Journal of Agricultural Chemistry and Environment
Vol.1 No.1(2012), Article ID:24942,7 pages DOI:10.4236/jacen.2012.11006
Production of reactive oxygen species by freezing stress and the protective roles of antioxidant enzymes in plants@NolistTemp#
Email: dzs@wsu.edu
1School of Biotechnology, Yeungnam University, Gyeongsan, South Korea;
2USDA/ARS, Department of Crop and Soil Sciences, Washington State University, Pullman, USA
Received 28 September 2012; revised 29 October 2012; accepted 9 November 2012
ABSTRACT
As one of the most severe environmental stresses, freezing stress can determine native flora in nature and severely reduce crop production. Many mechanisms have been proposed to explain the damage induced by freezing-thawing cycle, and oxidative stress caused by uncontrollable production of harmful reactive oxygen species (ROS) are partially contributed to causing the injury. Plants in temperate regions have evolved a unique but effective metabolism of protecting themselves called cold acclimation. Cold-acclimating plants undergo a complex but orchestrated metabolic process to increase cold hardness triggered by exposure to low temperature and shortened photoperiod and achieve the maximum freezing tolerance by a concerted regulation and expression of a number of cold responsive genes. A complicated enzymatic system have been evolved in plants to scavenge the ROS to protect themselves from oxidative stress, therefore, coldacclimating plants are expected to increase the de novo synthesis of the genes of antioxidant genes. Indeed, many antioxidant genes increase the expression levels in response to low temperature. Furthermore, the higher expression of many antioxidant enzymes are positively correlated to inducing higher tolerance levels against freezing. All the information summarized here can be applied for developing crop and horticultural plants to have more freezing tolerance for higher production with better quality. There have been extensive studies on the activities of antioxidant enzymes and the gene regulation, however, more researches will be required in near future to elucidate the most effective antioxidant enzymes to induce highest freezing tolerance in a crop plant in a transformation process or a breeding program.
Keywords: Antioxidant enzymes; Cold acclimation; Freezing stress; Manganese superoxide dismutase; Reactive oxygen species
1. FREEZING STRESS AND COLD ACCLIMATION
Environmental stresses can significantly reduce crop productivity all over the world [1]. Recently these stresses are getting serious due to the effect of global warming and intensive agricultural practices. Most severe stresses can include drought, salt, light and temperature stresses. Temperature stresses hold a high and a low temperature stress above or below adequate growth temperature. Especially low temperature can be divided into chilling stress and freezing stress. Freezing stress or sometimes called cold stress means a severe freezing, untimely frosts, or unseasonably cold weather can contribute to the injury [2-4]. The stress can damage crop production capacity as shown in the Pacific Northwest experienced a 70% yield loss due to winter-kill of wheat in 1991 [5]. Freezing stress also determines native flora in temperate regions and polar areas [2,6]. Damages due to freezing temperatures are common in crop and horticultural industries, therefore, producers throughout all over the world have suffered significant economic losses.
Freezing of plants at subfreezing temperature has two different manners of ice formation depending on the speed of cooling and the speed of withdrawal of water molecules from plant cells [3,7]. In extracellular freezing, ice is first nucleated outside the cell walls and ice crystals are initiated upon cooling of plant cells to subzero temperatures. Extracellular freezing is characterized by cellular dehydration, growing of ice crystals on the external cell surface and contracting cellular volume as water diffuses out of the cells. With intracellular freezing, crystallization of the water inside the cell may occur by internal nucleation or by penetration into the cell by an external ice crystal [7].
There are great variations in freezing tolerance, a multigenic trait, in different species in plants, at different growth stages, or even within population [2,4,8]. Most tropical plant species cannot tolerate even slight frost while twigs of mulberry were not injured even when cooled from 0˚C to –196˚C at a cooling rate of 0.5 K/min [2]. Plants can be classified into three different groups according to their freezing temperature tolerance [9]. The first group consists of frost tender plants, which have little or no freezing resistance. These plants are chilling sensitive and cannot become cold-acclimated. The second group consists of plants with some frost resistance, which can tolerate various amount of exposure to subfreezing temperatures. Plants in this group have a broad range of freezing tolerance from slightly below freezing in broad-leafed summer annuals to –30˚C in perennial grasses. The freezing tolerance is determined by the stage of cold acclimation, the rate and degree of temperature decline, and the genotypes. The third group consists of cold hardy plants that are mostly temperate woody species. These plants are very cold hardy after experiencing cold-acclimation.
Although most temperate plants have the ability to develop cold tolerance in response to acclimating conditions, the level of cold tolerance and the rate at which it is acquired by plants vary a great deal [10]. Plants undergo cold acclimation in response to acclimating conditions, which typically include temperatures slightly above 0˚C and short photoperiods [11]. While most plants typically need several weeks of acclimating conditions (low temperature) to induce the maximum level of cold tolerance, some plants such as Arabidopsis are rather unique in that they respond to acclimating conditions rapidly, and often maximum cold tolerance can be induced within 3 - 4 days of cold acclimation [12]. Cold acclimation is a complex process and appears to involve a concerted regulation and expression of a number of cold inducible genes [2,13-15]. A number of cold induced proteins may play a key role in inducing cold tolerance in plants [12, 14,16], and indeed, many of these cold responsive genes have been identified, cloned, and characterized in a number of plants [8].
Although no generally effective mechanisms can be defined to explain cell death and survival caused by a freeze-thaw cycle [8], maintenance of the plasma membrane integrity has been known to be the most critical cellular response during a freeze-thaw cycle [2,7,8]. During a freeze-thaw cycle, the plasma membrane experiences complex types of stresses, any of which may be responsible for the membrane damage. These factors may include a physical effect of the low temperature per se, freeze-induced reduction in the surface area and solute concentration effects, dehydration of the protoplasm, molecular packing of membrane constituents induced by cell shrinkage, changes in pH and ionic strength within and outside of the cell, and oxidative stress caused by reactive oxygen species (ROS) [2,17]. Of these factors, severe cellular dehydration occurring upon ice formation is considered as the most damaging part of freezing injury [8].
2. ROS INDUCED BY FREEZING STRESS
Because of their highly reactive nature, hydrogen peroxide (H2O2), superoxide anion (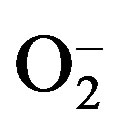 ), and hydroxyl radical (HO·) are called ROS [17]. ROS are generated on a regular basis in biological pathways as by-products or signal transducers [17,18], however, an excess of ROS can cause a damaging condition known as oxidative stress. This condition may be induced by a wide variety of environmental stresses and artificial harsh conditions [19- 21]. Oxidative stress can result in severe injury or plant death, if ROS accumulate to high levels due to excessive production or inefficient scavenging by antioxidant systems [19]. The cellular components susceptible to damage by ROS are lipids (peroxidation of unsaturated fatty acids in membranes), proteins (denaturation of enzymes), carbohydrates (breaking up of polysaccharides), and nucleic acids (“nicking”, cross-linkage and scission of DNA strands) [22,23].
), and hydroxyl radical (HO·) are called ROS [17]. ROS are generated on a regular basis in biological pathways as by-products or signal transducers [17,18], however, an excess of ROS can cause a damaging condition known as oxidative stress. This condition may be induced by a wide variety of environmental stresses and artificial harsh conditions [19- 21]. Oxidative stress can result in severe injury or plant death, if ROS accumulate to high levels due to excessive production or inefficient scavenging by antioxidant systems [19]. The cellular components susceptible to damage by ROS are lipids (peroxidation of unsaturated fatty acids in membranes), proteins (denaturation of enzymes), carbohydrates (breaking up of polysaccharides), and nucleic acids (“nicking”, cross-linkage and scission of DNA strands) [22,23].
Oxidative damage by ROS has been proposed to explain freezing injury [17,24-26]. The freezing process has been shown to generate free radicals in bacterial cells, which is dependent on the concentration of oxygen present [17]. An oxidative burst of superoxide radicals formed from oxygen and electrons leaking from the mitochondrial electron transport chain caused major injury to yeast cells during an aerobic freeze-thaw cycle [26]. Although superoxide anion or H2O2 has not been detected directly following freezing in plants, chemiluminiscence observed during freezing and thawing has been considered as a result of the formation of free radicals [24]. The amount of chemiluminescence in cryopreserved in vitro cultures of the shoot-tips of Brassica napus and cell suspension of Daucus carota was correlated with the degree of freezing injury, and the observed chemiluminiscence was considered to be the result from the production of free radicals, especially singlet oxygen [24]. Similarities between freezing injury and oxidative stress were reported in isolated alfalfa membranes exposed to the oxidative stress, strongly suggesting the involvement of ROS in freezing injury [27].
3. DIFFERENTIAL EXPRESSION OF ANTIOXIDANT ENZYMES DURING COLD ACCLIMATION
Plants have evolved very efficient but complicated antioxidant systems to scavenge ROS to protect themselves from oxidative stress [17,21,28]. The antioxidant systems can be divided into three groups: 1) antioxidant enzymes; 2) lipid soluble, membrane associated antioxidants, e.g. α-tocopherol, β-carotene, and ubiquinone); and 3) water soluble antioxidants (e.g., glutathione and ascorbate) [17, 22]. Among these systems, antioxidant enzymes are the most active and efficient protective mechanism [17,23]. Several kinds of antioxidant enzymes and frequently, several isozymes of a given enzyme, are known in plants [17,23]. The most active and important antioxidant enzymes are catalase (CAT), superoxide dismutases (SOD), peroxidases, and enzymes in the ascorbate-glutathione cycle (Figure 1). CAT (EC 1.11.1.6) degrades H2O2 into H2O and O2. SOD (EC. 1.15.1.1) catalyzes the dismutation of two superoxide (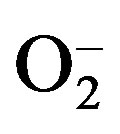 ) anions and water into H2O2 and O2. Three SOD proteins distinguished by their covalently linked catalytic metal ions and cellular locations have been identified in plants; manganese SOD (MnSOD), iron SOD (FeSOD), and copper-zinc SOD (Cu, ZnSOD). The different SOD isozymes in plants are actively transported to specific cellular locations. SOD isozymes probably arose due to the need to deactivate superoxide anions produced in those locations, because
) anions and water into H2O2 and O2. Three SOD proteins distinguished by their covalently linked catalytic metal ions and cellular locations have been identified in plants; manganese SOD (MnSOD), iron SOD (FeSOD), and copper-zinc SOD (Cu, ZnSOD). The different SOD isozymes in plants are actively transported to specific cellular locations. SOD isozymes probably arose due to the need to deactivate superoxide anions produced in those locations, because  is very reactive and cannot readily cross biological membranes [29]. MnSOD with Mn (III) at the active site, is synthesized in the nucleus and is transported to mitochondria for the activity [18]. FeSOD has Fe (III) at the active site and is found in chloroplasts [18,29]. Cu, ZnSOD has Cu(II) plus Zn (II) at the active site, and is found in the cytoplasm and in chloroplasts [18,29]. While MnSOD and FeSOD are quite obviously related
is very reactive and cannot readily cross biological membranes [29]. MnSOD with Mn (III) at the active site, is synthesized in the nucleus and is transported to mitochondria for the activity [18]. FeSOD has Fe (III) at the active site and is found in chloroplasts [18,29]. Cu, ZnSOD has Cu(II) plus Zn (II) at the active site, and is found in the cytoplasm and in chloroplasts [18,29]. While MnSOD and FeSOD are quite obviously related
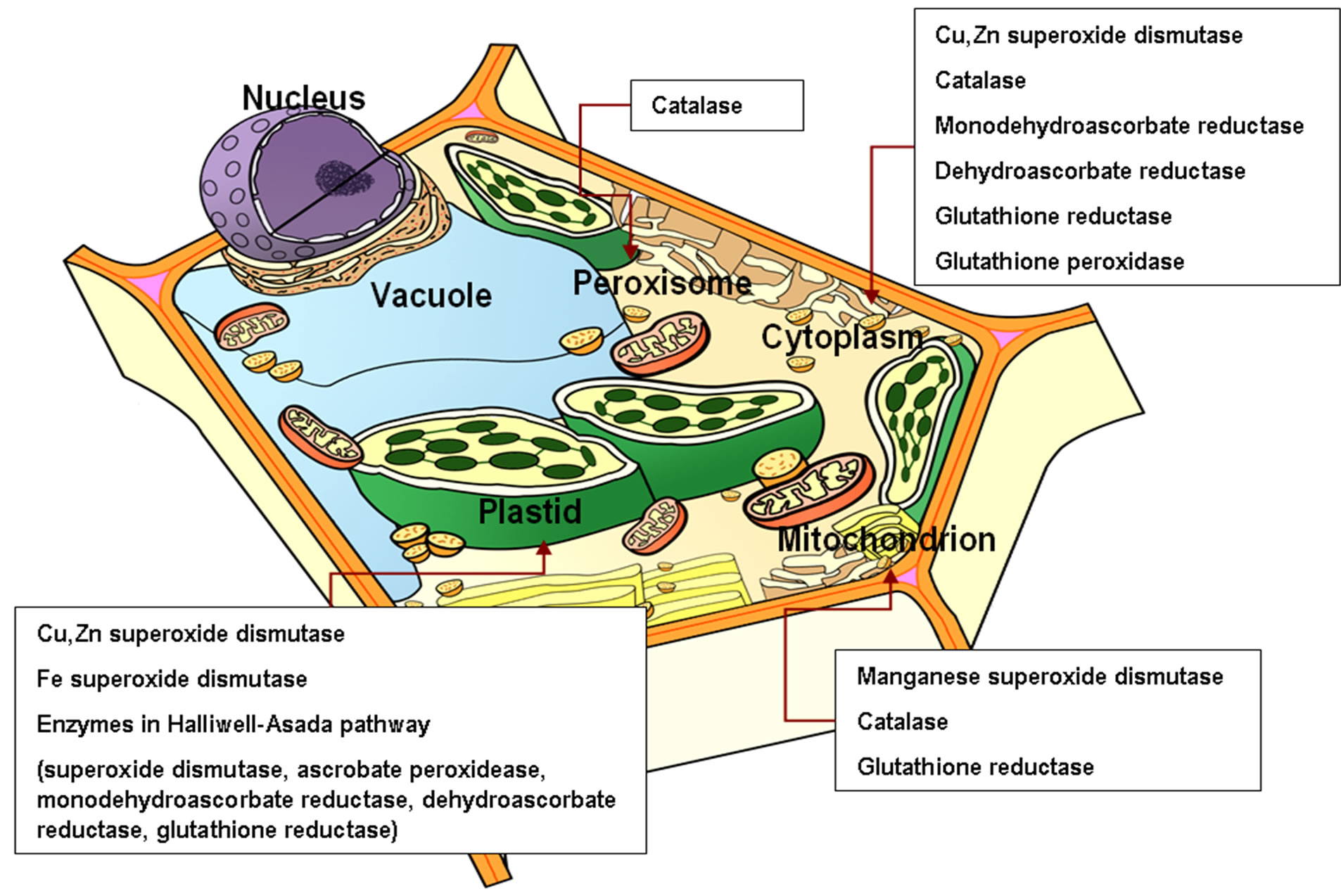
Figure 1. Subcellular locations of various antioxidant enzymes. Antioxidant enzymes are localized in the cytoplasm and many subcellular organelles to scavenge the evolved reactive oxygen species at the site efficiently.
by DNA sequence and catalyzing mechanisms [29], the MnSOD genes have an obvious eukaryotic history but the FeSOD genes are thought to have originated from the cyanobacteria-like symbiont that gave rise to the chloroplasts [30]. Cytosolic and chloroplast Cu, ZnSOD forms arose from a common gene, therefore, chloroplast Cu, nSOD is clearly the result of a duplication of the eukaryotic gene [30]. Peroxidases oxidize an organic substrate (R-H2) with H2O2 producing oxidized substrate and water. The ascorbate-glutathione cycle, also known as Halliwell-Asada pathway or water-water cycle, detoxifies ROS by the temporal and spatial regulation of the enzymatic activities of ascorbate peroxidase (APX), dehydroascorbate reductase (DHAR), glutathione reductase (GR), and monodehydroascrobate reductase (MDAR). APX detoxifies H2O2 into H2O by oxidation of ascorbate to monodehydroascorbate [31,32]. Monodehydroascorbate is regenerated by MDAR using NAD(P)H as reducing equivalents, and it can spontaneously be dismutated into dehydroascorbate. DHAR regenerates ascorbate driven by the oxidation of two gluthatione (GSH) into GSSG. Regeneration of GSH from GSSG can be mediated by GR using NAD(P)H as a reducing agent. This pathway occurs in chloroplasts and is involved in scavenging superoxide radicals and H2O2 along with the production of NAD(P)+ [31,32].
If these antioxidant systems are broken or weakened during freezing, the production of ROS could be evolved at the levels beyond control. To prepare the oxidative stress imposed by severe freezing, plants might develop antioxidant systems during cold acclimation for the protection. The oxidative burst characterized by the sudden increase of ROS by low temperature might be a signal of increasing the capacity of antioxidant systems. Relatively weak stimulation to plants by oxidative stress responded to low temperature might induce more active antioxidant systems against possible more severe oxidative stress imposed by freezing. ROS, especially H2O2, are proposed to perform multiple functions as a signal transducer in plant defense against both biotic and abiotic stresses including low temperature [33-36]. H2O2 is an ideal signal transducer because of its rapid response, high water-solubility, easy degradativity, transmembrane mobility, and the ability to be synthesized in or out of cells readily and to oxidize a target signal molecule [17]. Transgenic plants expressing high levels of H2O2 which were found to be resistant to pathogen attack [36]. Oxidative burst, the accumulation of active oxygen species including H2O2, is proposed as the first step in signal transduction in response to water stress [35]. H2O2 and menadione, a superoxide-generating compound induced chilling tolerance in 3 days-old chilling sensitive maize seedlings [37], and furthermore, acclimation, H2O2, and ABA protected mitochondria against chilling injury in maize seedlings [38]. Oxidative stress was induced in the callus tissue of Arabidopsis incubated at low temperature (4˚C) [34]. Therefore, cold-acclimating plants respond rapidly to low temperatures with oxidative burst which involves the accumulation of ROS including H2O2 within hours of exposure to low temperatures for signal transduction [34,37].
Up-regulated ROS scavenging systems is usually manifested as increased activities of antioxidant enzymes from de novo synthesis (Allen, 1995). To protect plants from injury due to the oxidative stress, plants have evolved very efficient and orchestrated antioxidant enzyme systems to scavenge the newly evolved ROS [17,21]. Exposure to environmental stresses can stimulate plants to enhance their ROS scavenging systems enhancing stress tolerance triggered by oxidative burst [21,39]. The increased activity of antioxidant enzymes may require de novo synthesis, initiated by an up-regulation of genes encoding antioxidant enzymes.
When the expression levels of total 9 antioxidant enzymes in wheat (Triticum aestivum) were measured during 4 weeks of cold acclimation, there were different responses among the genes [40]. Total 6 antioxidant enzymes increased the expression levels in response to low temperature. The genes of antioxidant enzymes were MnSOD, cytosolic MDAR, thylakoid-bound APX, chloroplastic DHAR, GR, and cytosolic GR. There were relatively constant expression levels shown in the genes of FeSOD and Cu, ZnSOD [40]. An interesting finding was that the expression levels of CAT gene decreased in response to low temperature, suggesting the roles of CAT on regulating the levels of H2O2 during cold acclimation [40]. Furthermore, during 4 weeks of cold acclimation, there were different patterns of increasing the transcripts of antioxidants. The genes of cytosolic MDAR, GPX, GR increased the transcripts by 2 weeks of cold acclimation, but the transcripts decreased after the time point, which facts might indicate the involvement of antioxidant enzymes in the metabolism of NAD(P)+ circulation. Near isogenic lines of winter wheat and spring wheat were used for the experiments, those lines only differ in the region of Vrn1-Fr1 on chromosome 5A, which regulates vernalization and freezing tolerance. These results suggest that the variation in a genomic region in wheat can affect both freezing tolerance and the expression levels of antioxidant enzymes.
Many plants have several copies of MnSOD genes [41-44], and the differential expression was reported in response to various stresses. Especially, the MnSOD transcripts increased significantly in both spring and winter wheat seedlings exposed to 2˚C, presumably leading to more MnSOD enzyme production in the mitochondria [40,43,44]. MnSOD genes in wheat are composed of a multigene family with slight differences in the nucleic acid and amino acid sequences [43,44]. Variants of MnSOD multigene family in wheat are expressed differentially at normal and low growth temperature [43,44], however, the differential expression was not related to the degree of stability of 3’ untranslated region [45], suggesting that there are other factors regulating the differential expression, e.g., promoter region variation.
4. PROTECTIVE ROLES OF ANTIOXIDANT ENZYMES AGAINST FREEZING STRESS
Antioxidant enzymes can alleviate freezing injury by scavenging effectively the overproduced ROS generated during freezing-thawing stress. There have been many reports increasing the stress tolerance by overexpressing various antioxidant enzymes [21]. In case of MnSOD, it is an important antioxidant enzyme protecting mitochondria from environmental stresses due to the first and the most efficient line of defense against ROS in mitochondria [18]. The importance of SOD has been well documented in the SOD-deficient mutants in both prokaryotes [46] and eukaryotes [47]; the mutants were hypersensitive to oxygen resulting in death when exposed to oxygen-rich environments. The overexpression of MnSOD increases tolerance for oxidative stress [48] and freezing stress [49]. Chlorella, expressing high amounts of MnSOD mRNA had more tolerance of chilling stress [50]. Transgenic alfalfa plants with tobacco MnSOD targeted to mitochondria or chloroplasts had more rapid regrowth following freezing stress than wild type alfalfa plants [49]. Transgenic Brassica napus plants with wheat MnSOD cDNA had normal phenotype, but increased resistance to oxidative stress and aluminium toxicity [51]. Transgenic maize plants with a tobacco MnSOD gene targeted to chloroplasts had clear effects on foliar tolerance to chilling and oxidative stress [52]. Over-expression of mitochondrial MnSOD in chloroplasts by changing only the transit peptide will be an important application to protect chloroplasts from oxidative stress because chloroplastic cupper/zinc-SOD is deactivated during exposure to severe oxidative stress [21].
Members of the MnSOD multigene family have slightly different sequences in mRNA and amino acids, however, the end products, MnSOD enzymes, have the same function but different enzymatic activities and locations in cellular organelles [42,53].When three sequence variant MnSOD1, MnSOD2, and MnSOD3 manganese superoxide dismutase genes isolated from wheat NA were transformed into E. coli strain QC 871, which is a SOD double-mutant, all QC 871 transformants grown at 37˚C expressed mRNA of MnSOD variants, but only MnSOD2 transformant had functional SOD activity [53]. At 22˚C, MnSOD3 and MnSOD2 expressed active SOD enzymes, but MnSOD2 had higher enzymatic activity than MnSOD3. There was only one amino acid variance detected between the sequence MnSOD2 and sequences MnSOD1 and 3, therefore, there was a newly made gene switching the amino acid sequence at the variation site. The newly made MnSOD variant expressed functional SOD activity, even at higher levels than the original MnSOD2 at all observed temperatures. All these experimental data suggest that many plants have multigene family of MnSOD and the gene products have different enzymatic activity during cold acclimation and the recovery period after the freezing-thawing injury.
5. CONCLUSIONS
Freezing injury of crop and horticultural plants is not limited to particular geographic regions but all temperate and polar regions, therefore, all producers throughout the regions have suffered significant economic losses. To lead to methods for increasing tolerance for freezing stress in crop and horticultural plants, the information regarding the genomic structures, the patterns of expression and degradation, and the enzymatic activities of the members of the antioxidant enzymes have been extensively studied.
Antioxidant enzymes are of considerable scientific interest and commercial value due to their protective roles for many environmental stresses in plants [21,54]. Furthermore, they are of special interest due to their ubiquitous occurrence and to their use of trace metal ions for their active site [17]. The over-expression of antioxidant enzymes in plants could have benefits, e.g. increasing tolerance for freezing stress since freezing stress imposes active production of oxidative stress, resulting in severe damage of cell or even death,. Many genes of antioxidant enzymes detoxifying ROS increased the level of expression significantly in response to low temperature. The up-regulated expression levels of the antioxidant enzymes during cold acclimation can protect plants from the over-produced ROS by freezing stress.
Studies of antioxidant enzymes can provide the information regarding to the genomic structures, the expression patterns, and the enzymatic activities of antioxidant enzymes, which knowledge would end up with increasing tolerance for environmental stresses, especially freezing stress. There are still researches required for determining the roles of antioxidant enzymes on the tolerance of freezing stresses as follows: 1) determining whether the up-regulated or down-regulated antioxidant genes forms a significant response of plants to exposure to cold temperature; 2) defining the physical variation among the genes of antioxidant enzymes; 3) ascertaining whether different forms of the multigene family of antioxidant enzymes differ in their regulation in response to cold; 4) determining whether the chromosome region conferring the regulatory behavior of antioxidant genes; and 5) elucidating the roles of antioxidant enzymes in cold acclimation metabolically, e.g., the involvement in signal transduction and NAD(P)+ circulation.
The different variants in the multigene family of an antioxidant enzyme may also have different scavenging capabilities for a ROS, and selection of the form with the highest activity will be a challenge for applying the antioxidant form to increase freezing tolerance. Therefore, there will be more researches required to discover the physical differences between cold-responsive, and less responsive members of the antioxidant multigene family, and to identify the variant of antioxidant enzyme with the highest activity. Ultimately, these researches will lead us to understand the physical basis of the differential responsiveness of members with the highest activities of the antioxidant enzyme. With the aid of this information, selection of antioxidant genes with the highest activity is expected to contribute to enhancing freezing tolerance by the application in transformation or breeding programs.
6. ACKNOWLEDGEMENTS
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2011-0008199).
REFERENCES
- Singh, K. and Purohit, S.S. (1995) Plant productivity under environmental stress. Vedams eBooks (P) Ltd, New Delhi.
- Sakai, A. and Larcher, W. (1987) Frost survival of plants. Springer-Verlag, New York. doi:10.1007/978-3-642-71745-1
- Yoshida, S. and Uemura, M. (1990) Responses of the plasma membrane to cold acclimation and freezing stress. In: Larsson C. and Moller, I.M. Eds., The Plant Plasma Membrane: Structure, Function, and Molecular Biology, Springer-Verlag, Berlin Heidelberg, 293-319.
- Viswanathan, C., Jianhua, Z. and Jian-Kang, Z. (2007) Cold stress regulation of gene expression in plants. Trends in Plant Science, 12, 444-451. doi:10.1016/j.tplants.2007.07.002
- Allan, R.E., Pritchett, J.A. and Little, L.M. (1992) Cold injury observation. Annual Wheat Newsletter, 38, 281.
- Burke, M.J., Gusta, L.V., Quamme, H.A., Weiser, C.J. and Li, P. H. (1978) Freezing and injury in plants. Annual Review of Plant Physiology, 27, 507-528. doi:10.1146/annurev.pp.27.060176.002451
- Guy, C.L. (1990) Cold acclimation and freezing stress tolerance: Role of protein metabolism. Annual Review of Plant Physiology and Plant Molecular Biology, 41, 187- 223. doi:10.1146/annurev.pp.41.060190.001155
- Thomashow, M.F. (2001) So what’s new in the field of plant cold acclimation? Lots! Plant Physiology, 125, 89- 93. doi:10.1104/pp.125.1.89
- Stushnoff, C., Fowler, D.B. and Brule-Babel, A. (1984) Breeding and selection for resistance to low temperature. In: Vose, P.B. Ed., Plant Breeding: A Contemporary Basis, Pergamon Press, Oxford, 115-136.
- Taiz, L. and Zeiger, E. (2006) Plant physiology. 4th Edition, Sinauer Associates, Inc., Sunderland.
- Salisbury, F.B. and Ross, C.W. (1991) Plant physiology. 4th Edition, Wadsworth Publishing Company, Beverly, 481.
- Gilmour, S.J., Hajela, R.K. and Thomashow, M.F. (1988) Cold acclimation in Arabidopsis thaliana. Plant Physiology, 87, 745-750.doi:10.1104/pp.87.3.745
- Tseng, M.J. and Li, P.H. (1987) Changes in nucleic acid and protein synthesis during induction of cold hardiness. In: Li, P.H. Ed., Plant Cold Hardiness, Alan R. Liss, Inc., New York, 1-28.
- Palva, E.T. and Pekka, H. (1997) Molecular mechanism of plant cold acclimation and freezing tolerance. In: Li, P.H. and Chen, T.H.H., Eds., Plant Cold Hardiness, Plenum Press, New York, 3-14.
- Trischuk, R.G., Schilling, B.S. Wisniewski, M. and Gusta, L.V. (2006) Freezing stress: Systems biology to study cold tolerance. In: Madhava Rao, K.V., Raghavendra, A.S. and Janardhan R.K., Eds., Physiology and Molecular Biology of Stress Tolerance in Plants, Springer, Dordrecht, 131-155.
- Seki, M., et al. (2002) Monitoring the expression profiles of 7000 Arabidopsis genes under drought, cold and highsalinity stresses using a full-length cDNA microarray. Plant Journal, 31, 279-292. doi:10.1046/j.1365-313X.2002.01359.x
- Halliwell, B. and Gutteridge, J.M.C. (2007) Free radicals in biology and medicine. 4th Edition, Oxford University Press, New York.
- Zelko, I.N., Mariani, T.J. and Folz, R.J. (2002) Superoxide dismutase multigene family: A comparison of the CuZnSOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) gene structures, evolution, and expression. Free Radical Biology and Medicine, 33, 337-349. doi:10.1016/S0891-5849(02)00905-X
- Fridovich, I. (1991) Molecular oxygen: Friend and foe. In: Pell, E.J. and Steffen, K.L. Eds., Active Oxygen/Oxidative Stress and Plant Metabolism, American Society of Plant Physiologists, Rockville, 1-5.
- Zhang, J. and Kirkham, M.B. (1994) Drought-stress-induced changed in activities of superoxide dismutase, catalase, and peroxidase in wheat species. Plant Cell Physiology, 35, 785-791.
- Allen, R.D. (1995) Dissection of oxidative stress tolerance using transgenic plants. Plant Physiology, 107, 1049- 1054.
- Monk, L.S., Fagerstedt, K.V. and Crawford, R.M.M. (1989) Oxygen toxicity and superoxide dismutase as an antioxidant in physiological stress. Physiologia Plantarum, 76, 456-459.
- Scandalios, J.G. (1993) Oxygen stress and superoxide dismutases. Plant Physiology, 101, 7-12.
- Benson, E.E. and Noronha-Dutra, A.A. (1988) Chemiluminiscence in cryopreserved plant tissue cultures: The possible involvement of singlet oxygen in cryoinjury. CryoLetters, 9, 120-131.
- McKersie, B.D. and Bowley, S.R. (1997) Active oxygen and freezing tolerance in transgenic plants. In: Li, P.H. and Chen, T.H.H., Eds., Plant Cold Hardiness, Plenum Press, New York, 203-214.
- Park, J.I., Grant, C.N., Davies, M.J. and Dawes, I.W. (1998) The cytoplasmic Cu, Zn superoxide dismutase of Saccharomyces cerevisiae is required for resistance to freezethaw stress. Generation of free radicals during freezing and thawing. Journal of Biological Chemistry, 273, 22921- 22928. doi:10.1074/jbc.273.36.22921
- Kendall, E.J. and McKersie, B.B. (1989) Free radical and freezing injury to cell membranes of winter wheat. Physiologia Plantarum, 76, 86-94. doi:10.1111/j.1399-3054.1989.tb05457.x
- Davies, K.J.A. (1995) Oxidative stress: The paradox of aerobic life. In: Rice-Evans, C., Halliwell, B., Lunt, G.G., Eds., Free Radicals and Oxidative Stress: Environment, Drugs, and Food Additives, Biochemical Society Symposium 61, Portland Press, London, 1-32.
- Fink, R.C. and Scandalios, J.G. (2002) Molecular evolution and structure-function relationships of the superoxide dismutase gene families in Angiosperms and their relationship to other eukaryotic and prokaryotic superoxide dismutases. Archives of Biochemistry Biophysics, 399, 19- 36. doi:10.1006/abbi.2001.2739
- Smith, M.W. and Doolittle, R.F. (1992) A comparison of evolutionary rates of the two major kinds of superoxide dismutase. Journal of Molecular Evolution, 34, 175-184. doi:10.1007/BF00182394
- Asada, K. (1999) The water-water cycle in chloroplast: Scavenging of active oxygens and dissipation of excess photons. Annul Review of Plant Physiology and Plant Molecular Biology, 50, 601-639. doi:10.1146/annurev.arplant.50.1.601
- Apel, K. and Hirt, H. (2004) Reactive oxygen species: Metabolism, oxidation stress, and signal transduction. Annual Review of Plant Physiology and Plant Molecular Biology, 55, 373-399.
- Levine A., Tenhaken, R., Dixon, R. and Lamb, C. (1994) H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell, 79, 583- 593. doi:10.1016/0092-8674(94)90544-4
- O’Kane, D., Gill, V., Boyd, P. and Burdon, R. (1996) Chilling, oxidative stress and antioxidant responses in Arabidopsis thaliana callus. Planta, 198, 371-377. doi:10.1007/BF00620053
- Shinozaki, K. and Yamajuchi-Shinozaki, K. (1997) Gene expression and signal transduction in water-stress response. Plant Physiology, 115, 327-334. doi:10.1104/pp.115.2.327
- Wu, G., Shortt, B.J., Lawrence, E.B., Fitzsimmons, J.L.K.C., Levine, E.B., Raskin, I. and Shah, D.M. (1997) Activation of host defense mechanisms by elevated production of H2O2 in transgenic plants. Plant Physiology, 115, 427- 435.
- Prasad, T.K., Anderson, M.D., Martin, B.A. and Stewart, C.R. (1994) Evidence for chilling-induced oxidative stress in maize seedlings and a regulatory role for hydrogen peroxide. Plant Cell, 6, 65-74.
- Prasad, T.K., Anderson, M.D. and Stewart, C.R. (1994) Acclimation, hydrogen peroxide, and abscisic acid protect mitochondria against irreversible chilling injury in maize seedlings. Plant Physiology, 105, 619-627.
- Mittler, R. (2002) Oxidative stress, antioxidants and stress tolerance. Trends in Plant Science, 7, 405-410. doi:10.1016/S1360-1385(02)02312-9
- Baek, K.H. and Skinner, D.Z. (2003) Alteration of antioxidant enzyme gene expression during cold acclimation of near isogenic wheat lines. Plant Science, 165, 1221- 1227. doi:10.1016/S0168-9452(03)00329-7
- Miao, Z. and Gaynor, J.J. (1993) Molecular cloning, characterization and expression of Mn-superoxide dismutase from the rubber tree (Hevea brasiliensis). Plant Molecular Biology, 23, 267-277. doi:10.1007/BF00029003
- Zhu, D. and Scandalios, J.G. (1993) Maize mitochondrial manganese superoxide dismutases are encoded by a differentially expressed multigene family. Proceedings of the National Academy Sciences of United States of America, 90, 9310-9314. doi:10.1073/pnas.90.20.9310
- Wu, G., Wilen, R.W., Robertson, A.J. and Gusta, L.V. (1999) Isolation, chromosomal localization, and differenttial expression of mitochondrial manganese superoxide dismutase and chloroplastic copper/zinc superoxide dismutase genes in wheat. Plant Physiology, 120, 513-520. doi:10.1104/pp.120.2.513
- Baek, K.H. and Skinner, D.Z. (2006) Differential expression of manganese superoxide dismutase sequence variants in near isogenic lines of wheat during cold acclimation. Plant Cell Reports, 25, 223-230. doi:10.1007/s00299-005-0073-6
- Baek, K.H. and Skinner, D.Z. (2006) Differential mRNA stability to endogenous ribonucleases of the coding region and 3’ untranslated regions of wheat (Triticum aestivum l.) manganese superoxide dismutase genes. Plant Cell Reports, 25, 133-139. doi:10.1007/s00299-005-0046-9
- Carlioz, A. and Touati, D. (1986) Isolation of superoxide dismutase mutants in Escherichia coli is superoxide necessary for aerobic life? EMBO, 5, 623-630.
- Van Loon, A.P., Pesold-Hurt, B. and Shatz, G. (1986) A yeast mutant lacking mitochondrial manganese-superoxide dismutase is hypersensitive to oxygen. Proceedings of the National Academy Sciences of United States of America, 83, 3820-3824. doi:10.1073/pnas.83.11.3820
- Bowler, C., Alliotte, T., Bulcke, M.V.D., Bauw, G., Vandekerckhove, J., Montagu, M. V. and Inze, D. (1989) A plant manganese superoxide dismutase is efficiently imported and correctly processed by yeast mitochondria. Proceedings of the National Academy of Sciences of United States of America, 86, 3237-3241. doi:10.1073/pnas.86.9.3237
- McKersie, B.D., Chen, Y., De Beus, M., Bowley, S.R., Bowler, C., Inze, D., D’Halluin, K. and Botterman, J. (1993) Superoxide dismutase enhances tolerance of freezing stress in transgenic alfalfa (Medicago sativa L.). Plant Physiology, 103, 1155-1163. doi:10.1104/pp.103.4.1155
- Clare, D. A., Rabinowitch, H. D. and Fridovich, I. (1984) Superoxide dismutase and chilling injury in Chlorella ellipsoidea. Archives of Biochemistry and Biophysics, 231, 158-168. doi:10.1016/0003-9861(84)90372-2
- Basu, U., Good, A.G. and Taylor, G.J. (2001) Transgenic Brassica napus plants overexpressing aluminum-induced mitochondrial manganese superoxide dismutase cDNA are resistant to aluminum. Plant, Cell, and Environment, 24, 1269-1278. doi:10.1046/j.0016-8025.2001.00783.x
- Van Breusegem, F., Slooten, V., Stassart, J.M., Botterman, J., Moens, T., van Montagu, M. and Inze, D. (1999) Effects of overproduction of tobacco MnSOD in maize chloroplasts on foliar tolerance to cold and oxidative stress. Journal of Experimental Botany, 50, 71-78.
- Baek, K.H. and Skinner, D.Z. (2010) Molecular cloning and expression of sequence variants of manganese superoxide dismutase genes from wheat. Korean Journal of Environmental Agriculture, 29, 77-85. doi:10.5338/KJEA.2010.29.1.077
- Holmberg, N. and Bülow, L. (1998) Improving stress tolerance in plants by gene transfer. Trends in Plant Science, 3, 61-66. doi:10.1016/S1360-1385(97)01163-1

