Journal of Behavioral and Brain Science
Vol.06 No.01(2016), Article ID:62977,11 pages
10.4236/jbbs.2016.61006
Behavioural and Neurophysiological Effects of a Stroke Rehabilitation Program on Emotional Processing in Tuberothalamic Infarct―Case Study
Katarzyna Kucharska1*, Ewelina Wilkos1, Roman Stefanski1, Grzegorz Makowicz1, Danuta Ryglewicz1, Ksenia Slawinska1, Ewa Piatkowska-Janko2
1Institute of Psychiatry and Neurology, Warsaw, Poland
2Institute of Radioelectronics and Multimedia Technlogy, Warsaw University of Technology, Warsaw, Poland

Copyright © 2016 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 24 November 2015; accepted 22 January 2016; published 25 January 2016
ABSTRACT
The aim of the study was to assess the neurophysiological and behavioural effects of a stroke rehabilitation involving Treatment As Usual (TAU) combined with Social Cognitive Training (SCT) in a right-handed patient suffered from tuberothalamic infarct compared to healthy controls (HCs) (n = 13). Methods: Both HCs and the patient were assessed by means of the following measures: Penn Emotion Perception Battery (ER40, EmoDiff40, PEAT40, PFMT),
Keywords:
Emotional Deficits, Thalamic Stroke, Social Cognitive Training, fMRI Changes

1. Introduction
The role of the thalamus in emotional processing has not received as much attention as other brain regions.
The thalamus is part of the limbic system, the region of the brain largely associated with the emotions, and is well recognised as the final relay station for perceptual data before it is passed on to the cerebral cortex. It receives input from diverse brain areas, primarily including all the senses except olfaction.
While a number of studies have examined emotion recognition [1] in damage of subcortical structures, mostly amygdala [2] [3] and basal ganglia [4] [5] ; few have considered the role of thalamus in emotional processing. In thalamic lesion, literature relatively little is known about the global aspects of emotional processing and the association between emotional deficits and treatment provided.
The involvement of the thalamus in emotional processing has long been postulated due to its connections to Papez’s circuit and through its close relationship with the prefrontal cortex through its integration in cortico-striatal-thalamo-cortical loops. This has recently been corroborated by Metzger and colleagues [6] [7] , who have used innovative high resolution fMRI on Tesla 7 in an attempt to improve the structural and functional understanding of thalamic circuitry. Despite the manifest thalamic involvement in emotional processing, there are relatively fewer researches studying the impact of thalamic lesions on emotional function. There are however a handful of human papers that document emotional deficits in subjects with thalamic lesions [1] [8] [9] .
Cheung and colleagues [8] compare the abilities of 19 healthy controls and 38 patients with subcortical strokes (6 of which had thalamic lesions) to recognise facial expressions using Ekman’s six basic emotion categories. The clinical participants perform significantly worse than the control group and those with thalamic lesions performed especially poorly at recognising sadness. As emotional recognition of facial expression is crucial in social functioning, it is evident that those with thalamic lesions may experience important social and emotional disability. This is consistent with the case report of a 63 year old male with a right thalamic infarct described by Vuilleumier et al. [1] 18 months after his stroke, this male is found to have a prosopoaffective agnosia, which is an impairment of the recognition of emotional facial expressions with sparing of the ability to discriminate facial identity. The role of the thalamus in regulating mood has also been recognised. Price and Drevets [10] implicate dysfunction of the limbic-cortical-striato-pallido-thalamic circuits as being involved in both bipolar affective disorder and clinical depression. This deficit matches with Veuilleumier’s patient, who has persistent hypomania after his thalamic infarct.
In a different case report, Clarke and colleagues [9] describe the cognitive and emotional functioning of a 54-year-old lady with an isolated left polar thalamic infarct. The patient develops acute global amnesia and slight frontal type dysfunction. Also noteworthy is the patient’s fluctuating mood and decreased motivational level. Furthermore, the patient struggles to understand the emotional content of her social interactions.
The rationale of this paper is to assess the neurophysiological and behavioural effects of comprehensive stroke rehabilitation involving Treatment As Usual (TAU) combined with Social Cognitive Training (SCT) in a thalamic-damaged patient compared to healthy controls.
The aim of recruiting of the healthy control group is to assess neural circuitry in healthy brains to find out more about thalamic involvement in emotional processes in comparison to a patient in acute thalamic stroke who undergo the same scanning procedure before implementing social cognitive training.
1.1. Subjects
A 52-year-old right handed male with a history of hypertension and obesity developed temporal disorientation and significant word finding difficulty with secondary comprehension and verbal memory problems [11] . On presentation he was alert, oriented in time, place and person. Neurologic evaluation revealed pronounced word finding difficulties, halting speech with frequent hesitations, and impaired word discrimination, auditory comprehension for complex commands and responsive naming. In striking contrast, repetition, reading and writing were intact. Digit recall, five-minute recall of three objects and long term memory was impaired. Cranial nerves, motor system, reflexes, cerebellar and sensory systems were normal. Basic biochemistry, full blood counter, liver and kidney function tests and thyroid function tests were normal.
Non-contrast computerized tomogram (CT) brain scan revealed a hypo-density in the left thalamus. Magnetic resonance imaging of brain (including sagittal and axial T1, T2, FLAIR, axial diffusion weighted images and coronal gradient echo images) showed infarction in the territory of the left tuberothalamic artery (19 × 10 mm in size) (Figure 1).
The patient was managed conservatively (enalapril was introduced at the dose of 5 mg once daily as a part of TAU), and his memory and speech problems gradually improved over the subsequent 9 days. The patient has never received psychiatric treatment and there were no mood or anxiety problems on initial assessment whilst he scored low on the following scales: STAI (state) 31, STAI (trait) 37, and Hamilton Depression Scale, HDS (3 points) [11] .
16 right-handed healthy controls initially participated after giving informed consent in time period 2012-2014. 13 out of 16 healthy volunteers (8 male, 5 female) took part in both behavioural assessment and brain scanning procedure. Approval for the study was obtained in 2012 from the Ethics Committee of the Institute of Psychiatry and Neurology in Warsaw, Poland. An inclusion criterion for all subjects was a minimum score of 23 on the Mini-Mental State Examination, MMSE [12] . People who were familiar with neuropsychological batteries and tests conducted in the study were also excluded to enhance sample cleanliness and the reliability of data analyses. Therefore the recruitment of healthy controls from medical staff or psychologists willing to volunteer was relatively limited from the Institute of Psychiatry and Neurology and the recruitment was mostly carried out from the general public. Exclusion criteria for all subjects included: habitual drug or alcohol abuse, secondary neurological disorders (e.g. epilepsy, dementia), learning disability, and other psychiatric diagnoses.
1.2. Methods
For research purposes, the researchers used standardized neuropsychological measurements that are delivered online via The University of Pennsylvania Computerized Neuropsychological Test Battery and The Social Emotional Cognition Test (online version) of CogState battery, and other individual tests delivered in paper form.
Components of social cognition were evaluated using the following tests:
1) Affect perception: a) Penn Emotion Recognition Test (ER40); b) Penn Emotion Discrimination Task (EmoDiff40); c) Penn Emotional Acuity Test 40 (PEAT40).
2) Facial memory: a) Penn Facial Memory Test (PFMT).
3) Theory of mind/empathy: a) Reading the Mind in the Eyes Test (revised version II); b) Toronto Alexithymia Scale (TAS-20).
In addition, the following clinical scales were used: the MMSE, Spielberger State-Trait Anxiety Inventory (STAI), and Hamilton Depression Scale (HDS) [11] .
Evaluative assessment was performed twice by a clinical psychologist (KS).
1.2.1. Social Cognitive Training (SCT)
Social Cognitive Training [13] constitutes an interdisciplinary program, which has been created on the basis of therapeutic interventions aimed at improving social functioning, mainly Social Cognition and Interaction Training,
Figure 1. T1-weighted MRI of a patient B.D with left tuberothalamic ischemic stroke.
(SCIT) by [14] . In its assumptions and methodology it draws from traditions of Cognitive Behaviour Therapy and incorporates elements of Rational Emotive Behaviour Therapy and Problem-Solving Therapy (PST). SCT consists of 12 therapeutic sessions delivered weekly for 60 minutes by a trained psychologist and psychotherapist (E.W.), blind to evaluative assessment, on ward or after discharge in out-patient clinic setting. SCT has been divided into five main stages of treatment: 1) introduction to SCT; 2) emotional education; 3) social cognition strategies training; 4) application of abilities in own life and; 5) summary of SCT.
The first five sessions have been developed to improve emotional functions and social perception. Other sessions are focused mainly on improvement of functions of the theory of mind and the attribution style. Throughout the whole training participants are encouraged to share personal and emotional difficulties or social ones; to analyze both their own behaviour and reactions of people presented in videos, pictures or text scripts. During the later sessions they learn helpful steps in solving problems in social interactions and communication with others.
1.2.2. Neuroimaging Methods
Structural and functional magnetic resonance imaging (fMRI) investigations in the control and the experimental group were performed on a 1.5 T clinical imaging system (Philips Achieva Nova Dual; whole-body system, Philips Medical Systems, Warsaw, Poland). The study utilized primary fluid-attenuated inversion recovery, diffusion weighted imaging, and T1 sequences for diagnosis of thalamic lesions and the exclusion of other pathologies of the brain. The scans were read by a radiology specialist with several years of experience. The criteria that were used to determine the presence of unilateral lesions were hyperintense changes in diffusion weighted imaging images and a corresponding decrease in the apparent diffusion coefficient maps, usually also with visible outbreak in fluid-attenuated inversion recovery in this area.
1.2.3. Image Acquisition
Structural 3D Sagittal MP-RAGE images were acquired with the following parameters: TE: 4 mms, TR: 8.67 ms, voxel 1 mm × 1 mm × 1 mm, image matrix: 240 × 240, 170 slices. Blood-oxygenation-level-dependent (BOLD) images were acquired with a reverse gradient-echo EPI sequence: 30 axial slices (4.5 mm thick, 0 mm gap; TR/TE = 3000/50 msec, FOV = 220 mm, matrix = 92 × 92), parallel to the AC-PC line and encompassing the entire cerebrum and the majority of the cerebellum.
1.3. fMRI Paradigm
Event-Related Fear Processing
An emotional face gender labelling event-related fMRI paradigm was used. It comprised an 8-minute event-re- lated neuroimaging task examining neural activity to neutral and 100% fearful facial expressions. All stimuli were gray-scale digitized photographs that were of fixed size (15 × 10.5 cm), cropped, and morphed using software to depict emotional expressions of 100% intensity of fear. Each stimulus was presented for 2 sec. with an inter-stimulus interval varied from 3 s to 8 s according to a Poisson distribution with average interval 6 s during which a fixation cross was displayed. In each experiment, participants viewed 20 faces of each intensities: neutral, and 100% intensities of fear (20 man/20 woman). Subjects were asked to respond with their index finger or thumb to indicate whether the actor in the picture was a man or a woman. They were also asked to respond as accurately as possible.
1.4. Data Preprocessing and Analysis
Data preprocessing was performed using SPM8 (http://www.fil.ion.ucl.ac.uk/spm). Functional individual data were realigned using the first slice as a reference, and unwrapped to correct for static inhomogeneity of the magnetic field and movement by inhomogeneity interactions. Functional volume was co-registered to the subject’s MPRAGE image. The subject’s MPRAGE image was segmented. Data were normalized to Montreal Neurological Institute standardized template and spatially smoothed with a Gaussian kernel of 8-mm full-width at half-maximum [11] . For each subject a general linear model (GLM) was constructed with emotion intensities (e.g., neutral, 100% fear) and fixation cross entered in the design matrix as separate regressors. Movement parameters from the realignment stage were entered as covariates of no interest to control for subject movement. The statistical parametric maps (SPM) were generated for neutral/fixation and 100% fear/fixation condition. The result is anatomical labelling using xjView toolbox (http://www.alivelearn.net/xjview) and visualized using Mricro tool [15] .
1.5. Statistical Analyses
Statistical Analyses were performed using SPM 8 [16] . Individual Brain Activation Map (IBAM) in first level analysis was calculated for each contrast neutral and 100% fearful facial expressions in comparison with fixation cross. One sample t-test for the HC group was calculated using IBAM. We presented our results for first level analysis for the patient, and results of group-level analysis for HC group. All results clusters were identified using False Discovery Rate (FDR) correction of p < 0.05.
2. Results
2.1. Emotional Processes
Patient B.D. underwent neuropsychological examination twice: three weeks after stroke onset and after 12 weeks of participation in SCT training.
In The Social Emotional Cognition Test of CogState battery, the patient’s results have improved slightly: 38 correct answers and 10 errors in the first test and 41 correct answers and 7 incorrect in the second (48 stimulation).
In The University of Pennsylvania Computerized Neuropsychological Test Battery by Penn the subject achieved improvement in all kinds of tests, both in face recognition (PFMT) and basic emotions. ER40 score showed an improvement in recognizing anger, in particular.
Patient’s responses were also more accurate on both PEAT40 (from very happy to very sad) as well as on EmoDiff40 (discrimination happy and sad). These differences may be related to an increase in the time needed to perform the tasks as well as some improvement in early visual processes. On TAS-20 scale, B.D. reached 42 points in the first measurement, and 22 points in the second one. Both results indicate no difficulty in identifying and describing his own emotions, lack of tendency to minimize the emotional experiences and focusing attention on the outside. The biggest differences concerned Externally-Oriented Thinking subscale (less 16 points). After SCT, the patient saw a greater need to focus on his own emotions and thoughts and analyze his own behavior.
The ability to recognize emotions in others, measured by the Reading the Mind in the Eyes Test, remained intact and comparable with the healthy controls (31 points―I exam; 30 points―II exam) which may suggest well- developed empathy and theory of mind (system of concepts, which allows conclusions about the state of mind others, e.g. correct interpretation intentions and desires).
Neuropsychological assessment in healthy controls was conducted once. Results data are presented in Table 1.
Table 1. Demographic and clinical data for the healthy controls (HC) and the patient (B.D) before and after therapy.
2.2. Emotional Processing as Compared with Baseline in B.D. before Therapy
2.2.1. Processing Neutral Faces
During the processing of neutral faces compared with baseline (12 foci), there was a significant activation in the following brain areas: temporal areas (left superior and middle temporal gyrus), visual areas (left calcarine, left lingual gyrus, left lateral occipitotemporal gyrus) and left pre- and postcentral gyri (cluster-level Familywise Error Rate FWE correction (p < 0.05); Table 2, Figure 2(a)).
Table 2. Brain activation during processing neutral and fearful faces in B.D before and after rehabilitation program (cluster-level FWE correction (p < 0.05)).
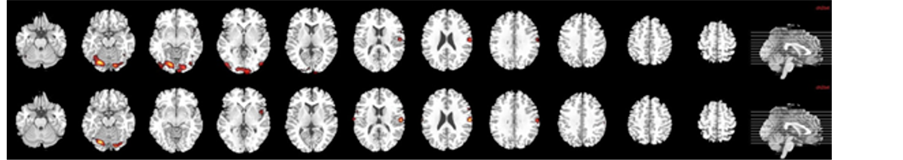
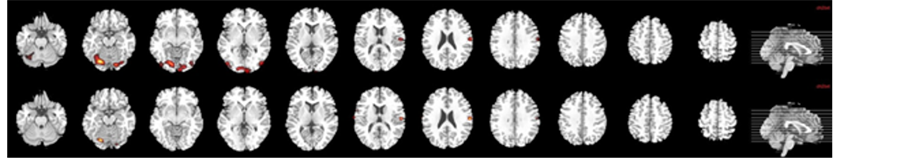
Figure 2. Brain map of neural activation in response to both neutral (a) and 100% fearful facial (b) expressions compared with fixation cross in B.D. in acute stroke before and after rehabilitation program.
2.2.2. Processing 100% Fearful Faces
The response to fearful stimuli recruited the following areas with significantly increased BOLD signal (9 foci): visual areas (left calcarine, left lingual gyrus, left fusiform gyrus), left pre and postcentral gyri and right cerebellum (cluster-level FWE correction (p < 0.05); Table 2, Figure 2(b)).
2.3. Emotional Processing as Compared with Baseline in B.D after Therapy
2.3.1. Processing Neutral Faces
The processing of neutral faces compared with baseline (8 foci) showed increased neural activation in the following regions: visual areas (left lingual gyrus, left fusiform gyrus), left precentral and right postcentral gyri and right cerebellum (cluster-level FWE correction (p < 0.05); Table 2, Figure 2(a)).
2.3.2. Processing Fearful Faces
After therapy of B.D., significantly increased brain activation was reported in the following brain areas (4 foci): bilateral postcentral gyrus, right cerebellum and left lingual gyrus (cluster-level FWE correction (p < 0.05); Table 2, Figure 2(b)).
2.4. Emotional Processing as Compared with Baseline in Healthy Controls
2.4.1. Processing Neutral Faces
The processing of neutral faces compared with baseline (21 foci) activated significantly: frontal areas (right medial frontal gyrus, right frontal inferior, bilateral precentral gyrus), temporal areas (right middle temporal and right temporal superior), parietal regions (right supramarginal gyrus, left superior parietal lobule, left postcentral gyrus), bilateral cerebellum, and subcortical area (left putamen and right globus pallidus) cerebellum (cluster p < 0.001, false discovery rate [FDR] p < 0.001; Table 3, Figure 3(a)).
2.4.2. Processing 100% Fearful Faces
The processing of fearful faces compared with baseline (24 foci) was associated with significant increase of BOLD signal in: the visual regions (left inferior occipital gyrus and left fusiform gyrus), temporal areas (right middle temporal and inferior gyri), parietal regions (left parietal superior, bilateral postcentral gyri, bilateral angular gyrus), and the bilateral cerebellum (cluster p < 0.001, false discovery rate [FDR] p < 0.001; Table 3, Figure 3(b)).
2.5. Neuroimaging Analyses
Hence, before therapy of B.D. recruitment was larger in the left hemisphere, both with neutral and fearful faces.


Figure 3. Brain maps of neural activation in response to both neutral (a) and 100% fearful facial expressions (b) compared with fixation cross in the healthy control group.
Table 3. Brain activation during processing neutral and fearful faces as compared to fixation in the healthy controls (cluster p < 0.001, false discovery rate (FDR) p < 0.001).
In response to neutral faces, visual cortex was significantly activated including calcarine and fusiform gyrus. Before treatment was provided, perception of 100% fearful expressions recruited cerebellum, precentral gyrus and visual regions but not amygdala.
B.D. showed fewer post-therapy activation maps compared with pretherapy ones and more significantly activated pre- and postcentral gyrus and right cerebellum in response to 100% fearful faces.
Interestingly, neither amygdala nor visual areas including fusiform gyrus and calcarine were activated as the response to fearful stimuli after therapy completed.
3. Discussion
This study examined the neural correlates of a promising rehabilitation program (TAU combined with SCT) in a patient with left thalamic lesion.
Compared to the examined group of healthy controls, the patient did present with mild deficits in emotional recognition, anger in particular, whilst recognition of other specific emotions including fear remained intact. Furthermore, B.D. scored lower on Penn Discrimination and Acuity Tests whilst his performance of Reading the Mind in the Eyes Test was comparable with the control group, suggesting his intact ability to empathize and mentalize.
The aim of the paper was to find out if the consequence of left tuberothalamic infarct was a deficit in emotion processing and to examine how the anatomical reorganisation post-stroke would affect emotion functioning in comparison to the healthy controls.
Up-to-date thalamus lesion literature did not show consistent results on thalamus involvement in affect recognition.
To the authors’ knowledge there were two studies published [8] [17] so far showing deficits in affect recognition on a relatively small sample size (respectively 6 and 8 patients).
Cheung et al. [8] reported patients’ difficulty in recognising sadness whilst Wilkos et al. [17] described the difficulty in performance of Emotion Discrimination Test and Reading the Mind in the Eyes Test in thalamic impaired patients. Other studies were case reports elaborating on clinical, psychological, and behavioural changes in patients with thalamic lesions.
A host of imaging studies had re-enforced the role of human fear conditioning [18] .
Before therapy of B.D. recruitment was larger in the left hemisphere, both with neutral and fearful faces. In response to neutral faces visual cortex was significantly activated, including calcarine and fusiform gyrus. Before treatment was provided, perception of 100% fearful expressions recruited cerebellum, precentral gyrus and visual regions but not amygdala. We found that processing of fearful faces in healthy controls was associated with increased activation in visual brain regions including fusiform gyrus, temporal and parietal areas and bilateral cerebellum; which again corresponded with robust neuroimaging data.
Functional neuroimaging literature highlighted the fusiform gyrus and inferior occipital gyrus as the key regions of the sensory cortex in response to visual emotion stimuli [19] .
B.D. showed a post-therapy contraction of activation pattern and more significantly activated pre-and postcentral gyrus and right cerebellum in response to 100% fearful faces. Interestingly, amygdala deactivation was found as the response to fearful stimuli before and after therapy was completed. This data did not correspond well with neuropsychological assessment, which stressed successful remediation of mildly impaired emotional functioning.
Animal and neuroimaging studies described that incoming sensory signals of fear travel from thalamus to amygdala via two neural pathways: a direct subcortical route and an indirect route via the sensory cortex [18] , [20] . It had been reported that amygdala received crude sensory input implicitly via a direct extrageniculostriate (superior colliculus and thalamic pulvinar) pathway [20] .
The tuberothalamic artery irrigated the ventral pole of the medial dorsal (MD) nucleus, mamillothalamic tract, ventral amygdalofugal pathway, ventral art of the internal medullary lamina and anterior thalamic nuclei [21] .
Hence, the lack of BOLD signal increase in amygdala of our patient with tuberothalamic infarct could be explained on the basis of disconnection involving the pathway between amygdala and anterior thalamic nuclei by damage to the amygdalothalamic projections passing through the internal medullary lamina.
Metzger and colleagues [6] substantiated the integration of the thalamic nuclei within cortico-striatal-thalamo- cortical pathways using high resolution functional MRI (hr-fMRI). They found that the MD nuclei was involved in the emotional processing of sexual stimuli and was coactivated with the pregenual anterior cingulated cortex (pgACC).
This supported the hypothesis of there being distinct segregated affective cortico-striato-thalamo-cortical “loops”.
To sum up, there is limited literature provided in this research domain to examine the specificity of the consequences of thalamic lesions depending on the vascular territory affected in the context of emotional processes. The majority of research so far was performed on a relatively small heterogeneous patient sample comprising a variety of either thalamic nuclei affected or damaged vascular territories within thalamus region.
Based upon literature review and our data, thalamus does not appear to play a key role in emotional process- ing as it was assumed.
To the authors’ knowledge this is the first study to administer the above-mentioned rehabilitation program in a case of unilateral thalamic stroke localized in left tuberothalamic territory.
Further research is needed to increase understanding about post-stroke anatomical reorganization after thalamic lesion in relation to emotion processing as well as the role of enhancing component of social cognitive training in efficacious thalamic rehabilitation as a whole.
Acknowledgements
This research project is supported by National Centre of Science (NCN) grant (Poland) 2011/01/B/NZ5/02838.
Many thanks to Dr Natasza Nalesnik from the Institute of Psychiatry in London for very helpful comments on the manuscript and Dr Simon Surguladze and Dave Gasston from the Institute of Psychiatry in London for their expert assistance in setting up fMRI experiments.
Disclosure
The authors declare that there is no conflict of interest regarding the publication of this paper.
Cite this paper
KatarzynaKucharska,EwelinaWilkos,RomanStefanski,GrzegorzMakowicz,DanutaRyglewicz,KseniaSlawinska,EwaPiatkowska-Janko, (2016) Behavioural and Neurophysiological Effects of a Stroke Rehabilitation Program on Emotional Processing in Tuberothalamic Infarct—Case Study. Journal of Behavioral and Brain Science,06,42-52. doi: 10.4236/jbbs.2016.61006
References
- 1. Vuilleumier, P., Armony, J., Clarke, K., Husain, M., Driver, J. and Dolan, R. (2002) Neural Response to Emotional Faces with and without Awareness: Event-Related fMRI in a Parietal Patient with Visual Extinction and Spatial Neglect. Neuropsychologia, 40, 2156-2166.
http://dx.doi.org/10.1016/S0028-3932(02)00045-3 - 2. Adolphs, R. and Tranel, D. (2004) Impaired Judgments of Sadness But Not Happiness Following Bilateral Amygdala Damage. Journal of Cognitive Neuroscience, 16, 453-462.
http://dx.doi.org/10.1162/089892904322926782 - 3. Adolphs, R., Tranel, D., Hamann, S., Young, A., Calder, A., Phelps, E., Anderson, A., Lee, G. and Damasio, A. (1999) Recognition of Facial Emotion in nine Individuals with Bilateral Amygdala Damage. Neuropsychologia, 37, 1111-1117.
http://dx.doi.org/10.1016/S0028-3932(99)00039-1 - 4. Phillips, M.L., Young, A.W., Senior, C., Brammer, M., Andrew, C., Calder, A.J., Bullmore, E.T., Perrett, D.I., Rowland, D., Williams, S.C., Gray, J.A. and David, A.S. (1997) A Specific Neural Substrate for Perceiving Facial Expressions of Disgust. Nature, 389, 495-498.
http://dx.doi.org/10.1038/39051 - 5. Sprengelmeyer, R., Rausch, M., Eysel, U.T. and Przuntek, H. (1998) Neural Structures Associated with Recognition of Facial Expressions of Basic Emotions. Proceedings of the Royal Society of London Series B: Biological Sciences, 265, 1927-1931.
http://dx.doi.org/10.1098/rspb.1998.0522 - 6. Metzger, C.D., Eckert, U., Steiner, J., Sartorius, A., Buchmann, J.E., Stadler, J., Tempelmann, C., Speck, O., Bogerts, B., Abler, B. and Walter, M. (2010) High Field FMRI Reveals Thalamocortical Integration of Segregated Cognitive and Emotional Processing in Mediodorsal and Intralaminar Thalamic Nuclei. Frontiers in Neuroanatomy, 4, 138.
http://dx.doi.org/10.3389/fnana.2010.00138 - 7. Metzger, C.D., van der Werf, Y.D. and Walter, M. (2013) Functional Mapping of Thalamic Nuclei and Their Integration into Cortico-Striatal-Thalamo-Cortical Loops via Ultra-High Resolution Imaging—From Animal Anatomy to in Vivo Imaging in Humans. Frontiers in Neuroanatomy, 7, 24.
http://dx.doi.org/10.3389/fnins.2013.00024 - 8. Cheung, C.C.Y., Lee, T.M.C., Yip, J.T.H., King, K.E. and Li, L.S.W. (2006) The Differential Effects of Thalamus and Basal Ganglia on Facial Emotion Recognition. Brain and Cognition, 61, 262-268.
http://dx.doi.org/10.1016/j.bandc.2006.01.008 - 9. Clarke, S., Assal, G., Bogousslavsky, J., Regli, F., Townsend, D.W., Leenders, K.L. and Blecic, S. (1994) Pure Amnesia after Unilateral Left Polar Thalamic Infarct: Topographic and Sequential Neuropsychological and Metabolic (PET) Correlations. Journal of Neurology, Neurosurgery & Psychiatry, 57, 27-34.
http://dx.doi.org/10.1136/jnnp.57.1.27 - 10. Price, J.L. and Drevets, W.C. (2012) Neural Circuits Underlying the Pathophysiology of Mood Disorders. Trends in Cognitive Sciences, 16, 61-71.
http://dx.doi.org/10.1016/j.tics.2011.12.011 - 11. Kucharska, K., Wilkos, E.., Stefanski, R., Makowicz, G., Ryglewicz, D., Slawinska, K. and Piatkowska-Janko, E. (2015) Psychological and Physiological Changes in Cognitive Functioning in Thalamic Lesion throughout the Rehabilitation Process. Journal of Neurology & Neurophysiology, 6, 1-7.
http://dx.doi.org/10.4172/2155-9562.1000318 - 12. Folstein, M.F., Folstein. S.E. and McHugh, P.R. (1975) “Mini-Mental State”. A Practical Method for Grading the Cognitive State of Patients for the Clinician. Journal of Psychiatric Research, 12, 189-198.
http://dx.doi.org/10.1016/0022-3956(75)90026-6 - 13. Kucharska, K., Wilkos, E. and Sawicka, M. (2013) Trening spolecznego poznania i neuropoznania: Podrecznik terapeuty. Instytut Psychiatrii i Neurologii, Warszawa.
- 14. Roberts, D.L., Penn, D.L., Labate, D., Margolis, S.A. and Sterne, A. (2010) Transportability and Feasibility of Social Cognition and Interaction Training (SCIT) in Community Settings. Behavioural and Cognitive Psychotherapy, 38, 35.
http://dx.doi.org/10.1017/S1352465809990464 - 15. Rorden, C. and Brett, M. (2000) Stereotaxic Display of Brain Lesions. Behavioural Neurology, 12, 191-200.
http://dx.doi.org/10.1155/2000/421719 - 16. Friston, K., Ashburner, J., Kiebel, S., Nichols, T. and Penny, W. (2007) Statistical Parametric Mapping: The Analysis of Funtional Brain Images. Elsevier/Academic Press, Amsterdam, Boston.
- 17. Wilkos, E., Brown, T.J., Slawinska, K. and Kucharska, K. (2015) Social Cognitive and Neurocognitive Deficits in Inpatients with Unilateral Thalamic Lesions-Pilot Study. Journal of Neuropsychiatric Disease and Treatment, 11, 1031-1038.
- 18. Le Doux, J. (2003) The Emotional Brain, Fear, and the Amygdala. Cellular and Molecular Neurobiology, 23, 727-738.
http://dx.doi.org/10.1023/A:1025048802629 - 19. Adolphs, R. (2002) Recognizing Emotion from Facial Expressions: Psychological and Neurological Mechanisms. Behavioral and Cognitive Neuroscience Reviews, 1, 21-62.
http://dx.doi.org/10.1177/1534582302001001003 - 20. Das, P., Kemp, A.H., Liddell. B.J., Brown, K.J., Olivieri, G., Peduto, A., Gordon, E. and Williams, L.M. (2005) Pathways for Fear Perception: Modulation of Amygdala Activity by Thalamo-Cortical Systems. NeuroImage, 26, 141–148.
http://dx.doi.org/10.1016/j.neuroimage.2005.01.049 - 21. Schmahmann, J.D. (2003) Vascular Syndromes of the Thalamus. Stroke, 34, 2264-2278.
http://dx.doi.org/10.1161/01.STR.0000087786.38997.9E
NOTES
*Corresponding author.




