American Journal of Plant Sciences
Vol.5 No.4(2014), Article ID:43259,7 pages DOI:10.4236/ajps.2014.54061
Selected Inorganic Nutrients in Black Tea from Three Tea Growing Agro-Ecological Areas in Kenya
![]()
1Biochemistry Department, Jomo Kenyatta University of Agriculture and Technology, Nairobi, Kenya; 2Tea Processing and Value Addition Programme, Tea Research Foundation of Kenya, Kericho, Kenya; 3Research, Production and Extension Department, Jomo Kenyatta University of Agriculture and Technology, Nairobi, Kenya.
Email: *kmoseti@tearesearch.or.ke, *kelvinmoseti@gmail.com
Copyright © 2014 Mangenya Thaddeus Mose et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accordance of the Creative Commons Attribution License all Copyrights © 2014 are reserved for SCIRP and the owner of the intellectual property Mangenya Thaddeus Mose et al. All Copyright © 2014 are guarded by law and by SCIRP as a guardian.
Received November 19th, 2013; revised January 7th, 2014; accepted January 19th, 2014
KEYWORDS
Black Tea; Inorganic Nutrients; Spectrophotometry; Kenya
ABSTRACT
The tea plant absorbs dissolved nutrients from soils for its normal growth and development, though to different extents. Nutrients play vital roles in various metabolic processes, their deficiency or excess being deleterious to living organisms. A study was carried out to quantitatively assess the inorganic nutrient content (K, P, Ca, Mn, Fe, Zn and Cu) of twelve black tea samples sourced from Murang’a, Meru and Kisii tea growing agro-ecological areas in Kenya. K and P were quantified using a flame photometer and a UV-Vis spectrophotometer respectively whereas Ca, Mn, Fe, Zn and Cu were quantified using an Atomic Absorption Spectrophotometer (AAS). The general accumulation pattern of the inorganic nutrients in the tea samples was established to be; K (1.6% ± 0.05% - 2.1% ± 0.01%) > P (0.30% ± 0.01% - 0.37% ± 0.04%) > Ca (0.16% ± 0.01% - 0.62% ± 0.03%) > Mn (0.07% ± 0.009% - 0.13% ± 0.004%) > Fe (136 ± 8 - 320 ± 5 µg/g) > Zn (27 ± 1 - 39 ± 7 µg/g) > Cu (10 ± 3 - 16 ± 1 µg/g). Statistically significant differences (p < 0.05) were observed in the inorganic nutrient contents of the black tea from the different tea factories as well as agro-ecological areas. These data demonstrate the tea plant’s ability to accumulate the studied nutrients, further underlining tea consumption as a potential dietary source of the nutritionally essential inorganic nutrients.
1. Introduction
Tea is the most popular non-alcoholic beverage world over [1-3] and is an infusion of processed young shoots of the tea plant (Camellia sinensis), that belongs to the plant family Theaceae [4]. It consists of three distinct varieties, China, Assam and Cambod, mainly distinguished by the nature of the leaf [2]. There are several types of tea products based on the method of manufacture of the tea leaves and consequently, their chemical composition [5]. The main types are green, oolong and black teas, where the nature and quality of a given tea product is mainly dependent on the chemical composition of the young tea shoots and the reactions they undergo during the manufacture process. The methods of manufacture of these tea products may be orthodox or non-orthodox and vary considerably with regard to their impact on the formative and degradative patterns of the various cellular components [6].
Kenya has varied climatical and geographical regions [7], with tea being grown on the foothills of the Aberdare ranges and Mount Kenya in the East of the Rift Valley and the Mau Ranges, Nandi, Kisii and Kakamega hills in the West of the Rift Valley [8]. The Kenyan tea industry is a rural based enterprise and specializes in the production of Cut, Tear and Curl (CTC) black tea [9], her leading foreign exchange earner and export commodity [10].
Several health benefits have been ascribed to the regular consumption of tea and include; serum cholesterol reduction, decreased risk of cancer and cardiovascular diseases [11], prevention of a number of diseases, including skin cancer [12], Parkinson’s disease [13], myocardial infarction [14], and coronary artery disease [15]. These health benefits have been attributed to the strong antioxidant activity of catechins that protect the body against free radical-induced oxidative stress [16]. Additionally, a biologically active group of tea components called polyphenols have been associated with amelioration of inflammation [17], inhibition of diabetes [18], prevention of intestinal damage and anti-diarrhoea properties [19], enhancement of oral health [20] and the potential to improve spatial cognitive learning ability [21]. Other compounds in tea that are beneficial to human health include methylxanthines such as caffeine, theophylline and theobromine and a number of inorganic nutrients, among which are calcium (Ca), potassium (K), iron (Fe), copper (Cu) and zinc (Zn).
The human body requires some inorganic nutrients (elements), within certain permissible concentrations for healthy growth and development [22] as well as proper functioning of the body [23]. The optimum values required vary widely from element to element, age, sex among other factors. Recent research findings have indicated that the content of essential elements in plants is conditional, mainly being affected by edaphic characteristics and the ability of plants to selectively accumulate some elements [24].
In this study, an attempt was made to quantify selected inorganic nutrients in Kenyan black tea to show how the inorganic nutrient content of teas varies with agro-ecological areas. Since a portion of these nutrients ends up in the tea liquor during the tea making process [25], such information (regional inorganic nutrient profile of teas) may be employed as a tool to market Kenyan tea.
2. Materials and Methods
2.1. Study Area Description
The study areas were Murang’a, Meru and Kisii tea growing agro-ecological areas in Kenya, a leading tea producer globally. The Murang’a and Meru areas lie to the East of the Great Rift Valley in altitudes between 2150 to 2300 m above mean sea level (amsl) on the windward and leeward sides of the snow-capped Mt. Kenya respectively as indicated in Figure 1. Rainfall in these areas is mainly relief and during the cold spell, usually between July and September, temperatures in these areas at times fall to 4˚C, corresponding to suppressed growth of tea shoots. Based on their location on the mountain, these two areas are regarded as different agro-ecologically.
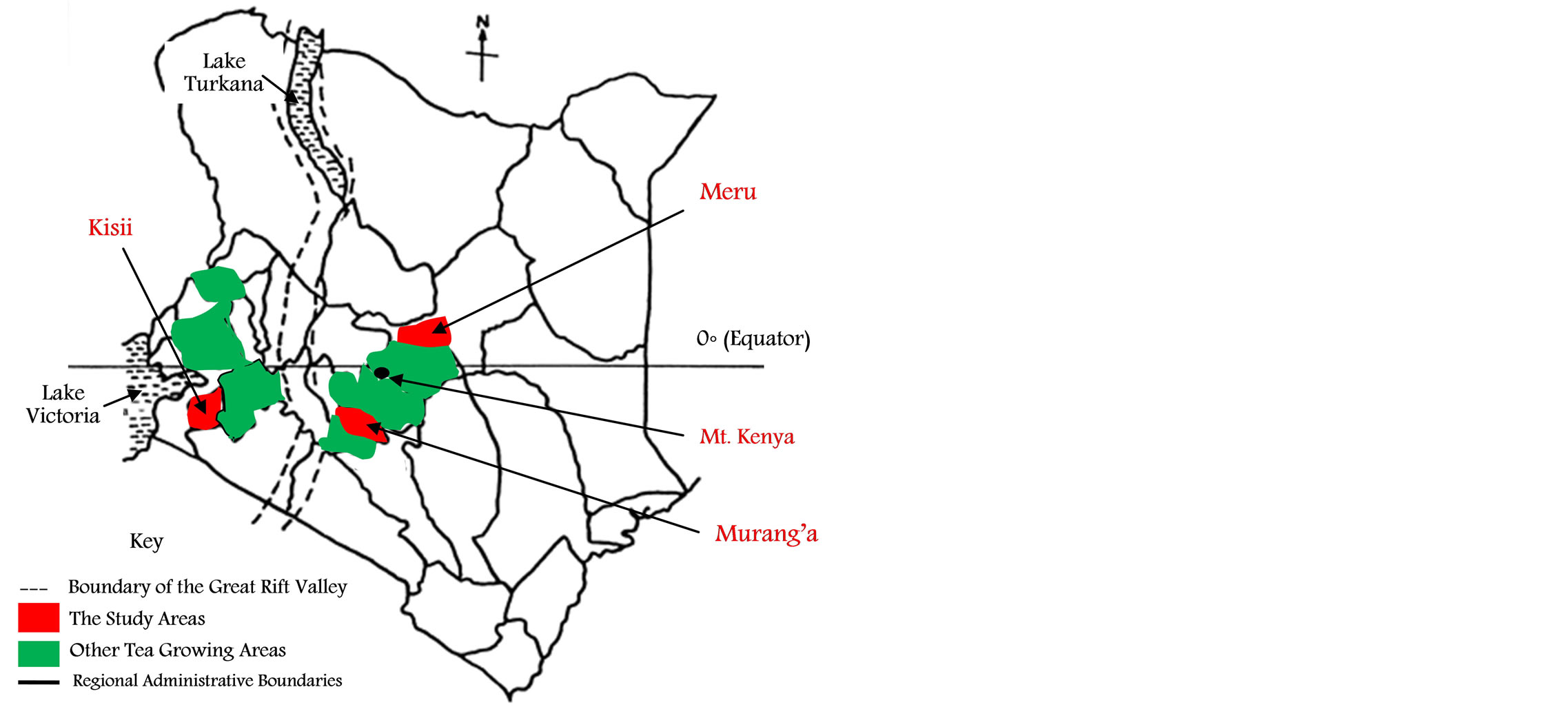
Figure 1. A map of Kenya showing the study areas.
On the other hand, the Kisii area lies to the West of the Rift Valley (Figure 1) and is lower in altitude than the Murang’a and Meru areas, with tea being cultivated at altitudes between 2100 and 2150 m amsl. Ambient temperatures in this area average between 20˚C and 25˚C whereas rainfall is mainly the convectional that precipitates as a result of the influence of Lake Victoria, and is well distributed all year, resulting in evenly distributed tea production with only relatively minor peak periods between April and May and October and November. Further, the warmer temperatures and evenly distributed rainfall correspond to faster growth rate of tea shoots unlike that in the Murang’a and Meru areas.
The Kenyan tea industry is characterized by two sub-sectors; the large-scale sub-sector (estate plantations) with production units larger than 20 ha and the smallscale sub-sector with smaller production units averaging 0.25 ha per farmer [26]. All the selected factories as indicated in Table 1 belong to the small-scale tea subsector, with each factory being served by hundreds of tea farmers within its catchment. Besides practicing different agronomic practices such fertilizer application rates, plucking standards and intensity, the farmers grow different clones of tea, with varying yield and quality characteristics. It is therefore expected that there will be variations in chemical composition of teas grown in these areas owing to the highlighted differences in environmental conditions.
2.2. Sample Collection and Pre-Treatment
Four (4) factories (Table 1) were randomly selected from each of the three agro-ecological areas described. A black tea sample was collected from each of the twelve (12) factories in triplicates, each replicate being picked from a different drier so as to obtain a representative

Table 1. Selected factories from the Murang’a, Meru and Kisii agro-ecological areas.
sample for the factory catchment area. The tea samples, were placed in clean, well labeled paper bags, sealed and transported to the Tea Research Foundation of Kenya (TRFK) laboratories at Kericho, Timbilil estate (latitude 0˚22'S, longitude 35˚21'E, altitude 2180 m amsl) where they were oven-dried at 80˚C (Memmert, 854 Schwabach, Germany) until a constant weight was attained. Once dry, the teas were ground into a fine powder using an electric blending device (Moulinex AR1043, China) with stainless steel blades to reduce the particle size and homogenize the sample.
2.3. Glassware and Reagents
All glassware were soaked in 50% HNO3 (Sigma Aldrich, UK) for 24 hours, thoroughly cleaned and subsequently rinsed with distilled water and oven dried before use. Distilled water was used for sample preparation and all the dilutions of both the samples and calibration solutions. All reagents and calibration standards used in this study were of analytical grade procured from Sigma Aldrich, UK.
2.4. Sample Preparation and Analytical Measurements
Fe, Zn and Cu contents of the tea samples were determined using Flame Atomic Absorption Spectrophotometry (FAAS), following the method described by [27].
To quantify K, P, Ca and Mn in the teas, 0.25g of each of the finely ground oven-dried tea samples were accurately weighed using an analytical balance (BL-3200 HL, Shimadzu, Japan) into separate, clean and dry specimen tubes and ashed in a muffle furnace (Gallenkamp, England) at 460˚C for 4.5 hours. The ashed samples obtained were allowed to cool followed by wet digestion using two parts of a 1:1 mixture of concentrated HNO3 (69% - 70.5%) and concentrated HCl (37%) to three parts of H2O2 under reflux to near dryness [28]. Each sample residue obtained was then dissolved in 25 ml of 0.05 M HCl solution. The clear solution was shaken thoroughly and allowed to stand for 4 hours prior to analysis.
Ca and Mn contents of the digests were then quantified using an Atomic Absorption Spectrophotometer (Varian, SPPA-880, EL98083516). P was quantified colorimetrically using a UV-Vis Spectrophotometer (UV-1800, Shimadzu, ENG240V, SOFT) fitted with a Shimadzu ASC-5 auto sample changer at 400 nm after complexing with a mixture of ammonium molybdite and ammonium metavanadate whereas K was quantified by flame emission using a flame photometer (Corning, 400, 2604) [29,30].
For quality assurance, two reference tea samples (A and B) were ashed, digested and analysed for all the inorganic nutrients of interest alongside every batch of samples daily throughout the analysis. Reagent and sample blanks treated in a similar way as the samples were also included. Moreover, to check the efficiency of the analytical methods, the reference samples A and B were spiked with known concentrations of the respective analytes and recovery tests performed.
2.5. Data Analysis
Data obtained were expressed as percentages (%) or parts per million (µg/g), as appropriate on a dry weight basis and subjected to analysis of variance (ANOVA) using MSTAT C [31], with the probability limit set at p ≤ 0.05. The effects of agro-ecological areas and factories of origin on the inorganic nutrient content of the black teas were analyzed using a two factor completely randomized design. Where significant treatment effects were observed, separation of means was performed using the least significant difference (LSD) test. The results were tabulated as a mean ± the standard deviation (Mean ± SD) of the triplicate determinations whereas graphical representations were done using Graph Pad Prism version 5.0.
3. Results and Discussion
3.1. Quality Assurance
Triplicate analyses of all the analytes in all the samples showed that the coefficients of variation (CV) of the results were within ±10% except for Cu whose results gave a CV of 17.5% as indicated in Table 2. Reagent blank analyses indicated no contamination or interferences from the reagents used and the calibration curves obtained for all the elements demonstrated adequate linearity (r2 > 0.999).
Being a fundamental element of method validation, the detection limit for each analyte was also estimated. Limit of detection (LOD) is the smallest mass of analyte that can be distinguished from the statistical fluctuations in a blank under specified test conditions [32]. LODs of the current method calculated as the concentration corresponding to a signal three (3) times of the blank analyses were 0.015, 0.031, 0.060, 0.025, 0.074, 0.013 and 0.043 for K, P, Ca, Mn, Fe, Zn and Cu respectively. The average percentage recovery for the two tea reference sam-

Table 2. K, P, Ca, Mn, Fe, Zn and Cu in black tea from Murang’a, Meru and Kisii agro-ecological areas.
ples (A and B) for K, P, Ca, Mn, Fe, Zn and Cu were 98% ± 2.3%, 98% ± 3.4%, 97% ± 2.8%, 96% ± 1.9%, 95% ± 2.6%, 98% ± 2.3%, and 92% ± 5.7% respectively demonstrating good accuracy of the data obtained.
3.2. Black Tea Inorganic Nutrient Content
Recent findings [33] revealed that tea leaf nutrients vary with clones and locations. The results presented herein (Table 2) for the inorganic nutrients studied (K, P, Ca, Mn, Fe, Zn and Cu) agree with this observation since variations with factory of production as well as the agro-ecological area were evident.
For each inorganic nutrient, the concentration ranges observed varied, demonstrating variations in the factors influencing their distribution in the different agro-ecological areas as well as the factories of origin [34] and their uptake and accumulation by the tea plant. For instance, the Fe content ranged between 136 ± 8 and 320 ± 5 µg/g whereas the Ca content ranged from 0.16% ± 0.01% to 0.62% ± 0.03%.
The differences in the levels of each inorganic nutrient in black tea from the 12 factories were statistically significant (p < 0.05) as can be seen in Table 2. This observation is in corroboration with [35] and [27,34], whose recent findings have demonstrated variations in the element content of black tea from different locations in India and different factories in Kenya respectively. Further, comparisons of the mean inorganic nutrient contents in black tea from the three tea growing agro-ecological areas reveal statistically significant differences (p < 0.05) in all the inorganic nutrients studied except for Zn and P as indicated in Table 2. Consequently, the highlighted differences result in significant (p < 0.05) interaction effects between the agro-ecological areas and factories for all the inorganic nutrients studied (Table 2).
Based on the current data, the inorganic nutrients studied can be arranged in descending order as K > P > Ca > Mn > Fe > Zn > Cu. K and Cu were the most and least abundant inorganic nutrients in the black teas with concentrations obtained being in the ranges 1.6% ± 0.05% - 2.1% ± 0.01% and 10 ± 3 - 16 ± 1 µg/g respectively (Table 2). Moreover, these data demonstrates the different extents to which different elements are accumulated in the tea shoots of the tea plant. K, P, Ca and Mn were present in the percent levels (Figure 2) whereas Fe, Zn and Cu were present in the parts per million levels (Figure 3) on a dry weight basis.
The Fe content obtained for the sample from the fourth factory from the Kisii agro-ecological area, Eberege (320 ± 5 µg/g) was significantly high compared to the obtained mean Fe contents of 202 and 180 µg/g obtained for black tea from the Kisii agro-ecological area and all the 12 tea processing factories under study respectively. In the same factory, a similar observation was made for
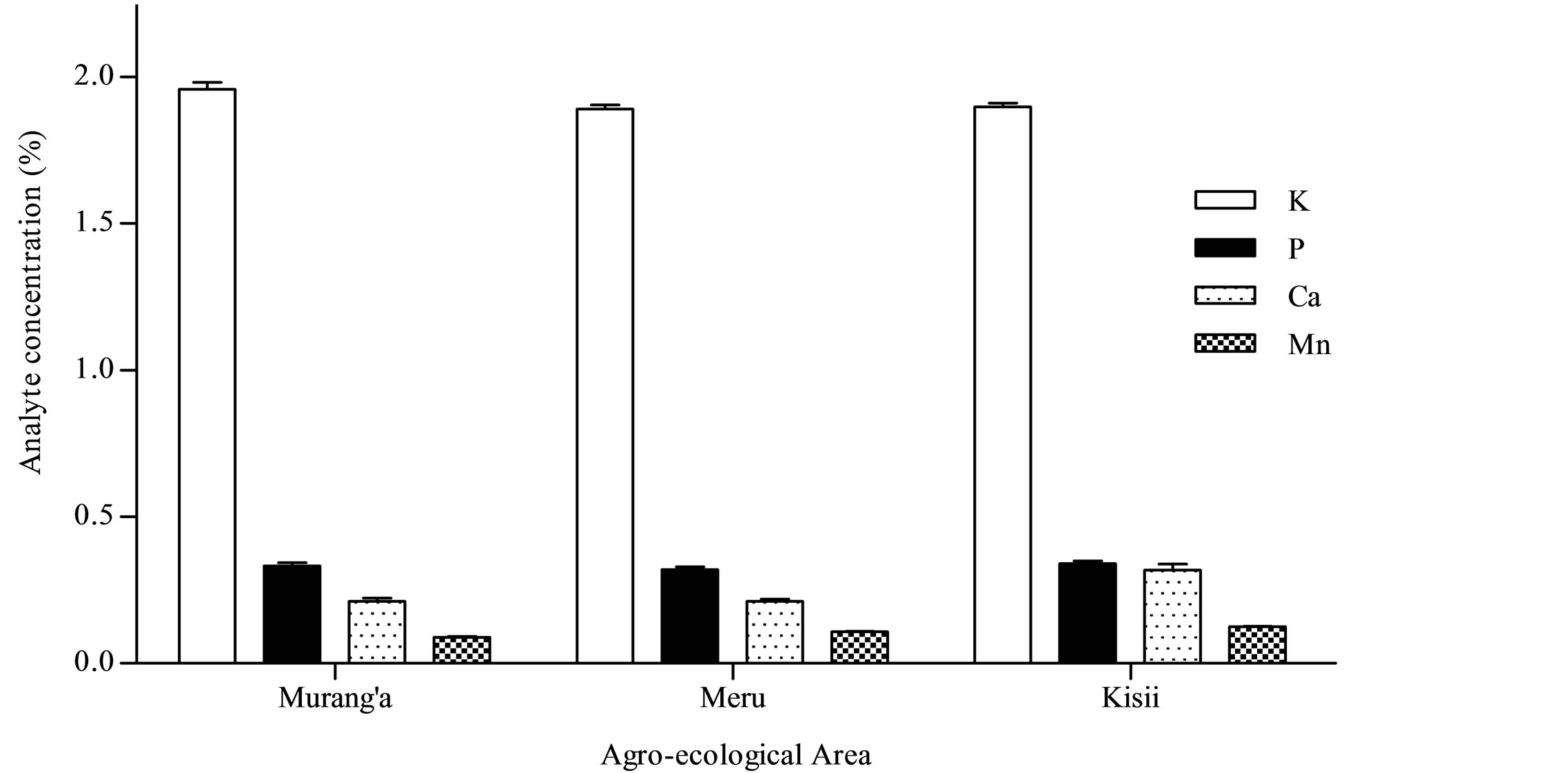
Figure 2. K, P, Ca and Mn in black tea from Murang’a, Meru and Kisii agro-ecological areas.
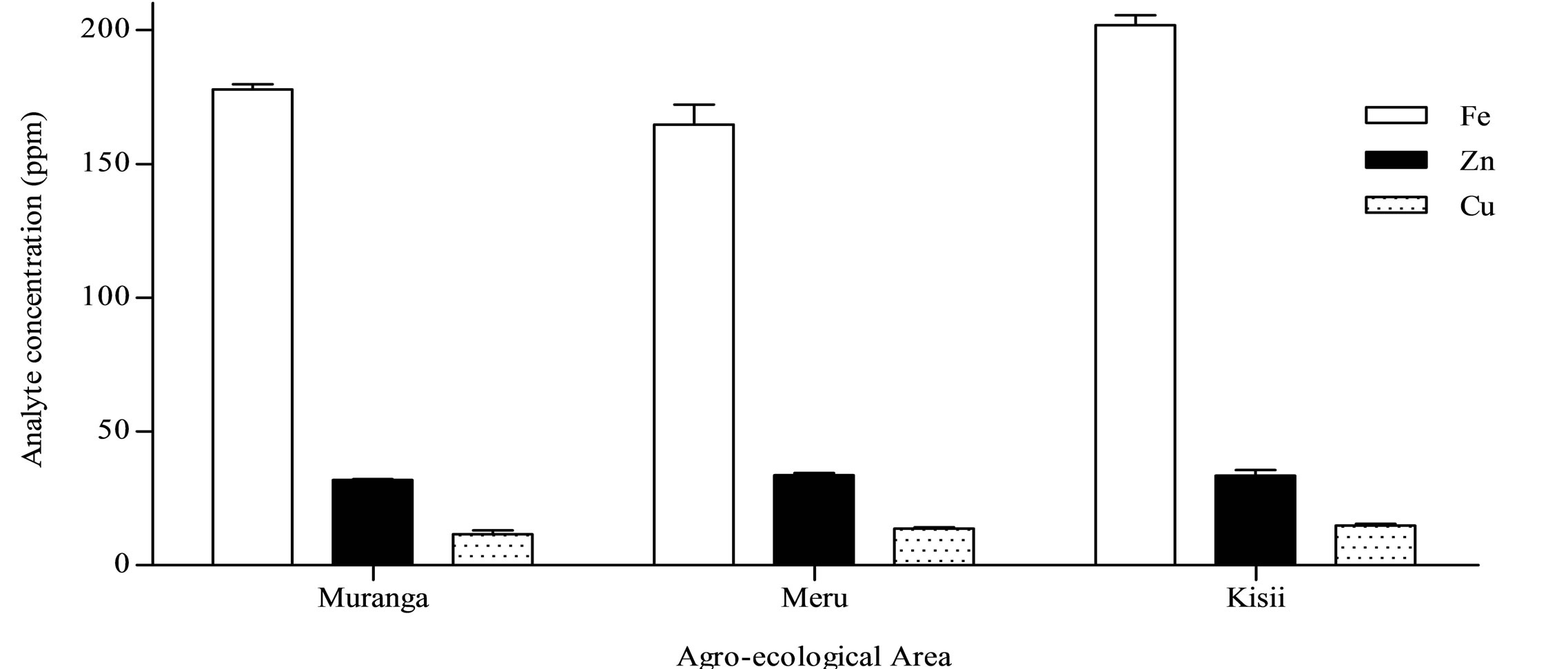
Figure 3. Fe, Zn and Cu in black tea from Murang’a, Meru and Kisii agro-ecological area.
Ca, where the level obtained (0.62 ± 0.03, Table 2) was significantly high compared than the mean Ca contents for the agro-ecological area (0.32 µg/g) and the grand mean for all the teas from all regions (0.25 µg/g).
High levels of Fe in tea leaves have been associated with mature leaf [36], implying that the high Fe content in black tea from Eberege factory may be attributed to coarse plucking standards and long plucking intervals among other factors hence the need for strict adherence to Good Agricultural Practices (GAP). Further, [27] observed the introduction of additional Fe along the CTC black tea processing chain. This is possibly due to wear and tear of the CTC segments during the rolling stage in tea manufacture [Personal Observation]. This observation further underlines the need for strict adherence to Good Manufacturing Practices (GMP) in the tea industry.
4. Conclusion
This study demonstrates the ability of the tea plant to absorb and accumulate the studied elements, though in varying proportions. It is evident that teas produced in different regions and processed in different factories have varying inorganic nutrient contents. This implies that tea consumers may potentially gain by being furnished with a number of inorganic nutrients necessary for various biological processes. However, the potential health benefits attributable to such nutrients will be due to the composition of the tea liquor consumed rather than the total element content in the black tea and as such dependent on the extent of extraction (“brew factor”) of the elements during the tea making process. To have a clear idea of the potential beneficial effects of tea, it is necessary to evaluate the “brew factor” of the elements of interest, an aspect we shall consider in our future investigations.
Acknowledgements
This study was supported by the National Council for Science and Technology (NCST), Kenya. This paper is published with permission of the director, Tea Research Foundation of Kenya (TRFK).
REFERENCES
- J. Ruan and M. H. Wong, “Accumulation of Fluoride and Aluminium Related to Different Varieties of Tea Plant,” Environmental Geochemistry and Health, Vol. 23, No. 1, 2001, pp. 53-63. http://dx.doi.org/10.1023/A:1011082608631
- T. K. Mondal, A. Bhattacharya, M. Laxmikumaran and P. S. Ahuja, “Recent Advances of Tea (Camellia sinensis) Biotechnology—Review of Plant Biotechnology and Applied Genetics,” Journal of Plant Cell, Tissue and Organ Culture, Vol. 76, No. 3, 2004, pp. 195-254. http://dx.doi.org/10.1023/B:TICU.0000009254.87882.71
- S. Seenivasan, N. Manikandan, N. M. Muraleedharan and R. Selvasundaram, “Heavy Metal Content of Black Teas From South India,” Food Control, Vol. 19, No. 8, 2008, pp. 746-749. http://dx.doi.org/10.1016/j.foodcont.2007.07.012
- N. S. Mokgalaka, R. I. McCrindle and B. M. Botha, “Multielement Analysis of Tea Leaves by Inductively Coupled Plasma Optical Emission Spectrometry Using Slurry Nebulization,” Journal of Analytical Atomic Spectrometry, Vol. 19, No. 10, 2004, pp. 1375-1378. http://dx.doi.org/10.1039/b407416e
- S. G. Reeves, P. O. Owuor and C. O. Othieno, “Biochemistry of Black Tea Manufacture,” Tropical Science, Vol. 27, 1987, pp. 121-133.
- K. C. Wilson and M. N. Clifford, “Tea: Cultivation to Consumption,” Chapman and Hall, London, 1992, p. 792. http://dx.doi.org/10.1007/978-94-011-2326-6
- R. M. Gesimba, M. C. Langat, G. Liu and J. N. Wolukau, “The Tea Industry in Kenya; the Challenges and Positive Developments,” Journal of Applied Sciences, Vol. 5, No. 2, 2005, pp. 334-336. http://dx.doi.org/10.3923/jas.2005.334.336
- A. Anandacoomaraswamy, W. A. J. M. De Costa, H. W. Shyamalie and G. S. Campbell, “Factors Controlling Transpiration of Mature Field-Grown Tea and Its Relationship with Yield,” Agricultural and Forestry Meteorology, Vol. 103, No. 4, 2000, pp. 375-386. http://dx.doi.org/10.1016/S0168-1923(00)00134-9
- Kenya Tea Development Agency, “From Bush to Cup (Tea Life Cycle),” 2011. http://www.ktdateas.com/index.php/the-tea/from-bust-to-cup
- Tea Board of Kenya, “Surviving the Economic Meltdown,” 2012.
- F. L. Chung, J. Schwartz, C. R. Herzog and Y. M. Yang, “Tea and Cancer Prevention: Studies in Animals and Humans,” The Journal of Nutrition, Vol. 133, No. 10, 2003, pp. 3268-3274.
- P. Katharine, “Yet More Roles for Tea in Disease Prevention,” Trends Pharmacology Science, Vol. 22, No. 10, 2001, p. 501. http://dx.doi.org/10.1016/S0165-6147(00)01870-8
- R. Richard, “Green Tea Extract May Have Neuro-Protective Effects in Parkinson’s Disease,” Lancet, Vol. 358, No. 9279, 2001, p. 391. http://dx.doi.org/10.1016/S0140-6736(01)05594-5
- T. O. Cheng, “Why Did Green Tea Not Protect against Coronary Artery Disease but Protect Against Myocardial Infarction,” The American Journal of Cardiology, Vol. 91, No. 10, 2003, pp. 1290-1291. http://dx.doi.org/10.1016/S0002-9149(03)00103-6
- R. Hirano, et al., “Comparison of Green Tea Intake in Japanese Patients with and without Angiographic Coronary Artery Disease,” The American Journal of Cardiology, Vol. 36, 2003, pp. 64-70.
- F. Pourmorad, S. J. Husseinimehr and N. Shahabimajd, “Antioxidant, Phenol and Flavanoid Contents of Some Selected Iranian Medicinal Plants,” African Journal of Biotechnology, Vol. 5, No. 11, 2006, pp. 1142-1145.
- S. M. Karori, R. M. Ngure, F. N. Wachira, J. K. Wanyoko and J. N. Mwangi, “Different Types of Tea Products Attenuate Inflammation Induced in Trypanosoma brucei Infected Mice,” Parasitology International, Vol. 57, No. 3, 2008, pp. 325-333. http://dx.doi.org/10.1016/j.parint.2008.02.003
- M. C. Sabu, K. Smitha and R. Kuttan, “Anti-Diabetic Activity of Green Tea Polyphenols and Their Role in Reducing Oxidative Stress in Experimental Diabetes,” Journal of Ethnopharmacology, Vol. 83, No. 1-2, 2002, pp. 109-116. http://dx.doi.org/10.1016/S0378-8741(02)00217-9
- S. Asfar, et al., “Effect of Green Tea in the Prevention of and Reversal of Fasting Induced Intestinal Mucosal Damage,” Nutrition, Vol. 19, No. 6, 2003, pp. 536-540. http://dx.doi.org/10.1016/S0899-9007(02)01097-3
- C. D. Wu and G. X. Wei, “Tea as a Functional Food for Oral Health,” Nutrition, Vol. 18, No. 5, 2002, pp. 443- 444. http://dx.doi.org/10.1016/S0899-9007(02)00763-3
- A. M. Haque, M. Hashimoto, M. Katakura, Y. Tanabe, Y. Hara and O. Shido, “Long Term Administration of Green Tea Catechins Improves Spatial Cognition Learning Ability in Rats,” Journal of Nutrition, Vol. 136, No. 4, 2006, pp. 1043-1047.
- S. Ahmad, et al., “Determination of Mineral and Toxic Heavy Elements in Different Brands of Black Tea of Pakistan,” African Journal of Pharmacy and Pharmacology, Vol. 6, No. 15, 2012, pp. 1194-1196.
- M. T. Soomro, E. Zahir, S. Mohiuddin, A. N. Khan and I. I. Naqri, “Quantitative Assessment of Metals in Local Brands of Tea in Pakistan,” Pakistan Journal of Biological Sciences, Vol. 11, No. 2, 2008, pp. 285-289. http://dx.doi.org/10.3923/pjbs.2008.285.289
- U. Divrikli, N. Horzum, M. Soylak, and L. Elci, “Trace Heavy Metal Contents of Some Spices and Herbal Plants from Western Anatolia, Turkey,” International Journal of Food Science and Technology, Vol. 41, No. 6, 2006, pp. 712-716. http://dx.doi.org/10.1111/j.1365-2621.2005.01140.x
- A. Mehra and C. L. Baker, “Leaching and Bioavailability of Aluminium, Copper and Manganese from Tea (Camellia sinensis),” Food Chemistry, Vol. 100, No. 4, 2007, pp. 1456-1467. http://dx.doi.org/10.1016/j.foodchem.2005.11.038
- Tea Board of Kenya, “Fact Sheet on Kenya Tea. Tea Board of Kenya Statistics,” 2007.
- K. O. Moseti, et al., “Fe, Zn, Cu, Pb and Cd in Tea Grown and Marketed in Kenya; a Quantitative Assessment,” International Journal of Environmental Protection, Vol. 3, No. 6, 2013, pp. 24-30.
- K. Sitienei, P. G. Home, D. M. Kamau and J. K. Wanyoko, “Nitrogen and Potassium Dynamics in Tea Cultivation as Influenced by Fertilizer Type and Application Rates,” American Journal of Plant Sciences, Vol. 4, No. 1, 2013, pp. 59-65. http://dx.doi.org/10.4236/ajps.2013.41010
- R. Thomas, R. W. Sheard and L. P. Moyer, “Comparison of Conventional and Automated Procedures From Nitrogen, Phosphorous and Potassium Analysis of Plant Material Single Digest,” Agronomy Journal, Vol. 59, No. 3, 1967, pp. 240-243. http://dx.doi.org/10.2134/agronj1967.00021962005900030010x
- A. G. Spencer, “Flame Photometry,” Lancet, Vol. 256, No. 6639, 1950, pp. 623-627. http://dx.doi.org/10.1016/S0140-6736(50)91586-8
- MSTAT C, “A Micro-Computer Program for the Design, Management and Analysis of Agronomic Research Experiments, MSTAT Distribution Package,” MSTAT Development Team, Michigan State University, USA, 1993.
- D. A. Armbruster and T. Pry, “Limit of Blank, Limit of Detection and Limit of Quantification,” Clinical Biochemistry Reviews, Vol. 29, Suppl. 1, 2008, pp. 49-52.
- B. O. Kwach, P. O. Owuor, D. M. Kamau and J. K Wanyoko, “Evaluation of Foliar Analysis as a Diagnostic Tool of Predicting Nutrient Deficiencies of Clonal Tea in Kenya,” Asian Journal of Biological and Life Sciences, Vol. 1, No. 1, 2012, pp. 8-18.
- W. N. Omwoyo, et al., “Availability of Some Inorganic Micronutrients and Effects of Grading on Their Levels in East African Black Teas and Their Infusions,” Asian Journal of Biological and Life Sciences, Vol. 2, No. 1, 2013, pp. 42-49.
- A. Kumar, A. G. C. Nair, A. V. R. Reddy and A. N. Garg, “Availability of Essential Elements in Indian and US Tea Brands,” Journal of Food Chemistry, Vol. 89, No. 3, 2008, pp. 441-448. http://dx.doi.org/10.1016/j.foodchem.2004.03.003
- J. K. Wanyoko and C. K. Njuguna, “Variation of Foliar Nutrients of Some Kenyan Tea and Their Relationship with Yield,” Tea, Vol. 4, No. 1, 1983, pp. 37-43.
NOTES
*Corresponding author.

