Journal of Minerals and Materials Characterization and Engineering
Vol.2 No.1(2014), Article ID:41965,8 pages DOI:10.4236/jmmce.2014.21006
2k Factorial Experiments on Factors that Influence the Recovery of Gold during the Upgrade of Ilesha-Itagunmodi Gold Ore through Froth Flotation
1Prototype Engineering Development Institute (PEDI), Ilesa, Nigeria
2Materials Science and Engineering, Obafemi Awolowo University (O.A.U.), Ile-Ife, Nigeria
3Department of Chemical Engineering, Landmark University, Omu-Aran, Nigeria
Email: kayoxemak@yahoo.com
Copyright © 2014 Kayode Emmanuel Oluwabunmi et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accordance of the Creative Commons Attribution License all Copyrights © 2014 are reserved for SCIRP and the owner of the intellectual property Kayode Emmanuel Oluwabunmi et al. All Copyright © 2014 are guarded by law and by SCIRP as a guardian.
Received September 13, 2013; revised October 20, 2013; accepted November 4, 2013
KEYWORDS
Factorial; Froth Flotation; Collector; Frother; Concentration Surface Graph; Conditioning Time
ABSTRACT
A low-grade gold ore from Ilesha-Itagunmodi, south western Nigeria was panned, dried and ball-milled before sieving into fractions. The morphological, mineralogical and chemical composition was studied by optical microscopy (Reflected and Transmission), X-ray diffraction (XRD) and Energy Dispersive X-ray fluorescence (XRF), respectively. The sieved fractions were subjected to chemical analysis (AAS). The +106 μm sieve had the highest concentration of the mineral and was then selected for the upgrade through froth flotation using standard reagents. In this paper a report on a 2k factorial experiment that provides an understanding of the impact of operational variables on the quantity of gold mineral obtained from the ore during froth flotation is presented. Analysis results showed that Ilesha-Itagunmodi gold ore is non-refractory with fine grain particles, amenable to froth flotation and contained about 20 other associated minerals, gold had a concentration of about 0.0024%. A combination of P-Xanthate and amine glycol collectors at a pH of 9.2 only produced a considerable increase in gold yield. This translated to about 87.13% increase in recovery of gold from the ore. Analysis of variance (ANOVA) was carried out and the model equation obtained was subsequently optimised to obtain a model equation that could be used in predicting the recoverable quantity of gold, indicating that F11,1-values for Collector concentration, Frother concentration, pH and Conditioning time were 156.86, 6.96, 43.81, and 56.77 respectively. A model with an F value of 88.41 was obtained which indicated that the model was significant. The model equation obtained was subsequently optimised to be able to predict the recoverable quantity of gold. A “Pred R-Squared” value of 0.9365 (93.65%) was also obtained and is in reasonable agreement with the “Adj R-Squared” value of 0.9534 (95.34%). It was established that Ilesha placer gold ore is amenable to froth flotation using standard reagents.
1. Introduction
One of the most striking differences between the ores of gold and those of all other metals lies in the extremely small proportion which the desired material bears to the worthless gangue by which it is accompanied [1]. The greater part of the vein gold now being produced is derived from ores containing only about one part of gold in 100,000 or even less [2]. However, due to the increase in demand for gold which has led to its rising global price, there is a need to develop suitable mineral processing routes to intensely upgrade placer gold deposits for effective gold recovery of placer deposits in Nigeria. The gold belt in the southwestern Nigeria is situated within Ife and Ilesha and is reputed to contain about 2 million ounces of gold that can be found to a depth of 3000 meters [3]. This Placer deposits contain low concentrations of gold [4]. They are non-refractory and can be processed using various techniques such as flotation [5]. Froth flotation which is being widely accepted as more economical and environmentally friendly than other processing routes such as amalgation and cyanidation, and has been successfully applied to deposits from various ore sources around the world [6-8]. The main purpose of this work is to determine the response of Ilesha-Itagunmodi gold to froth flotation using standard reagents. This will be of great economic benefit to the nation if properly exploited.
1.1. Materials and Methods
1.1.1. Materials and Sample Preparation
Pit samples were collected from 3 pits in Ariye, a village in Itagunmodi town. The first 2 pits had the same opening which narrowed down and had a corridor at the bottom that was at 90 degrees to the main pit, they had the following GPS reading Latitude 7˚30.673′ North and Longitude 4˚40.131′ East. The approximate depth was about 12 feet, with an approximate diameter of about 7 feet. The third sample was collected from a separate stand alone pit which was dug to an approximate depth of about 7 feet with an approximate width of about 6 feet. GPS reading of Latitude 7˚30.850′ North and Longitude 4˚40.077′ East. 120 kg of the samples were obtained at the stone base.
1.1.2. Ore Preparation
The sample was panned and the heavy metal ores (black sand) was recovered. This was dried and ball milled in batches for fifteen minutes using a Denver ball mill that was set at 60 rpm. Particle size analysis was done in phases using Endecott standard sieves to separate the grounded concentrate into different size fractions. Sieve sizes of 710 μm, 500 μm, 355 μm, 250 μm, 106 μm, 75 μm, 63 μm and 45 μm were used. The sieves were arranged using geometric progression based on √2 with the sieve having the largest aperture on top [9]. 500 g of the black sand concentrate was used for each sieving set. The sieves were taken apart and the ore retained on each sieve was weighed (Table 1).
Each of the fractions was subjected to Atomic Absorption spectroscopic tests to determine the fraction with the highest concentration. 1 g of the concentrate was heated with 10 ml of aqua-regia (HCl and HNO3 in the ratio 3 to 1) on a hot plate in a Teflon crucible at a temperature of about 100˚C. The filtrate was diluted according to atomic absorptions spectrometer (AAS) analysis requirements. Buck Scientific equipment Atomic Absorption Spectrometer (AAS) model 210 V was used and the experiment was done in triplicates. The +106 µm was found to have the highest concentration and was chosen for subsequent experiments (Table 2).
1.1.3. Description of Froth Flotation Process
A standard matrix (Table 3), showing the codes and the values, was constructed to establish a relationship between the recovery of gold mineral and the operational variables (collector concentration, frother concentration, pH and conditioning time). The four factors were operated at two levels for the 2k factorial design of the experiment. The choice of the factors to be studied was done on the basis of the theoretical data about several factors that determine the amount of the gold mineral that is

Table 1. Sieve Analysis of the Ilesha-Itagunmodi black sand concentrate.

Table 2. AAS result of the gold concentrations in the sieved fractions before flotation.

Table 3. Matrix for the design of the flotation experiment.
recoverable during an ore upgrade via froth flotation. Flotation experiments were conducted on the +106 µm a Denver size D12 laboratory type flotation machine. Tap water was used in experimental work. Pulp pH was adjusted with sodium Silicate to 9.2 and 10 using a Horiba pH meter for the 2 levels worked with. The flotation experiment was performed under certain experimental conditions that 16 experimental sets were generated and each experimental set was repeated twice to give a total of 48 responses, the resulting average of each set of experiment was obtained (Table 4). The experimental results and the
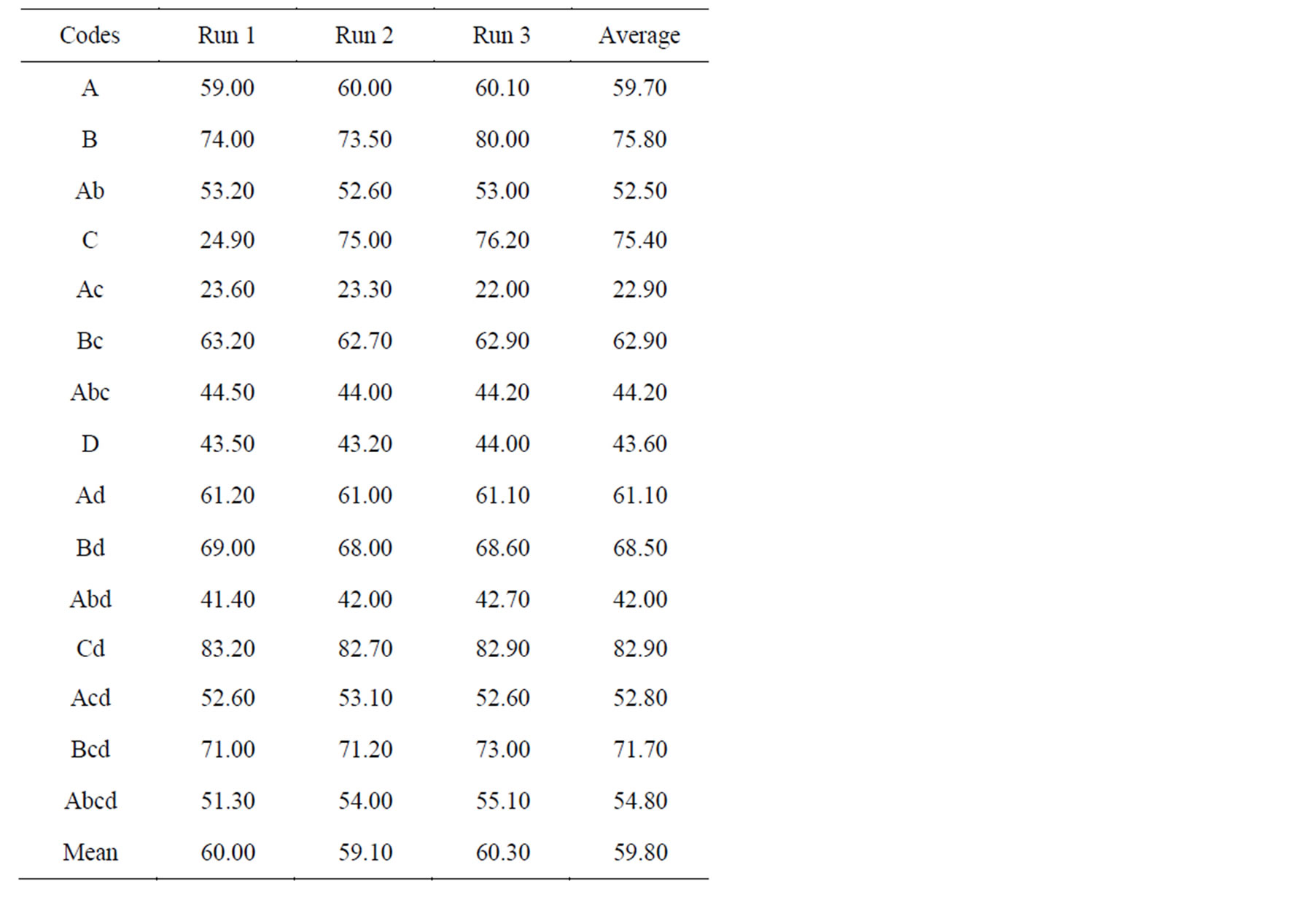
Table 4. Results of the froth flotation experiment quantity of gold (g/t).
effects of the factors at two levels and interactions that have a significant effect on the yield of gold were analyzed using two way analysis of variance (ANOVA) with replicates. The 2k factorial tool on the Design Expert 8.0.3 software (by Stat-Ease, Maryland) was used to achieve this. The sum of squares of factor A (SA), factor B (SB), error (SE) and total (ST) as well as the comparison of the F values (Fcalc) with the critical F values (F table) for (1 − α) confidence (or for significance level of α) were all generated by the software. If the calculated value is greater than the critical one, then the independent variable has significant influence on the dependent variable at a level of 100 × (1 − α) % confidence [1,10]. The probability (p) values of independent variables were also obtained using this software. The calculated “p” value is lower than the significance level α, thereby making the influence of independent variable on the dependent variable statistically significant. A mathematical model was generated from the results of the 2k factorial experiments and a 3D surface graphs to show the interaction of the factors and their effect on gold yield were obtained.
2. Results and Discussion
2.1. Bulk Mineralogy Results
Chemical and Bulk mineralogical compositions of the sample were studied by X-ray fluorescence (XRF) using a ED-XRFS MiniPAL 4 Model (2005), Xray diffraction (XRD) analysis using Radicon-10 diffractometer and Optical Microscopy was carried out using a Leica Reflected light microscope model number DMRX/MP60 respectively. XRF confirmed the presence of gold in the form of Au2O3 with a concentration of about 0.0024% other minerals of significant concentrations include Ti occurring as TiO2 and Ti2O3 and Fe occurring as Fe2O3 with 36.5%, 0.03% and 35.67% concentrations respectively (Table 5), while the XRD analysis showed that the gold oxide phase had a range of (32.4606 - 54.0942) for 2Ɵ values (Figure 1) and was sparingly and non-uniformly distributed within its ore [11]. The reflected light microscopy (Plates 1a, 1b) showed brass yellow coloured grains which was suspected to be the gold bearing materials, grey to colourless grains which represents siliceous minerals or quartz, and black grains which were suspected to be sulphide minerals like pyrites, arsenopyrites and chalcopyrite [12].
2.2. Results of Froth Flotation Process
The recovery of the gold mineral ranges from 83.2 g/t and 22 g/t (Table 4). The least response occurred when the pH value was at high values (3rd run of experiment number 5). This result suggests that pH is a negative factor when increased beyond the optimum range and this reduces the yield of gold. This indicates that the higher the value of the pH factor the lower the yield of gold. The significance of this factor is observed when experiment 12 results are compared with the results of experiment 5. The difference between the averages of these two experiments was about 60 g/t. Experiments 5, 11, 13 and 15 also show that in most cases, maximum response value occurred when collector concentration, frother concentration and conditioning time were at high values. Experiment 12 shows that A, B, D are significant factors in the recovery of gold mineral. This result also suggests that Collector concentration, frother concentration and conditioning time are positive factors and improve the quantity of gold recovered.
The highest yield of gold obtained during the 12th experimental set with an average value of 82.90 g/t, can be attributed to the following factors; that the pH was maintained at an optimum value of (9.2), despite the increase in collector concentration, frother concentration and conditioning time. However, the least yield obtained during the 5th experimental set with an average value of 22.90 g/t occurred because there was an increase in pH value outside the optimum range which brought about an increase in the contact angle, thereby preventing sustained flotation of the gold mineral. The significance of these is observed from the 3D surface graphs. The results of Analysis of variance (ANOVA) are shown in Tables 6 and 7.
The ANOVA results yielded a model with a F-value of 88.4 which was remarkably significant and had a Pvalue < 0.0001. There is only a 0.01% chance that a “Model F-Value” this large could occur due to noise. Values of “Prob > F” less than 0.0500 indicate model terms are significant. Also A, B, C, D, AB, AC, AD, BC, CD, ABC, ACD are seen as significant model terms. Theses values revealed that this regression is statistically significant, only 4.7% of the total variations was not explained by this regression model. Meanwhile, the R2 indicated a high reliability between the observed values and the predicted values. The lack of fit was not significant, which revealed that the model was quite significant for the response. The value of R2 and adjusted R2 was 96.4% and 95.3% respectively. Therefore it can be used for the theoretic prediction of the yield of gold during the flotation of Ilesha-Itagunmodi gold ore. A non homogenous cubic regression equation was obtained from the ANOVA Where Y is the gold mineral yield and 58.21 is the intercept.

Table 5. Result of the analysis done by ED-XRF on the + 106 µm sieve fraction of the Ilesa-Itagunmodi Gold ore.

Figure 1. X-ray diffractogram of Ilesha-Itagunmodi black sand concentrate.
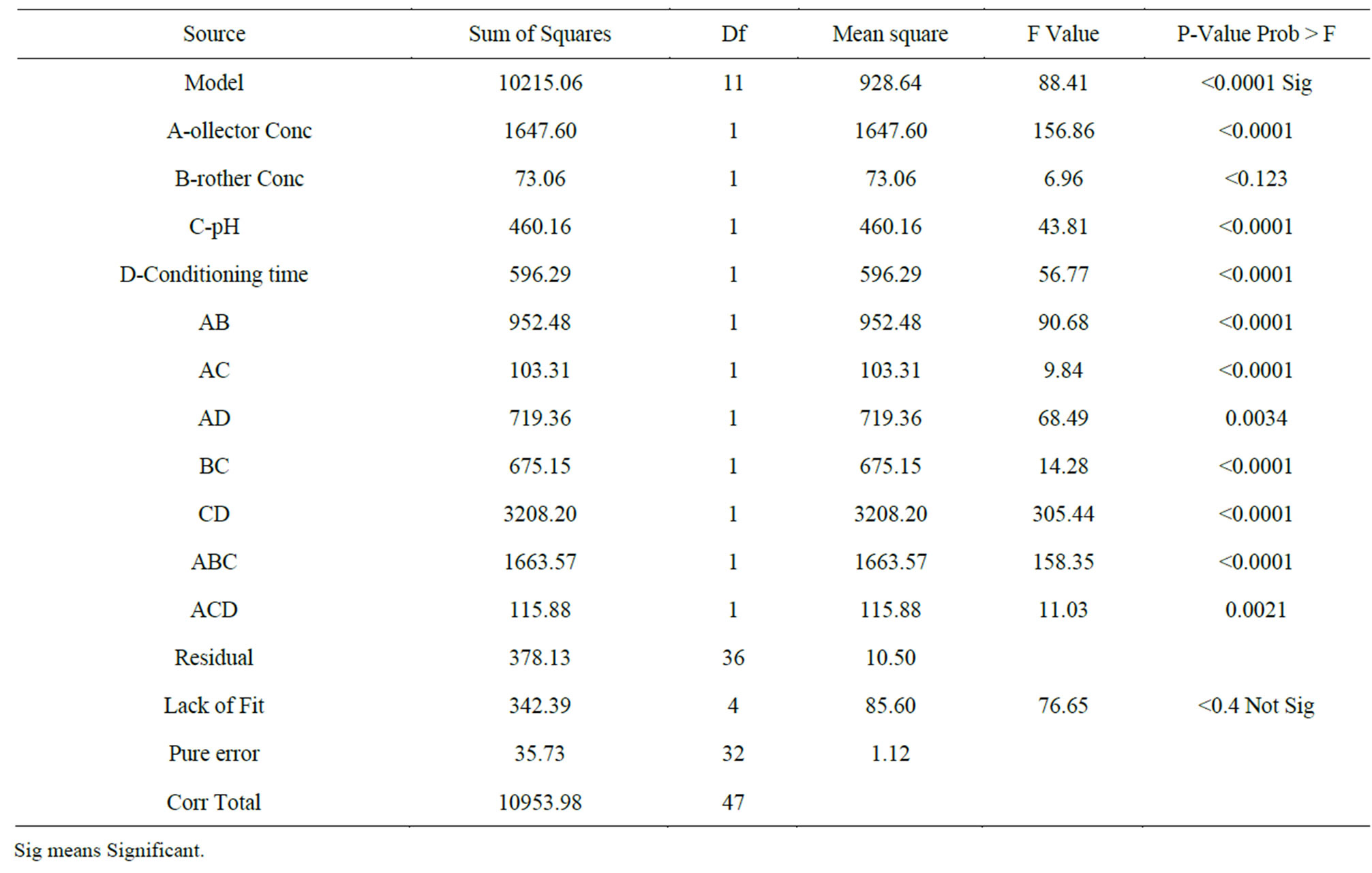
Table 6. Results obtained from the ANOVA analysis.
 (a)
(a) (b)
(b) (c)
(c) (d)
(d) (e)
(e) (f)
(f) (g)
(g) (h)
(h) (i)
(i) (j)
(j)
Figure 2. (a-j) 3D surface graphs showing influence of factors on gold yield and contour line showing the mutual interaction of the parameters.
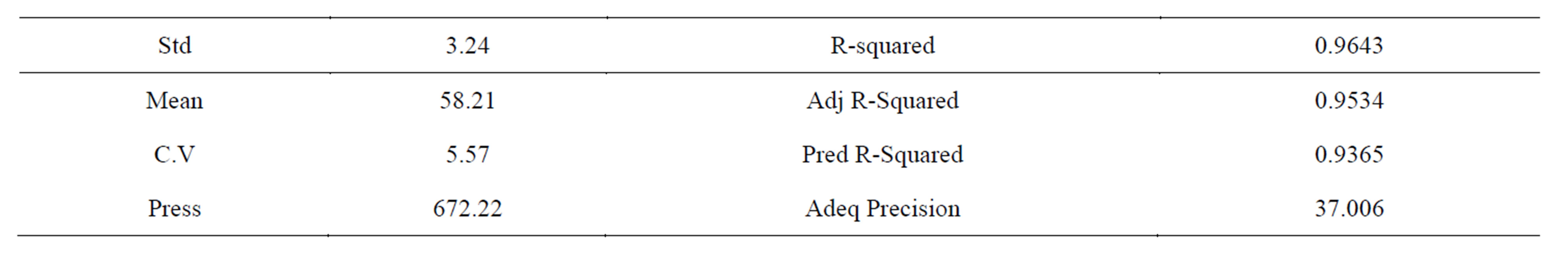
Table 7. Results obtained from the ANOVA analysis (contd.).

Table 8. Optimization values.
Y = 58.21 + 5.86A + 1.23B − 3.10C + 3.52D + 4.45AB − 1.47AC − 3.87AD + 3.75BC + 8.18CD − 5.89ABC − 1.55ACD Optimization of these given values was then carried out with software to obtain values for carrying out the experiment under ideal conditions. 14 sets of values for the 4 factors and their gold yield values under ideal conditions were generated by the software. The first set was selected (Table 8) and worked with in the laboratory.
The maximum yield of gold in theory was 83.825 g/t in theory, when the concentration of collector was 4 g, concentration of frother was 0.9 ml, pH was 9.2 and conditioning time was 5 minutes. On this condition, experiments were carried out in triplicate and the yield of the gold mineral was 83.82 g/t.
Such a graph as this can provide a kind of visual method to observe responsive value and also carry out tests for prameter level relation. The 3D surface graphs and its contour line by means of the above mentioned regression mathematic equation is shown below. Figures 2(a)-(j) show 3D surface graphs and contour plots for the following (a, b) effect of collector conc. and frother (AB) and their mutual interaction. (c, d) effect of collector concentration and pH (AC) and their mutual interaction. (e, f) effect of collector concentration and conditioning time (AD) and their mutual interaction. (g, h) effect of Concentration of frother and pH and their mutual interaction. (i, j) effect of pH and conditioning time and their mutual interaction.
3. Conclusion
The two-level factorial experiments used in this study enabled the determination of the effect of the significant experimental parameters influencing the quantity of gold mineral recovered during the froth flotation of Ilesha Itagunmodi gold ore. It could be used to forecast the quantity of gold yield. The 24 factorial experimental design results showed that an average yield of about 82.90 g/t of gold after flotation compared to 44.30 g/t obtained before flotation. This translated to about 87.13% increase in recovery of gold from the ore for a pulp density of 200 g/L and Optimum yield of gold at 83.82 g/t was obtained when Collector concentration was 4 g, the frother concentration was 0.9 ml, the pH value was 9.2 and conditioning time was 5 minutes.
REFERENCES
- S. A. Hussien, “Flotation of Low Grade Gold Ores,” Ph.D. Thesis, Swiss Federal Institute of Technology, Zurich, 1951, pp. 1-95.
- S. M. Bulatovic and D. M. Wyslouzil, “Flotation Behaviour of Gold during Processing of Porphyry CopperGold Ores and Refractory Gold-Bearing Sulphides,” In: 2nd International Gold Symposium, Vol. 1, Academy Press, Lima, 2000, pp. 2964-2975. www.worldgoldcouncil.com
- Nigerian Mining Corporation (NMC), “Mining Activites in Nigeria,” 2001. www.community.vanguardngr.com
- J. A. Adekoya, O. O. Kehinde-Phillips and A. M. Odukoya, “Geological Distribution of Mineral Resources in Southwestern Nigeria. Prospect for Investment in Mineral Resources of Southwestern Nigeria,” Nigerian Mining and Geosciences Society (NMGS), 2003, 180p. www.scirp.org/journal/PaperInformation
- J. A. Ajayi and S. A. Awe, “Recovery Efficiency Study on Ilesha Placer Gold Ore by Flotation Using Locally Sourced Frothers and Collectors,” XXV International Mineral Processing Congress: IMPC 2010, “Smarter Processing for the Future,” Brisbane, 6-10 September 2010, 8p. www.ausimm.com.au/publications/publication.aspx
- R. W. Lehne, “Treatment and Microscopy of Gold and Base Metal Ores,” Geneva University, Geneva, 2006. www.scribd.com
- J. M. W. Mackenzie and P. J. Cabassi, “Froth Flotation Process for the Recovery of Minerals and a Collector Composition for Use Therein,” Patent No: 4908125, 1990. www.patentstorm.com
- W. Wang and G. W. Poling, “Methods for Recovering Fine Placer Gold,” CIM Bulletin, Vol. 76, No. 860, 1983, pp. 47-56. www.nora.nerc.ac.uk
- B. A. Wills, “Mineral Processing Technology: An Introduction to the Practical Aspect of ore Treatment and Mineral Recovery,” Pergamon Press, Oxford, 1998.
- T. Guler and U. Akdemir, “Statistical Evaluation of Flotation and Entrainment Behavior of an Artificial Ore,” ScienceDirect, 2011, pp. 1-7. www.sciencedirect.com
- C. J. Mitchell, E. J. Evans and M. T. Styles, “British Geological Survey, Technical Report. WC/97/14. A Review of Gold Particle Size and Recovery Method,” Mitigation of Mining Related Mercury Pollution Hazards, Keyworth, Nottingham, BGS, 1997, pp. 1-34. www.nora.nerc.ac.uk
- A. Emmanuel, et al., “Application of the Principal Component Analysis on Geochemical Data: A Case Study in the Basement Complex of Southern Ilesha Area, Nigeria,” Saudi Society for Geosciences, 2010, pp. 239-248. www.arab.j.geosciences.org/pdf

