectious Diseases, Japan) for the technical transfer of the focus assay.
REFERENCES
- Y. Kurosawa, A. Yamamoto, I. Kurane and M. Nakayama, “Development of a Purification Method for Japanese Encephalitis Virus Particles Using Ceramic Hydroxyapatite Chromatography,” Medicine and Biology, Vol. 156, No. 6, 2012, pp. 410-416.
- Y. Kurosawa, I. Kurane and A. Yamamoto, “Observation of Japanese Encephalitis Virus Particles on Ceramic Hydroxyapatite by Scanning Electron Microscopy,” Medicine and Biology, Vol. 153, No. 12, 2009, pp. 607-610.
- Y. Kurosawa, A. Yamamoto and T. Ogawa, “Purification of Japanese Encephalitis Virus Particle by Ceramic Hydroxyapatite Chromatography,” 1st International Conference on Hydroxyapatite & Related Products, March 1999, Lyon, p. 8.
- K. Z. Thant, K. Morita and A. Igarashi, “Sequence of E/NS1 Gene Junction from Four Dengue 2 Viruses of Northeastern Thailand and Their Evolutionary Relationships with Other Dengue-2 Viruses,” Microbiology and Immunology, Vol. 39, No. 8, 1995, pp. 581-590.
- A. Igarashi, “Isolation of a Singh’s Aedes albopictus Cell Clone Sensitive to Dengue and Chikungunya Viruses,” Journal of General Virology, Vol. 40, No. 3, 1978, pp. 531-544. doi:10.1099/0022-1317-40-3-531
- Y. Okuno, A. Igarashi and K. Fukai, “Neutralization Tests for Dengue and Japanese Encephalitis Viruses by the Focus Reduction Method Using Peroxidase-AntiPeroxidase Staining,” Biken Journal, Vol. 21, No. 4, 1978, pp. 137-147.
- C. Lim, T. Takasaki, A. Kotaki and I. Kurane, “Vero Cell-Derived Inactivated West Nile (WN) Vaccine Induces Protective Immunity against Lethal WN Virus Infection in Mice and Shows a Facilitated Neutralizing Antibody Response in Mice Previously Immunized with Japanese Encephalitis Vaccine,” Virology, Vol. 374, No. 1, 2008, pp. 60-70. doi:10.1016/j.virol.2007.12.021
- U. K. Raemmli, “Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4,” Nature, Vol. 227, No. 5259, 1970, pp. 680-685. doi:10.1038/227680a0

