American Journal of Plant Sciences
Vol.09 No.08(2018), Article ID:86074,11 pages
10.4236/ajps.2018.98121
Taxonomic Significance of Foliar Epidermal Characters in Azanza garckaena (F. Hoffm.) Exell & Hillc in Tula, Kaltungo Local Government Area of Gombe State, Nigeria
H. M. Abba, H. Daniel, S. Sale, D. A. Zhigila
Department of Biological Sciences, Botany Programme, Gombe State University, Gombe, Nigeria

Copyright © 2018 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: May 18, 2018; Accepted: July 17, 2018; Published: July 20, 2018
ABSTRACT
Leaf epidermal microscopy of Azanza garckeana was studied in search of stable taxonomic characters and delimitation of the plant. Fresh leaves were obtained from 3 accessions in Tula: Tula Wange, Tula Baule and Tula Yiri/Bwaile respectively. Leaf epidermal peels of both surfaces of the plant were made using free hand sectioning and maceration methods. Temporary slides were prepared for observations under light microscope to examine their stomatal features, epidermal cell shapes, and anticlinal cell-wall patterns. The result revealed epiamphistomatic leaves, Stomatal complex type (SCT) was exclusively anomocytic, epidermal cell shapes were all irregular in shape, with curved anticlinal cell wall patterns. There was no significant difference on the abaxial surface across all the parameters at all accessions but significant difference exists (P ≤ 0.05) on some characters on the adaxial surfaces. The Stomatal density was the highest on the adaxial surface of accession I (23.67 ± 4.04a) and the lowest on the adaxial surface of accession II (13.33 ± 2.08b mm2). The Stomatal sizes of abaxial surface of accession I was the highest (45.49 ± 3.73 µm), while adaxial surface of accession III showed the lowest (24.98 ± 2.27a μm). The Stomatal index was the highest (7.42 ± 2.30a) on the adaxial surface of accession I, and the lowest (4.23 ± 0.20b) on the abaxial surface of accession II. It was concluded that there were similarity of anomocytic type of stomata, small and large stomatal sizes, presence of dendritic trichomes, irregular and curved epidermal cells on both leaf surfaces, and variations in the stomatal index within all the accessions. Although these variations are good and reliable taxonomic features of the accessions. More taxonomic tools are required for the delimitation of the accessions into distinct taxa.
Keywords:
Azanza garckeana, Dendritic Trichome, Epiamphistomatic, Anomocytic

1. Introduction
Azanza garckeana (F. Hoffm.) Exell & Hillc. belongs to the family Malvaceae. It is popularly known as Afrikaans (Snot apple); Bemba (Chinga, Mukole); English (Azanza, tree hibiscus, Snot apple, Wild hibiscus, African chewing gum); Lozi (Muneko); Lunda (mukole); Ndebele (Uxhakuxhaku); Nyanja (Mkole); Shona (Mutohwe); Swahili (Mtobo); Tongan (Muneko); Tswana (Morajwa), Goron Tula in Hausa. It is widely distributed in Eastern and Southern Africa [1] [2]. The specific countries where the species is found are Botswana, Kenya, Nigeria, South Africa, Tanzania, and Zimbabwe. In Nigeria, they are found in North-West Katsina State in Kankiya, Daggish, in the middle belt of River Benue. In Northern Nigeria, they are found in Tula area of Kaltungo Local Government Area of Gombe State, Kali hills of Zah district of Michika Local Government Area of Adamawa State [3] [4] stated that the species grows naturally in all types of woodlands from about 1700 m above sea level.
Description of the Plant
Azanza garckeana is a semi-deciduous tree/shrub with a rounded medium crooked stem [5] [6]. The tree can grow to a height of 3 to 15 metres (Plate 1) depending on the environment in which it is growing [2] [4] [6] [7] [8] [9]. The tree is multi stemmed with straight or crooked stem, which is sometimes forking from the base, and the stem can reach up to 20 - 25 cm in diameter at breast height. The bark is rough and greyish-black to brown, fibrous with longitudinal fissures [1] [4] [8] [9]. The twigs are hairy when young but becomes smoothly with age and branches have wooly hairs [1] [2].
The leaves are distinctively rounded 8 by 12 cm on long stalks. They are alternatively simple, alternate and roundish. The leaves have 3 - 5 lobes. Usually bluntly pointed or rounded. The base of the leaves is heart shaped and is 5 - 7 nerved. The young leaves are bronze in color and velvety [2]. Flower is large up to 60 cm long, solitary on long pedicels in axils of uppermost leaves [2] [6] [9]. The flowers have a conspicuous cup like calyx with which the calyx is fused. The flowers have many stamens and 5 petals, which are yellow in color with dark purple or dark red center [6]. The flowers are bisexual with floral part in five’s [9]. They are showy yellow, ageing to purplish in color with a maroon patch at the base of each petal. Its flower in wet season and fruits in dry season (April through August) [9]. The fruit are spherical and woody, 2.5 cm to 4 cm in diameter long with short hairs. The fruits are divided into 4 to 5 section. They are yellowish to brownish green and hairy when mature and tardily dehiscent [1] [6]. Fruit are edible from November. The seed are hemispherical in shape up

Plate 1. Azanza garckeana.
to 10 mm long and 7 mm thick with brownish and wolly floss [2] [5] [9] the wood is greyish in color and the heartwood is dark. It is hard durable and strong. The tree is susceptible to leaf defoliation by insect (Empoasca) and fungi (Phakopsora) [10] reported that tree could be attacked by leafhoppers both in the field and Nursery. Azanza garckeana is a host to the cotton stainner and black beetle and therefore should not be planted in cotton producing areas [2] [4] [5]. The fruit are the most useful resources of Azanza garckeana. Fruits are eaten while slightly green or when ripe. They are persistent, therefore are picked on ripening. Some people dry them and reconstitute them later [10]. Despite their hard woody nature, ripe fruit carpels are edible and have an energy content of 8.10 kJ/g. A sweet mucilage comes out when chewed like chewing gum producing glutinous slime [2] [10]. The fruit can be soaked in small amount of water to make jelly [2]. They can also be boiled and used as relish or made in to porridge [5]. The fruit pulp at 52% dry matter contains 35% carbohydrates, 45% fibre, 1% fat, 12% crude proteins and 21 mg/100 g ascorbic acid [11].
Leaves of Azanza garckeana are used for making relish and can be burned to produce salt [5] [12]. The tips of the leaves when matured are used to improve land productivity/increasing fertility. The leaves are also eaten as vegetable and could also be used as manure or mulch [1] [6] [9]. The tree provides fodder to animals. The timber is a source of firewood. The roots are eaten orally for pain relief to treat cough and chest pains. Several studies, have been carried out in various aspects of the plant, but so far no macro-morphological studies have been carried out, so the need for this studies. The present work, which forms part of the investigation of epidermal studies, is an attempt to fill in the gap in our knowledge and to provide a baseline information for understanding the Taxonomic characters of the plant.
2. Materials and Methods
2.1. Plant Collection and Microscopic Examination for Epidermal Study
Fresh matured leaves of Azanza garckeana were obtained from three different sites in Tula: Tula Wange, Tula Baule and Tula Yiri/Bwaile. The specimens wereidentified in the University Herbarium, Department of Biological Sciences, Gombe State Universiy, Gombe, Nigeria. Leave samples were then fixed in Formalin Acetic Acid (FAA) and preserved in 70% ethanol. Epidermal peels of the surfaces of the plant were made using the method of [13]. The adaxial and abaxial surfaces of the leaves were carefully sectioned from the median portion of the leaves with razor blade (free hand section) and placed on a microscope clean glass slide. The preparation were stained with 1% aqueous solution of saffranin for 4 to 8 minutes, and rinsed carefully in water to remove excess stain and then mounted in 10% glycerol and observed under a light microscope and the leaf epidermal features were then examined using 35 fields of view at × 40 objective as quadrats. The numbers of subsidiary cells per stomata was noted to determine the frequency of the different stomatal complex types and was expressed as percentage occurrence of such complex types based on all occurrences [14]. Terminologies for naming stomatal complex types followed [15]. The Stomatal size/guard cells area of a species was determined as the product of length and breadth. The mean stomatal sizes/ guard cell area were determined by Francos constant method (Stomatal size/Guard cells area = length × breadth (width) × 0.7854) of guard cells using an ocular eye-piece micrometer and finally converted by the ocular constant with respect to the power with which they were taken. Samples of 35 stomata were used. The method followed those of [16] [17]. The stomatal densities were determined by counting the number of stomata permicrometer view quadrat. Stomatal Index was determined as number of stomata per square millimeter divided by number of stomata plus number of epidermal cells per square millimeter multiplied by 100 [15]. The stomata observed were viewed with the light microscope and were calculated in unit area using the stomatal Index (S.I) formulae as determined according to [18] as shown below.
where S.I = Stomatal Index
S = Number of stomata per unit area
E = Number epidermal cells in the same unit area.
2.2. Statistical Analysis
All data for epidermal study were processed using Analysis of Variance (ANOVA), Duncan’s Multiple Range Test (DMRT) was used. A probability value of 0.05% was used as bench mark for significant difference between parameters.
3. Results and Discussions
Leaf epidermis of Azanza accessions were studied anatomically (Plates 2-7). Focus was on such leaf micromorphological characters such as stomata types, epidermal cell types and anticlinal cell wall patterns and presence of trichomes. The three accessions studied had epiamphistomatic leaves i.e. (leaves having more stomata on the adaxial surface) this was similar to the findings of [13] in his study of Capsicum annuum. Stomatal complex type (SCT) was exclusively anomocytic within all the accessions. The homogeneous nature of the SCT on the leaves was responsible for its 100% frequency occurrence. The 100% frequency of anomocytic stomata in Azanza garckeana is noteworthy because specific stomatal types can be used to distinguish species: This also suggests that the stomatal system of Azanza garckeana in having no subsidiary cells coupled with epiamphistomatic feature is likely to be water-conserving. This view was corroborated by [14] in their study of some Citrus species, in which species with few subsidiary cells are more water―conserving than those with many subsidiary cells. A similar finding was reported by [19] [20] in their study of Talinumtriangulare in stomatal complex types, stomatal size, density and index of some vegetable species in Nigeria and in some Six species of Anacardiaceae and Lannea species. [21] also reported that large number of subsidiary cells per stoma may be responsible for a more precise and rapid regulation of stomatal opening. Duncan Multiple Range Test (DMRT)―one-way ANOVA revealed that there was no significant difference on the abaxial surface across all the parameters at all accessions, but significant difference exists (P ≤ 0.05) on some densities on the adaxial surface across all the parameters (Table 1). DMRT of the Stomatal sizes in the 3 accessions were shown in (Table 1). However, the stomatal size was highest (45.49 ± 3.73 µm) on the abaxial surface of accession 1 and lowest value (24.98 ± 2.27a µm) on the adaxial surface of accession III). In this study stomatal sizes were small on the adaxial and large on the abaxial surfaces. Stomatal
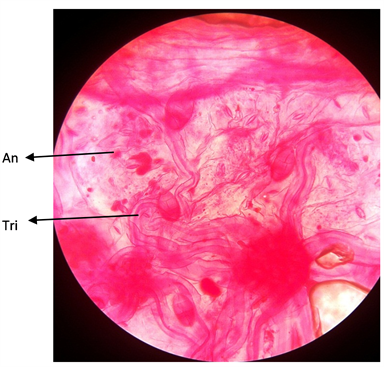
Plate 2. Adaxial surface of A. garckeana showing anomocytic stomata ×400.
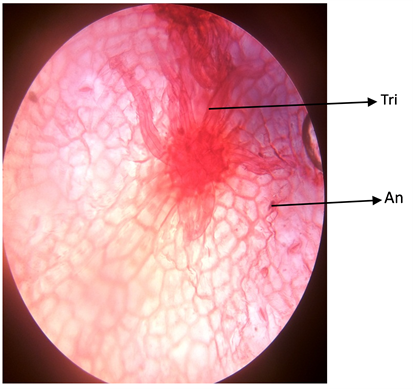
Plate 3. Abaxial surface of A. garckeana showing anomocytic stomata ×400.
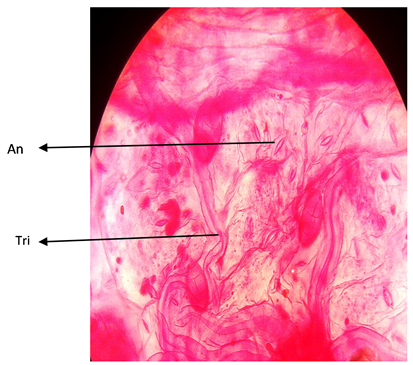
Plate 4. Adaxial surface of A. garckeana showing anomocytic stomata ×400. KEY; Tri = Trichomes.
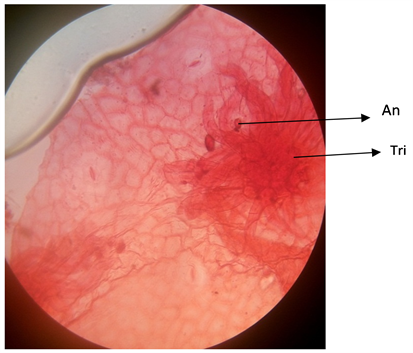
Plate 5. Abaxial surface of A. garckeana showing anomocytic stomata ×400. An = Anomocytic stomata.
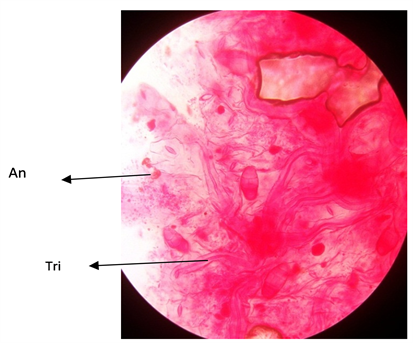
Plate 6. Adaxial surface of A. garckeana showing anomocytic stomata ×400.
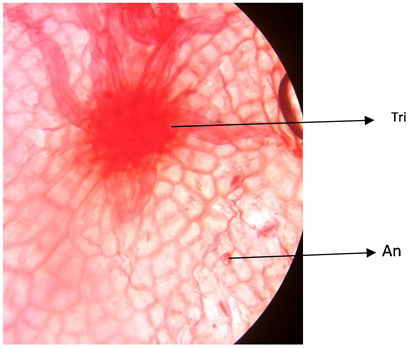
Plate 7. Abaxial surface of A. garckeana showing anomocytic stomata ×400.
Table 1. Stomatal features in three accessions of Azanza garckeana in tula.
Means followed by the same letter in a column are not significantly different at 5% probability level using Duncan Multiple Range Test Key to Accession 1 = Tula Wange; II = Tula Baule; III = Tula Bwale/Yiri.
size is often correlated with stomatal density such that small stomata give
high density and large stomata give low density [22] and [23]. Stomatal size (guard cell area and stomatal index) also provides values that would serve as parameters for comparison among taxa which can also be useful for identification of the studied taxa. [24]. This study was not in line with the findings from the above workers. It is however in correlation with the works of [13] , where he observed an exception in accession Adaw-ding where stomatal size was larger but with higher stomatal density. For this study, the stomatal index was significantly higher at accession I (7.42 ± 2.30a) on the adaxial surface and lowest value (2.45 ± 0.24) on the abaxial surface of accession II. This shows that stomata occupied larger proportion of the leaf surface in the adaxial surface and smaller proportion on the abaxial surface (Table 1). The variations in stomatal index observed in this study can be reasonably employed in delimiting Azanza species (Table 1). This is consistent with the works of [25] [26] who reported that the stomatal index indicates the proportion of stomata relative to leaf surface and is also a reliable taxonomic character. It is highly constant for any given species and the value is more uniform upon the abaxial than the adaxial surface except in isobilateral leaves. [23] also reported that stomatal index is independent of the changes in epidermal cells size brought about by environmental factors. The role of stomatal index in systematic work to separate species has also been reported by [19] [27] [28]. The stomatal density was highest (23.67 ± 4.04a) on the adaxial surface of Azanza garckeana in accession 1 and lowest value (13.33 ± 2.08b) on the adaxial surface of accession II. [29] reported that stomatal density may depend on the environmental factor.
The differences in stomatal density and stomatal index of a species are often a reflection of physiological responses to combination of environmental factors which could be useful in delimitation at the species level.
Contrary to their observation, since Stomatal Index, Stomatal density, and Stomatal Size on both surfaces may be inherent characters as the plant was subjected to the same treatment. This may have adaptive implication for water conservation in the plant since the leaf surfaces are responsible for loss of water through the stomata [13]. Most of these features agreed with the accounts of the previous workers [30] [31]. Non-glandular dendritic type of trichomes was present on both leaf surfaces. This is of great diagnostic interest. The multicellular hairs in them serve to reduce the rate of transpiration in the plant. The importance of trichomes in taxonomy has been highlighted by [32] [33]. [34] suggested that the primary function of stinging trichomes might be the regulation between plant and the environment. This is consistent with the works of [18] who noted trichomes as good diagnostic tool in taxa delimitation. No variability existed within the epidermal cell shape pattern within all the accessions. They were all irregular in shape, with curved anticlinal cell wall pattern (Table 2). According to [29] , epidermal cell characteristics are under strong genetic control, hence are stable traits and therefore proofed to be a better tool for the delimitation of Azanza than stomatal traits. These findings suggest that to a certain extent and with verification from other taxonomic characters, and epidermal cell density can make contribution in delimiting this species.
Table 2. Epidermal Features in Accession of Azanza garckeana in Three Different Sites, in Tula.
Means followed by the same letter in a column are not significantly different at 5% probability level using multiple Range Test. Key to Accession: 1 = Tula Wange; II = Tula Baule; III = Tula Bwale/Yiri.
4. Conclusion
The epidermal features such as 100% frequency, small stomatal sizes on the adaxial and large stomatal sizes on the abaxial leaf surfaces, anomocytic stomatal types, dendritic type of trichomes, variability in stomatal index, curved anticlinal and irregular shapes of the epidermal cell wall pattern, within the accessions all contributed to the delimitation of Azanza garkaena. However, more taxonomic tools are required for the delimitation of the accessions into distinct taxa.
Cite this paper
Abba, H.M., Daniel, H., Sale, S. and Zhigila, D.A. (2018) Taxonomic Significance of Foliar Epidermal Characters in Azanza garckaena (F. Hoffm.) Exell & Hillc in Tula, Kaltungo Local Government Area of Gombe State, Nigeria. American Journal of Plant Sciences, 9, 1677-1687. https://doi.org/10.4236/ajps.2018.98121
References
- 1. International Centre for Research in Agro Forestry (I.C.R.A.F.) (1992) A Selection of Useful Trees and Shrubs for Kenya. Notes on Their Identification. Propagation and Management for Use by Farming and Pastoral Communities, Nairobi.
- 2. Palmer, E. and Pitman, N. (1972) Trees of Southern Africa, Covering All Known Indigenous Species in the Republic of South Africa, South-West Africa, Botswana, Lesotho and Swaziland, 3 Volumes. Balkema, Cape Town, 2235.
- 3. Michael, K.G., Onyia, L.U. and Jidauna, S.B. (2015) Evaluation of Phytochemicals in Azanzagarckeana (Gorontula) Seed. Journal of Agriculture and Veterinary Science, 8, 71-74.
- 4. Mulofwa, J., Simute, S. and Tengas, B. (1994) Agroforestry Manual for Extension Workers in the Southern Province, Zambia. Technical Handbook No. 4. Regional Soil Conservation Unit/SIDA, Nairobi.
- 5. Storrs, A.E.G. (1995) Know Your Trees: Some Common Trees Found in Zambia. Regional Soil Conservation Unit (RSCU), Nairobi.
- 6. Palgrave, K.C. (1988) Trees of Southern Africa. Struik Publishers, Cape Town.
- 7. Hutchinson, J. and Dalzeil, J.M. (1972). Flora of West Tropical Africa. 2nd Rev. Edition. Vol 1-III. Millbank, London.
- 8. White, F. (1962) Flora of Northern Rhodesia. Oxford University Press, London.
- 9. F.A.O. (1983) Food and Seed Bearing Forest species. Examples from East Africa. Forestry Paper, FAO, Rome.
- 10. Taylor, F. and Kwerepe, B. (1995) Towards Demonstration of Indigenous Fruit Trees in Botswana. In: Maghebe, J.A., Ntulpanyama, Y. and Chirwa, P.W., Eds., Improvements of Indigenous of Miambo Wood Land of South Africa, ICRAF, Nairobi.
- 11. Mshelia, B.H., Watirahyel, E.M., Maigari, A.U., Christopher, Y. and Ishamail, F. (2016) Cytoxicity and Antioxicidant Activity of Stem Bark Extract of Azanza garckeana. European Journal of Pure and Applied Chemistry, 3, 16-24.
- 12. Mateke, S.M., Kamara, C.S. and Chikasa, P. (1995) Ripening Periods of Edible Indigenous Fruits of Zambia: Implications of Utilisation and Domestication. In: Magehembe, J.A., Ntupanyama, Y., and Chirwa, P.W., Eds., Improvement of Indigenous Fruit Trees in the Miombo Woodlands of Southern Africa, ICRAF, Nairobi.
- 13. Zhigila, D.A., Sawa, F.B.J., Aluko, T.A., Oladele, F.A. and Abdul Rahaman, A.A. (2015) Leaf Epidermal Anatomy in Five Varieties of Capsicum annuum L. Solanaceae. America Journal of Experimental Agriculture, 5, 392-399. https://doi.org/10.9734/AJEA/2015/12825
- 14. Obiremi, E.O. and Oladele, F.A. (2001) Water Conserving Stomatal System in Selected Citrus Species. South African Journal of Botany, 67, 258-260. https://doi.org/10.1016/S0254-6299(15)31127-3
- 15. Dilcher, D.L. (1974) Approaches to the Identification of Angiosperm Remains. Botanical Review, 40, 1-157. https://doi.org/10.1007/BF02860067
- 16. Franco, C. (1939) Relation between Chromosome Number and Stomata in Coffea. Botanical Gazette, 100, 817-827. https://doi.org/10.1086/334832
- 17. Wilkinson, H.P. (1979) The Plant Surface (Mainly Leaf), Part 1: Stomata. In: Metcalfe, C.F. and Chalk, L., Eds., Anatomy of Dicotyledons, Vol. 1, 2nd Edition, Clarendon Press, Oxford, Chapter 10, 113.
- 18. Metcalfe, C.R. and Chalk, L. (1950) Anatomy of Dicotyledon. Clarendon Press, Oxford, Vol. 11, 421-440.
- 19. Abdulrahaman, A.A. and Oladele, F.A. (2003) Stomatal Complex Types, Stomatal Size, Density and Index in Some Vegetable Species in Nigeria. Nigerian Journal of Botany, 16, 144-150.
- 20. Abdulrahaman, A.A., Kolawole, O.S. and Oladele, F.A. (2014) Leaf Epidermal Features as Taxonomic Characters in Some Lannea Species (Anacardiaceae) from Nigeria. Phytologia Balcanica, 20, 227-231.
- 21. Carr, S.G. and Carr, D.J. (1990) Cuticular Features of the Central Australia Bloodwoods Eucalyptus Section Corymbosae (Myrtaceae). Botanical Journal of the Linnean Society, 102, 123-156. https://doi.org/10.1111/j.1095-8339.1990.tb01872.x
- 22. Mbagwu, F.N., Nwachukwu, C.U. and Okoro, O.O. (2007) Comparative Leaf Epidermal Studies on S. macrocarpum and S. nigrum. Research Journal of Botany, 3, 45-48.
- 23. Abdul Rahaman, A.A. and Oladele, F.A. (2004) Types, Densities and Frequencies of Trichomes in Some Nigerian Vegetable Species. Nigerian Journal of Pure and Applied Science, 19, 1653-1658.
- 24. Essiet, U.A., Edet, N.I. and Illoh, H.C. (2011) Leaf Epidermal Studies of Two Species of Laportea in Southern Nigeria. International Journal of Botany, 24, 245-255.
- 25. Metcalfe, C.R. and Chalk, L. (1998) Anatomy of Dicotyledon. 2nd Edition, Oxford University Press, Oxford, 97-117.
- 26. Adedeji, O. and Jewoola, O.A. (2008) Importance of Leaf Epidermal Characters in the Asteraceae Family. Notulae Botanicae Horti Agrobotanici Cluj-Napoca, 36, 7-16.
- 27. Isawumi, M.A. (1989) Epidermal Studies in the Species of Jatropha L. (Euphorbiaceae) Found in Nigeria. Nigerian Journal of Botany, 23, 94-100.
- 28. Aworinde, D.O., Nwoye, D.U., Jayeola, A.A., Olagoke, A.O. and Ogundele, A.A. (2009) Taxonomic Significance of Foliar Epidermis in Some Members of Euphorbiaceae Family in Nigeria. Research Journal of Botany, 4, 17-28. https://doi.org/10.3923/rjb.2009.17.28
- 29. Alege, G.O., Mustapha, O.T., Ojo, S. and Awosemo, B.M. (2013) The Morphological, Proximate and Mineral Responses of Sesame to Different Nutrient Sources. Global Journal of Bio-Science and Biotechnology, 2, 12-16.
- 30. Inamdar, J.A. and Gangadhara, M. (1977) Studies on the Trichomes of Some Euphorbiaceae. Feddes Repertorium, 88, 103-111. https://doi.org/10.1002/fedr.19770880105
- 31. Raju, V.S. and Rao, P.N. (1972) Variation in Structures and Foliar Stomata in Euphorbiaceace. Botanical Journal of the Linnean Society, 94, 84-90.
- 32. Stace, C.A. (1980) Plant Taxonomy and Biosystematics. Edward Arnold Limited, London, 74-83.
- 33. Illoh, H.C. and Inyang, U.E. (1998) Foliar Epidermis and Petiole Anatomy in Some Nigeria Solanum Linn. Species in the Sub-Genus Leptostemonum (Bitt). Dun. Glimpses in Plant Research, 12, 73-86.
- 34. Corsi, G. and Bottega, S. (1999) Glandular Hairs of Salvia Officinalis: New Data on Morphology, Localization and Histochemistry in Relation to Function. Annals of Botany, 84, 657-664. https://doi.org/10.1006/anbo.1999.0961


